D-Alloisoleucine: Tracing the Road from Curiosity to Critical Knowledge
Historical Development and Context
Digging into the story of D-Alloisoleucine brings to mind how scientific exploration often stirs up more questions than answers. D-Alloisoleucine is not just a chemical oddity—it's a mirror into the workings of chirality and the fine line separating nature’s patterns from what humans create in the lab. The recognition of D-amino acids as more than synthetic curiosities started decades back when researchers peeled away assumptions about proteins in living organisms containing only L-isomers. Until advances in chromatography and analytical chemistry in the mid-20th century, D-amino acids stayed mostly overlooked, sidelined as contaminants or rare artifacts. Only with more precise tools did biochemists appreciate their quiet but sometimes critical appearances in microorganisms, peptides, and certain metabolic disorders. This finding forced a big rethink about what it means to be “essential” or “natural” in biology and medicine.
Product Overview
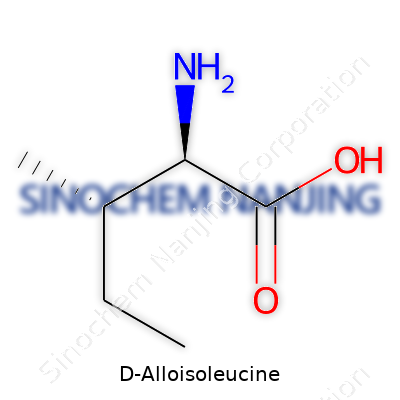
D-Alloisoleucine stands as a D-isomer of isoleucine, a branched-chain amino acid. While L-isoleucine supports muscle metabolism and energy production, its D-version slips into focus when researchers explore rare inborn errors of metabolism, use as standards in laboratory assays, or probe bacterial cell wall structure. Pharmacies, biotech labs, and academic research groups might use D-Alloisoleucine for peptide synthesis or to track isomerization in complex systems. In most natural proteins, it’s absent or extremely rare, but in certain bacteria, D-forms of other amino acids play roles in cell wall architecture. Understanding D-Alloisoleucine’s place in this puzzle matters as much for who needs to avoid it—patients with metabolic disorders—as for those seeking to exploit its strange shape for new therapies or diagnostics.
Physical and Chemical Properties
This compound looks like a white crystalline powder, which sounds mundane unless you spend days sifting through sample after sample in a busy lab. The molecular structure mirrors L-isoleucine’s backbone, just flipped to the D-configured mirror image across the chiral center. Water solubility is moderate, lines up with other amino acids, and it carries the expected behavior in different pH environments—acidic in strong solutions, neutral at its isoelectric point. Keeping D-Alloisoleucine stable means storing it in tightly sealed containers, away from moisture and sunlight. Oxidation or racemization can skew results, turning careful experiments into wild goose chases, which is why anyone working with it learns early to keep things cool and dry.
Technical Specifications & Labeling
In the field, clarity around labeling saves both time and money. D-Alloisoleucine comes specified by its purity, chiral excess, and solubility, but what sets the bar is stereochemical purity—a tiny slip in chiral content can undermine a whole research batch. Well-run labs check for batch consistency and audit supply chains to ensure nothing picks up a hint of L-isoleucine or D-leucine, which could contaminate results. Regulatory rules in regions like the European Union and North America set minimum standards for labeling and documentation, often demanding details about source, intended use, and recommended storage.
Preparation Method
The synthesis of D-Alloisoleucine typically calls for asymmetric methods—enzymatic resolution or chiral catalysis dominate the landscape. Early methods relied on fractions of microbial enzyme systems or on the use of chiral auxiliaries to split racemic mixtures. These techniques often required patience—sometimes tedious stepwise purification or repeated crystallization rounds. In modern practice, directed biocatalysis has allowed more targeted, scalable methods, letting researchers tweak yields, selectivity, and environmental impact. Sourcing enough pure D-Alloisoleucine usually comes down to balancing available technology with demand—pharma rarely needs metric tons, but analytical labs expect batch-to-batch consistency. Waste management matters, too. Unreacted solvents, discarded racemates, and byproducts demand safe handling and careful disposal.
Chemical Reactions & Modifications
For those diving into the chemistry of D-Alloisoleucine, the focus often falls on its reactivity as an alpha-amino acid. Derivatization with fluorophores or isotopic labeling allows scientists to track its journey in metabolic pathways or complex mixtures. The carboxyl and amino groups invite classic reactions—amide bond formation for peptide synthesis, esterification, and salt formation. Modifying the side chain helps researchers tune physical properties, like hydrophobicity or charge, opening up a toolkit for probe design or molecular recognition studies. In the hands of a skilled chemist, even a molecule as small as D-Alloisoleucine offers up avenues for innovation.
Synonyms and Product Names
D-Alloisoleucine bears a handful of alternative names, which can cause confusion. You’ll see it denoted as (R)-2-amino-3-methylpentanoic acid, or sometimes as D-α-amino-β-methylvaleric acid. In catalogues, it may pop up under CAS numbers or abbreviated as D-AIle, among others. Careful pharmacies and researchers keep track of these to avoid costly ordering errors—a simple mix-up between D-Alloisoleucine and its L-counterpart, or even a positional isomer, can derail analytical runs.
Safety & Operational Standards
Lab safety in handling D-Alloisoleucine aligns with general amino acid protocols—gloves, dust masks if powder gets airborne, and good air circulation. Eating, drinking, or smoking in the lab risks accidental ingestion. Cleanup procedures, waste disposal, and emergency protocols should always follow local and institutional rules, since trace exposure at normal levels doesn’t raise alarms, but concentrated accidental ingestion or improper chemical mixing can challenge even seasoned technicians. Occupational health guidelines stress regular training and access to clear, up-to-date safety data.
Application Areas
Research interest in D-Alloisoleucine swells most when clinicians see rare metabolic conditions that throw up elevated D-amino acids in patient fluids, sometimes signaling a breakdown in amino acid processing or renal function. Routine screening in newborns traces D-isomers for early spotting of maple syrup urine disease. Outside of diagnostics, D-Alloisoleucine becomes a valuable internal standard for chromatography, helping analysts untangle complex peptide mixtures. In synthetic peptide chemistry, swapping in D-isomers lets researchers explore stability and resistance to enzymatic breakdown, a central question in drug design. The world of bacterial cell walls, full of D-amino acids, hints at future discoveries where D-Alloisoleucine could play a cramped yet important role.
Research and Development
Discussion about amino acids usually drifts toward L-forms, but D-Alloisoleucine’s place in research stays secure among those chasing rare biomedical questions. Current research uses it as a biomarker for inherited metabolic disorders, which are rare enough that only major pediatric and genetic centers see them often. As analytical detection becomes more precise—think mass spectrometry and high-performance liquid chromatography—the window for D-Alloisoleucine measurement opens wider. Improved synthesis methods now let chemists produce more consistent, purer forms for experiments, giving researchers confidence to trust their controls. In the pharmaceutical realm, tweaking peptides by adding D-Alloisoleucine helps drug hunters improve stability against degradation in the bloodstream, extending the half-life of candidate molecules. There’s ongoing debate about the potential for D-amino acids in controlling peptide folding or mimicking certain biological actions, with D-Alloisoleucine sometimes starring in the supporting cast.
Toxicity Research
Toxicological data for D-Alloisoleucine point mostly toward its low inherent risk under controlled use in labs, though accidental ingestion, particularly in vulnerable populations like infants or those with existing metabolic disorders, can cause trouble. The rare metabolic pathways handling D-amino acids fall short in people lacking key enzymes, leading to unwanted accumulations that can show up as neurological symptoms or kidney strain. Animal studies do not flag dramatic acute toxicity, but disruption in metabolic pathways always draws attention from clinicians watching at-risk patients. For routine lab exposure, the usual amino acid handling guidelines suffice, but clinical practitioners pay close attention to lab values and patient symptoms when D-Alloisoleucine appears outside expected contexts.
Future Prospects
Prospects for D-Alloisoleucine rest at the crossroads of rare disease research, refined analytical chemistry, and peptide engineering. Metabolomics, a fast-growing field, will keep shining a light on D-amino acids that signal subtle changes in health or disease. In drug development, using D-isomers to protect therapeutics from breakdown shows real promise, even as researchers puzzle over how these molecules behave in actual human bodies. Environmental microbiology may unearth fresh uses too, where D-variants in bacterial systems help unravel resistance mechanisms and push new antibiotic designs. Keeping up with advances in synthesis and affordable, high-quality supply will make sure research doesn't stall. Expect more surprises as international teams dig into the role of rare amino acids in both health and disease.
What is D-Alloisoleucine used for?
The Role of D-Alloisoleucine in Medicine
People often overlook the lesser-known amino acids. D-Alloisoleucine, for example, doesn’t pop up often in everyday health conversations. It stands out because it's not the version of isoleucine you get from protein-rich food like meat and eggs. Instead, it belongs to the “D” side of amino acids, which act differently in the body than the natural “L” forms found in most proteins. Scientists have learned that D-Alloisoleucine has a key role in identifying serious metabolic conditions, particularly maple syrup urine disease (MSUD).
How D-Alloisoleucine Became a Diagnostic Marker
D-Alloisoleucine doesn’t appear in healthy people. Its presence in blood or urine rings alarm bells for doctors and lab specialists. In MSUD, the body can’t break down certain amino acids, leading to toxic buildup. D-Alloisoleucine emerges in high amounts as one of these byproducts. Labs run special tests and use high-tech equipment, such as tandem mass spectrometry, to catch this silent warning sign early on. Early detection saves lives — newborn screening programs rely on markers like this to spot trouble before symptoms turn severe.
Why Early Detection Shapes Lives
Growing up with a friend who had a rare inherited disease taught me firsthand how tough life can become without proper diagnosis. Parents often struggle for answers before someone finally puts a name to their child’s condition. Early signs of MSUD mimic common baby illnesses, so families might not recognize the danger in time. Tests that include D-Alloisoleucine change this story. They pull the hidden diagnosis to the surface, letting families and doctors step in fast with special diets or medical care. These quick actions make the difference between lifelong disability and a shot at a healthy childhood.
Research and Future Uses Beyond MSUD
D-Alloisoleucine has scientists curious about possible uses beyond rare disease testing. Some researchers see potential in tracking gut bacteria changes or exploring its role in aging and neurological disorders. The idea comes from ongoing studies that link abnormal amino acid profiles to various health problems, from muscle loss in seniors to the progression of certain cancers. At the moment, these links stay mostly in the research realm, but new findings could spark innovative treatments or earlier warning systems for broader health issues.
Challenges in Accessibility and Awareness
Not every hospital has the gear or expertise to detect D-Alloisoleucine. Rural areas and low-resource settings often miss out on early, accurate testing. I have met doctors frustrated by this gap — they know the science exists but can’t offer it to patients in need. Improving access starts with more investments in public health labs and training for young technicians. Education about rare diseases also matters. People, especially parents, learn to ask the right questions once they recognize signs that don’t fit simple explanations.
Hope Through Collaboration
When specialists, families, and community groups unite, they push governments and research groups to invest in better testing and treatment options. Support networks exchange knowledge about where advanced testing is available and share stories that remind policy makers every rare disease case has a face. By raising awareness about markers like D-Alloisoleucine, the path to faster diagnosis and care becomes clearer for everyone dealing with these tough health journeys.
Is D-Alloisoleucine safe for human consumption?
What Stands Behind D-Alloisoleucine?
D-Alloisoleucine, an uncommon form of the amino acid isoleucine, rarely finds itself in food or medicine compared to its better-known sibling, L-isoleucine. Most amino acids people get from diets come in the “L” form, which the body uses for everything from muscle repair to immune strength. The “D” form, like D-Alloisoleucine, pops up rarely in nature and for decades hasn’t held much attention outside of scientific labs or specialized testing.
How D-Alloisoleucine Appears in the Human Body
Doctors sometimes see D-Alloisoleucine show up in tests run on children suspected of having maple syrup urine disease (MSUD)—a rare disorder tied to how the body breaks down certain amino acids. In these rare cases, D-Alloisoleucine leaks into blood and urine, hinting at metabolic issues rather than providing any benefits. This background tells us the body isn't built to handle large amounts of D-Alloisoleucine; its presence sends up red flags for disease, not health.
No Long-Term Research on D-Alloisoleucine in Diets
Scientists have studied the common L-amino acids for decades, so health agencies know what they do and how much is safe. D-Alloisoleucine has not had the same spotlight. I searched medical journals and safety reviews, but there’s little to no data on humans eating supplements containing D-Alloisoleucine. This lack of evidence leaves open questions no one can answer with certainty. Even for rats and other lab animals, studies stay thin.
Possible Risks from D isomers
D-amino acids, as a group, don’t play much of a role in people’s nutrition. Some D-forms show up as impurities in synthetic supplements and food additives, so food safety agencies in the US, EU, and Japan pay attention during production. L-isoleucine is safe, but D-alloisoleucine could potentially interfere with regular metabolic processes. The science suggests that bodies may not process D-forms properly, so they could build up and cause problems for the brain or kidneys. In MSUD, for instance, high D-amino acid levels damage the brain if left untreated. The only small hint of safety comes from rare cases where people with MSUD tolerate tiny amounts without immediate deadly effects—hardly reassuring for general snacking.
What Could Build Trust?
People reading labels deserve transparency. Companies or researchers eager to add D-Alloisoleucine to foods or supplements would need to run animal studies, then controlled human trials, covering short and long-term use. Until researchers do that and make the results public, regulators can’t say D-Alloisoleucine belongs on supermarket shelves. Nutritionists already warn about less-researched amino acids, asking people to stick with proven, safe forms unless under direct medical guidance.
The Practical Route: Trust What’s Proven
Millions stay healthy by eating foods rich in L-amino acids—nuts, dairy, whole grains, fish—knowing these have decades of research behind them. Novel amino acids with little data walk a risky path. People want to hold onto the belief that new ingredients showing up in formulas get some sort of vetting. For D-Alloisoleucine, the story remains unfinished, but right now, every fact points to caution and sticking with ingredients everyone knows are safe.
What is the difference between L-Alloisoleucine and D-Alloisoleucine?
Looking Beyond the Labels: Two Very Different Amino Acids
Amino acids can trip up even experienced biochemists. Flip a molecule a certain way, and suddenly the body treats it as an outsider. That’s what goes on with L-Alloisoleucine and D-Alloisoleucine. They look quite similar on paper, but their behavior in living things couldn’t be more different.
The “L” and “D”: Nature’s Left and Right Hands
Proteins in all plants, animals, and microbes build themselves mainly from “L” forms of amino acids. This “L” stands for “levorotatory,” and in L-Alloisoleucine, the atoms arrange in a pattern the body can weave into muscles, tissues, and enzymes. This isn’t just a chemical trivia fact. Doctors use L-Alloisoleucine in rare disease diagnosis—especially for maple syrup urine disease—because people’s bodies can start accumulating it when something goes wrong with their metabolic machinery.
D-Alloisoleucine doesn’t show up naturally in the body’s protein-making. This “D” variety, called “dextrorotatory,” mirrors the “L,” but the orientation of its side chains means enzymes can’t use it for building and repairing tissue. Some bacteria pull off tricks using the “D” forms, which end up in cell walls, but most higher animals barely touch them.
Why the Difference Matters: Biochemistry at Work
The consequences show up far beyond the laboratory flask. Proteins have strict rules about which building blocks fit. L-Alloisoleucine slips into these structures naturally, folding into the right shapes. D-Alloisoleucine gets rejected. For anyone using protein supplements, only “L” forms can play an effective part in supporting muscle recovery or nutrition. D-forms just float around doing little, or, in rare cases, disrupting normal processes.
During my time in the hospital lab, I’ve seen confused parents panicking over metabolic disorders when doctors order amino acid panels. L-Alloisoleucine spikes in those rare inborn errors, giving early warning of trouble that trained eyes can spot. D-Alloisoleucine never raises its head; its presence would usually mean contamination or a mistaken reading.
Looking for Clarity in the Supplement Industry
Supplement companies sometimes try to blur these distinctions. It makes a difference in purity and benefit. If the tub on the shelf lists just “alloisoleucine” without an “L” or “D,” buyers ought to ask questions. Reputable suppliers pledge the “L” form and can show independent lab data. Without that, there’s no way to know what’s inside.
More Than a Chemical Curiosity: Health and Research Implications
Scientific research uses specific amino acid forms to untangle life’s puzzles, but that knowledge should trickle down to people’s everyday understanding of what goes into the body. The “L” and “D” thing seems minor—just a letter—but it influences how the body performs, how doctors diagnose disease, and what researchers uncover next.
So, L-Alloisoleucine and D-Alloisoleucine might share a name and a backbone, but in real life, they head down different paths. One plays an essential role in life’s daily chemistry; the other mostly stays in the background, interesting mainly to academics and a few microbes.
How should D-Alloisoleucine be stored?
The Focus on Purity and Freshness
Working with delicate amino acids like D-Alloisoleucine has taught me that little things can make big differences. Some folks overlook storage and wonder later why they get inconsistent results. D-Alloisoleucine isn’t something you leave lying around. It attracts moisture, and humidity is its worst enemy. Even a little bit in the air can spoil your supply. Exposure to air can also cause oxidation, slowly changing its character in ways you don’t want—especially if you use it for analytical work, research, or pharmaceuticals.
Imagine buying your chemicals in top condition and, a few months later, discovering they’re not performing as expected. Researchers talk about this all the time. They trace the trouble back and often find out the bottle was left open or sat too long at room temperature. All that money and effort, thrown away because of careless storage.
Temperature and Light Aren’t Just Details
Every time I worked in a lab without a dedicated cold storage, I saw more waste and more frustration. Stores room temperatures will speed up degradation. Heat, whether from equipment or sunlight through a window, can break down D-Alloisoleucine before you use up the container. Keeping it below -20°C in a freezer not only slows down deterioration but also helps the chemical stay the same as it was the day you got it. It means research isn’t compromised months down the line. Also, light—especially direct sunlight—can trigger all sorts of changes in the compound. Storing your supply in a dark container inside a freezer has made a world of difference in my experience.
Moisture and Contamination: Hidden Problems
It’s easy to underestimate how quickly water from the air sneaks into open bottles or poorly sealed bags. Once inside, moisture can cause D-Alloisoleucine to stick together or even react with other compounds in the mix. Lab coordinators now push for airtight vials, sometimes with silica gel to suck up stray moisture. These steps aren’t difficult, yet they cut risk for everyone who depends on the chemical down the chain—researchers, students, even doctors who rely on the accuracy of reference standards.
Handling Tips for the Real World
If you want reliability, start by dividing large quantities into smaller, single-use vials. That way, you only open what you need, leaving the rest untouched by light and air. Use gloves and clean scoops to avoid skin oils or other cross-contaminants. Always label the date of opening and store unopened supplies back in the freezer right away. Simple routines like these keep quality high and expenses low.
Industry Practices and Room for Improvement
Labs often set up storage with failsafes, but people still make mistakes when moving supplies or working in a hurry. Automated alerts for fridge temperatures or inventory checks could really help, and more training on chemical stability wouldn’t hurt. Even the best compound can’t perform if it’s mistreated. Careful storage makes all the difference, and isn’t just good practice—it protects budgets, experiments, and trust in results.
What is the purity level of the D-Alloisoleucine product?
Purity isn’t just a number on a certificate
D-Alloisoleucine, a less-common cousin of the protein-building amino acids, rarely gets the spotlight. Still, for anyone who’s worked in a lab or industry where trace impurities can throw off an entire project, the value of knowing the real purity percentage hits home fast. Most commercially available D-Alloisoleucine products claim purity levels that reach above 98%, often even stretching to 99%. This can sound perfect, but those last decimal points tell a meaningful story.
Why chemists and researchers obsess over this stuff
A reliable purity reading is more than a quality badge; it means experiments work out the same across countries and months. In my experience, working with amino acids that fall below top purity levels usually introduces noise—tiny chemical “accents” that can mess with analysis, reactions, or bioactivity assessments. For something like D-Alloisoleucine, often used in advanced peptide synthesis or metabolic research, even a small wobble from contaminants such as L-alloisoleucine, D-isoleucine, or trace solvents can skew a researcher’s results.
Facts about testing and standards in the real world
Producers rely on tools like HPLC (high-performance liquid chromatography) and chiral analysis to gauge purity, and the best vendors publish these results on batch-specific Certificates of Analysis. Some labs push beyond, testing for endotoxin contamination or cross-checking optical rotation to rule out hidden racemates. Having run extra controls in past projects, I can say without hesitation that “99% pure” sometimes glosses over tiny but significant differences, especially at industrial or clinical scales where every fraction of a percent builds up.
The stakes for scientists and companies
Researchers working on metabolic disorders or designing new peptide drugs need more than assurances—they need rock-solid, reproducible building blocks. If you’re trying to make a peptide chain that folds just so, even a trace of a wrong isomer or leftover synthetic catalyst will get in the way. These impurities can show up as background noise in mass spectrometry or biological assays, muddying data and wasting time. Years back, I saw an entire drug screening botched because a supplier swapped a 98.0% product for a 99.8% one mid-stream; only after weeks of troubleshooting did the real source emerge.
Getting to the real number
No matter how impressive a data sheet looks, the daily realities of handling D-Alloisoleucine come down to running your own quality check, especially ahead of any critical or clinical application. Researchers who order from high-quality suppliers—those open about their analytical methods—get better results and fewer headaches. Ask for full HPLC traces, check for comprehensive impurity profiling, and keep your guard up for older stock, where time alone can change purity.
Raising the standard across the industry
For all the talk about progress and efficiency, the single most useful habit is transparency between supplier and customer. Companies should show both purity data and the raw chromatograms, not just a summary on paper. Labs, especially those doing sensitive work, benefit from supplementing the supplier’s numbers with their own quick checks. An open conversation about what “purity” should mean, step by step, serves both the bottom line and the final results in the lab.
| Names | |
| Preferred IUPAC name | (2S,3S)-2-amino-3-methylpentanoic acid |
| Other names |
(2R,3S)-2-Amino-3-methylpentanoic acid Allo-Isoleucine allo-Ile 2-Amino-3-methylpentanoic acid |
| Pronunciation | /ˌdiː ˌæl.oʊˌaɪ.səˈluː.sin/ |
| Identifiers | |
| CAS Number | 35148-24-4 |
| 3D model (JSmol) | `/showmol.cgi?sid=8402524509&mol=JSmol&cid=67928` |
| Beilstein Reference | 1537605 |
| ChEBI | CHEBI:15576 |
| ChEMBL | CHEMBL88230 |
| ChemSpider | 3476748 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.131.066 |
| EC Number | EC Number: 204-561-7 |
| Gmelin Reference | 67799 |
| KEGG | C06425 |
| MeSH | D-Alloisoleucine MeSH: C030986 |
| PubChem CID | 439530 |
| RTECS number | WH3010000 |
| UNII | 6T8S17326B |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID60713704 |
| Properties | |
| Chemical formula | C6H13NO2 |
| Molar mass | 145.19 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.22 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | -2.2 |
| Acidity (pKa) | 2.29 |
| Basicity (pKb) | pKb: 10.79 |
| Dipole moment | 5.9275 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | “-528.4 kJ mol⁻¹” |
| Std enthalpy of combustion (ΔcH⦵298) | -2181.2 kJ/mol |
| Pharmacology | |
| ATC code | A16AA23 |
| Hazards | |
| Main hazards | Hazardous if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | No hazard statements. |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P321, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Flash point | 225.2 °C |
| Autoignition temperature | Autoignition temperature: 540°C |
| LD50 (median dose) | Mouse oral LD50: >4 g/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 µg |
| Related compounds | |
| Related compounds |
Isoleucine L-alloisoleucine L-isoleucine D-leucine D-norleucine |

