Cystamine Dihydrochloride: A Deep Dive from Lab Bench to Daily Impact
Historical Development
Looking back, Cystamine Dihydrochloride didn’t enter the scientific world overnight. Researchers noticed its unique structure over half a century ago, at a time when the hunt for new antioxidants and radical scavengers was just kicking off. Early on, chemists, eager to discover sulfur-containing molecules, experimented with disulfide compounds as potential radioprotectants. Cystamine Dihydrochloride’s twin amino ends linked by a disulfide bond stood out, and scientists started using it in labs by the 1960s, mainly in basic redox chemistry. Since then, its uses widened. Over the years, new production techniques made it more accessible, cheaper, and of a higher purity, encouraging both academic and industrial groups to test it in fresh applications, from medical models in rats and mice to large-scale fermentation processes in biotech companies. Even now, its story keeps unfolding: today’s studies look further into neuroprotection and rare genetic diseases, showing that progress rolls on, step by step.
Product Overview
Cystamine Dihydrochloride usually comes as a white crystalline powder that dissolves easily in water, making it practical for application in both wet and dry laboratory settings. It’s a small molecule, meaning it can travel through cell membranes and act inside the body. This chemical works as a reducing agent—handy for neutralizing harmful oxidants and balancing redox reactions in biological and industrial chemistry. Rather than sitting on the shelf, this compound finds its way into gene therapy labs, radiobiology clinics, and manufacturing lines for pharmaceutical intermediates. Wide interest in this chemical mirrors its adaptability: scientists use it for everything from treating cellular stress to modifying proteins, all thanks to its straightforward yet powerful molecular structure.
Physical & Chemical Properties
Cystamine Dihydrochloride’s most obvious features come down to its appearance and solubility. At room temperature, it forms solid crystals that dissolve rapidly in water and, to a lesser extent, in alcohols. This property fits many experimental protocols where water-based solutions matter. The compound’s molecular formula, C4H14Cl2N2S2, highlights its double amino groups and central disulfide linkage—a structural motif that helps it break and form bonds with other sulfur-containing molecules. Its melting point runs in the range of 220-225°C (with decomposition), making it brittle under heat. It smells faintly sulfurous, and the hydrochloride salts enhance stability and shelf life in air-exposed conditions, something any lab manager can appreciate.
Technical Specifications & Labeling
Labs host cystamine dihydrochloride in tightly labeled containers, often specifying a purity over 98%, which meets most research and industrial criteria. Labels ought to highlight both the chemical structure and relevant safety data, like the batch number, expiry dates, and precise storage conditions—usually cool, dry shelves away from direct sunlight. Technical datasheets describe not only the basic stats, such as molecular weight (227.16 g/mol), CAS number, and standardized test results for chloride content, but also stipulations on heavy metals or organic impurities. In my own work, clear labeling creates confidence among team members, especially during regulatory inspections or when troubleshooting unexpected results during synthesis.
Preparation Method
Chemists produce cystamine dihydrochloride through a controlled oxidation of cysteamine hydrochloride. The process relies on forming the disulfide bond at the heart of the molecule, typically using mild oxidizing agents under acidic conditions to prevent unwanted side reactions. Post-reaction workup includes steps to isolate the pure dihydrochloride salt, ensuring that no excess reagent or byproducts contaminate the finished powder. Years back, I remember how temperature adjustments and careful pH monitoring during synthesis made a huge difference—not just in yield but in the consistency that lets suppliers deliver the same batch quality time after time.
Chemical Reactions & Modifications
Cystamine Dihydrochloride excels at playing both donor and acceptor in disulfide exchange reactions. Drop it into a mixture with other thiols, and bonds start to shuffle. This characteristic makes it popular for crosslinking proteins or modifying polymers in biochemical experiments. Breaking the disulfide bridge with reducing agents like DTT or TCEP regenerates cysteamine, a reaction you’ll witness in many protocols to study enzyme activities or molecular signaling. The molecule also tolerates further N-alkylation and acylation on its amino groups, unlocking the door to a suite of derivatives with tailored properties for specific therapeutic or diagnostics needs. Years of bench work taught me that the reactivity of the disulfide bond can prove both an asset for rapid chemical manipulation and a hazard if stability under storage or use gets overlooked.
Synonyms & Product Names
Ask three chemists, and they might give three names for cystamine dihydrochloride. Some call it Dicystamine dihydrochloride, while others refer to it as 2,2'-Dithiobis(ethylamine) dihydrochloride, or simply cystamine hydrochloride in catalogs. Recognizing alternate names matters, especially during literature searches or cross-referencing product lots for different vendors. Mismatches between synonyms cause confusion and cost precious time, something I’ve experienced firsthand coordinating multi-lab projects where each team used their own shorthand.
Safety & Operational Standards
Cystamine dihydrochloride lands on the list of chemicals requiring respect and diligence. Direct contact with skin or mucous membranes may trigger irritation, and inhalation of airborne powder can prompt respiratory discomfort or more serious symptoms for those sensitive to dust. Lab protocols should always include gloves, lab coats, and well-ventilated workspaces. Emergency eyewash stations and spill kits are not just window dressing—they save the day when a powder cloud or accidental spillhead runs the risk of turning routine work into a health hazard. Any disposal must follow local chemical waste rules, as improper release into drains or trash creates risks not only for lab workers but also for the wider community.
Application Area
Cystamine dihydrochloride makes its mark in both research and applied science. In medicine, it’s been trialed for treating rare genetic disorders like cystinosis and holds promise for curbing neurodegenerative damage in animal studies. It also works in radioprotection, absorbing harmful radiation effects in experimental models. Beyond medicine, it acts as a crosslinking agent in manufacturing stable protein gels or biopolymers, bridging academic curiosity and downstream industrial relevance. Over the years, its twin role—as both a research tool and a therapeutic lead—showed scientists how basic chemistry can reach into hospital clinics and industrial vats with tangible benefits.
Research & Development
Active research into cystamine dihydrochloride spans multiple disciplines. Drug developers target its potential to counteract diseases tied to oxidative stress and protein misfolding. Some teams focus on crafting new delivery vehicles or modifying the molecule to improve selectivity and half-life in living systems. I’ve seen collaborations between chemists, biologists, and engineers lead to patent filings and pre-clinical studies, all chasing the next breakthrough. Data sharing and preprint archives speed up cycles, turning isolated findings into building blocks for larger projects. These efforts, rooted in experimental evidence and case studies, create learning loops, ensuring that insights don’t stay trapped in one lab or company.
Toxicity Research
Safety trials run deep for cystamine dihydrochloride, both in test tubes and living animals. Acute toxicity studies find that it can cause adverse reactions at higher doses, including seizures or organ stress in murine models. Scientists have mapped these risk zones and refined dosing regimens, minimizing side effects for both clinical candidates and research animals. Teams rigorously assess metabolites to rule out carcinogenic or mutagenic breakdown products. My earliest work in toxicology reinforced the value of transparency and repeatability in these tests—poorly reported side effects ten years ago led to lessons about careful record keeping and publishing raw data.
Future Prospects
Few compounds carry as much potential, especially as understanding of oxidative stress and cell damage grows. Researchers target cystamine derivatives for new therapies in areas like Parkinson’s, Huntington’s, and radiation injury, backed by robust clinical and preclinical experiments. The compound may also see use in stabilizing novel biomaterials—something clean-tech startups and big biotech firms push as the race for sustainable production lines heats up. Scientific and regulatory teams focus more on improving purity and reducing environmental impact during manufacturing. In years to come, more refined analogs and delivery systems should open fresh applications, both in treating disease and in advancing bio-industrial processes. Engaging collaborations, open data, and constant vigilance on safety shape this compound's journey—reminding anyone in the field that progress depends on both skill at the bench and clarity in communication.
What is Cystamine Dihydrochloride used for?
Beyond Chemistry Classrooms
Cystamine dihydrochloride might sound like an ingredient you’d only find in a high-level chemistry lab, but its uses stretch far beyond textbooks. People like me who dig through pharmaceutical and biomedical research often see this compound pop up when scientists search for solutions against nerve damage, radiation effects, and even certain genetic disorders. The work I do keeps introducing me to ways cystamine dihydrochloride walks the line between promise and practicality in medical research.
Fighting Neurological Disorders
Researchers often explore cystamine dihydrochloride’s potential in the treatment of neurodegenerative diseases. In Huntington’s disease, for example, the brain cells begin to die off due to a faulty protein. Cystamine dihydrochloride seems to help by blocking specific enzymes (transglutaminase) that play a part in the damaging process. I remember reading a study out of Massachusetts General Hospital, where scientists treated mice with the compound and saw the animals maintain better movement and coordination. They lived longer, too. The compound doesn’t cure, but it helps keep some of the worst symptoms at bay in early trials.
Shielding Against Radiation Damage
No one likes to think about accidents in nuclear plants or patients facing harsh cancer treatments, but radiation sickness isn’t just science fiction. Cystamine dihydrochloride attracts attention because it can mop up harmful molecules known as free radicals. During radiation, cells get hit with these damaging particles, and cystamine dihydrochloride steps in to neutralize some of them. The U.S. Department of Defense spent time in the ‘80s and ‘90s looking for ways to protect workers and soldiers, and this compound came up over and over because it can slow down some devastating tissue damage if given ahead of exposure.
Building Better Drug Delivery
Anyone who’s had a stubborn infection or needed long-term antibiotics knows that getting medicine into the right spot isn’t always simple. Cystamine dihydrochloride often ends up in research about nanocarriers—tiny vehicles designed to deliver drugs directly where the body needs them. The molecule’s unique properties help stabilize these carriers. In practice, this might someday mean fewer side effects and better outcomes for tough-to-treat diseases.
Supporting Knowledge with Research
I like to check my sources before repeating health claims. A 2019 review from Journal of Cellular and Molecular Medicine highlighted cystamine dihydrochloride’s proven ability to boost levels of antioxidant molecules like glutathione, a major player in protecting cells from stress. Another group from the University of Cambridge shared data in 2021 showing reduced cellular death in Parkinson’s models after adding cystamine dihydrochloride to treatment.
Challenges and Improvements
Working with cystamine dihydrochloride brings up real hurdles. At high levels, it can be toxic—balance matters. Researchers have choices to make: how much is safe, how often to give it, what happens with long-term use. Clinical trials in humans still drift in early phases. One answer lies in smarter, targeted drug delivery, which would mean cystamine dihydrochloride does its job precisely where needed and doesn’t linger where it can cause harm. Better measurement tools and real-world studies can help untangle possible risks.
The Road Ahead in Health Science
Cystamine dihydrochloride may not grab headlines like aspirin or penicillin. Still, it carries big potential for folks working on tough diseases and scenarios where classic drugs fall short. As I follow the progress in journals and conferences, I notice real hope for harnessing its benefits while finding ways to dodge its downsides. Guided by detailed research, collaboration, and honest risk assessment, cystamine dihydrochloride might carve a place as a helpful ally in modern biomedical science.
What is the chemical structure and formula of Cystamine Dihydrochloride?
A Closer Look at Cystamine Dihydrochloride
Cystamine dihydrochloride doesn’t pop up much outside of research labs, but its structure hides a lot of potential. Underneath the white powder sits a backbone of two carbon chains linked by a single sulfur-sulfur bond. Anyone studying biochemistry probably remembers that disulfide bonds play a big part in holding proteins together. Cystamine taps into that chemistry, but it’s been tweaked for different uses.
The chemical formula for cystamine dihydrochloride is C4H14N2S2·2HCl. Those hydrochloride parts matter a lot for its stability and how it behaves. When hydrochloride salts get added to amine compounds, they often become easier to dissolve in water. That makes cystamine dihydrochloride valuable for scientists needing a quick, even solution, especially in biological tests.
Breaking Down the Structure
This compound features two -NH2 (amine) groups on each end of a two-carbon chain, bound in the middle by a sulfur-sulfur link (disulfide bond). Picture it like this: H2N-CH2-CH2-S-S-CH2-CH2-NH2. Each amine group grabs a proton from hydrochloric acid, forming a dihydrochloride salt. That’s what leads to its official name and formula.
Chemists see the disulfide bridge as a switching point. In biological systems, disulfide bonds break and reconnect during all sorts of cellular repairs and reactions. Cystamine, thanks to its structure, plays a role in controlling these changes. It can either snap apart into two cysteamine molecules under reducing conditions, or hold firm and link things together.
Why the Structure Matters
Despite looking similar to amino acids like cysteine, cystamine’s extra ethylene groups make it act differently. Its disulfide bond lets it rupture under certain conditions, letting the compound latch onto or release various molecular partners. In my own research days working with cell cultures, cystamine showed up as a reagent to block some oxidative reactions. It often protected sensitive proteins from breaking down, which often happens when too many free radicals bounce around a test tube.
Lab work with cystamine dihydrochloride isn’t just academic. It connects with studies about neurodegenerative diseases, cell protection, and even tissue regeneration. Researchers have tested its abilities to limit damage from radiation and slow the breakdown in cell models of Huntington’s disease. A lot of this power comes straight from the stability and reactivity of its chemical structure. Unlike some more fragile compounds, cystamine dihydrochloride keeps its form under normal lab conditions, and dissolves easily once water hits it.
Addressing Safety and Storage
Storing cystamine dihydrochloride requires attention to moisture and air. That hydrochloride salt holds water easily, so any container needs to stay tightly sealed. The powder gives off a smell a bit like rotten eggs, which hints at its sulfur content. Safety data urges handling it with gloves and goggles, since contact can leave skin or eyes irritated. After all, those chemical groups don’t know friend from foe — any strong nucleophile might react if given the chance.
Moving Forward
Several suppliers now make cystamine dihydrochloride accessible for labs worldwide. Quality and purity matter most, since trace contaminants change how tests turn out. Lab teams watch for consistent, highly characterized material that matches the published formula exactly. And as studies broaden, understanding the fine details of cystamine’s chemistry equips researchers to dream up new applications, possibly in medicine, biochemistry, or even materials science.
How should Cystamine Dihydrochloride be stored?
Why It Matters
Cystamine dihydrochloride shows up in labs, manufacturing, and medical research for a reason: it brings unique properties to the table. Its reactive nature, though, makes safe storage more than a box to tick. Skipping careful handling can damage ongoing research, compromise patient health, and risk employee safety. I’ve handled my share of chemicals, and a lapse as simple as leaving a cap loose has made me appreciate the rules. Let’s skip the formalities and focus on what genuinely works.
Environmental Factors and Real-Life Risks
Anyone who’s set foot in a storage room knows: moisture, sunlight, and volatile chemicals don’t mix well. Cystamine dihydrochloride absorbs water straight from the air, turning clumpy and even decomposing faster under the wrong conditions. This transformation isn’t just annoying—it can affect how it reacts in future experiments or processes. Picture opening a container to find a solid mass rather than an even powder. That waste feels both frustrating and expensive.
Direct sunlight brings extra hazards. Light exposure can trigger chemical changes, sending the compound down unpredictable paths. Even a short spell near a sunny window shortens its shelf life and adds unknowns into formulations. My early days in research taught me – a closed, dark cabinet pays off every single time. Fewer headaches, fewer emails asking what went wrong.
Practical Storage Tips from the Trenches
Start with airtight, high-quality containers—preferably glass with a strong seal. Plastic might absorb chemicals or let in tiny bits of moisture over time. Keep the original container if it’s sturdy, but transfer to a better one if the lid wobbles or doesn’t close tightly. Label everything clearly, not just for yourself but for everyone else walking into that storage space. Confusion breeds mistakes.
Cool, dry storage away from heat sources keeps the compound stable. Most labs rely on dedicated chemical fridges or shelving that’s locked and far from direct sunlight. If refrigeration suits your setup, don’t place cystamine dihydrochloride next to food or drink. Cross-contamination risks should not get shrugged off, and regulators have hefty punishments for mixing chemical storage with personal items.
Safety and Regulatory Considerations
Gloves, goggles, and proper respirators make a difference. Accidental spills with cystamine dihydrochloride can irritate skin and eyes quickly. Adequate ventilation in storage spaces, along with spill kits nearby, turns small slip-ups from major emergencies into minor clean-ups. Any time I’ve seen people skip this step, the cleanup costs and downtime hit much harder.
Disposal practices deserve the same level of care. Local regulations dictate proper procedures—never pour leftover solution down the drain. Facilities should train team members on chemical hazards, using safety data sheets as a reference. Audits help spot any lapses, with accountability built in. These moments of discipline keep bigger disasters at bay.
Better Solutions for the Long Term
Digital monitoring for temperature and humidity can flag subtle shifts before problems develop. Automated alerts let staff act quickly, rather than discovering losses at the next inventory check. Making storage and handling guidelines part of regular safety briefings ensures every team member, new or veteran, stays sharp on protocols. Sharing firsthand stories of near-misses drives home best practices better than any memo.
Safe cystamine dihydrochloride storage demands more than just a good shelf. Respect for its quirks, a vigilant team, and a willingness to invest in sturdy storage pay off miles beyond the costs—protecting people, research, and reputations all at once.
What are the safety precautions for handling Cystamine Dihydrochloride?
Understanding the Risks
Cystamine dihydrochloride lands in many labs, from biochemistry to medical research. The risks it poses aren’t always obvious to newcomers. The white powder looks unassuming, but like many chemicals in research work, it carries hazards that call for more respect than fear. I’ve seen folks ignore warnings, maybe because they’ve never had a bad encounter. Direct skin contact leads to irritation fairly quickly, and inhaling dust can leave your nose burning for days. The safest lab I ever worked in nailed the simple mantra: know what you’re handling.
Personal Protection Makes the Difference
The lab coat acts as the first line of defense, not just a uniform. I always wear gloves, and goggles are a must — not afterthoughts on the benchtop. The powder clings to skin and clothes. Even small spills become big headaches if you touch your face or eat lunch without careful hand washing. Disposable gloves, changed often, cut the risk almost to zero. In a warm room, the temptation to work without goggles grows, but a single dust flake on the eye tells the story for weeks. Some forget respiratory masks. I put mine on, especially during weighing and transferring, because there’s no way to guarantee the air stays clear of fine particles.
Ventilation Can’t Be Ignored
A well-ventilated fume hood isn’t just for handling sulfuric acid or chloroform. In my early days, I underestimated the value of good air flow. Cystamine dihydrochloride doesn’t produce clouds, but all it takes is brushing powder off a balance to send it airborne. The best labs I’ve seen place balances and transfer stations under hoods, letting the blower suck away risk. A simple fan on a bench does nothing. Engineers I trust recommend testing hoods twice a year. Filters stop working in dusty environments, and workspace corners gather the invisible stuff that settles overnight.
Safe Storage and Clear Labeling
Mix-ups in the storeroom account for too many close calls. Cystamine dihydrochloride stores safely in a sealed, dry jar. Moisture clumps the powder and makes the cap harder to open next time. I’ve seen someone mistake it for a benign buffer because the label wore off—tape and fading pens don’t cut it where safety’s involved. A fresh, permanent label gives everyone confidence. Shelving matters, too, away from acids or bases that start unwanted chemical reactions. Organization stopped spills before they started, and routine checks catch broken seals before a mess grows.
Training, Teamwork, and Incident Plans
Most labs talk a good game about safety, but putting plans on paper doesn’t save anyone who panics. Simple steps, like a five-minute safety huddle, reinforce what to do if something goes wrong. Everyone on the team should know the location of eye-wash stations and showers. I ran drills with coworkers and found weaknesses — blocked access, hard-to-open exits. Reporting mistakes and near-misses makes the workplace safer for everyone next time. Relearning the basics after each incident isn’t failure; it’s how you build a culture where accidents grow rare. With new chemicals, nobody should work alone.
Practical Precautions, Real Protection
Handling cystamine dihydrochloride calls for practical steps: label everything clearly, always protect your eyes and skin, keep a clean, organized workspace, and never skimp on training. Risks aren’t always visible, but experience and the right preparation keep the laboratory safe for work that matters.
Is Cystamine Dihydrochloride soluble in water?
Understanding the Basics
Cystamine dihydrochloride rarely grabs headlines outside laboratory circles, but ask a chemist about its properties and the conversation turns practical quickly. Water solubility comes up fast. This compound, with its simple structure, holds two hydrochloride salts on a cystamine backbone. Scientists and clinicians cross paths with it when researching cystinosis, investigating gene editing, or developing chemical probes. Knowing if it mixes well with water makes or breaks an experiment—or treatment plan.
What Real-World Experience Tells Us
I spent time in an academic lab preparing small molecules for cell culture work. Sometimes, products proved stubborn, stubborn enough to put your timeline days behind, if not weeks. The one thing you count on in a formulation project: will that white powder actually dissolve in water? Cystamine dihydrochloride answers, yes—dissolves readily. Not just a little. We’re talking about grams per milliliter at room temperature. The high solubility isn’t some incidental bonus. It means you can prepare concentrated stock solutions and dilute as needed. This brings flexibility for university researchers and pharmaceutical professionals alike.
Why Solubility Matters in the Lab
Taking shortcuts with solvent choice usually causes downstream trouble. Poorly soluble compounds mean hazy solutions and wasted chemicals. In enzyme kinetics, for instance, clarity means reliability. Water solubility opens the door for straightforward laboratory protocols. No need to fiddle with organic solvents, no need to heat or ultrasonicate just to force things into solution.
In clinical research, cystamine dihydrochloride’s water solubility means improved consistency. It enters biological systems cleanly when dissolved in saline or other aqueous buffers. This supports better reproducibility and reduces side reactions, both in animal models and cell culture. The science community pays close attention here. If you want data that speaks beyond one laboratory, control over how a drug or research tool behaves in solution comes first.
Safety and Environmental Aspects
Working safely with chemicals takes top priority. Highly soluble materials mix quickly, but they also spread faster in the event of a spill. A researcher handling cystamine dihydrochloride needs gloves, goggles, and solid ventilation—nothing out of the ordinary, but worth restating. The easy water solubility means cleanup is more manageable but also means rapid dispersal into drains. Waste management should never take a back seat. Even trace amounts making it to wastewater streams can pose environmental questions, especially if research ramps up.
Solutions Beyond the Bench
Sometimes a solution to a chemistry problem doesn’t need another reagent. It’s about practical choices. With cystamine dihydrochloride, water does the heavy lifting. If one works with large quantities—or in a setting with tight environmental controls—setting up closed systems with built-in filtration keeps both scientist and surroundings safer. Academic labs could borrow procedures from bigger pharmaceutical settings, treating water waste before disposal. Basic practices like double-checking concentration, logging amounts used, and sharing standard protocols make research smoother for everyone.
Evidence in the Literature
Peer-reviewed journals and data sheets from suppliers like Sigma-Aldrich confirm cystamine dihydrochloride’s water solubility. Chemical catalogs list it with water solubility exceeding 200 mg/mL at room temperature. You can see the clear solution settle within seconds as you stir. Years of practical lab use, alongside these data, keep confidence high among academic, clinical, and industrial users.
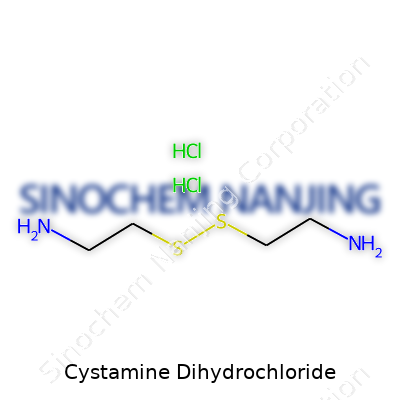
| Names | |
| Preferred IUPAC name | 2,2′-Disulfanediyldiethanamine dihydrochloride |
| Other names |
2,2′-Diaminodiethyl disulfide dihydrochloride Cystamine 2HCl Cystaminum dihydrochloridum |
| Pronunciation | /saɪsˈtæm.iːn daɪ.haɪˈdrɒk.ləˌraɪd/ |
| Identifiers | |
| CAS Number | 56-17-7 |
| Beilstein Reference | 64894 |
| ChEBI | CHEBI:64102 |
| ChEMBL | CHEMBL17838 |
| ChemSpider | 14047 |
| DrugBank | DB04528 |
| ECHA InfoCard | 100.022.368 |
| EC Number | 222-311-2 |
| Gmelin Reference | 75492 |
| KEGG | C00558 |
| MeSH | D08HL73F52 |
| PubChem CID | 60946 |
| RTECS number | MI8580000 |
| UNII | 3M6E033W02 |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID2020409 |
| Properties | |
| Chemical formula | C4H14Cl2N2S |
| Molar mass | 175.17 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 0.633 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -2.0 |
| Acidity (pKa) | 9.35 |
| Basicity (pKb) | 6.71 |
| Magnetic susceptibility (χ) | -7.8 × 10⁻⁶ cm³/mol |
| Dipole moment | 4.44 D |
| Pharmacology | |
| ATC code | V03AB92 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P264, P270, P280, P301+P312, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-3-4-W |
| Lethal dose or concentration | LD50 Oral Rat 165 mg/kg |
| LD50 (median dose) | LD50, Mouse, intraperitoneal: 180 mg/kg |
| NIOSH | PY3675000 |
| PEL (Permissible) | PEL (Permissible exposure limit) for Cystamine Dihydrochloride: Not established |
| REL (Recommended) | 10-50 µM |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Cysteamine Cystine Glutathione Dithiothreitol Dithioerythritol |

