Cyclohexyl Isocyanate: Charting Evolution, Use, and Safety
Historical Development
Tracing back, cyclohexyl isocyanate came out of the crowded field of organic isocyanates in the early 20th century—a period bustling with chemists pushing the boundaries of material sciences. Isocyanates quickly staked their claim as important intermediates in the synthesis of polyurethanes and unique polymers. The cyclohexyl variant earned notice due to its reactivity profile and manageable volatility, setting it apart from its aromatic relatives. Post-World War II, new manufacturing methods and stricter safety legislation pushed both production and handling to new levels, creating demand that didn’t just ride on technical features. Research in dye intermediates and agrochemicals kept cyclohexyl isocyanate in active labs, even as alternatives sometimes gained the spotlight for specific applications.
Product Overview
Cyclohexyl isocyanate delivers reactivity as an aliphatic isocyanate, bridging gaps in industrial and research spaces where aromatic isocyanates—like phenyl isocyanate—don’t cut it. It’s mainly valued for how it incorporates into synthesis pathways for ureas, carbamates, or heterocyclic systems, giving the end compounds robust hydrolytic stability and reduced yellowing under UV. Producers place it in the hands of chemists in labs and plant-scale operations, keeping careful watch on purity specs. The product shows up as a colorless to pale yellow liquid with a sharp, distinct odor, making it easy to identify but impossible to ignore safety.
Physical & Chemical Properties
Cyclohexyl isocyanate holds a molecular formula of C7H11NO, tipped in at a molecular weight of about 125.17 g/mol. At room temperature, the compound remains a mobile, colorless to slight yellow liquid, boiling between 168°C–170°C. Vapor pressure sticks low enough for controlled handling but rises significantly with heat. Solubility plays out best in typical organic solvents—toluene, acetone, ether—but water contact causes brisk hydrolysis, kicking out carbon dioxide and forming cyclohexylamine or its derivatives. Combined with its flash point above room temperature, handling requires an eye for both flammability and chemical splash. Density figures hover around 0.98 g/cm³, and keen chemists remember the strong, pungent odor as a clear warning sign.
Technical Specifications & Labeling
Industrial supply sticks to tight technical specification sheets. Standard purities benchmark above 98.0%, with small allowances for isomeric or solvent residues. Labels must show UN hazard codes (often UN2282), isocyanate warnings, and safeguards against inhalation or skin exposure. Chemical registries pin cyclohexyl isocyanate with CAS number 3173-53-3, and shipments always demand correct DOT class 6.1 toxic material labeling. Material Safety Data Sheets (MSDS) become a constant companion, breaking down handling routines, spill protocols, first aid, and storage rules. Precision meets practicality on these docs, as regulatory agencies keep manufacturers on alert for compliance slip-ups.
Preparation Method
Bulk production typically starts with cyclohexylamine fed through phosgenation—a direct, well-trodden route that balances cost with yield. Modern green chemistry hints at non-phosgene alternatives, but industrial inertia and cost keep the classic approach dominant. Some labs push for rearrangement reactions or the use of carbonyl diimidazole analogs, yet scale and reliability often steer companies back to phosgene. Isolation requires deft fractionation, since moisture ruins the yield and safety margins drop sharply with mishandling. As with many isocyanates, plants running cyclohexyl isocyanate lines split production and packaging into isolated zones, going heavy on containment.
Chemical Reactions & Modifications
Organic chemists ride cyclohexyl isocyanate into a wide range of synthesis opportunities. Mixing with alcohols gives urethanes; water or amines push toward ureas, biurets, or complex heterocycles. Acidolysis isn’t a go-to, but strong nucleophiles or heat can crack the isocyanate group, revealing underlying amines or carbon dioxide in the process. Modified cyclohexyl isocyanates also feed into polymer chemistry, producing elastomers with toughness and stable color. In specialty labs, researchers sculpt custom molecules for pharmaceuticals by leveraging the isocyanate for selective linking—especially vital where aromatic head groups create toxicity or instability. Some surface treatments in plastics even rely on these reactive ends, locking coatings tight to substrate or prepping for further cross-linking steps.
Synonyms & Product Names
In technical literature and trade catalogs, the term cyclohexyl isocyanate goes hand in hand with names like Cyclohexyliocyanate, Isocyanatocyclohexane, and CyHEX-NCO. Chemists juggling catalogs from different suppliers bump into German or Japanese nomenclature, but CAS and UN numbers anchor the identity—avoiding mix-ups with multi-isocyanate versions or aromatic lookalikes. Some polymer and adhesive brands market compounds pre-modified with cyclohexyl isocyanate as curing or crosslinking agents, folding the name into proprietary blends, though keeping the basic isocyanate warning up front.
Safety & Operational Standards
Working with cyclohexyl isocyanate isn’t just about wearing gloves and goggles—long experience in the chemical industry has shown that running a tight operation means real engineering controls. Proper ventilation remains non-negotiable. Respirators, splash-proof suits, and face shields come standard in bulk transfer zones. Leaks demand quick containment, as inhalation or skin exposure triggers strong irritation—sometimes full-blown sensitization. Storage spaces need climate control for temperature and humidity; water presence triggers runaway hydrolysis. The Occupational Safety and Health Administration (OSHA) and similar agencies mandate exposure monitoring, spill drills, and special first aid kits. Engineers in the field talk about walkers on a shop floor needing to understand both short-term and chronic risks—not just ticking off safety checklists. In a pinch, quick access to emergency showers and ventilation beats fancy containment.
Application Area
Polyurethane makers, adhesive formulators, and agrochemical producers pull cyclohexyl isocyanate into production lines that also process complex polymers, crosslinkers, or specialty coating agents. It plays a role in creating flexible foams, tough coatings for floors, non-yellowing lacquers for wood, and even durable sealants in aviation and automotive repair. Chemists assembling pharmaceutical intermediates put it to work in making custom urea linkages, especially when aromatic isocyanates would drag in solubility or safety headaches. Research labs value it in the hunt for new crop protection agents, using it to tweak bioactive molecules for better uptake or environmental profile. In electronics manufacturing, the demand for robust, low-color polyurethanes brings cyclohexyl isocyanate into high-purity blends, tightening process tolerances as device sizes shrink.
Research & Development
Some of the brightest lights in industrial R&D keep chewing on new catalysts or greener prep methods to cut phosgene from the process. Green chemistry projects chase lower emissions, renewable starting materials, and new coupling partners for the isocyanate group. Advances in reaction monitoring—like in-line spectroscopy—hand companies ways to quickly adjust for purity, safety, and conversion in real-time. On the product front, surface chemists test modified cyclohexyl isocyanates for non-stick coatings with added durability or flexibility. Pharmaceutical research continues to scout for bioorthogonal reactions where this isocyanate can slip new functions into complex macromolecules. Agrochemical R&D plugs cyclohexyl isocyanate into combinatorial screens, always searching for the next step change in selective herbicides or safer crop protection. Tech transfer—bridging lab and pilot plant—often proves the hard part, demanding strong collaboration between process chemists, environmental teams, and plant managers.
Toxicity Research
Researchers working with cyclohexyl isocyanate know the risks from decades of animal studies and real-world exposure data. Inhalation irritates eyes, throat, and lungs, sometimes leading to asthma-like sensitivity with repeated hits. Chronic skin contact opens the door for dermatitis or allergic responses. Acute exposure brings headaches, nausea, and potential nervous system effects. Studies in rodents confirm tissue damage at high doses; metabolism in humans leans on detoxification through urea-cycle pathways, but diagnostics after exposure track changes in blood or breath. Regulators peg work-site limits low—often under one part per million—to head off both acute and long-haul effects. Many labs invest in training on spot testing, rapid response, and occupational health support to cover what ventilation and PPE can’t catch.
Future Prospects
Cyclohexyl isocyanate’s future lies at the crossroads of sustainability and performance. The chemical industry faces stronger pressure every year to shrink environmental footprints; this ripples through to isocyanate manufacturing, driving investment in alternative synthesis, emission control, and recyclability in finished products. As regulatory frameworks tighten—especially in regions like the EU—producers tackle the challenge of keeping process economics viable while lifting worker and consumer safety. For end users, the push for low-VOC coatings, longer-lasting sealants in construction, or biocompatible ureas and carbamates in pharma keeps demand solid. Researchers who get their hands dirty with scale-up and production know that innovation on the shop floor beats lab theory every time; large plants shift toward more automated, enclosed handling and digital spill monitoring. As for the molecule itself, as more specialized applications pop up in microelectronics or medical coatings, expect cyclohexyl isocyanate to keep evolving—slow and steady, rather than spectacular, but with real impact felt in tough, safer materials and tighter standards.
What is Cyclohexyl Isocyanate used for?
An Inside Look at a Key Chemical for Industry
Chemists and manufacturers work with cyclohexyl isocyanate because it brings a distinct mix of reactivity and structure to the table. I’ve watched this compound find its way into some surprising corners of chemical synthesis, touching everything from specialized plastics to medicines, long before the finished products hit shelves.
How Cyclohexyl Isocyanate Shapes Polyurethane and Coating Industries
Factories that produce adhesives and coatings often lean on cyclohexyl isocyanate to create polyurethanes. Think of furniture with seamless finishes or high-performance paints: chances are good that compounds building their backbone came from reactions kickstarted by this isocyanate. Polyurethanes made with cyclohexyl isocyanate aren’t just about sticking two things together. These materials set the bar for flexibility and toughness, withstanding temperature changes and mechanical stress that would crack cheaper alternatives. In paint chemistry, these isocyanates play a behind-the-scenes role, fighting off scratches and sunlight damage. It matters, because it means products last longer — less waste and fewer frequent replacements in homes, schools, and factories.
Pharmaceuticals: Small Molecule, Big Contributions
Moving from plastics to pharmaceuticals might seem like a leap, but here’s where cyclohexyl isocyanate stands out again. Researchers in medicinal labs use this chemical as a building block for drug molecules. Through my work with researchers, I’ve seen it help create urea derivatives — compounds that sometimes serve as antiviral or anticancer agents. Isocyanates offer precise chemistry, allowing new drugs to be assembled with a kind of reliable “click” assembly. One example: certain antimalarial drugs draw on building blocks that require cyclohexyl isocyanate at key steps. By enabling efficient, predictable reactions, it cuts down on waste and accelerates development timelines, which matters when new diseases threaten public health.
Challenges: Worker Safety and Environmental Concerns
Every tool comes with risks, and cyclohexyl isocyanate is no different. Ask anyone who has worked in a polymer plant or R&D facility: exposure calls for respect. Without strict handling and ventilation protocols, this chemical can trigger respiratory or skin irritation. Data from regulatory agencies support that exposure above recommended limits can harm workers. Stories circulate about accidents where poor storage led to accidental release and hospital trips. So, safety programs aren’t paperwork — they’re the front line. Training, strong engineering controls, and real investment into safer workspaces ensure people don’t pay with their health for industrial innovation. Some companies already champion automated systems and closed reactors to remove most risk factors, setting examples others can follow.
Moving Forward: Greener Chemistry and New Approaches
As society pushes for sustainability, the chemical industry faces big questions. Cyclohexyl isocyanate stands as a valuable tool, but chemists keep searching for alternatives with lower toxicity or higher biodegradability. Biobased feedstocks and cleaner synthesis methods are picking up steam; these offer a chance to keep the performance but shrink the environmental footprint. Real change depends on investment and cooperation between chemical manufacturers and academics. Looking out for both product quality and worker health, while meeting modern environmental demands, marks the way forward for cyclohexyl isocyanate and everything it helps build.
What are the safety precautions when handling Cyclohexyl Isocyanate?
Why Respect Matters in the Lab
Cyclohexyl isocyanate isn’t something to treat lightly. This chemical packs a punch, especially if you breathe it in or get it on your skin. Working with it, I’ve learned that respect for the process keeps everyone breathing easy. Too many stories out there of someone setting up a reaction without double-checking ventilation, only to feel that burn in the back of the throat a few hours later. People get comfortable. That’s the real threat.
Keeping Clean Air a Priority
Fresh air can feel like a luxury in some older labs. I remember the old fume hoods at my first workplace—they rattled and whined, but they did their job. Fume hoods are non-negotiable with cyclohexyl isocyanate. This chemical can irritate your eyes, nose, throat, and lungs before you realize what’s happening. If you feel dizzy or your eyes start to water, you’ve already been exposed. The Centers for Disease Control (CDC) warns about potential lung damage and even asthma from this compound. Open windows don’t cut it. Chief lesson: always prep and use materials inside a well-maintained fume hood, cleaning the workspace before and after.
The Right Gear Saves Skin
I used to think a simple lab coat and gloves covered everything. Then I watched someone spill just a few drops of cyclohexyl isocyanate on their arm—red, irritated skin, a trip to the medic, and months of being a case study in safety talks. Nitrile gloves, lab goggles, and a solid face shield add layers between chemical and skin. Don’t rely on memory—check that gloves don’t have pinholes. Long sleeves, buttoned cuffs, and pants with no gaps at the ankles or wrists make a difference when working with stuff that reacts fast.
The Quiet Importance of Preparation
Rushing leads to mistakes. My mentor’s rule holds up: always walk through the process before opening a single bottle. Cyclohexyl isocyanate reacts with water—even with humidity in the air—and releases toxic gases. Dry containers, dry tools, and sealed waste bins help stop accidents before they begin. Every time I think I have the steps memorized, I force myself to check the safety data sheet (SDS) again. The breakdown on proper storage—cool, dry, away from acids, bases, and incompatible chemicals—ends up saving more than just time.
What If Something Goes Wrong?
Nobody forgets their first chemical spill. Immediate action beats hesitation. I keep the location of eyewash stations and showers clear in my mind. Speed matters—every second reduces the risk. A good spill kit—complete with neutralizer, absorbents, and protective clothing—belongs at arm’s reach, not in a locked closet down the hall. I learned the hard way that shouting for help instead of handling things alone brings better outcomes. The Environmental Protection Agency (EPA) recommends all waste go into labeled, sealed containers. Clean-up, reporting, and medical attention should follow a clear plan.
Building a Safety Culture from Experience
Nobody works in a bubble. I’ve seen the best results where everyone in the lab checks in with each other. Short meetings before work and a willingness to ask simple questions prevent big problems. Veteran chemists pass on tips about handling cyclohexyl isocyanate—like double-gloving for long tasks or taping glove cuffs to sleeves. Small steps add up to a safer space.
Better Training, Fewer Accidents
Clear, hands-on training works better than any poster on the wall. Watching a demonstration, then practicing under supervision, helps the safety lessons stick. Laboratories can blend real-world stories with guidelines from OSHA, CDC, and local experts. The more open people are about mistakes and lessons learned, the stronger the safety net becomes.
What is the chemical formula and CAS number for Cyclohexyl Isocyanate?
Chemical Formula and CAS Number
Chemists identify Cyclohexyl Isocyanate by a straightforward formula: C7H11NO. Its CAS number is 3173-53-3. For anyone who spends time with chemicals, these codes are like a passport, unmistakable and essential for research, regulations, and safety. Every bottle on the laboratory shelf carries this number, linking it to databases, hazard sheets, and trade systems across the globe.
Why Naming Matters
The specifics on a label can make the difference between a productive day and a dangerous one. Cyclohexyl Isocyanate isn’t just a mouthful, it’s a critical raw material in the formation of custom-made ureas, pesticides, and pharmaceuticals. In my graduate research days, accuracy in labeling mattered. A single digit off in a CAS number meant a wasted order, or worse, unpredictable test results. One vial with the right number gets you certainty. It also means the safety information you’re reading matches what’s in your hands.
Chemical’s Role in Industry
Real-world application for Cyclohexyl Isocyanate extends from polyurethane foams to specialty chemicals for crop protection. Anyone who’s worked near polyurethane manufacturing knows how much these isocyanates influence product quality and safety measures. Manufacturers turn to materials like this one for making tailored reactions work, such as in protective coatings or specialty adhesives. There’s a reason chemical engineers memorize specifics like C7H11NO and 3173-53-3 — plant floors and lab benches reward the detail-oriented.
Safety at the Forefront
Experience has taught me caution with isocyanates. Cyclohexyl Isocyanate poses real health hazards if handled carelessly. It’s volatile and can irritate eyes, skin, and lungs quickly. Knowing exactly what you’re working with means you reach for the right gloves, the fitted respirator, and you keep proper ventilation running. It’s not about paranoia, it’s about avoiding emergency room visits or regrettable incidents. Community standards, like the Globally Harmonized System for classification, depend on precise identifiers to protect people.
Tracking Down the Right Data
Regulatory compliance officers and supply chain managers both lean heavily on numbers and formulas. When inspectors walk into a facility, they check those little numbers printed in black. Too many companies learn the hard way that using ambiguous chemical names can tangle up registrations or block imports. With a correct CAS number — 3173-53-3 — you get straight answers in customs paperwork and Safety Data Sheets.
Paths Toward Safer Chemicals
The chemical industry faces calls for greener alternatives and safer materials every day. Researchers keep digging for ways to replace hazardous isocyanates while holding onto performance. Sometimes the answer is improved handling rather than elimination. Worker training grows more important, especially when people understand the risks tied to ingredients like Cyclohexyl Isocyanate. My time in chemical safety training revealed that most accidents link back to confusion over what’s in a bottle or drum.
Staying Informed
Everyone from bench chemists to logistics staff benefits from knowing what those names and numbers mean. Cyclohexyl Isocyanate remains an example of why precise identification cuts through confusion. Whether you work in a laboratory, along the supply chain, or at a regulatory desk, consistent naming protects both lives and livelihoods. Information turns into power only when it’s accurate — and in chemical work, accuracy starts with the label.
How should Cyclohexyl Isocyanate be stored?
Understanding Its Hazards
Cyclohexyl isocyanate ranks high among chemicals that demand full respect in both lab and industrial settings. Exposure carries real risks—not just sneezing or a mild rash, but deep lung trouble or serious skin burns. My years working around reactive chemicals have taught me that short-term shortcuts often lead to long-term regrets. Improper storage doesn’t just mean financial loss. It endangers everyone nearby, including people never directly handling the substance.
Location Matters More Than You Think
It’s tempting to stash hazardous chemicals on any available shelf. But with cyclohexyl isocyanate, that move turns storage rooms into potential accident sites. I’ve seen facilities try to cut corners by keeping reactive compounds with less volatile products. That choice can cause dangerous reactions—especially if leaks develop or containers fail. Separate storage—away from moisture, acids, and alcohols—truly saves lives. That means a well-ventilated, cool, and dry spot, away from direct sunlight. Never underestimate the chemical’s sensitivity to spills or drips when sorting out a storage plan.
Containers: The Right Choice Prevents Disaster
Not all bottles or barrels work for storing reactive liquids. Polyethylene and steel both play a role, but only if clean, intact, and clearly labeled. Fresh, appropriate seals matter as much as choosing the right door lock for your house. Forgetting to check a stopper or letting a lid corrode risks exposure. I’ve learned that double-checking seals and labels before each use saves trouble, and clear signage never hurts.
Controlling Temperature—A Small Investment, Big Payoff
People in charge of storage sometimes overlook temperature guidelines, figuring heat is just an inconvenience. In reality, a rise above recommended levels can boost pressure inside containers. I once witnessed a storage area become an emergency site, all because no one tracked the room temperature during a summer heat wave. Regular checks create peace of mind. Keep cyclohexyl isocyanate between 2°C and 8°C whenever possible. That small step prevents pressure buildups and leaks that can harm everyone.
Moisture: The Silent Enemy
Even a drop of water spells real danger. Cyclohexyl isocyanate reacts violently with water, forming gas that worsens an already risky spill. I always push for silica gel or other desiccants in storage cabinets handling reactive chemicals. Keeping lids tight and containers dry stops disaster before it starts. If spill kits sit nearby and everyone knows how to use them, quick response turns a near-miss into a story—not a health crisis.
Training: It’s Worth Repeating
Technical skills matter, but clear instruction makes the biggest difference. I never forget how new staff, unsure of proper storage, can unknowingly put everyone at risk. Regular training and visible signage help keep storage safe. Even veterans need refreshers because routines cause forgetfulness. Clean gloves, goggles, and aprons send the message that safety isn’t just a rulebook—it's a culture.
Planning for Emergencies
Even airtight storage rooms don’t remove all risk. Quickly locating safety showers, eyewash stations, and spill neutralizers prevents small mistakes from becoming big health problems. My experience says checklists work. Regular drills and visible emergency numbers keep readiness high. Neighbors, visitors, and other workers all stand safer when people know what to do if something goes wrong.
Last Thoughts
Storing cyclohexyl isocyanate affects everyone in a building, not just the folks working with the substance. Smart storage—through careful location, right containers, strict control over temperature and moisture, ongoing training, and emergency planning—protects health, investments, and business reputation. Regulation exists for a reason, but personal commitment to safety creates the strongest defense.
What are the potential health hazards of Cyclohexyl Isocyanate exposure?
What Cyclohexyl Isocyanate Means for the Body
Cyclohexyl isocyanate doesn’t have the sort of reputation most people bump into at the grocery store, but plenty of people work with chemicals just like it in labs, factories, and sometimes right down the supply chain in manufacturing. Many chemicals in this family come packed with danger, but few walk in with as many hidden risks. Once this stuff hits the air, it brings irritation to the eyes, nose, throat, and lungs. Walking into a cloud of it feels a lot like what I’ve experienced near paint or resin fumes at an old job—eyes start to water, breathing gets tricky, and headaches creep in fast.
Short-Term Problems Hit Fast and Hard
No matter how tough someone thinks they are, Cyclohexyl isocyanate takes hold quickly if it gets on your skin or in your eyes. Redness, rashes, and even small blisters can pop up after a short touch. If someone doesn’t remember gloves or goggles, that first mistake can bring a painful lesson. Breathing even a bit of its vapor at close range triggers intense coughing, chest tightness, and sneezing so strong it’s hard to ignore. People I’ve spoken to in the local auto body industry remember small spills that sent coworkers to emergency rooms for oxygen. An immediate wash-down sometimes helps, but once those fumes hit the lungs, recovery can take days.
Long-Term Risks Stack Up Quietly
The risks don’t clock out with a clean-up. Over months or even years, repeated hits from low levels of Cyclohexyl isocyanate add up. People exposed over time may notice breathing problems showing up unexpectedly—think asthma, wheezing, and a cough that doesn’t go away. Studies from the American Journal of Industrial Medicine lay it out clear, showing a much higher rate of occupational asthma in workers dealing with isocyanates. Memory from friends in manufacturing jobs sticks with me—folks too young to be short of breath felt like their lungs aged decades too soon.
Chronic skin contact brings its own set of troubles. Skin gets sensitized, so the smallest touch later might spark a reaction much worse than before. Medical community consensus says respiratory sensitization is the bigger threat. Once triggered, the body’s immune system never quite forgets, and small exposures later bring on big breathing problems.
What Can Be Done About It?
Solutions actually start before someone picks up a chemical drum. Every workplace dealing with Cyclohexyl isocyanate needs sturdy rules and safety gear. I’ve watched teams spend extra time doubling up on gloves, swapping out torn suits, and running good air filtration—all steps that keep these vapors out of the lungs. Well-maintained extraction fans cut down on airborne chemicals. There’s a strong case for monitoring: regular lung function tests help catch changes before symptoms spiral out of control. When it comes to spills, quick action with proper equipment—eyewash stations, spill containment kits, emergency showers—prevents simple accidents from turning life-changing.
There’s no silver bullet, but clear training goes further than any manual on the shelf. A team that practices spills, changes filters, and checks each other’s personal gear avoids the silent creep of chemical risks. I’ve seen shop floors where every worker looked out for the next, no matter how busy it got, and those places kept their teams healthier.
The Choice to Care
Cyclohexyl isocyanate doesn’t belong in the hands of anyone unprepared for its hazards. Making sure one more worker goes home safely comes down to diligence, respect for risk, and treating every exposure as something that matters now and in the future. The numbers tell their story, but the real lesson comes from those who’ve faced the aftermath.
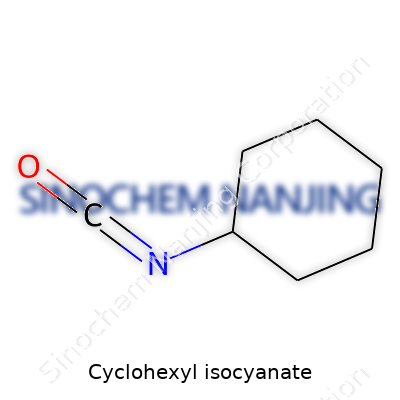
| Names | |
| Preferred IUPAC name | N-cyclohexylidenecarbamidoazane |
| Other names |
Isocyanatocyclohexane Cyclohexylcarbyl isocyanate Hexahydrobenzene isocyanate Cyclohexylisocyanide 1-Isocyanatocyclohexane |
| Pronunciation | /saɪ.kloʊˈhɛk.sɪl aɪ.soʊˈkaɪ.ə.neɪt/ |
| Identifiers | |
| CAS Number | 3173-53-3 |
| 3D model (JSmol) | `CCC1(CCCCC1)N=C=O` |
| Beilstein Reference | 1278721 |
| ChEBI | CHEBI:132962 |
| ChEMBL | CHEMBL49794 |
| ChemSpider | 12014 |
| DrugBank | DB08573 |
| ECHA InfoCard | 100.033.685 |
| EC Number | 211-534-7 |
| Gmelin Reference | 8189 |
| KEGG | C06581 |
| MeSH | D003480 |
| PubChem CID | 8754 |
| RTECS number | GV2875000 |
| UNII | MK6U60K7UA |
| UN number | UN2282 |
| CompTox Dashboard (EPA) | DTXSID5054639 |
| Properties | |
| Chemical formula | C7H11NO |
| Molar mass | 155.22 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Pungent odor |
| Density | 0.94 g/mL at 25 °C (lit.) |
| Solubility in water | Decomposes |
| log P | 2.8 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 14.5 |
| Basicity (pKb) | 13.16 |
| Magnetic susceptibility (χ) | -5.36×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.446 |
| Viscosity | 2.26 mPa·s (20 °C) |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -116.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3898.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H314, H317, H330, H334, H335 |
| Precautionary statements | P210, P261, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-3-1-W |
| Flash point | 58 °C |
| Autoignition temperature | 420 °C |
| Explosive limits | Explosive limits: 1.1% - 10.4% |
| Lethal dose or concentration | LD50 oral rat 662 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 316 mg/kg |
| NIOSH | MA8050000 |
| PEL (Permissible) | PEL: 0.005 ppm |
| REL (Recommended) | REL (Recommended): 0.005 ppm |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Isocyanic acid Cyclohexylamine Phenyl isocyanate |

