Cyclohexyl Formate: More Than Just a Chemical Name
Tracing the Path: Historical Development
Looking back at the chemical industry, it's easy to spot the big names like benzene or acetone, but tucked away in laboratories and factories, smaller players like cyclohexyl formate often shape progress behind the scenes. Its story stretches back to a time when chemists turned their attention to esters, not just for their scents but also for their practical uses in synthesis. Researchers in the twentieth century started exploring how combining cyclohexanol with formic acid gave birth to this molecule, opening new doors in organic chemistry. Over the decades, improvements in distillation methods and catalyst design helped shift production from limited research settings to broader commercial markets. Cyclohexyl formate didn't make the front pages, but chemists who work with flavors, solvents, and specialty intermediates came to count on its unique properties—quietly forging a legacy built on reliability and performance.
Product Overview and Chemical Character
Most folks will never hear about cyclohexyl formate unless they dig into the world of esters. This compound carries the CAS number 2865-23-6, a signpost for chemists but meaningless to most. Its molecular formula, C7H12O2, tells a story of seven carbon atoms linked in a ring, paired up with a formate group. That combination hands cyclohexyl formate two hats to wear—one as a mild, sweet-smelling liquid used in fragrance composition, and another as a building block for chemical synthesis. Markets don’t make noise about it, but anyone who’s dabbled in blending perfumes or designing new organic reactions has probably seen it in a reagent bottle or on a data sheet before.
Physical and Chemical Properties
If you ever lift a vial of cyclohexyl formate, you notice a liquid that flows easily, looks clear, and gives off a scent you’d find familiar from faint floral or apple-blossom-like notes. Its boiling point sits in the range of 170-175°C, making it stable enough for most industrial applications yet simple to distill without advanced setups. Insoluble in water but mixes readily with organic solvents, this ester often finds a place on the go-to list for chemists seeking to modify the character of their reactions or final products. Its physical stability and moderate reactivity serve up predictability. Nobody wants unknowns in a factory or laboratory, and this molecule rarely throws surprises, either when blending it into a formula or heating it under mild conditions.
Technical Specifications & Labeling in Practice
Handling cyclohexyl formate doesn't call for elaborate paperwork, but keeping the basics right makes a difference. Labels usually mark its formula and hazard symbols, and companies use industry-standard concentrations to avoid slips. Analytical chemists use techniques like gas chromatography to check its purity, making sure off-odors or by-products don’t spoil the intended application. Regulations mean that packaging guarantees tamper-evidence and chemical compatibility, so there's little risk of spoilage during storage. In practice, it's hard to spot an error when companies stick to routine quality control and proper labeling—good habits that anybody in the business should already have in place.
Preparation Method: From Bench to Batch
Producing cyclohexyl formate hinges on a simple esterification between cyclohexanol and formic acid, almost like preparing a classic recipe. The trick comes from balancing reactant ratios, maintaining gentle temperatures, and often removing water as the reaction proceeds, which shifts the equilibrium toward more product. Some labs add acid catalysts like sulfuric acid to drive the reaction, but others experiment with alternative catalysts to boost yield or reduce waste. After the main step, purification by distillation ensures that the end product meets technical requirements. For larger batches, chemical engineers optimize these basics to scale up efficiently, saving time and energy on every kilogram produced. Staying sharp on small process tweaks often spells the difference between a clean product and one bogged down with leftovers or side-reactions.
Chemical Reactions & Modifications
Cyclohexyl formate doesn't grab headlines for reactivity, but it has a toolkit that niche chemists appreciate. Under acidic or basic conditions, it hydrolyzes back to cyclohexanol and formic acid, making it handy as a temporary protecting group or an intermediate in multi-step synthesis. Researchers sometimes exploit its ester bond to blend or block reactions while constructing more complex molecules. Given its relative stability, it travels safely through a range of chemical operations—including reduction, oxidation, and transesterification—before revealing its unique value in fragrance chemistry or specialty synthesis. There’s a reason it keeps popping up in advanced organic textbooks; versatility matters more than flashiness in most industrial labs.
Synonyms & Product Names
Like many specialty chemicals, cyclohexyl formate masquerades under a variety of names. Among chemists, names like formic acid cyclohexyl ester or simply cyclohexyl methanoate do the rounds. In the perfume industry, it might even go by trade names crafted to boost marketing appeal. These alternate names can trip up less experienced buyers, so resting on a firm understanding of chemical structure prevents mistakes in sourcing. Seasoned suppliers and technicians know the value of cross-checking these synonyms, especially in international trade or when tracking down safety data sheets.
Safety & Operational Standards: Hard Lessons from the Field
People handling chemicals long enough always have stories about missed steps—a splash here, a broken container there—that underline how important proper safety really is. Cyclohexyl formate doesn’t rank as a top hazard, but it can irritate eyes and skin, and inhaling its vapors isn’t pleasant. Ventilated workspaces, gloves, and eye protection keep those risks low. Storage guidelines don’t come out of thin air; keeping it cool and away from open flames prevents small accidents that can spiral into big costs or legal trouble. Industrial history proves that following proper disposal, spill management, and emergency procedures stops small oversights from turning into emergencies. It’s too easy to let routine breed carelessness, so keeping safety fresh in everyone’s mind pays off for teams and communities alike.
Application Area: Real-World Uses
Chemists and engineers have found cyclohexyl formate fits best in a few focused corners. Most notably, the fragrance and flavor industry turns to it for subtle apple-like or floral notes in perfumes and specialty food products. Sometimes it gets blended into solvents or used as an intermediate in pharmaceutical and agrochemical synthesis, where its mildness and stability offer predictable results. While it rarely plays a starring role in major consumer products, it’s often working quietly behind the scenes, giving other chemicals or formulas the right smell, performance, or structure they need. Its chemical backbone lets it blend into existing manufacturing processes without forcing companies to reinvent workflow or invest in high-dollar upgrades.
Research & Development Pushing Boundaries
Much of the research around cyclohexyl formate circles back to making its production cleaner, cheaper, or more sustainable. Scientists constantly experiment with greener catalysts, better yields, or solvent-free conditions. These small tweaks compound over time—the sort of progress that adds up both financially and for environmental benchmarks. There are also ongoing efforts to harness its scent or reactivity in novel formulations, aiming to meet consumer demand for natural-smelling, safe, and biodegradable fragrance ingredients. Research teams look beyond today’s markets, hunting for new ways to tweak the molecule or use it as a platform for smart design in next-gen pharmaceuticals and specialty materials. It’s easy to overlook incremental advances, but in the long run, steady R&D helps companies and customers both.
Toxicity Research: Looking Past Common Perceptions
No chemist ever stops short at assuming a material’s safe—especially with esters, where minor tweaks can swing toxicity wildly. Cyclohexyl formate lands at a low hazard level, but animal studies still suggest routes of exposure and the importance of ventilation. Chronic exposure hasn’t shown dramatic risks so far, yet regulatory agencies keep a close watch and keep safety data up to date. Ongoing toxicity monitoring gives manufacturers and workers the confidence to handle it with the right routines. Companies who invest in toxicity research not only protect their people, they futureproof their products against tightening regulations or shifting public expectations. Sometimes it takes years for surprises to show up, making up-to-date research essential for companies playing the long game.
Future Prospects: Charting the Road Ahead
Looking forward, cyclohexyl formate stands poised to hold onto its quiet importance, especially as industries shift to cleaner, safer, and more sustainable chemistries. As fragrance and flavor houses compete to build safer, more appealing products, molecules like this one win new attention for their balance of performance and gentle hazard profile. Continued focus on green chemistry and efficient processing also makes cyclohexyl formate an appealing target for researchers hoping to squeeze more value out of every reaction while cutting environmental impact. Regulatory shifts, consumer expectations, and industry collaborations will shape exactly how and where it finds its place, but its legacy as an unassuming but essential player seems secure. For anyone tracing the threads of chemistry’s evolution, keeping an eye on practical esters like cyclohexyl formate brings a deeper appreciation of the small building blocks supporting whole branches of innovation.
What is Cyclohexyl Formate used for?
Everyday Chemistry in Fragrance and Flavors
Cyclohexyl formate never sparkles on a store shelf, yet it shapes experiences with fragrances and flavors. You’ll find its trace in the perfume industry, where perfumers turn to it for a clean, fruity, apple-laced aroma. As someone who’s spent hours testing fragrances, I find notes from cyclohexyl formate especially clear in fresh-scented sprays and some colognes designed for summer days. It bridges green and fruity tones without overpowering other ingredients, and that’s tough to pull off. In addition, products like air fresheners, scented candles, and even some soaps leverage its pleasant character.
Behind the Scenes in Food and Beverage Production
Walk into a candy workshop, and you might catch hints of what cyclohexyl formate can do in food applications. It works as a flavoring agent, lending subtle apple or pear touches to confections and drinks. Regulatory groups such as the FDA and the Flavor and Extract Manufacturers Association (FEMA) keep a close eye on food additives. Cyclohexyl formate has passed safety evaluations for use in trace amounts, which is all you need for effect. Lab experience taught me that compound purity matters — a stray note or excess can ruin a whole batch, so professionals measure carefully. Blenders in beverage companies seek this compound for light, refreshing flavors in juice mixes or candy-like flavors in sodas.
Industrial Solvent Applications
Beyond tastes and scents, cyclohexyl formate gets tapped as a solvent in coatings and paints. Its chemical structure dissolves certain polymers and resins, which helps paints dry evenly and gives coatings a glossy look. I saw cyclohexyl formate in use at a small paint plant, where techs praised it for balancing flow and evaporation rates without releasing strong, harsh odors common to other solvents. This means a safer environment for workers and a cleaner result for finished surfaces.
Component in Synthesis
Cyclohexyl formate also acts as an intermediate in chemical synthesis. Manufacturers use it to help build more complex molecules, including pharmaceuticals and specialty materials. I worked with synthetic teams who valued reliable sources of intermediates, since batch inconsistencies or contaminations can create serious setbacks. By producing cyclohexyl formate with high purity, companies support not just their own output but also quality in downstream products like medicines and plastics.
Considerations for Safety and Environment
Handling chemical ingredients takes caution. Cyclohexyl formate is considered low-toxicity, though it can cause mild skin and eye irritation in concentrated form. Manufacturers train staff to use gloves and eye protection, keep good ventilation, and store compounds away from heat or spark sources. Regulations in the US and Europe require labeling, tracking, and efficient disposal. Responsible companies establish spill response plans and review supplier certifications, keeping both workers and the environment in mind.
What Could Improve?
While cyclohexyl formate has valuable uses, industries face challenges in sourcing and sustainability. Overreliance on petroleum-derived chemicals raises concerns. Researchers look for ways to synthesize flavor ingredients like cyclohexyl formate from renewable feedstocks or by greener methods, often in partnership with biotech firms. Supporting innovation and transparency in the supply chain can give buyers greater confidence in how their fragrances, foods, or coatings are produced. This helps fulfill growing consumer demand for both safety and environmental responsibility.
What is the chemical formula of Cyclohexyl Formate?
Getting to Know Cyclohexyl Formate
Many households and hobbyists keep an eye on the safe use of chemicals, both at home and in workplaces. Cyclohexyl formate turns up in more spots than most people realize. This compound carries the formula C7H12O2, showing it is made up of carbon, hydrogen, and oxygen. Cyclohexanol and formic acid react to create it, sewing together the cyclohexyl group and the formate group. Over the years, I’ve come across its mild, fruity aroma in some perfumes—yes, people spray these molecules without pausing to think about what makes that “fresh” note possible.
Understanding Its Importance
Building blocks like cyclohexyl formate don’t stay confined to one corner of science. They show up across agriculture, perfume manufacturing, and food flavoring. The world relies on formulas like C7H12O2 not just because chemists say so, but because these molecules carry distinct characteristics: gentle volatility, appealing scents, and stable chemistry. Safety comes up for discussion since the difference between a sweet-smelling compound and a hazardous fume gets measured in small amounts. Organizations like the Food and Drug Administration and international fragrance associations set guidance on levels allowed in consumer goods.
Real-World Experience
Students in undergraduate labs usually remember their first foray into esterification—the process behind cyclohexyl formate’s creation. I remember spending afternoons swirling flasks, watching acids and alcohols transform with the right push from heat and a drop of catalyst. Beyond textbooks, I noticed how these sorts of esters don’t just end up in beakers. The sanding of wood sometimes brings out a similar fruity scent, a reminder that nature sets the bar for aromas, and chemists do their best to bottle it.
Safety and Environmental Considerations
Chemical use today demands strict attention to safety. Cyclohexyl formate, with its mild scent, could trick people into underestimating it. Direct exposure irritates skin and eyes. In the workplace, proper gloves and goggles remove guesswork. At home, using flavorings or perfumes that contain it calls for storing them away from children and pets. Disposal takes effort—pouring leftovers down a drain poses environmental risks, including contamination of waterways and impact on aquatic organisms.
Organizations have started tightening rules about chemical disposal. Unlike in the past, I now drop off chemicals like cyclohexyl formate at dedicated facilities. Cities issue guidance, and manufacturers list recommended disposal practices, reflecting the realization that each small bottle shapes the bigger environmental picture.
Supporting Responsible Chemistry
Science classrooms and businesses benefit from open talk about responsible chemical handling. Simple labels on products with their chemical formula, C7H12O2, encourage users to investigate what’s inside. Education can demystify chemicals, giving people the confidence to read safety sheets and store materials properly. Community programs, from school science nights to local hazardous waste collection events, give everyone a hand in keeping neighborhoods safe while still enjoying the benefits that chemistry brings to daily life.
What are the storage requirements for Cyclohexyl Formate?
Understanding Cyclohexyl Formate and Its Hazards
Cyclohexyl formate is a clear, oily liquid with a fruity smell. It sees use in fragrances and flavorings. Unlike common household solvents, it is slightly flammable, produces irritating vapors, and breaks down under some conditions into substances that are not safe to inhale. Unfortunately, many users treat it casually, ignoring the risks that come with improper handling or sloppy storage. Based on real-world lab experience and supported by chemical safety agencies, keeping this compound in a safe state involves both knowledge and thoughtful action.
Choosing the Right Storage Area
Most users overlook the importance of a cool, dry, and well-ventilated area for Cyclohexyl formate. Heat ramps up decomposition and vapor formation, which, in turn, raises fire and exposure risks. From personal experience, facilities that stored this in hot, cramped spaces often faced issues with pressure buildup inside containers. Experts agree—temperatures should stay below 30°C (86°F). Fumes should never build up in the air. Good airflow reduces the chance of vapors collecting, especially if there’s a small leak or during dispensing. Flammable solvents belong far from ignition sources like heaters, open flames, or sparking electrical equipment.
Container Selection and Labeling
Use containers crafted from metal or tough plastics that do not react with formate esters. Thin plastics warp and leak. Old or reused bottles sometimes carry residues that react with Cyclohexyl formate, creating dangerous compounds. Mark every container clearly with its name and hazard warnings. Emergency crews or coworkers can’t guess what’s inside if time is short—good labeling has saved lives during spills.
Protection from Moisture and Sunlight
Direct sunlight speeds up chemical reactions and weakens lots of storage plastics. I’ve seen clear plastic jugs turn brittle and fail in sunlit storage rooms. Moisture control matters, too. Water doesn’t mix with Cyclohexyl formate, but droplets inside a poorly sealed bottle lead to slow breakdown of the chemical, sometimes producing heat and pressure. Store far from sinks, wet floors, and leaky pipes. A desiccant nearby helps in humid climates.
Keeping Storage Areas Safe
Never stack containers higher than three layers, since falls and breaks produce puddles that evaporate into hazardous fumes. Plan for spills: keep absorbent pads and neutralizing materials close by. OSHA reports show that rapid response saves not only cleanup bills but also employee health. In my own work, a secondary tray or spill pallet added extra peace of mind under larger drums, especially when restocking.
Regular Checks and Inventory Management
Routine inspection catches swollen, damaged, or corroded containers before they become an emergency. It’s easy to get careless over time—as I’ve seen in more than one lab—but checking monthly, including a sniff test (with caution), spots problems quickly. Rotate your stock: oldest goes out first. Never let expired or decaying product sit around. Disposal gets more expensive and riskier the longer it waits.
Final Thoughts on Safety Culture
No product or production schedule matters more than health. Safety agencies like the CDC and European Chemicals Agency offer simple bullet-point guides that work in any workshop, and I’ve found them reliable every time. Employees follow the example set by supervisors, and it’s up to management to make safe storage part of daily work, not just a binder on the shelf.
Is Cyclohexyl Formate hazardous or toxic?
Looking at Cyclohexyl Formate in the Real World
Walking through an industrial lab, you come across hundreds of chemicals with names that sound like tongue twisters. Cyclohexyl formate is one of those chemicals. It shows up in perfumes, flavorings, and sometimes in industrial fluids. The big question crops up: How risky is it for people and the environment?
Understanding Its Basic Risks
Cyclohexyl formate carries a faint fruity smell. Labs often use it for fragrances and flavor additives. Most people outside the lab won’t come across it in high amounts, but accidents do happen. Skin contact usually brings mild irritation; it leaves skin a bit red, sometimes itchy. Breathing in a lot of its vapors can bring on headaches, dizziness, or even nausea. The U.S. National Library of Medicine lists it as an irritant, not a severe toxin. Mixing it up or spilling it on your desk doesn’t give you an immediate threat like strong acids or cyanide, but personal experience in the lab reminds me that not every hazard comes from immediate reactions—some chemicals take time to show their effects.
What Science Says About Its Toxicity
Toxicology reports show it has low acute toxicity by ingestion or skin contact. If a person swallows a little, the body usually handles it without major damage. That said, drinking a bottle of it can spark stomach pain, nausea, and vomiting, much like drinking any household solvent. Getting it in your eyes stings, and it doesn’t mix well with open wounds.
Animal studies report a pretty high threshold for lethal effects—the LD50 in rats sits above 2000 mg/kg, so you’d need a huge quantity to reach dangerous exposure. This doesn’t mean people should handle it without care, but it’s not as insidious as heavy metals or some pesticides.
Occupational Use and Real Hazard
Workers sometimes underestimate how much skin absorption matters. Handling cyclohexyl formate daily without gloves can dry out skin. Ventilation matters, too, since breathing in high vapor concentrations brings on discomfort. Safety sheets recommend goggles and gloves, along with common-sense ventilation.
You don’t find widespread reports of cyclohexyl formate poisoning. In lots of years messing around with dozens of solvents, I never found a case where cyclohexyl formate sent someone to the hospital, or left them with long-term damage. The real risk comes from poor chemical handling: spilled containers, closed-up workplaces, or eating lunch without washing hands.
Environmental Impact and Safe Use
Cyclohexyl formate doesn’t last long out in the open. It breaks down fairly fast, and most environmental agencies haven’t flagged it as a persistent pollutant. Small spills don’t tend to travel far or poison local water sources, but dumping barrels of it straight into a creek will hurt fish and insects. Every chemical presents risk at high enough levels, so safe storage and disposal keep it out of places where it could build up.
Real safety means sticking with gloves, proper storage, using fume hoods or vented rooms, and giving up the habit of eating or touching your face in the lab. Teaching these kinds of habits stops minor risks from turning into major incidents.
Moving Forward with Awareness and Facts
Cyclohexyl formate lands in the “use with care” category, not the “panic if exposed” one. Respect common sense guidelines, limit contact, and stay aware of where it ends up. Safety training and good habits carry more weight here than hazard labels. For most people, its risks stay manageable. Our workplaces get safer not just from chemical bans, but from paying attention day by day and owning the responsibility for what happens on our watch.
What is the CAS number for Cyclohexyl Formate?
Understanding Cyclohexyl Formate
Cyclohexyl formate isn’t a chemical most people talk about around the dinner table, but in labs and factories, those three words spark a very specific thought. Chemists rely on precise language and numbers because a single molecule out of place changes everything. The CAS number for cyclohexyl formate is 3152-61-2, and behind that string of digits sits a level of reliability that the global industry trusts every day.
The Story Behind CAS Numbers
Every compound ever registered has its own Chemical Abstracts Service number. This registry keeps mistakes low and traceability high. I remember talking with an old friend who manages a paint factory. She once told me a shipment of cyclohexyl formate with a typo in the documentation nearly stalled production for a week. Only by cross-referencing the CAS number did the team sort things out. It protected jobs, saved money, and kept their paint formula on track.
Getting Beyond Names: The Importance of the Numbers
A lot of chemicals bear lookalike names, especially in flavors, fragrances, and solvents. Cyclohexyl formate, used in everything from perfumes to cleaning products, needs careful handling. CAS 3152-61-2 closes the door on any mixups. Product recalls cause headaches for everyone involved—retailers, manufacturers, and consumers alike. Clear identification speeds up auditing, simplifies import checks, and reduces the risk of hazardous errors.
Sourcing and Trust
Global supply chains have grown complex. Shipments pass through many hands and borders. CAS numbers provide a shorthand trusted worldwide. This is more than bureaucracy; it’s the frontline defense against counterfeit goods and mislabeling. Counterfeiting incidents, especially in the fragrance and food sectors, often trace back to incorrect or omitted CAS numbers. Regulatory agencies such as the FDA and ECHA depend on these numbers to monitor substances and keep products safe.
Protecting Health and the Environment
Accurate chemical identification doesn't just protect business interests. It shields workers, communities, and the environment from accidental exposures. If cyclohexyl formate gets mislabeled and ends up somewhere it doesn’t belong—maybe even in a non-compatible process or shipped to the wrong facility—the outcome can be contamination or unsafe reactions. Familiarity with the CAS 3152-61-2 number cuts risk. My uncle, who works as an industrial hygienist, once helped halt a processing line due to a mismatched CAS number on a delivery. His quick action meant nobody got hurt.
Building Good Practices Into Every Step
Companies that invest in staff training about how to read and verify CAS numbers see fewer supply chain disruptions. There’s real value in regular audits and inventory checks using these identifiers. Digital databases have made verification faster, but the principles remain the same as they did decades ago—know what you’re getting, up front and without confusion.
Clear Answers Make Solid Decisions
Looking up cyclohexyl formate and confirming its CAS number isn’t busywork. It’s a simple, effective way to keep production, safety, and compliance in balance. The journey from raw material to finished product depends on a shared language, and these numbers bring clarity to a world where little mistakes can turn costly or dangerous. That’s why CAS 3152-61-2 stays at the center of every label, manifest, and inspection form where cyclohexyl formate plays a part.
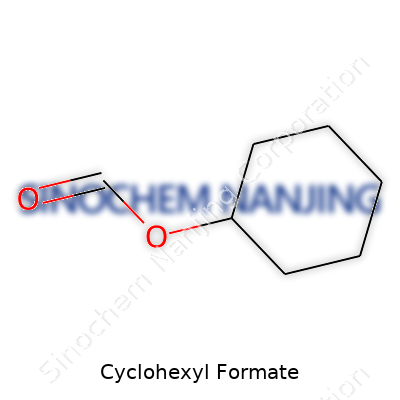
| Names | |
| Preferred IUPAC name | Cyclohexyl methanoate |
| Other names |
Formic acid cyclohexyl ester Cyclohexyl methanoate |
| Pronunciation | /saɪ.kloʊˈhɛk.sɪl ˈfɔːr.meɪt/ |
| Identifiers | |
| CAS Number | 28647-72-3 |
| Beilstein Reference | 1462378 |
| ChEBI | CHEBI:82227 |
| ChEMBL | CHEMBL444410 |
| ChemSpider | 14218 |
| DrugBank | DB14163 |
| ECHA InfoCard | 04de90a3-97eb-4980-9e64-0ed1b935265d |
| EC Number | 203-688-2 |
| Gmelin Reference | 63059 |
| KEGG | C10442 |
| MeSH | Cyclohexanecarboxylic Acids |
| PubChem CID | 96349 |
| RTECS number | GU5950000 |
| UNII | 0H110P1RE8 |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID7077556 |
| Properties | |
| Chemical formula | C7H12O2 |
| Molar mass | 142.20 g/mol |
| Appearance | Colorless liquid |
| Odor | pleasant, fruity |
| Density | 0.976 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.8 |
| Vapor pressure | 0.38 mmHg (at 25 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -67 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4470 |
| Viscosity | 3.2 mPa·s (25 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 298.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -380.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3916.7 kJ/mol |
| Pharmacology | |
| ATC code | Cyclohexyl Formate does not have an ATC code. |
| Hazards | |
| GHS labelling | GHS07, Warning |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | > 74°C (Closed cup) |
| Autoignition temperature | 220 °C |
| Lethal dose or concentration | LD50 (oral, rat): 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 3000 mg/kg |
| NIOSH | BCH7365000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 0.5 – 5 |
| Related compounds | |
| Related compounds |
Formic acid Methyl formate Ethyl formate Propyl formate Isopropyl formate Butyl formate Cyclohexanol |

