Cyclobutyl Chloroformate: Chemistry Shaping Industry, Safety, and Progress
Historical Development
Looking back, the road that led chemists to synthesize cyclobutyl chloroformate tells an interesting story about how organic chemistry shifted in the late twentieth century. The chemical world once revolved around more familiar groups like methyl, ethyl, and phenyl, but as pharmaceutical companies reached for more targeted drugs and materials science demanded unique polymers, less obvious building blocks like cyclobutyl rings started turning heads. Cyclobutyl chloroformate emerged as organic chemists adapted old phosgene reactions for new ring systems, a sign of the cat-and-mouse pace between demand for specialty fine chemicals and the skills of academic and industrial labs. The discovery and subsequent adoption of this compound reflect a persistent drive to expand molecular toolkits. Each new functional group, each new reactive species, fits a gap in synthesis that previously forced chemists to take longer or less selective routes. By the early 1970s, cyclobutyl chloroformate had made its way from specialized research contexts to the shelves of chemical suppliers, helping connect cyclobutyl building blocks with broader worldwide innovation.
Product Overview
Ask anyone in chemical R&D circles, cyclobutyl chloroformate stands out for what it brings to the table—a reactive chloroformate moiety joined with a cyclobutyl group. This combination means reactivity with amines and alcohols, carving out a spot in the synthesis of carbamates and urethanes, as well as cyclobutyl derivatives that crop up across medicinal chemistry. As someone who has moved precursors and lab reagents, the practical aspects also matter. Cyclobutyl chloroformate, often clear or slightly yellow liquid, carries a pungent odor and demands respect from those who handle it. The molecule’s structure gives just enough strain from the four-membered ring to alter reactivity patterns when compared with open-chain analogs, an ongoing interest for researchers who track what makes cyclobutyl chemistry distinct.
Physical & Chemical Properties
Those white-knuckling a pipette know first-hand that properties aren’t just trivia—they shape every step of handling and use. Cyclobutyl chloroformate’s liquid state at room temperature, volatility, and solubility in organic solvents like dichloromethane or toluene push users to work in well-ventilated hoods. Like most acyl chlorides, this compound reacts quickly with water, releasing corrosive hydrogen chloride gas. Simple numbers like boiling point and density may sound dry, but for anyone writing up a synthetic scheme, they make the difference between a clean reaction and an unintended hazard. The ring tension in cyclobutyl contributes to higher energy, sometimes lending reactivity that more stable rings don’t match, a fact that echoes through yield tables and reaction screens on many a chemist’s bench.
Technical Specifications & Labeling
Quality control ties directly to safe and productive use. Purity counts. Analytical details—like GC or NMR profiles—come up in every discussion with suppliers, as trace impurities in cyclobutyl chloroformate can poison sensitive steps in drug synthesis and specialty materials. Regulatory rules force clear labeling: the chemical name, hazard codes for corrosivity and toxicity, handling precautions, and storage conditions. Those symbols and signal words, dry as they may seem at first, are the first line of defense in day-to-day operations. In my own experience, good stewardship starts with good information, visible on every bottle, matched by full documentation.
Preparation Method
Few synthetic moves are as central—and as fraught with risk—as turning cyclobutyl alcohol into its chloroformate. Most routes bring phosgene or similar chlorinating agents into play, and phosgene’s reputation as a deadly gas means process safety hovers over every scale-up. Chemists learned to use substitutes like triphosgene, which trade off convenience and risk, but the fundamentals stay the same: neat stoichiometry, low temperatures, and scrupulous ventilation prevent uncontrolled reactions and minimize unwanted side-products. Tinkering with solvents, tweaking rates of addition, and constant monitoring of temperature let operators maximize yield without trading away safety. From my own time working in chemical process development, every batch distills down to a series of risk vs. reward decisions. And in making cyclobutyl chloroformate, vigilance always outranks raw throughput.
Chemical Reactions & Modifications
Organic chemists value versatility, and cyclobutyl chloroformate fits the bill—reacting cleanly with alcohols and amines, generating esters and carbamates that push the boundaries of medicinal screening libraries and polymer building blocks. The chloroformate functional group plays gatekeeper, activating the cyclobutyl ring for incorporation into more complex molecules. In my lab days, we’d run these reactions under anhydrous conditions, using slow addition and external cooling to keep side-products at bay. The cyclobutyl piece isn’t just window dressing; it nudges selectivity and can dial in physical features—like molecular rigidity—that more common alkyl groups can’t provide. Chemists studying new reaction pathways pay close attention to how cyclobutyl patterns influence substitution and addition, generating data that feed back into process improvements and mechanistic models.
Synonyms & Product Names
A stroll through chemical catalogs reveals cyclobutyl chloroformate mentioned under names like cyclobutyl carbonochloridate, cyclobutanecarbonyl chloride, and carbonochloridic acid cyclobutyl ester. These alternate names carry over from IUPAC and CAS conventions, but practical lab folk stick to trade names or shorthand. Clarity counts, especially when ordering or managing inventory, so seasoned researchers double-check CAS numbers to avoid mix-ups that can torpedo entire projects. It’s a reminder that, despite a field awash in abbreviations and cataloging systems, clear communication still beats any automated purchase order.
Safety & Operational Standards
The word on the street in every chemical lab I’ve worked is simple: respect cyclobutyl chloroformate. That pungency in the air says more than any label ever could—it reminds everyone that personal protective gear, gloves, and face shields aren’t optional. Rigorous training on proper waste handling and spill response keeps minor slip-ups from turning into medical emergencies. Regulations require storage in cool, dry areas, segregated from bases and incompatible reagents, while process safety managers push for regular refreshers on hazardous materials handling. My time on safety audit teams showed me the hard truth: no shortcut or missed glove changes ever pays off when dealing with reactive liquids like these. Best practices have evolved in pace with stricter environmental and health safeguards, and today’s standards draw directly on accumulated lessons from decades of near-misses and close calls in the chemical trades.
Application Area
Cyclobutyl chloroformate’s main job shows up in chemical synthesis, especially for those pursuing new drugs, crop protection agents, or specialty polymers. The unique quirks of the cyclobutyl group have made it a favored choice for structural modification, aiming to change molecular shape or add metabolic stability. In practice, this means medicinal chemists reach for the compound when building prodrugs, enzyme substrates, or novel scaffolds for screening campaigns. In materials research, polymer chemists use it to pop in rigid cyclobutyl linkages where flexibility isn’t wanted—tweaking properties or unlocking new processing conditions. In my conversations with colleagues, the consensus holds: cyclobutyl chloroformate often fills specialized roles where more common chloroformates fall short, and for those who solve hard problems, that edge makes the difference.
Research & Development
Research pushes forward through tools like cyclobutyl chloroformate, whose role in expanding the available chemical space can’t be overstated. Teams developing new synthetic methods still look to ring systems as ways to bypass patent obstacles or tune receptor binding in pharmaceutical candidates. The push to minimize hazardous by-products has led to greener synthesis pathways, using milder chlorination conditions or alternative activating agents. At conferences and in journal clubs, the chatter always circles back to efficiency, selectivity, and creative new uses. Graduate students, postdocs, and industrial chemists alike treat every new application as a test of their ability to extract maximum utility from specialty reagents like this one. For the next round of patents and market advances, the incremental improvements in process chemistry or downstream modifications continue to build on these sometimes-overlooked intermediates, sparking collaborations and spurring the hunt for safer, more cost-effective methods.
Toxicity Research
You can’t talk about chloroformates without addressing safety. Cyclobutyl chloroformate’s hazard profile isn’t just a line in the MSDS—it affects job safety, public health, and even the process choices researchers make. Like other acyl chlorides, it can damage skin and eyes, cause respiratory irritation, and trigger dangerous reactions with moisture. Toxicological studies have focused on both the parent compound and its breakdown products, as chemical exposures in both lab and industrial settings have prompted stricter workplace monitoring. The call for more and deeper research grows louder as regulatory bodies update permissible exposure limits and seek data on long-term health impacts. In the end, proper design of engineering controls—fume hoods, scrubbers—and mandatory PPE reduce risk, but vigilance remains the norm. Facilities that handle reactive organochlorine species know full well the risks, and apply a multi-layered approach blending compliance training, incident drills, and real-world feedback from exposure monitoring.
Future Prospects
Cyclobutyl chloroformate may look like just another specialty reagent, but its trajectory signals broader changes in the chemical industries. The demand for more structurally diverse small molecules keeps climbing, especially as AI-driven drug design and advanced materials call for unique, hard-to-access motifs. Researchers also know that pressures for cleaner, more sustainable processes will continue to influence how this and similar compounds get made and used. You see more partnerships forming between academic groups, contract manufacturing organizations, and green chemistry consortia aiming to refine synthesis methods, recycle hazardous by-products, and cut environmental footprint. Regulatory momentum towards tighter controls on hazardous chemicals will drive both improvements in safety management and the gradual replacement of riskier process options. In my view, cyclobutyl chloroformate won’t just hang on as a legacy chemical; it will adapt along with shifting priorities in synthesis and manufacturing, shaped by advances in science and guided by a sharper focus on safe, responsible stewardship.
What is Cyclobutyl Chloroformate used for?
Inside the World of Chemical Synthesis
Labs chase innovation at a relentless pace. Each new compound brings a mix of curiosity and cautious respect, especially with building blocks like cyclobutyl chloroformate. This molecule doesn’t make headlines outside expert circles, but it plays a quiet role in the design and manufacture of new medicines and specialty materials. Small changes in a chemical structure often create big breakthroughs. Cyclobutyl chloroformate takes up space in this conversation as a solid choice for modifying other molecules, helping researchers find new paths to chemical bonds that turn inspiration into outcomes.
Building Block for Pharmaceutical Research
Think about the journey from test tube to tablet. Scientists constantly hunt for structures that bind, block, or support processes in the body. Cyclobutyl chloroformate shows up repeatedly as a tool for crafting these structures. It gets used in the development of carbamate and urethane groups, staples in the medicinal chemist’s toolkit. These groups often help tune a drug’s effectiveness or safety. This compound reacts cleanly, helping chemists control exactly what sticks where on a molecular scaffold. Good chemistry reduces byproducts, cuts waste, and saves time, which translates into lower risk and better outcomes down the line.
Beyond Medicine: Fine Chemicals and Materials
Cyclobutyl chloroformate doesn’t stop at pharmaceuticals. Makers of specialty chemicals turn to it for crafting unique agrochemicals, dyes, and advanced polymers. Demand for more specific, sustainable, and stable materials keeps growing. An efficient chemical like this one can lower the number of steps involved in synthesis, which often drives down both cost and energy use. Fewer steps mean less chance for something to go wrong—a constant struggle in chemical scale-up.
The Safety Challenge
Anyone who’s worked with chloroformates knows they come with hazards. Cyclobutyl chloroformate can release toxic gases upon contact with water or alcohols, and direct exposure to skin or eyes spells trouble. Years spent handling risky substances teach the value of layering protections: chemical fume hoods, gloves, and training by experienced hands. Accidents happen faster than you think, often when someone skips a step or overlooks a warning. Chemists push for safer reagents, but until safer alternatives deliver, vigilance stays front and center.
Supply Chain and Regulatory Pressures
Sourcing high-purity chemicals never gets easier. Unexpected delays turn meticulous research timelines upside down. Stricter regulations around transport and storage keep labs on their toes, especially with reactive substances. Anyone ordering cyclobutyl chloroformate has probably faced questions about intended use and storage plans. These gatekeepers help prevent misuse but can slow down research progress, especially for small labs. Consistent supply and clear documentation help, but it takes ongoing work to keep projects moving forward.
Paths Toward Safer Chemistry
People in the lab always look for ways to make science safer and greener. Some teams experiment with less hazardous alternatives, but results can fall short of what’s needed for high-value synthesis. Better engineering controls, clear protocols, and investment in training remain effective shields against known risks. Larger networks between academic groups and manufacturers help share best practices and foster more transparency. Bringing together knowledge from every corner of chemistry will set the stage for new solutions.
What is the chemical formula of Cyclobutyl Chloroformate?
Understanding Cyclobutyl Chloroformate
Chemistry class left me with a habit of double-checking formulas, and Cyclobutyl Chloroformate gave me that reason again. Its chemical formula is C5H7ClO2. That’s five carbons, seven hydrogens, one chlorine, and two oxygens. This compound finds its spot in labs, not in household under-sink cabinets. It forms the backbone for reactions that build up medicines and modify molecular surfaces, which means accuracy in naming and formula matters a lot more than folks might guess. Mixing up a formula here could set off a chain of lab headaches, ruined resources, and even safety risks.
Why the Correct Formula Matters
Anyone who’s worked through a long organic synthesis knows how a single wrong atom can mess up everything. Students in college labs who jot down an incorrect formula see it when their yields tank; so do professionals who rely on chemical supply catalogs and digital inventory software. Cyclobutyl Chloroformate, with its blend of a cyclobutyl group and a reactive chloroformate group, acts as a building block. Chemists use it for adding protective groups to amines or making carbamates. Start with the wrong formula, and your pathway crumbles.
In my early days, watching an experiment fizzle because a reagent was misidentified gave a lesson that stuck. Official safety sheets, research journals, and procurement orders tie back to these chemical identities, supporting responsible work and clear results. Without exact details, chemists risk repeating mistakes, not catching hazards, and misreporting their findings. For new students or technical staff, learning the language of chemical formulas builds credibility and keeps their teams out of harm’s way.
Supporting Facts: Real-World Impact
Around the world, organizations like the American Chemical Society and the National Institutes of Health publish reminders highlighting the hazards of mislabeling. In some documented cases, mix-ups led to costly shutdowns and safety investigations. Cyclobutyl Chloroformate’s reactivity means handling, storage, and shipment follow strict guidelines. The right formula keeps suppliers, logistics teams, and customs inspectors in sync; it keeps hazardous chemicals out of the wrong hands or from being stocked in the wrong warehouse. Small errors here can have outsized impacts in the lab and market.
Reducing Mistakes: A Call for Better Practices
To keep things moving forward, companies and institutions can invest in digital inventory tools that match chemical names with their formulas. Training for lab staff shouldn’t treat formula verification as an afterthought. Researchers benefit from peer checks, clear documentation, and ongoing reference updates. In schools and universities, tying every hands-on experiment to its correct molecular formula lays a foundation that sticks as students move into industry or academia. Errors don’t just waste time—they put people, budgets, and scientific progress at risk.
How should Cyclobutyl Chloroformate be stored?
Understanding the Risks
Cyclobutyl chloroformate lives in a class of chemicals that simply refuse to play nice if not treated right. The risk of serious problems, such as skin burns or dangerous vapors, climbs if storage gets sloppy. In real life, chemists have found themselves frantically evacuating labs after a careless seal let fumes escape. It seems so basic, yet attention to everyday details like container tightness or room ventilation often makes the difference between a safe workspace and a disaster.
Choosing the Right Container
A lot rides on the choice of container. Glass bottles with PTFE-lined caps handle cyclobutyl chloroformate without reaction or leakage. It pays off to skip old plastic, since this chemical likes to chew through weak spots. I’ve watched a seemingly sturdy plastic bottle sweat and crack after a few weeks, leaving an ugly puddle and a room full of nasty fumes. There's no cutting corners here: glass stands up to the challenge.
Safe Environment Matters
Temperature control doesn't get enough respect. Cyclobutyl chloroformate breaks down if things get too warm, especially above room temperature. Many facilities keep it in a cool, dry chemical refrigerator set far from food or flammable solvents. It’s best to leave the crisper drawer for your lunch. Explosive reactivity with water or alcohols makes it critical to keep moisture away, which means a desiccator or a dry cabinet isn’t just for show.
Labeling and Segregation
Mislabeling has led to serious mix-ups. I once saw a bottle passed off as “organic solvent,” when it actually held cyclobutyl chloroformate. That oversight landed two people in the ER. Bright, bold labels with both the full name and a hazard warning cut the confusion. In any chemical storeroom, separate the chlorinated compounds from acids, bases, and anything flammable. Shelving maps may seem like overkill, but they help prevent accidents caused by stacking incompatible bottles together.
Personal Safety in Storage
Loading and unloading containers calls for basic respect. Eye protection and gloves rated for organochlorines become non-negotiable. Nothing ruins a shift like a chemical burn from a hidden drip along the outside of a cap. Spills aren't just messy—they release corrosive and toxic gas. Emergency protocols, spill kits, and accessible eyewash stations need to be part of the routine.
Preparing for the Unexpected
Even with perfect storage practices, surprises happen. Weekly inspections keep forgotten leaks or damaged lids from becoming big problems. In my own work, teams run drills on handling a spill or accidental exposure—no one wants to learn amid chaos. Facilities that build in regular checks catch issues before anyone gets hurt.
Moving Forward With Responsibility
Chemists and lab managers must treat cyclobutyl chloroformate with the gravity it deserves. Trustworthy storage isn’t about ticking boxes for compliance; it’s about keeping colleagues alive and well, protecting investments, and guarding the environment from another careless mistake. With solid containers, cool and dry conditions, respect for labels, sharp organization, and steady training, it’s possible to keep this challenging compound in check.
Is Cyclobutyl Chloroformate hazardous or toxic?
Why Cyclobutyl Chloroformate Draws Caution
If you have spent any time in a chemistry lab, you know how quickly chemicals can swing from useful tools to safety hazards in the blink of an eye. Cyclobutyl chloroformate stands out in this category, not just because of its use in making specialty chemicals and pharmaceuticals, but also for the health risks it brings to the table. Most people outside the lab circle rarely read about it, which makes our conversation about its hazards worth having.
Just by its structure, cyclobutyl chloroformate belongs to a class of chemicals known as chloroformates. In direct terms, these compounds carry a high reactivity that helps in making medicines and agrochemicals. This reactivity also means the substance demands respect and sturdy safety culture among everyone handling it. If this compound touches your skin, trouble starts with a stinging burn. Inhaling its vapors can irritate the nose, throat, and lungs. Exposures can lead to coughing, headaches, and even dangerous respiratory issues down the line. People exposed without proper gear could face corneal burns or long-term health issues.
Sources of Danger and Real Workplace Stories
I’ve seen stories and heard first-hand accounts of why cyclobutyl chloroformate needs handling with care. One chemist described a time a reactive bottle cap failed, releasing fumes she could smell before anyone sounded an alarm. Her symptoms didn’t seem dramatic—watery eyes, a slight cough—but chemical exposures often don’t show their full hand right away. Health agencies flag accidents like these because even small exposures can sneak up on the body. OSHA details how many chloroformates, including cyclobutyl chloroformate, can corrode tissue and disrupt breathing. Most of us underestimate just how quickly a minor spill or splash can alter a life forever.
As a rule, the compound reacts with water or moisture, giving off phosgene gas—a substance historically used in warfare and acutely toxic in small doses. Modern labs should have proper ventilation, fume hoods, and staff trained to deal with emergencies. I remember my university lab running safety drills not as afterthoughts but as part of daily routine. Every bottle got labeled, every procedure written and checked twice. This approach stems not from paranoia but from a deep respect for the risks common with chemicals like cyclobutyl chloroformate.
Steps for Safer Handling
Knowledge goes a long way in keeping people safe. Reading the Material Safety Data Sheet (MSDS) isn’t a box to tick, it gives you the facts and numbers needed to stay out of danger. PPE—face shields, gloves, goggles—serves as your first line of defense. Having eyewash stations and showers close at hand can turn a life-threatening exposure into a manageable scare. It makes sense to keep only small amounts on-site and store bottles away from direct sunlight or humid areas, since temperature spikes or leaks can make a bad day worse fast.
Disposal often gets ignored, yet improper discarding can send hazardous vapors through the air or contaminate water. Years ago, I saw colleagues double-check waste containers every day, making sure the lids fit tight and nothing mixed by accident. It may sound like extra work, but stories of near-misses and lessons shared at safety meetings underline that even seasoned professionals take nothing for granted with chemicals like cyclobutyl chloroformate.
Final Thoughts and the Path Forward
The risks with cyclobutyl chloroformate can’t be glossed over. It holds value for industry but only for those who match its potential with an equal measure of awareness and preparation. Lives often depend, not on raw intelligence or technical tricks, but on habits built from respecting the hazards these compounds bring into the lab.
What precautions should be taken when handling Cyclobutyl Chloroformate?
Getting Real About Chemical Risks
Cyclobutyl chloroformate is no household item. I’ve spent a fair share of hours in different labs, and few things demand respect like acyl chlorides. You learn quick that ignoring protocols puts you or your team at real risk. One careless move could send you to the hospital. This is not just about ticking boxes. It’s about avoiding days or weeks with chemical burns, struggling through shortness of breath, or explaining to a manager how an evacuation got triggered.
Personal Gear Isn’t Optional
Anyone who’s ever had even a pinhole in their gloves during a synthesis knows the hassle of skin irritation, and with cyclobutyl chloroformate, that risk jumps a few notches. Splash goggles stop a routine transfer from ruining your vision for good. Lab coats and chemical-resistant gloves (not just any old latex, but butyl or nitrile) close the gaps that splashes target. The face shield? That’s just peace of mind every time concentrated acid and acyl chloride start hissing in the flask.
Good Ventilation Is Non-Negotiable
Why do seasoned chemists swear by well-ventilated fume hoods? Cyclobutyl chloroformate mixes "pungent" with "unstable"—if you’ve ever whiffed its vapors, you understand. Inhalation can burn your airway, cause coughing fits, or trigger symptoms you only want to read about, not feel. Keeping everything under a fume hood isn’t just comfort—it’s clear-headed hazard management. Fume hoods make accidental inhalation much less likely. You get to go home with your lungs in the same shape as you brought them in.
Never Trust Improvised Storage
I once watched a bottle of reactive chemical eat straight through a plastic lid—no one forgot that lesson. Cyclobutyl chloroformate demands tight, sealable containers of glass or high-grade compatible plastics. The storage spot needs a cool, dry shelf, marked with hazard labels so no one grabs it thinking it’s safe. Humidity or heat can make these bottles vent pressure or form gases. Proper secondary containment, like a protective bin, gives backup if a leak starts. A spill kit nearby lets you clean up accidents without turning a stinky drop into a lab shutdown.
Know Your Reactions and Incompatibilities
Mixing cyclobutyl chloroformate with water kicks up clouds of corrosive and noxious gases, especially hydrochloric acid and phosgene traces. Questionable moves—like disposing down the sink or mixing with bases and amines without thought—could fill the workspace with fumes. You need familiarity with material safety data sheets and reaction pathways before touching the stuff. Predict what might happen if something goes wrong, and plan your response.
Training Saves Nerves and Lives
Formal training in handling dangerous chemicals isn’t boring theory if you care about walking away unharmed. Teams that review spill response and first aid handle emergencies with less panic—knowing exactly what to do. Eye wash stations and safety showers should be within sprinting distance, not down the hallway. Practicing with mock scenarios helps people reach for the right solution, not just freeze or run.
Safer labs are built on experience and preparation. Stepping up personal responsibility and awareness brings a culture of respect, keeps everyone out of trouble, and ensures that hazardous work stays productive instead of dangerous. Mistakes with cyclobutyl chloroformate get costly fast, but most are preventable with practical precautions and honest respect for the risks involved.
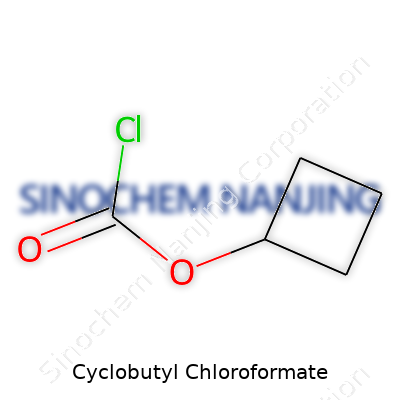
| Names | |
| Preferred IUPAC name | Cyclobutyl carbonochloridate |
| Other names |
Cyclobutanecarbonyl chloride Cyclobutyl carbonochloridate |
| Pronunciation | /ˌsaɪ.kləˌbjuː.tɪl ˈklɔː.rəˌfɔː.meɪt/ |
| Identifiers | |
| CAS Number | 2713-33-9 |
| Beilstein Reference | 1912455 |
| ChEBI | CHEBI:132214 |
| ChEMBL | CHEMBL135478 |
| ChemSpider | 573867 |
| DrugBank | DB22056 |
| ECHA InfoCard | 100.043.868 |
| EC Number | 227-586-6 |
| Gmelin Reference | Gmelin Reference: **82818** |
| KEGG | C18806 |
| MeSH | D017209 |
| PubChem CID | 70037 |
| RTECS number | GN2100000 |
| UNII | 84T36538UE |
| UN number | UN3277 |
| Properties | |
| Chemical formula | C5H7ClO2 |
| Molar mass | 120.56 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 1.222 g/mL at 25 °C |
| Solubility in water | Decomposes in water |
| log P | 1.9 |
| Vapor pressure | 0.9 mmHg (20 °C) |
| Acidity (pKa) | 1.3 |
| Basicity (pKb) | Basicity (pKb) = -1.66 |
| Magnetic susceptibility (χ) | -6.07 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.446 |
| Viscosity | 2.38 cP (20°C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 370.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -218.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -589.6 kJ/mol |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302 + H332: Harmful if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P210, P261, P280, P301+P330+P331, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 64 °F |
| Autoignition temperature | 170 °C |
| Lethal dose or concentration | LD50 oral rat 632 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 113 mg/kg |
| NIOSH | FG6300000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Cyclobutyl Chloroformate: Not established |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate Isopropyl chloroformate Isobutyl chloroformate N,N-Dimethylcarbamoyl chloride Benzyl chloroformate |

