Cyanuric Chloride: Why One Chemical Tells a Bigger Story
Historical Development Steers Industrial Growth
Cyanuric chloride didn’t just wander onto the chemical scene. Its development tracks closely with the rise of industrial chemistry in the early 20th century, especially as synthetic methods opened doors to compounds nobody found in nature. Early chemists eyed this triazine ring with excitement, seeing practical uses in dyes and pesticides during a time when industry hungered for new materials. Production really gathered steam around World War II, as nations spent big on chemical intermediates, pointing to broader shifts in how societies pushed chemistry to solve everyday problems. Watching old patents and literature, it’s clear that cyanuric chloride isn’t just a footnote. It played a role in transforming how we think about material science, coloring technology, and later, the electronics we now stash in our pockets.
Product Overview and Why Chemists Keep Coming Back
A pale or white crystalline powder, cyanuric chloride looks innocuous on a lab bench, but it serves as a lynchpin for making everything from herbicides to water treatment materials. Its structure holds a triazine ring with three chlorine atoms attached, laying the groundwork for a wild array of downstream chemical applications. Plenty of bulk chemicals claim versatility, but few match the reach of this one. Dyestuffs, disinfectants, optical brighteners, and even plastic additives owe their roots to cyanuric chloride’s backbone. You find its fingerprint across textiles, swimming pool maintenance, and more sophisticated spaces such as pharmaceuticals and advanced materials research.
Physical and Chemical Properties: Not Just a Dry Description
Cyanuric chloride melts at roughly 145 degrees Celsius and boils near 190 degrees. This moderate thermal stability shapes how it’s handled in factories and labs. It doesn’t dissolve in water, but it will react with moisture, so handling requires care to avoid hazardous byproducts like hydrogen chloride gas. Its molecular weight, about 184.41 g/mol, gives you a sense of heft in reactions, and its strong, choking odor signals trouble if you spill it without ventilation. Watch how it interacts with common solvents; it easily dissolves in organic ones like acetone or acetonitrile, again reminding us these chemical traits shape usage and safety alike.
Technical Specifications and Honest Labeling
In practice, inquiry about quality digs deeper than purity. Users check for residual moisture, intended particle size, and free acid content. Standard specifications often demand purity above 99 percent, mainly to suppress unwanted and dangerous side reactions, particularly when the output winds up in things like pharmaceuticals or water purification agents. Industry labeling stresses hazards: corrosive to eyes and skin, noxious if inhaled. Sentiment around cyanuric chloride safety isn’t paranoia—these harsh warnings save lives and equipment, especially as downstream industries have to answer to stricter regulatory eyes.
Preparation Method Reflects Changing Technology
Chlorination of cyanuric acid forms the dominant synthesis route. Industrial plants use phosphorus pentachloride or sulfur dichloride to yank hydrogens and substitute chlorine in their place, shifting a simple ring into a highly reactive platform. This process grew in importance as environmental guidelines cracked down on emissions and unnecessary reagents. Still, legacy processes from decades past can pop up, especially in less regulated regions. Efficiency here matters because byproducts build up quickly, and waste treatment involves real money and safety headaches. Methods keep evolving as companies hunt for cheaper, cleaner, and more efficient ways to pump out massive quantities year after year.
Chemical Reactions and Why Chemists Love Modifications
Cyanuric chloride’s real party trick hails from those three reactive chlorine atoms. Each one acts as a potential site for nucleophilic substitution—stick an amine, phenol, or thiol in, and you cook up a tailored triazine with properties fit for dyes, cross-linkers, or pharmaceuticals. These reactions take place stepwise, and each substitution tweaks reactivity, so synthetic chemists stay busy figuring out how to grab control over yield and selectivity. This flexibility means a single kilogram of cyanuric chloride in a research lab can set off dozens of different syntheses, forming the foundation for new products, materials, and even medicines.
Synonyms and Product Names Spin a Web
Nobody agrees on what to call this stuff, which adds confusion when tracing its use. You’ll run into names such as 2,4,6-trichloro-1,3,5-triazine and trichlorotriazine, plus trade names that stretch across countries and regulatory frameworks. This jumble can frustrate safety auditors, customs agents, and anybody checking supply chains for hazards. I’ve watched more than one shipment get stuck at a border because a labeling term didn’t show up in a computer database, which shows that better consensus on standardized naming isn’t simply academic—it saves time and money across international borders.
Safety and Operational Standards: More Than Red Tape
Cyanuric chloride carries real health risks. Every safety sheet lists skin burns, respiratory trouble, and eye damage as top concerns. If inhaled, workers risk lung inflammation and scarring. Companies install elaborate extraction and containment gear, and proper training means more than ticking boxes. Gloves, face shields, and full-body suits mark daily routines. Any slip-up leads to trips to the emergency shower or medical wing. Factories must also monitor airborne concentrations and keep antidote stations within reach. Regulation doesn’t stop at the factory; even in transport and storage, leaks can trigger hazardous materials protocols and downstream contamination events.
Application Area Points to Everyday Impact
The reach of cyanuric chloride spills over into places few realize. Along with common pesticides, the biggest use sits in light fast dyes for fabrics. Ever wear a colored shirt that keeps its shade after many cycles in the wash? You probably owe some thanks to compounds built on cyanuric chloride. It enters the pool business too, where it stabilizes chlorine so backyard water stays clean longer. Recent years have seen advanced applications take off—in electronics manufacturing, triazine rings shape performance in laminates and flame retardants. Its molecular versatility keeps scientists busy, proving that an old chemical still has miles to run in modern supply chains.
Research, Development, and Real-World Challenges
Academic and industrial researchers circle cyanuric chloride as a launch pad for novel molecules. The race for greener syntheses means scientists are chasing ways to use less hazardous reagents, cut down on hazardous waste, and power up selectivity. Data from the last decade points toward solvent-free reactions and microwave-assisted methods, but scaling from lab to kilo-scale batches brings headaches. Beyond process optimization, some pharmaceutical and agricultural companies tackle the challenge of selective functionalization, looking for ways to pull more useful structures out of less starting material. Research funding from both public and private sources follows sustainability and clean chemistry trends, as public opinion puts heat on companies releasing persistent toxins and greenhouse gases.
Toxicity Research Keeps Raising New Questions
Toxicity sits at the center of any discussion about this compound. Both acute and chronic exposures lead to notable health issues in humans and animals. Animal studies have mapped out inhalation and oral hazards, showing tissue damage in lungs and liver with repeated contact. Water treatment plants worry about downstream byproducts and environmental persistence. For every positive property, researchers and regulators keep finding new challenges and questions. Some studies even focus on the breakdown of cyanuric chloride in the environment, pointing to slow degradation and buildup risks. If regulators set stricter discharge limits or define new safe exposure levels, companies may face expensive upgrades and researchers would scramble to build in greener alternatives.
Future Prospects Demand Both Innovation and Responsibility
Cyanuric chloride probably won’t fade into chemical obscurity anytime soon. Its established utility keeps demand steady, while its chemistry lets specialists tinker with structures and applications. The next big challenge lies in finding and deploying cleaner synthesis strategies that ease the burden on workers and the environment. Bioprocessing, recycling, and better containment tech all make the list of priorities for future research funding. As sustainability and health issues dominate public debate, everyone—from scientists to industry managers—must rethink risk, accountability, and how to build cleaner bridges between chemistry and daily life.
What is cyanuric chloride used for?
Core Chemical for Everyday Products
Cyanuric chloride might sound like a lab-only oddity, but this compound ends up in more household and industrial products than most realize. I first saw its name unpacked when helping a friend with a pool care business and later noticed it popping up in agricultural conversations too. In both cases, cyanuric chloride played a big role without flashy headlines.
The Backbone of Herbicides
Farmers often depend on triazine herbicides to manage relentless weeds. The production of chemicals like atrazine and simazine depends on cyanuric chloride. Without it, those herbicides just wouldn’t come together. Crops like corn and sugarcane count on triazine herbicides for consistent yields, especially with increasing resistance to other weed management tools. Globally, these herbicides support food security by guarding fields against aggressive, invasive plants. According to a USDA report, triazines account for a major chunk of the world’s herbicide demand.
Used in Pool Water Stabilizers
Spend time around swimming pools, and people talk about chlorine, not cyanuric chloride. But this chemical makes safe swimming possible. It acts as a building block for cyanuric acid, which stabilizes chlorine in pool water. Sunlight breaks down chlorine rapidly, so cyanuric acid keeps it active longer. That means families get safer water and spend less on chemicals. From personal experience opening community pools in spring, no pool operator overlooks this step. It’s an everyday example of a specialty chemical creating public health value.
Dye and Pigment Manufacturing
Textiles, paints, inks—each area uses dyes made using cyanuric chloride. It links dye molecules with fiber, helping colors hold fast through washes and weather. Industrial dye processes take full advantage of its reactive nature. The European Chemicals Agency highlights its importance for vibrant, durable coloration in everything from clothing to signage. Over the years, the move toward higher-quality, longer-lasting colors has only boosted demand for this chemical in the pigment sector.
Pharmaceuticals and Specialty Chemicals
Medicines rely on complex chemistry for both effectiveness and safety. Cyanuric chloride acts as a key building block, especially in synthesizing antiviral agents and other high-value drugs. Its three chlorine atoms allow for versatile chemical reactions, letting pharmaceutical researchers fine-tune molecules for targeted treatments. Better, safer medicines depend on this kind of foundational chemistry.
Challenges and Safer Handling
Despite its versatility, cyanuric chloride poses safety challenges many in the industry know well. Direct exposure can irritate the skin and lungs, so proper storage and handling aren’t optional. The American Chemical Society strongly recommends gloves, goggles, and exhaust ventilation for anyone handling it. Accidents during transport or spills can put workers and groundwater at risk.
Regulations keep tightening. Suppliers now label their shipments in line with global standards, and many companies invest in training workers on safe practices. A personal takeaway: never cut corners on protective gear or skip refresher training—real people rely on these safeguards.
Innovation and Sustainability
Driven by demand for greener manufacturing, chemists are searching for safer alternatives or improved handling protocols. Some firms have started recycling cyanuric chloride from production waste, a choice that lowers environmental impact and saves money. Although no single magic-bullet replacement exists yet, the chemical industry’s push toward more sustainable processes feels tangible.
Conclusion
Cyanuric chloride touches daily life in more ways than expected, from the food we eat to the water we swim in. Its benefits come with real risks, but science and sound policy continue to make its use safer and more responsible.
Is cyanuric chloride hazardous to health?
Digging Into Cyanuric Chloride
Cyanuric chloride often comes up in chemical plants, industrial settings, or water treatment discussions. Most people won’t find a bottle of it under their kitchen sink, but its uses pop up in places that matter, like swimming pools, herbicides, and as a building block for pesticides. This chemical might seem far away from daily life, but its handling and health effects deserve attention.
Understanding the Risks
Breathing in cyanuric chloride dust or fumes can hit the airways hard. Workers in factories sometimes talk about nose and throat irritation, coughing, eye burning, and even shortness of breath after exposure. Splashes on bare skin can trigger redness and itching. Studies have shown that folks regularly handling this substance, without strong safety measures, can end up with chronic coughs or slowly weakening lungs.
The reason boils down to its reactive nature. Cyanuric chloride reacts with water—yes, even the moisture in your eyes or nose—to release hydrogen chloride gas. This gas can irritate or damage tissue. Animal studies point to the risks of repeated exposure, which only strengthens what many workers describe from experience.
Long-Term Worries
Extended contact doesn't just make someone feel lousy during the workday—it can creep up on overall health. Consistent exposure tends to tip the scale toward respiratory problems. Chronic bronchitis or asthma-like symptoms can haunt those who face this chemical all week long. Nobody wants to face these conditions just for clocking in.
Lab research has not flagged cyanuric chloride as carcinogenic, but the verdict is not completely settled. Still, its ability to break down and produce hydrochloric acid fumes inside the body means the inflammation and tissue damage are real.
How to Stay Protected
Good ventilation goes a long way in factories or labs. Nobody enjoys wearing heavy masks on humid days, but respirators, gloves, and full goggles protect eyes, nose, and skin—especially in places where powder can fly or liquid can spill. Training sessions for new hires don't just tick a regulatory box; they're one line of defense. Stressing these basics and double-checking safety routines keeps cyanuric chloride from turning a workplace into a health hazard.
Outside the Workplace
In public pools, cyanuric chloride usually appears as a stabilizer for chlorine. By the time it hits the diving board crowd, it’s transformed into another compound, which holds less risk for swimmers. Still, pool operators must respect dosing guidelines and avoid overdoing it. Swimmers shouldn’t panic about every pool, but anyone handling products containing this chemical should follow directions and keep it out of reach of kids and pets.
Toward Safer Solutions
Safer alternatives to some industrial uses are available. Engineers and chemists in the chemical industry stay on the lookout for materials that offer similar benefits without the bite. Finding options with a gentler impact means less trouble for lung health and fewer worries over the long run.
Whether in a factory or pool supply closet, understanding the hazards of cyanuric chloride beats ignoring them. Real knowledge, real equipment, and a focus on safer materials help avoid preventable illness. Health always comes first, even in the most chemical-heavy industries.
What are the storage requirements for cyanuric chloride?
Understanding the Chemical
Cyanuric chloride doesn’t sound like something that belongs outside a lab, but this compound plays a key role in agriculture and the chemical industry. As someone who’s seen the inside of both large chemical warehouses and small research facilities, the way folks store cyanuric chloride tells you plenty about their approach to safety. This compound reacts quickly with water and gives off toxic fumes. Run into even a small spill in a poorly ventilated room and you’ll remember the smell and burn in your throat for the rest of the week.
Don’t Play Fast and Loose With Heat and Humidity
Cyanuric chloride needs to stay bone dry. This is not about convenience, but about preventing nasty reactions. Any humidity in the storage area starts a slow chain reaction that doesn’t just waste product but can trigger accidents. Warehouses storing this chemical use dedicated desiccators or sealed rooms that run on dehumidifiers around the clock. Forget to check those machines for a few days, and you face crusted valves, discolored drums, and sometimes corroded shelving.
Heat makes things worse. Cyanuric chloride breaks down quickly if temperatures creep above 30°C. This isn’t an abstract issue from a safety lecture—one summer, I saw an overheated storeroom cause several drums to crack open. Rooms with steady air conditioning, or cool basements insulated from outside heat, keep the chemical stable and prevent staff from hazardous surprises.
Choose Solid Containers and Label Clearly
Leaky bags and broken lids don’t just waste product, they poison the air. Cyanuric chloride belongs in airtight plastic drums or metal cans lined to prevent corrosion. Even after years of use, you’ll find stains on the outer packaging if storage slips. Workers trust labels on containers to warn of the risks inside. Storage bins often carry the red-and-white diamond symbol for “Toxic” and clear worded warnings about avoiding water and keeping away from acids and oxidizers.
Reliable inventory systems prevent old or damaged packages from lurking forgotten on high shelves. Too often, a missing or faded label has left someone guessing at the contents, often with regrettable results. Barcode scanners and strict logs make it easier for teams to track every batch on the premises, flagging the oldest stocks for disposal before leaks and accidents happen.
Ventilation and Emergency Planning
If fumes escape during transfer or storage, the air must move quickly to prevent toxic build-up. Fans and vents pull air from storage rooms and exhaust it safely outside. I’ve seen places that skip this basic step—after one small leak, employees spent hours dealing with burning eyes and nausea. Emergency showers, chemical spill kits, and respirators stay close to the storage area. Trained teams learn how to contain a spill by blocking drains and using absorbent powders, not by improvising with a mop and rag.
Treat Cyanuric Chloride With Respect
Storing cyanuric chloride safely isn’t just about rules, it’s about coming home at the end of the shift in good health. Good practices, the right storage, and ongoing attention keep chemical incidents to a minimum. People’s lives depend on care, preparation, and the discipline to check every step, every time.
How is cyanuric chloride manufactured?
The Basics Behind Production
Chemistry runs deep in the world of industrial manufacturing, and sometimes it takes a hard look at the process to realize what happens behind closed doors. Cyanuric chloride comes out of a process that starts with simple molecules and cranks up the pressure—literally. Factories take hydrogen cyanide, a substance with its own risks, and react it with chlorine gas. The setup needs high temperature, often over 400°C. This isn’t your backyard project. The molecules join forces in stainless steel reactors, into a ring-shaped structure that forms the base compound of cyanuric chloride.
After the reaction shakes out, the crude product drops as a solid. Workers and automated systems separate this from byproducts like ammonium chloride. You don’t just bag it and send it out the door, though. There’s more involved. The mixture heads over for distillation and purification before the finished cyanuric chloride meets the world. Because of its reactivity, the chemical usually leaves the plant as flakes or white powder, carefully packed and labeled. The process demands respect, both for the volatile raw ingredients and the heat involved.
Why Bother? The Uses Carry Weight
Chemists landed on cyanuric chloride because of what it allows in other fields. For years, dyestuffs, optical brighteners, and herbicides drew from its unique setup. The triazine ring—just six atoms, three carbons and three nitrogens—offers sharp reactivity once the chlorine atoms on the molecule swap out with other groups. The flexibility these reactions enable supports the agriculture sector, keeping crops strong and weeds at bay through products like atrazine. In the textile world, colorfast dyes wouldn’t stick as well without it. Even swimming pool owners see the chemical in the stabilization of chlorine tablets, though the linkage is less direct.
Safety and the Environment: Taking Risks Seriously
There’s no glossing over the risks. Cyanuric chloride and its raw materials, especially hydrogen cyanide and chlorine, demand tight controls. I’ve seen what happens when safety gets taken for granted: releases of these gases endanger workers and everyone nearby. History offers harsh lessons from accidents. Strong ventilation, automatic leak detectors, and hands-on safety training pay off. Factories have built in extra safeguards—scrubbers to remove stray gases, emergency drills, and isolation zones around reactors. Regulatory agencies inspect these facilities more closely than most, because mistakes cut deep both in human and environmental terms.
Waste poses a real challenge. Effluent from the process includes chlorinated compounds, some of which linger if not handled well. Modern plants push wastewater through neutralization steps, followed by treatment to break down hazardous residues. Local aquatic life benefits from extra diligence here. Companies face not only legal pressure but also community expectations to limit contamination and greenhouse gas emissions. Embracing automation and closed-system tech reduces worker exposure and lowers accidents. Even so, the global market keeps growing, which means these conversations keep getting louder.
Where Do We Go From Here?
Progress in this space often moves fast. Green chemistry efforts take a swing at replacement processes using less hazardous reagents, or alternative catalysts that function at lower temperatures. Labs around the world hunt for ways to upgrade gear and reduce the footprint, both for safety and sustainability. Some fixes start to show up—better waste capture, more efficient thermal management, and energy recovery systems. The price tag for these upgrades sometimes slows adoption, especially in countries where regulations look loose. Consumer demand—and public pressure—encourages stricter oversight. For now, tough rules and serious investment let responsible players keep this business running, while chemists keep pushing the boundaries safely.
What are the safety precautions when handling cyanuric chloride?
Understanding Cyanuric Chloride’s Risks
Cyanuric chloride pops up in everything from pool stabilizers to herbicides. I’ve watched chemists and warehouse staff work with it, so it’s easy to see how dangerous it gets once you open a drum. That acrid odor hints at trouble—its fumes attack the nose and eyes fast. Even one slip on a hot day can send someone scrambling for fresh air. Exposure burns skin, irritates lungs, and in heavy doses causes real damage. That's not a maybe; MSDS sheets and incident reports back it up. It reacts hard with water, letting off hazardous hydrogen chloride. Splash some on a damp glove and watch it eat through. Simple mistakes stack up fast in a lab or a loading dock.
Setting Up for Safety
You can’t roll the dice with personal protective equipment. Nitrile or neoprene gloves protect hands better than latex, which breaks down. Safety goggles with side shields matter—a standard pair of specs won’t block splashes. Good labs call for face shields and long-sleeve lab coats that get washed right after use. I remember seeing a tech skimp on a face shield once; his glasses fogged, and he leaned in too close to measure. That’s all it takes.
Ventilation strips away much of the risk. Fume hoods do more than make the work fancy—they pull vapors out before they reach your lungs. Heavy users of this chemical fit their rooms out with negative pressure systems and tight airlocks. In storage, cyanuric chloride lives in dry, airtight drums away from acids, bases, or anything moist. Shelf labels mark warnings in bright ink, since even sealed containers need checking in humid weather.
Prepared for Spills and Accidents
No lab or plant should treat spills like a routine mop job. I joined a drill once where a rookie dropped a small bag. The team closed off the area—the spill plan lived on the wall, not just in the plant manager’s head. Spill kits for cyanuric chloride include neutralizers that won’t simply add more fumes. Only trained staff attack the mess, suited up and working fast. Down the hall, emergency showers and eyewash stations keep close. I haven’t seen anyone need them for real, but I notice every visitor checks for those green stations on a tour.
Training Makes the Biggest Difference
Hazard communication means more than a sign or a memo. Managers teach what to watch for, from that faint chlorine whiff to irritation hours after exposure. Regular drills, up-to-date safety sheets, and open talk during team meetings sink lessons deeper than any email reminder. Medical kits nearby, fast communication lines, and an emergency plan help limit harm.
Building a Culture of Safety
Companies in agriculture, chemical manufacturing, and pool supply chains face real risks with cyanuric chloride. They can’t only enforce policies—they have to encourage questions and double-checks. I’ve seen veterans show new staff how to test gloves, or how to run a hood for an extra minute. Small details like fresh gloves and a steady ventilation hum beat out shortcuts. Sharing stories of close calls pushes the point through better than a warning on paper.
Precaution takes teamwork, not just protocols. Most accidents I’ve heard about didn’t come from bad luck, but from someone skipping gear or ignoring strange smells. Cyanuric chloride rewards preparation, equipment checks, and honest talk about mistakes. That’s what keeps everyone upright at the end of a shift.
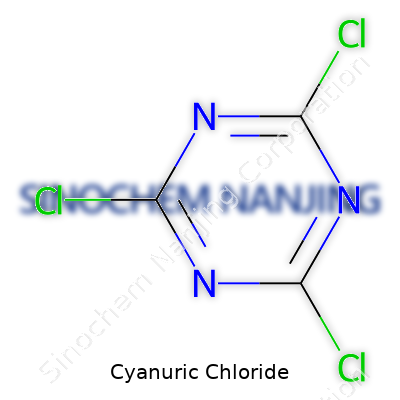
| Names | |
| Preferred IUPAC name | 2,4,6-Trichloro-1,3,5-triazine |
| Other names |
2,4,6-Trichloro-1,3,5-triazine Cyanuric trichloride Trichloro-s-triazine Synergex |
| Pronunciation | /saɪˌæn.jʊr.ɪk ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 108-77-0 |
| Beilstein Reference | 136-23-2 |
| ChEBI | CHEBI:31441 |
| ChEMBL | CHEMBL141722 |
| ChemSpider | 21106379 |
| DrugBank | DB11262 |
| ECHA InfoCard | 07c6d681-1acc-4d45-8ac7-39b47cba7dd5 |
| EC Number | 208-772-0 |
| Gmelin Reference | 79098 |
| KEGG | C01472 |
| MeSH | D003545 |
| PubChem CID | 9321 |
| RTECS number | GB6300000 |
| UNII | M65I73010E |
| UN number | UN2671 |
| CompTox Dashboard (EPA) | DTXSID2046796 |
| Properties | |
| Chemical formula | C3Cl3N3 |
| Molar mass | 184.41 g/mol |
| Appearance | White crystalline powder |
| Odor | Pungent |
| Density | 1.92 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.71 |
| Vapor pressure | 0.0133 hPa (20 °C) |
| Acidity (pKa) | 1.0 |
| Basicity (pKb) | 13.5 |
| Magnetic susceptibility (χ) | -69.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 1.45 mPa·s (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -199.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1082 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301, H310, H314, H317, H330, H334, H335 |
| Precautionary statements | P261, P264, P271, P301+P330+P331, P304+P340, P305+P351+P338, P309+P311, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Autoignition temperature | 570 °C (1060 °F; 843 K) |
| Lethal dose or concentration | LD50 oral rat 1400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat): 1400 mg/kg |
| NIOSH | SN1575000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Cyanuric Chloride: "0.5 mg/m³ (as dust, OSHA PEL) |
| REL (Recommended) | REL (Recommended Exposure Limit) of Cyanuric Chloride is "0.5 mg/m3". |
| IDLH (Immediate danger) | 20 mg/m3 |
| Related compounds | |
| Related compounds |
Cyanuric bromide Cyanuric fluoride Cyanuric acid Melamine |

