Cyanoacetic Acid: Chemistry, Challenges, and Opportunities
A Look Back: Historical Threads
Digging into the story of cyanoacetic acid, it's impossible to ignore how central it’s been to synthetic chemistry. Early chemists found new tools in its unusual structure, sporting both a cyano group and a carboxylic acid sitting right next to each other on a two-carbon chain. This combination created a building block with plenty of reactivity up its sleeve. Through the late nineteenth century, folks working in organic labs discovered they could coax a surprising range of compounds out of cyanoacetic acid, which eventually made a mark in pharmaceuticals, pesticides, and dye industries. Its manufacturing and handling got more scientific muscle over time, with process optimization and regulations picking up in the twentieth century. Chemicals like barbiturates and malonic acid derivatives owe their birth to cyanoacetic acid, and older research papers read like a roadmap to the steady, persistent refinement of its production and use. Today, the substance still attracts attention for reactions in fine chemicals and niche agrochemical products.
Product Overview: More Than Just a Raw Material
Cyanoacetic acid isn’t just one more bottle on a shelf. Chemists know it as a sharp tool, combining two functional groups in a single, compact molecule. The molecule packs a wallop with its acidity, making it stand out in reactions that want something more active than regular acetic acid. Because both the cyano and carboxyl groups offer sites for transformation, cyanoacetic acid shows up in plenty of syntheses. Drug researchers lean on it to craft active ingredients or intermediates with complex ring structures. Manufacturers tap its versatility for flavoring agents, artificial sweeteners, and adhesives. In my time handling cyanoacetic acid during research, I saw both its creative power and its complications—good yield in the right hands, mishaps in careless ones.
Physical and Chemical Properties: What Stands Out
Cyanoacetic acid sits as a white, crystalline solid with a strong sour smell. Its melting point is usually reported just under boiling water temperature, making it easy to handle in solid or liquid reactions. Chemically, the acid stands out because the cyano group next door pulls electron density, hiking up the acidity compared to other small acids. It dissolves well in water and polar organic solvents, and its reactivity lets it participate in condensation, alkylation, and coupling reactions. Given its dual functionality, it slides into more roles than most simple acids. The molecule does tend to darken with air and moisture, so storage conditions matter for keeping purity.
Technical Specifications and Labeling: An Insider’s View
On the technical side, cyanoacetic acid usually comes labeled for purity, moisture content, and trace impurities like chloride or heavy metals. Suppliers lean toward a minimum 98 percent assay for most industrial uses, though research-grade stuff gets even cleaner. Laboratories track the color, form (fine or granular crystals), and sometimes residual solvents if it’s freshly recrystallized. Proper labeling is critical because confusion between cyanoacetic acid and something like malonic acid can ruin complex synthesis workflows or even trigger accidents. Regulations require full hazard statements, since the acid hits safety limits with both skin corrosion and acute toxicity. Whether you pick up a small bottle in a teaching lab or a barrel in a manufacturing plant, the labeling reminds you about safe handling and correct storage—all the more important given the chemical’s risks and sensitivity to moisture.
Preparation Methods: How Industry Makes It
I’ve watched cyanoacetic acid take shape through two main processes. The old-school method mixes sodium chloroacetate and sodium cyanide in water, driving substitution to yield the sodium salt of cyanoacetic acid. Treating that with a mineral acid like hydrochloric then precipitates the finished acid, ready for collection. Newer methods swap chloroacetic acid for bromoacetic acid and tinker with phase-transfer catalysts for efficiency. The reaction always requires care—because cyanide wastes generate hydrogen cyanide gas, a killer even in small doses. Industrial operations build plenty of containment, scrubbers, and automated handling, reflecting pain learned from early accidents. Quality depends on tight pH control and precise temperature management, since too much heat or squeezing the acidity the wrong way triggers unwanted byproducts. The finished acid then gets filtered, dried, and sometimes recrystallized for higher purity or specialty applications.
Chemical Reactions and Molecular Tweaks
Once cyanoacetic acid lands on a chemist’s bench, its story only gets more interesting. The acidity lets it act as a nucleophile in Knoevenagel condensations, which crank out molecules used in dyes and drugs. Alkylation at the methylene carbon fills out even more of the chemical landscape, making cyanoacetic acid a favorite for building up carbon chains in research settings. In the presence of ammonia and heat, it can morph into amino acids or help construct aromatic rings, making it key in some pharmaceutical synthesis routes. Simple base treatment can trigger decarboxylation, freeing cyanoacetic acid’s carbon for further transformation. I’ve had frustration when small changes in pH or solvent type swing these reactions from product-rich to tarry mess, so there’s little room for autopilot. The same flexibility that makes cyanoacetic acid useful also means a heavy workload on method development for custom molecules or scale-up.
Names and Aliases: Navigating the Jargon
Cyanoacetic acid carries more than one label, and in different corners of science, you’ll spot names like 2-cyanoacetic acid, α-cyanoacetic acid, or even carboxymethyl cyanide in older papers. CAS registry number 372-09-8 often sits alongside it in regulatory files. Chemists need to stay sharp here because the cyano prefix and similar-sounding cousin compounds create confusion. Even trade catalogs sometimes mix up the synonyms, so double-checking nomenclature avoids waste, returns, and safety hitches on the job.
Playing It Safe: Handling, Hazards, and Best Practices
Handling cyanoacetic acid always demands vigilance. It bites both the skin and airways, and even small exposures trigger burning. On the toxicology side, animal studies put LD50 values squarely in a range that calls for gloves, goggles, and fume hoods—never casual handling. The acid’s reactivity means it should never go near basic solutions or hot surfaces without oversight, since toxic fumes can result. From the cleanup side, I learned from mentors to clean up crystals and dust with water-dampened materials, never dry sweeping, to avoid airborne particles. Emergency protocols suggest copious rinsing of spills and a direct line to eyewash and showers. Instructions on every drum, bottle, and datasheet spell out storage in cool, dry, well-ventilated spots, away from incompatible chemicals or open flames. For industrial operators, ongoing training, updated hazard communication, and auditing make up the difference between an uneventful day and a trip to the emergency room.
Where It Works: Application Areas
Cyanoacetic acid leaves fingerprints across several industries. Agrochemical groups use it as a springboard for herbicides and fungicides—the molecule’s carbon backbone helps shape actives with precise toxicity profiles. Pharmaceutical giants use it as an intermediate for anti-epileptic drugs and antibiotics, while perfumery leans on its reactivity to graft on complex esters for scents. Even adhesives and coatings engineers pull it into resin and polymer production, chasing specific chemical features that only readily form through cyanoacetic acid intermediates. For years, researchers looked at it as a candidate for making synthetic amino acids and flavor compounds, and its fast, versatile reactions keep it popular in new compound library searches in medicinal chemistry. Its physical form suits blending and scale-up in fine chemicals, though handlers always weigh benefits against the health and regulatory burdens.
On the Research Front: New Approaches and Persistent Hurdles
Much of the excitement around cyanoacetic acid comes from its role in new synthetic routes that aim to sidestep harsher conditions or lower-cost raw materials. Green chemistry puts pressure on the industry to cut out cyanide-based feedstocks, which prompts investigation into biocatalytic or alternative chemical pathways. Researchers continue to probe for new reactions using cyanoacetic acid skeletons in multi-component condensations, hoping to unlock new medicine candidates in less time. At the same time, material scientists search for fresh uses in specialty plastics or conductive polymers. Alongside, advances in process engineering make the preparation safer and more energy-efficient, with automated monitoring to slash accident rates. My experience reading through current literature suggests persistent interest in reaction optimization, purification tech, and waste minimization—because small improvements can ripple out to lower environmental and financial costs.
Toxicity Research: What the Data Warns
Toxicologists haven’t let cyanoacetic acid off easy. Lab tests show acute oral and dermal hazards, underscored by warnings on standard safety sheets. Chronic exposure data suggest potential organ involvement, though gaps linger for precise long-term effects in humans. Animal studies point to metabolic disruption, which aligns with cyanoacetic acid’s ability to interfere with crucial biochemical pathways. Researchers highlight areas for more study, especially in low-dose, industrial, and environmental scenarios. I’ve found that, despite decades of research, modern regulatory frameworks keep updating limits and controls based on emerging evidence. Health professionals track workplace exposures and focus on improving on-site monitoring, immediate decontamination techniques, and design changes that physically isolate workers from hazards.
Where the Road Leads: Future Prospects
The future for cyanoacetic acid pulls in different directions. Strict regulations around cyanide handling push the industry toward cleaner preparations and safer packaging. Green chemistry principles push old processes aside, searching for renewable starting materials or lower-energy synthesis. Application growth depends on how quickly researchers can find safer derivatives or recycle cyanoacetic acid effectively. In pharmaceuticals and specialty materials, new reactions built around its active methylene group could spawn treatments or materials nobody’s considered yet. In regulatory spaces, expect tighter controls, more training, and greater transparency on product labeling. Industry success and worker safety rely on steady progress in process improvement, detection methods, and practical, hands-on safety protocols. Real innovation grows when the right balance gets struck between chemical utility, risk, and environmental impact, and cyanoacetic acid’s future likely hinges on how well the chemical world navigates those trade-offs.
What is cyanoacetic acid used for?
The Unsung Workhorse in Laboratories
Step into a university chemistry lab or most industrial research centers, and you’ll find bottles labeled “cyanoacetic acid” tucked between more famous names. It doesn’t attract much attention, but this sharp-smelling, crystalline solid powers so many reactions. Most chemists remember puzzling over its formula during their studies. What makes it interesting is its reactive cyano and carboxyl groups, which open the door to a huge range of uses.
Building Blocks for Pharmaceuticals
Drug discovery moves fast, and chemists often turn to cyanoacetic acid as one of their tools. The compound acts as a key player in synthesizing important medications. Take barbiturates, for example—used for sedation, anesthesia, and even seizure management. Cyanoacetic acid provides the backbone for molecules like these, helping shape the active part of the drug. Its reactivity makes it perfect for adding or swapping chemical arms, building complexity where the pharmaceutical industry demands it.
Agrochemicals and Protecting Crops
People working in agriculture rarely hear the name cyanoacetic acid, but they depend on it all the same. This compound forms the starting point for some modern herbicides and pesticides. Glyphosate synthesis, for example, starts from reactions involving cyanoacetic acid. The science goes beyond the field: without such foundations, the range of options farmers hold would shrink. That’s not just important for crops, but also for keeping food prices stable and kitchens stocked with plenty of choice.
Modern Materials and Dyes
A textile factory or materials lab uses cyanoacetic acid in ways most consumers never suspect. Dyes for clothing, plastics, and some pigments for printers or paints rely on the sort of chemical bonds cyanoacetic acid can help form. In fact, many high-performing dyes in synthetic fabrics or technical uses wouldn’t exist without this simple acid’s chemistry making their vivid colors possible. For materials scientists, it helps adjust strength and flexibility of advanced resins and plastics.
Challenges and Safer Practices
Getting all these benefits out of cyanoacetic acid demands careful handling. The compound is toxic if inhaled or swallowed and can irritate the skin. Anyone who’s worked with it knows the sharp smell and the warning labels. Over the last decade, stricter regulations and better lab training cut back on accidental exposure and pollution. Chemical manufacturers invest in exhaust systems and sealed reactors, and most now publish safety data so smaller shops can avoid nasty surprises.
Greener Solutions on the Horizon
As calls for greener chemistry grow, some researchers look at ways to produce cyanoacetic acid with fewer harmful byproducts. Bio-based synthesis methods and recyclable catalyst systems offer some hope, though the path from lab bench to industrial scale takes time and investment. Strong environmental rules push this work forward, and the market for sustainable chemicals keeps widening. It’s a sign that responsible production is not only possible but can become the standard if economic and social incentives point the way.
What All This Means for the Future
Everyday products and lives improve with chemicals like cyanoacetic acid working behind the scenes. The benefits can’t come at the expense of safety or health. People in the field—lab workers, farmers, factory staff—learned that years ago. As technology improves the process, society gets the most out of chemistry’s potential while keeping risks in check. With eyes on both innovation and responsibility, cyanoacetic acid can keep serving quietly at the heart of medicine, agriculture, and materials for years to come.
What are the safety precautions when handling cyanoacetic acid?
Respect the Hazards
Working with cyanoacetic acid takes more than just wearing a lab coat. You can’t ignore its risks: skin burns, eye damage, and possible poisoning if inhaled or swallowed. Even small spills make your throat scratchy and your eyes water. Breathing too much of its vapor during an experiment once left me with a pounding headache. This isn’t the kind of job you can do with half your attention.
Use Personal Protective Equipment
Before even unsealing the bottle, slip on nitrile gloves that won’t break down quickly. Latex breaks down with organics, but nitrile stands up better. Swap out gloves at the first sign of weakness; cyanoacetic acid eats through worn-out material. Choose thick, chemical splash goggles over safety glasses—I used to think glasses would suffice, until a tiny splash during weighing ran along my cheekbone. Face shields go further, blocking unexpected spatters from flasks. Those with asthma or breathing issues do well to fit a respirator with an organic vapor cartridge, especially in low-ventilation setups.
Ventilation Matters
Decent airflow dilutes the fumes that sneak up fast. Standard rooms just don’t cut it—use a chemical fume hood with a sash kept low and body tucked back. Fans blowing fumes into the hallway won’t help you or anyone else. My first months in a university lab taught me that even a quick open-air transfer releases vapors that blanket the workspace. Spend the extra time walking work into a hood or, if you can, setting up a local exhaust system.
Prepare for Spills and Accidents
Spills don’t announce themselves. Have sodium bicarbonate or another neutralizer close at hand, scoops and absorbent pads within arm’s reach. Investing a few minutes up front means you don’t scramble later, which I learned after one bottle slipped from condensation. The acid pooled across the bench, but neutralizer kept harm at bay. Safety showers and eyewash stations belong within ten seconds of your hands: in those moments, even a short detour feels endless.
Label and Store Properly
Sharp labeling with the hazard pictograms takes guesswork out of the equation. In one forgetful spell, I tucked cyanoacetic acid between benign buffers—never again. Deadly chemicals need isolation in corrosive-proof cabinets, capped tightly and away from heat or direct light. Check containers for cracks or discoloration. Rotate your stock so aged containers leave before they leak or degrade. Mixing up the order brought me grief once when an expired sample crusted over and vented fumes.
Train and Communicate
Fresh hands on deck? Take them through each risk and safe practice, don’t just hand off the safety data sheet and call it good. Walk through what to do in a spill, where to find protective gear, and how to safely dispose of waste. Open communication lowers the odds of risky shortcuts. Regular drills with my co-workers paid off; in moments where panic tempts haste, habits kick in.
Final Thoughts
Cyanoacetic acid brings serious hazards to any workplace, large or small. Simple, consistent habits—using proper gear, maintaining airflow, keeping clean-up supplies handy, and never skipping real-life training—form your best defense. Respect for the risks and a focus on basics make all the difference between a routine day and an emergency call.
What is the molecular formula of cyanoacetic acid?
Understanding Cyanoacetic Acid
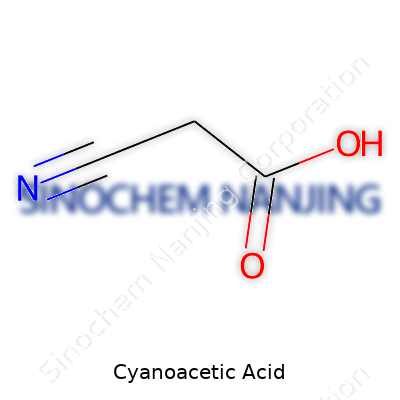
Cyanoacetic acid brings together two distinct groups—cyano and carboxylic acid—within one compact molecule. Its molecular formula sits as C3H3NO2. This formula points to three carbon atoms, three hydrogens, a nitrogen, and two oxygens. I’ve held small vials of cyanoacetic acid in my hands back in my undergraduate chemistry days, sometimes with a sense of caution. Once, a professor pointed out, “this compound’s formula is simple enough, but its impact ripples far.” That stuck with me. You’ll hear about acetic acid and even cyanide in everyday conversations, but mix the concepts, and you get a new beast.
Why the Molecular Formula Matters
Clarity in a molecular formula helps chemists avoid costly mistakes. Mixing up one letter or number—say, confusing C3H3NO2 with C2H3NO2 (glycine)—could completely derail a reaction or produce a plastic monomer instead of a pharmaceutical intermediate. Accuracy in molecular formulas, simple as it looks on paper, can impact jobs, the safety in a lab, and the effectiveness of manufactured goods.
I recall a friend’s story from a pharmaceutical lab where someone mistyped the material request as “cyanoacetamide” instead of cyanoacetic acid. Two weeks vanished in confusion before they pieced it together—because a molecular formula wasn’t double-checked. Mistakes can turn expensive, especially for companies mass-producing fine chemicals or custom APIs.
Applications Driving Demand for Precision
Cyanoacetic acid features in adhesives, amino acid synthesis, dyes, even certain antihypertensive drugs. I’ve read in industrial journals that its nitrile and carboxylic acid combination gives it unique reactivity. For example, pharmaceutical chemists rely on cyanoacetic acid to build more complex molecules. Even in undergraduate organic synthesis courses, instructors lean on it to teach newer students about nucleophilic substitution reactions and malonic ester-like chemistry.
For people working in manufacturing, a slip in identifying the right chemical can stop production. Synthesizing beta-amino acids, for example, uses cyanoacetic acid at key stages—a missed batch could mean failing to deliver a critical drug component on time.
Quality and Safety Factors
Getting the right formula also connects to health and safety. Cyanoacetic acid shows strong acidity and can release toxic gases if mishandled. Workers trust labels and safety sheets, which all start with knowing the correct chemical formula. Even small research teams embrace standards set by organizations like the FDA or European Medicines Agency; regulatory bodies pull up exact formulas to trace ingredients in everything from over-the-counter painkillers to agricultural products.
Demand for transparent supply chains has grown while governments and consumers expect tighter oversight of chemical manufacturing. Traceability begins with something as basic as a formula—C3H3NO2 lets regulators, quality controllers, and import/export teams verify what’s moving through ports and factories. In my own experience, minor gaffes get magnified as batches move from bench scale to tonnage.
Building on Scientific Trust
Years of working with chemicals have taught me that trust in research starts by getting the basics sorted out. The molecular formula of cyanoacetic acid might look like a few dry letters and numbers at the surface, but mistakes here ripple into issues far beyond a single laboratory bench. Diligent work and constant verification keep things moving—whether in a high school classroom or an industrial scale-up.
How do you store cyanoacetic acid safely?
The Realities of Handling a Sensitive Chemical
I’ve seen enough chemistry labs over the years to know some compounds demand extra respect. Cyanoacetic acid falls into this camp. It offers plenty of value for work in pharmaceuticals and organic synthesis, but let’s not forget its risks: corrosive, flammable, and nasty if mishandled. If you’ve ever seen the aftermath of a forgotten bottle of the stuff leaking fumes into a storeroom, you know cutting corners does not mix with acids like this one.
The Right Kind of Container Makes All the Difference
Cyanoacetic acid does not play well with just any old plastic or metal. Too porous, and you risk leaks. Too fragile, and you’re one careless bump away from a spill emergency. Amber glass bottles with air-tight screw caps beat all the rest, especially since this acid degrades under light. Amber or brown-tinted glass means fewer breakdown products floating in the air or ruining your next experiment. I always label my containers with bold, clear warnings, plus the date of opening: no guessing games needed. If you need to store more than a few ounces, sturdy secondary containers save the day—never underestimate a good spill tray.
Temperature and Humidity: More Than Afterthoughts
Walk into any chemical storeroom and you can spot problems pretty quick: temperature swings, high humidity, poor ventilation. Throw cyanoacetic acid in those conditions, and you ask for trouble. My best results come from rooms kept cool—ideally under 25°C (77°F)—and bone-dry. Temperature spikes boost the risk of decomposition, at worst producing toxic fumes that no regular lab fan can scrub fast enough. A sealed desiccator takes storage up a notch; it locks out moisture and holds temperature steady. Tossing packets of molecular sieves inside helps—these little desiccants mop up stray water and keep the acid dry for months.
Keep It Away From Incompatibles
Chemistry teaches harsh lessons if you store acids close to strong oxidizers or bases. With cyanoacetic acid, even organic solvents can start unwanted reactions, releasing gases or heat. Pay attention to separation rules in your storeroom. If you keep other reactive acids, flammable liquids, or even just common lab bleach nearby, block off a dedicated space for the cyanoacetic acid. I separate by shelf and add obvious warning tape—saves confusion for anyone in a rush or new on the job.
PPE: Not Just for Big Spills
A quick splash can eat through cotton, skin, and even the best wool dress pants. Goggles, thick nitrile gloves, a lab coat, and good closed shoes are my minimum every time I open a bottle. I keep a spill kit close, stocked with absorbent pads and neutralizers. Anyone storing cyanoacetic acid should run a drill on using these kits: in a real spill, every second counts. I keep the emergency eye-wash and shower free of clutter, just in case.
Training and Documentation Save More Than Samples
New lab workers often assume the label warnings are just legal padding. I spend real time walking trainees through Material Safety Data Sheets (MSDS) for cyanoacetic acid. We talk real-life accidents and lessons learned. Detailed logbooks for opening dates, storage checks, and disposal records help keep everyone honest—no more out-of-date bottles hiding in the back. Controlled access to the storeroom keeps prying hands and accidents to a minimum.
Disposal: Respect Goes to the Finish Line
Expired cyanoacetic acid should never go in regular trash or down the sink. I’ve seen labs fined or shut down from sloppy disposal practices. I collect old or contaminated acid in thick, labeled containers and hand it off to certified waste handlers. No shortcuts, no exceptions—regulatory agencies like the EPA take this seriously, and so do I. Responsible disposal closes the circle of safety around this tricky chemical.
What are the potential health hazards of cyanoacetic acid?
Understanding Cyanoacetic Acid in the Real World
Cyanoacetic acid often shows up in conversations about lab safety and chemical handling, but most people outside science circles hardly hear about it. I learned about its risks the hard way while assisting with a small-scale chemistry demonstration. The sharp, vinegar-like odor hit me right away, and the instructor firmly warned about the dangers. That lesson stuck with me, so let’s talk about what’s at stake with this chemical.
Main Health Risks
Cyanoacetic acid has a sneaky way of creating problems if you’re not paying close attention. Just a small amount on bare skin can trigger redness and painful blisters. Absorption through skin contact doesn’t only cause localized burns; it opens up the door for more problems since cyanoacetic acid moves quickly into the body’s systems.
Inhaling the dust or fumes triggers coughing, chest tightness, and sometimes a raw feeling in the lungs. After direct exposure, people sometimes develop headaches and feel nauseous for hours. Researchers know the substance can irritate the respiratory tract in a matter of minutes, so working with proper ventilation makes a world of difference.
Swallowing cyanoacetic acid by accident causes a stinging sensation in the throat and mouth, followed later by abdominal pain, vomiting, and—in rare but serious cases—convulsions. Poisoning from this acid doesn’t always show up as a dramatic emergency right away. So medical experts urge anyone who comes in contact with even a modest amount to seek timely medical help instead of shrugging it off.
Potential Long-Term and Systemic Effects
Given the presence of both cyano and carboxyl groups in the molecule, this acid can wreak havoc further down the road. The cyano group, in particular, is notorious for releasing cyanide ions inside the body during metabolism. Cyanide blocks the body’s ability to use oxygen on a cellular level. Every major system gets hit in a severe poisoning event, and once oxygen gets cut off, the brain and heart don’t last long. Even small exposures over time can quietly sap energy and weaken concentration, something scientists studying industrial exposure have documented for decades.
Health and Environmental Safety for Workers
People who handle cyanoacetic acid at work, especially in pharmaceuticals and chemical manufacturing, face bigger risks. In my own experience on a factory walkthrough, I noticed some employees didn’t have protective goggles or gloves. They had gotten used to rushing the process to meet production goals, thinking “what’s a little splash?” Years of examples, though, show even brief exposure can disrupt someone’s health. Direct skin contact causes chemical burns—a real risk in busy production lines.
In poorly ventilated spaces, gases and vapors build up and hang around. That’s a recipe for chronic health issues, especially for the more vulnerable workers. Pregnant employees and people with existing health problems face higher risks from both direct and indirect exposure. Science backs these concerns: repeated exposure sometimes leaves a mark on the liver, kidneys, and nervous system.
Improving Safety—Without Cutting Corners
Effective solutions involve more than issuing a list of instructions. Companies and schools need to build safety into their regular routines—routine ventilation checks, stocking up on gloves and face shields, and making sure everyone leaves contaminated clothing behind in the workroom. Routine health screening for workers remains a practical step. Transparency about what cyanoacetic acid exposure can do helps workers respect those warning labels, not just follow them out of habit.
Finally, safe disposal of waste and regular drills on what to do in case of a spill keep both people and the environment safer. I’ve seen firsthand that a culture of safety—where people feel comfortable reporting problems—saves a lot of trouble down the line. The reason cyanoacetic acid deserves respect is simple: it only takes one careless slip for permanent harm to follow.
| Names | |
| Preferred IUPAC name | 2-cyanoacetic acid |
| Other names |
Cyanacetic acid 2-Cyanoacetic acid 2-Cyanoethanoic acid |
| Pronunciation | /ˌsaɪ.ə.noʊ.əˈsiː.tɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 372-09-8 |
| 3D model (JSmol) | `C[CH](C(=O)O)C#N` |
| Beilstein Reference | 635875 |
| ChEBI | CHEBI:50587 |
| ChEMBL | CHEMBL50290 |
| ChemSpider | 54670 |
| DrugBank | DB04315 |
| ECHA InfoCard | ECHA InfoCard: 100.003.233 |
| EC Number | 203-806-2 |
| Gmelin Reference | 7663 |
| KEGG | C00183 |
| MeSH | D003570 |
| PubChem CID | 986 |
| RTECS number | GF9625000 |
| UNII | 5A067OQW1J |
| UN number | UN2698 |
| Properties | |
| Chemical formula | C3H3NO2 |
| Molar mass | 85.06 g/mol |
| Appearance | White crystalline powder |
| Odor | Pungent |
| Density | 1.266 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.0 |
| Vapor pressure | 0.0117 mmHg (25°C) |
| Acidity (pKa) | 2.5 |
| Basicity (pKb) | 8.98 |
| Magnetic susceptibility (χ) | -33.5e-6 cm³/mol |
| Refractive index (nD) | 1.432 |
| Viscosity | 19.1 mPa·s (25 °C) |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −213.7 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -870.7 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H312, H314 |
| Precautionary statements | P210, P261, P264, P280, P301+P312, P330, P302+P352, P305+P351+P338, P304+P340, P312, P370+P378, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 127 °C |
| Autoignition temperature | > 300 °C (572 °F; 573 K) |
| Explosive limits | 3.4–?% |
| Lethal dose or concentration | LD50 oral rat 1,510 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1,510 mg/kg |
| NIOSH | SN2075000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Cyanoacetic Acid: "No OSHA PEL established |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Acetic acid Malonic acid Cyanoacetamide Cyanacetylnitrile Cyanoacetyl chloride |

