Pseudocumene: More Than Just a Chemical Compound
A Journey Through Its Historical Development
Pseudocumene, known in some circles as 1,2,4-trimethylbenzene, has lingered in the background of industrial chemistry for generations. Dating back to the discovery of aromatic hydrocarbons in the 19th century, chemists curious about coal tar found this molecule among the complex web of ring compounds. As the chemical industry expanded, the need for versatile solvents and intermediates grew. Pseudocumene came into wider use, especially after advancements in petroleum refining and hydrocarbon separation in the early and mid-20th century. No pomp or ceremony, just a steady presence in paint factories and research labs, quietly underpinning more visible products and solutions.
Unwrapping Its Product Reality
Those who have worked in chemical plants or industrial labs know the distinct, somewhat sweet aroma of aromatic hydrocarbons. Pseudocumene, a clear liquid at room temperature, fits right in, sometimes mistaken for xylene or other methylbenzenes by the untrained nose. Most commonly, it's part of commercial mixtures, especially those used as solvents in coatings, dyes, and specialty fuels. Several suppliers distribute it under names like "trimethylbenzene" or even "antidetonant" when adding it to fuel blends. Despite its relatively low profile, the compound underpins key industrial formulations, hiding in the lineup of ingredients on data sheets.
Physical and Chemical Identity
Pseudocumene isn’t flashy—clear, nearly colorless, with a density a bit less than water, and a boiling point that offers easy separation from heavier fractions during distillation. Every chemist learns early that methyl substitutions change not just nomenclature, but also subtle aspects of reactivity and solubility. With three methyl groups tucked onto a benzene ring at the 1, 2, and 4 positions, the molecule resists most forms of simple oxidation but can undergo sulfonation and halogenation in controlled settings. Low water solubility means it doesn’t simply wash out of spills or leaks, suggesting a need for care in waste handling.
Getting Down to the Lab Bench: Preparation and Chemical Transformations
Large-scale facilities rely on the methylation of toluene or the controlled catalysis of xylene streams from crude oil cracking to obtain pseudocumene. Those who have witnessed an industrial reformer in action know the importance of process controls—one slip in temperature or catalyst performance, and output ratios shift, leading to more by-products than practical. On the lab scale, students experiment with Friedel-Crafts reactions, learning that positional selectivity isn’t always predictable, and that yields of the desired 1,2,4 arrangement depend on reaction conditions and clean separation methods. In the wild world of chemical modifications, pseudocumene serves as a precursor for trimellitic anhydride and other intermediates, acting as a bridge to resins, plasticizers, and specialty compounds.
Looking at the Label: Names and Regulations
Anyone with a background in regulatory compliance will recognize the jumble of synonyms attached to compounds in the aromatic family. Alongside pseudocumene, labels might read '1,2,4-TMB', 'trimethylbenzene', or local language variants. Tracking chemical shipments or keeping up with international labeling standards means tracing these synonyms through customs databases and regulatory lists. Safety data frequently reference accepted limits from OSHA or local labor ministries, with strict guidelines on exposure and storage due to volatility and flammability risks. Unlike household paints or fuels, working with pseudocumene demands respect for the paperwork and for the risks involved.
Using It Wisely: Application Beyond the Obvious
My years in research taught me the difference between bench chemistry and real-world utility. Pseudocumene serves as a heavy-duty solvent, thinning durable coatings and reducing resin viscosity in ways that simpler solvents cannot replicate. Paint manufacturers and specialty ink companies value its ability to dissolve complex polymers, knowing that a little goes a long way. In analytical chemistry, it pops up as a calibration standard for detecting volatile organics. Less visibly, it plays a key role in neutrino detection experiments, where its high transparency and specific chemical reactivity allow for fine-tuned detectors. Each use case stretches the boundaries of what a basic hydrocarbon molecule can accomplish.
Digging Into Safety and Health Impacts
Anyone who's handled aromatic solvents learns quickly that casual exposure invites health issues. Pseudocumene volatilizes readily, so inhalation poses the biggest risk in workplaces with poor ventilation. Headaches, dizziness, or respiratory irritation are early warning signs; longer-term studies have raised flags about potential impacts on liver and kidney function with chronic exposure. Flammability is another red line—its vapors form explosive mixtures with air, demanding robust storage practices and proper grounding of containers during transfer. Personal protective equipment becomes habitual, not just for compliance but for basic self-preservation. Safer work environments depend not solely on rules, but on the habits workers absorb from experience and training.
Research, Development, and Rethinking Its Role
Innovation usually springs from necessity, and the same holds for molecules like pseudocumene. In the last couple of decades, research labs explored less-toxic solvents and green chemistry alternatives, partly in response to the environmental persistence of aromatic hydrocarbons. Yet, demand persists, particularly in high-performance applications where substitutes come up short. Laboratories have studied how pseudocumene’s photophysical properties enhance detection techniques, pushing the envelope in particle physics and spectroscopy. These applications capture how chemists adapt and repurpose even well-understood compounds to solve new challenges.
Seeking Answers: Toxicity and Environmental Fate
Concerns about the toxicology of pseudocumene emerge regularly in environmental monitoring. Studies confirmed moderate acute toxicity to mammals and aquatic species, with the molecule tending to bioaccumulate in fat tissues. Biodegradation in the environment follows a slow path, thanks to the stability of the aromatic ring and its side chains, raising questions over runoff and groundwater contamination. Regulatory pressure continues to mount, with tighter controls on industrial releases and growing expectation for companies to shift toward less persistent alternatives.
Future Prospects and Where to Go Next
Change ripples through the chemical industry, not just from legal mandates but from shifting market preferences. Pseudocumene’s fate seems tied to the broader question of how much risk society accepts in return for high-performance solvents and specialty chemicals. Research keeps chipping away at new, less-harmful formulations, exploring bio-based aromatics, and improving containment and recovery methods. The balance between utility and safety will likely move further toward human health and sustainability, pushing old workhorses like pseudocumene toward more specialized or controlled roles. For those on the ground, this shift means keeping skills current and learning to adapt as tools and materials evolve.
What is Pseudocumene used for?
A Glance at Pseudocumene’s Identity
Pseudocumene, also called 1,2,4-trimethylbenzene, shows up in more places than you might guess. Most days, I don’t handle tricky-sounding chemicals, but this one often pops up in industries around me. People with jobs in refineries or those working with specialty chemicals know it by its strong, sweet odor and its stubborn presence in many science labs.
Pseudocumene’s Place in Industry
Oil refineries pull out pseudocumene during the process of making gasoline. Refineries need to squeeze every drop from crude oil, and compounds like pseudocumene help boost the octane rating. A higher rating keeps engines from knocking, and it’s not just high-performance cars that benefit. Reliable transportation makes a difference to families, businesses, and the food on store shelves.
Another side to pseudocumene’s story comes from specialty chemical manufacturing. Synthetic resins, production of dyes, and paint thinners often rely on this compound. Chemists value pseudocumene because its ring-shaped structure opens up different paths for creating new products. In the paint world, for example, solvents need to do more than just thin things out. They need to dry at the right speed, not leave behind gunky residues, and stay affordable for all the local contractors trying to make a living.
Pseudocumene in Science and Research
Science teams searching for clues about the universe sometimes turn to pseudocumene. Large neutrino detectors, like Borexino in Italy, use it as a liquid scintillator. In simple terms, this means the chemical helps catch flashes of light released by elusive neutrinos, a kind of invisible particle that passes through just about everything. Without pseudocumene’s special glow, many experiments on cosmic mysteries would end before they even begin.
Safer ways to handle pseudocumene keep communities and researchers protected from risk. Leaks or spills can cause headaches and dizziness, especially in people who work long hours near strong-smelling solvents. Good ventilation, protective gear, and strong spill response plans mean workers head home healthy and ready for their own lives.
Pseudocumene and the Air We Breathe
Nobody likes to think about toxins in the air, but pseudocumene lands on lists of substances worth watching. As regulations get tougher, businesses can’t ignore their responsibilities. Control equipment like carbon filters traps fumes before they drift out of the stacks. Where I live, efforts to clean up industrial emissions trace back to cases like this: regulators push for lower release limits because it matters to neighbors, families, and the workers themselves.
Environmentally safer chemical management helps limit exposure, protects water supplies, and builds trust between companies and the local neighborhoods. It’s not just about fines or paperwork. Healthier air means fewer sick days for families and cleaner parks where kids play each afternoon.
Looking Down the Road: Smarter Uses and Substitutes
Next steps include more research into substitutes or new techniques that reduce the need for pseudocumene in fragile settings. Green chemistry attracts talent and drives investments—universities and businesses scan for ideas that trim hazardous waste and still deliver strong product performance.
Getting there means sharing information transparently and acting on feedback from everyone affected. I’ve seen conversations shift from behind closed doors to open meetings with clear reporting. Room for improvement always exists, but as more people pay attention to what goes into our air and products, the future looks a little safer and a lot more cooperative.
Is Pseudocumene hazardous to health?
Understanding Pseudocumene
Pseudocumene isn’t a chemical name that pops up in most homes, yet it finds its way into industries like petrochemicals and paint manufacturing. People who work in or close to chemical plants may recognize the strong, sweet odor long before learning the word itself. Many ask if it’s truly hazardous. Experience working near chemical storage facilities tells me: familiarity with an industrial smell rarely equals safety.
Evidence of Harm
Pseudocumene, or 1,2,4-Trimethylbenzene, can harm the body in ways that many don’t expect. Inhaled fumes from leaks or poor ventilation can irritate the nose, throat, and lungs. I remember a co-worker once complaining about coughing fits and dizziness after working near a tank where traces of pseudocumene leaked. We checked the Safety Data Sheet. It clearly spelled out the risks — headaches, nausea, skin rashes after exposure, and potentially more severe long-term effects if not managed properly. The National Institute for Occupational Safety and Health (NIOSH) cautions workers not to take these symptoms lightly.
Concern for Chronic Exposure
Acute effects like dizziness or eye irritation usually lead exposed people to leave the area fast. Chronic exposure often gets overlooked. Research in toxicology points to impacts on the liver, kidneys, and central nervous system if inhaled over long periods, even at lower concentrations. This isn’t theoretical; people who spent years in jobs with regular pseudocumene contact experienced these problems, as documented in occupational health reports.
Environmental Danger
Spills don’t stay put. Pseudocumene can seep into soil or run off into streams. Wildlife pays the price, as aquatic life doesn’t tolerate this chemical well. Data from the EPA notes its toxicity in fish and invertebrates. Those who grew up fishing in rural rivers see firsthand that invisible pollution deserves attention long before wildlife turns up dead or sick. Farmers have sometimes faced contaminated wells, discovering too late that improper chemical handling upstream placed families and animals at risk.
Making Workplaces Safer
Decent ventilation, leak detection systems, and up-to-date personal protective gear matter. Having spent time in both union and non-union worksites, I saw how places that invested in these measures suffered fewer accidents and health complaints. Employers should offer real education, not just a quick safety briefing. OSHA sets permissible exposure limits — but best results come from aiming for levels far below the legal ceiling. Simple steps, like maintaining seals and pumps, save headaches — literally.
The Need for Stronger Oversight
Government agencies and industry watchdogs kick in the most value through routine checks and fines for unsafe handling. I learned to trust a plant’s commitment to safety only after watching how often they conducted drills, cleaned up spills, and supplied replacement respirator filters. Communities near chemical plants need public reporting on leaks and drift, not just reassurances. Buying air quality monitors isn’t a luxury when your health is at stake.
Common Sense Steps
If you live near chemical operations or work with solvents, use odor as an early alarm. Insist on transparency, read public health notices, and push for strict standards. Simple respect for chemicals like pseudocumene goes much further than panic — especially when it's paired with protective gear and clear information. People deserve to know what’s in the air, and to act before a sore throat turns into something far worse.
What are the physical properties of Pseudocumene?
A Close Look at Pseudocumene
Pseudocumene, usually known in the lab as 1,2,4-trimethylbenzene, has followed me through years of science classes and do-it-yourself projects. This liquid finds its way into more conversations than one might expect, especially once you notice how it pops up in industrial plants and research labs. At room temperature, it shows up as a colorless liquid with a sharp, sweetish odor that you’ll recognize if you’ve ever been near fresh paint or solvents.
Liquid at Everyday Temperatures
Heat and cold both reveal quite a bit about its behavior. Pseudocumene has a melting point not far below freezing, landing around -43°C. So if you keep a sample in most home freezers, it will still pour. Boiling sets in at 169°C, which is hotter than water on the stovetop but not extreme for organic liquids. These numbers matter for anyone moving or using the material. Let’s say you are working in a chemical plant: spills can vaporize into the air fairly quickly above room temperature. Once in vapor form, it’s heavier than air, so it can collect in low spots, creating a real need to focus on safety, ventilation, and prompt cleanup practices.
Solubility Tells a Story
Pseudocumene does not like water. Add a few drops to a glass of water, and you’ll see oily globules float or sit at the top. Water solubility is practically zero. Yet mix it into organic solvents – ethanol, ether, acetone – and it dissolves without trouble. It reminded me of cleaning oil-based paintbrushes as a kid, watching mineral spirits dissolve sticky paint, leaving clear rinse water untouched. The chemical’s behavior means you can’t rely on water for cleanup, raising stakes for environmental safety. After spills, it can seep into soil or run off into waterways, sticking with other oily pollution.
Density and Appearance in Practice
Dipping a finger into the safety side of things, pseudocumene is lighter than water with a density of about 0.88 g/cm³. If you’re pouring it into a tank, you’ll see it float before it spreads into a rainbow sheen. This low density helps with containment and skimming in spill response. Precision counts here. In research settings, even small differences in mass and volume can throw off results. A lack of color helps in some quality control operations, though the strong odor alerts people long before any physical symptoms arrive.
Volatility and Workplace Considerations
Pseudocumene gives off enough vapor to create air quality problems in unventilated spaces. Workers in storage and transportation use gas detectors to stay ahead of risky concentrations. The flash point, at about 44°C, is well below what you’ll hit inside a parked car on a hot day, which brings real concerns around fire safety. In my own experience working with volatile organics, a whiff can sometimes stick to clothes and linger for hours. Employers and safety officers keep eyes open for leaks, maintain grounded containers, and train new workers early on best storage habits.
Solutions for Safer Handling
Personal protective equipment like gloves and goggles offers a first step, but engineering controls stand as the real backbone: fume hoods, proper storage, and ongoing education. Taking the time to double-check containers for cracks turns into a regular habit. Spill prevention planning is not just regulatory red tape—it stems from seeing how quickly a forgotten puddle can become a workplace hazard. Responsible disposal and ongoing monitoring around storage sites help reduce risks to soil and water supplies.
Responsible Use Matters
Pseudocumene’s physical quirks—the volatility, the slight oily feel, the way it floats—call for respect but not fear. Its properties inform everything from fire safety to environmental care. In both professional labs and industrial fields, a little experience goes a long way in making sure its benefits are realized without unnecessary harm.
How should Pseudocumene be stored and handled?
Understanding the Stakes
Pseudocumene, also known as 1,2,4-trimethylbenzene, shows up in labs and industry for a reason: it helps in making solvents, resins, and certain plasticizers. But this stuff doesn’t behave politely. Its vapors catch fire even with a small spark, and it can mess with your lungs and skin if things go sideways. Not everyone realizes how easily careless storage lets a hazard slip through the cracks. I’ve seen some companies lose valuable time and money by cleaning up leaks because plastic drums cracked or weren’t grounded. More importantly, missing even a single step can put lives on the line.
Safe Spaces for Dangerous Liquids
Pseudocumene calls for real respect. Metal drums with tight seals stand up better to chemical attack than plastic, which has a knack for wearing out before you expect. Every container needs a clear, no-nonsense label. Workers don’t always double-check, especially on long shifts, so a fresh, bold marking cuts confusion. If a drum sits in the wrong spot, such as next to heat or sunlight, pressure inside builds up and the odds of a disaster climb fast. Keep it in a cool, ventilated spot. One warehouse I managed swapped out a few flickering bulbs for proper explosion-proof lights and trimmed the risk from electrical shorts. Small details pay off.
Fire Hazards Can’t Be Ignored
No one forgets their first time seeing a fire jump from static on a barrel. Pseudocumene belongs nowhere near open flames or even a careless smoker. Flammable liquids demand grounding cables so static doesn’t sneak up and cause trouble. Inside warehouses, certified flammable storage lockers give a layer of protection if a spill happens. We ran drills twice a year and found people often underestimated how quickly the situation can spin out of control. Keeping fire extinguishers suited for chemical fires within arm’s reach isn’t just smart; it keeps your insurance guy comfortable, too.
Handling Matters—Protect Yourself
Gloves and goggles can feel awkward at first, but they’re non-negotiable. Pseudocumene doesn’t just hurt your eyes or skin—it can take your breath away before you realize it. I’ve heard of experienced workers suffering headaches after just a short exposure to the vapors, especially when ventilation corners were cut. A splash of the chemical on skin feels slick, but it burns later. A chemical-resistant apron pulls its weight, and no one has ever regretted putting on a proper mask or face shield. Spill kits aimed at hydrocarbons, not just water-based messes, should sit within reach and see real drills. During one staff meeting, walking through a spill clean-up routine made clear who understood the risk and who needed more training.
A Culture of Respect Beats Carelessness
Pseudocumene never rewards shortcuts. The most reliable safety strategy comes from building a culture where everyone looks out for mistakes. Pop quizzes, checklists, and walk-arounds pick up bad habits before they turn into emergencies. Regular training refreshes what’s at stake, and reporting every near-miss gives more data for improvement. Auditing storage areas every month exposes leaks and keeps ventilation clear. Someone always has to take responsibility for staying on top of things, and there’s no room for complacency in a warehouse dealing with flammables.
Better Policies—A Safer Outlook
Companies leading the way set the bar high not just with equipment, but with attitudes. Encouraging workers to speak up, providing up-to-date material safety sheets, and investing in modern detection systems cut risks and show employees their well-being matters. It pays off; fewer incidents, less downtime, and a team that knows their safety matters. Storing and handling pseudocumene isn’t just about rules on a wall—it’s about turning those words into habits that save lives.
What is the chemical formula of Pseudocumene?
Understanding Pseudocumene
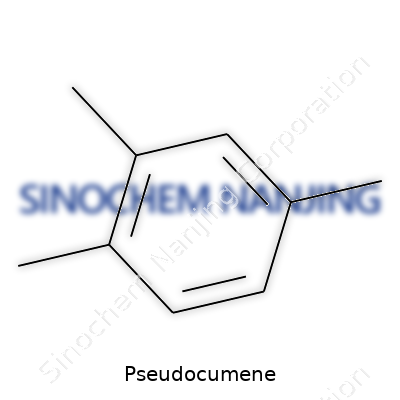
Pseudocumene carries the chemical formula C9H12. It comes with a fancier name, too—1,2,4-trimethylbenzene. That means you start off with a benzene ring and stack three methyl groups onto it, at positions 1, 2, and 4. This formula holds a lot of weight in both chemistry class and in big industries, like those that churn out products for fuels, solvents, and even tracer solutions for physics experiments.
Why C9H12 Carries Real-World Weight
My first brush with pseudocumene came in a college organic lab. One whiff is enough to tell you it’s not a toy—strong, sweet, and surprisingly stubborn when you try to wash your hands. Companies don’t just use it for the aroma, though. That structure of three methyl groups on a benzene ring makes it behave differently from plain benzene or even from its cousins, like mesitylene (which throws all three methyls next to each other).
Industries use pseudocumene for many reasons. Its chemical stability means it doesn’t break down easily under normal lab or industrial conditions. Scientists pump it into liquid scintillator detectors to track faint signals in dark matter and neutrino experiments. Oil refineries use it as a raw material to make other compounds, like trimellitic anhydride, which ends up in plasticizers for flexible PVC.
Risks Tied to C9H12
Pseudocumene’s formula isn’t just an answer on a test. Knowing what C9H12 means reveals real safety risks. It evaporates easily, so breathing in too much can cause headaches or dizziness. The Environmental Protection Agency (EPA) keeps an eye on it and has flagged it as a hazardous air pollutant. Prolonged exposure may affect organs like the liver and kidneys, as research from the National Institute for Occupational Safety and Health (NIOSH) points out.
Local fire departments treat tanks of it with respect. It forms vapors heavier than air, so it can settle in low spots—without good ventilation, you get a recipe for trouble. If it leaks or spills, it can also run into rivers and groundwater, sparking environmental alarms. Safety data sheets warn against letting it get anywhere near drinking water sources or soil.
How to Handle Pseudocumene Responsibly
Good handling starts with knowing the hazards, then taking real steps to limit exposure. Workers need gloves, goggles, and masks, and industrial plants invest in proper ventilation. Storage means metal drums locked tight, away from sparks or heating elements. Training helps, but attitude counts too—treating every drum like it might bite saves trouble later.
Dumping pseudocumene down the drain counts as both illegal and dangerous. Waste disposal teams use technologies like incinerators designed for hazardous organics. Regulators stay on watch, and plant managers update records and check storage tanks for leaks.
People sometimes forget that behind every chemical formula lives a host of decisions that ripple out to health, safety, and even climate. C9H12 may seem simple on paper, but safe, responsible use protects both workers and neighborhoods. It pays to do more than just memorize its formula—understanding what it leads to, both good and bad, can push science and industry in better directions.
| Names | |
| Preferred IUPAC name | 1,2,4-Trimethylbenzene |
| Other names |
1,2,4-Trimethylbenzene ps-cymene TMB |
| Pronunciation | /ˌsuːdoʊˈkjuːmiːn/ |
| Identifiers | |
| CAS Number | 95-63-6 |
| Beilstein Reference | 1361119 |
| ChEBI | CHEBI:17590 |
| ChEMBL | CHEMBL1409 |
| ChemSpider | 5466 |
| DrugBank | DB02244 |
| ECHA InfoCard | ECHA InfoCard: 100.003.605 |
| EC Number | 201-851-2 |
| Gmelin Reference | 7577 |
| KEGG | C06425 |
| MeSH | D018428 |
| PubChem CID | 7909 |
| RTECS number | XS5250000 |
| UNII | F5D430IPB2 |
| UN number | 1307 |
| Properties | |
| Chemical formula | C9H12 |
| Molar mass | 120.19 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic odor |
| Density | 0.876 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.7 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 6.97 |
| Magnetic susceptibility (χ) | -8.6e-6 |
| Refractive index (nD) | 1.505 |
| Viscosity | 0.66 mPa·s (20°C) |
| Dipole moment | 0.31 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −24.5 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | –3893.5 kJ/mol |
| Pharmacology | |
| ATC code | V04CX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H304, H315, H319, H335, H411 |
| Precautionary statements | P210, P243, P261, P264, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P337+P313, P370+P378, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 52 °C |
| Autoignition temperature | 536 °C (997 °F; 809 K) |
| Explosive limits | 1.2–6% |
| Lethal dose or concentration | LD50 oral rat 6,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 6.3 g/kg |
| NIOSH | US7845000 |
| PEL (Permissible) | 25 ppm |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Benzene Toluene Cumene Mesitylene Durene Hemimellitene Isodurene Prehnitene |

