Copper Selenite: History, Properties, Uses, and Prospects
Historical Development
Copper selenite started cropping up in the late 1800s, back when chemists first learned to isolate selenium from ores in mining regions. Copper smelters often stumbled on selenium-rich byproducts, setting the stage for new compounds as curiosity about selenium grew. Through much trial, error, and laboratory heat, researchers finally coaxed selenium to form stable compounds with copper, noticing the vivid colors and unique reactivity right away. Old academic journals mention copper selenite crystals as subjects of study for both mineralogists and early inorganic chemists, with attention paid to both their analytical chemistry and possible industrial uses as synthetic methods improved through the mid-twentieth century.
Product Overview
Copper selenite falls under the category of inorganic copper salts, typically appearing as blue-green powders or crystalline masses. Most of the time, the commercial product includes traces of water in the structure, which changes its weight and solubility. Its chemical formula comes in variants, but the most referenced form remains CuSeO3·2H2O. Production runs in academic-scale labs and niche manufacturing for glass, pigment, and occasional electronics applications, given the compound’s specialized role and health risks.
Physical & Chemical Properties
Pure copper selenite carries a blue-green tint, and the crystals dissolve sparingly in cold water but more readily in hot water, breaking down to copper and selenium ions. High sensitivity to acids means it releases selenium dioxide on contact with strong mineral acids, so chemical gloves and eye shields become standard equipment around it. The taste, not that anyone should ever attempt tasting, would be extremely bitter, and even a pinch carries risks because of selenium’s toxicity. About density: most measurements sit near 3.4 g/cm3, aligning with readings found in comprehensive chemical handbooks. In the open air, it shows good stability but breaks down under harsh reducing environments or sustained heating.
Technical Specifications & Labeling
Manufacturers ship copper selenite with specifications for selenium and copper content, noting impurity levels for heavy metals, sulfate, and chloride residues. Packaged in airtight plastic or amber glass, the labeling emphasizes the danger from both its copper and selenium content—every shipment marked with warning icons, hazard statements like H301 (toxic if swallowed), and GHS pictograms. Certificates of analysis specify batch purity and crystalline phase, as different applications, from research reagents to pigment production, demand tailored forms. Shelf-life rarely becomes an issue so long as humidity is kept in check.
Preparation Method
Making copper selenite demands a careful hand and well-ventilated setup. Chemists usually dissolve either selenious acid or sodium selenite in distilled water, then introduce copper sulfate—sometimes copper nitrate—to the mix. Gentle stirring, sometimes with a warming mantle, brings about a double displacement reaction, and blue-green precipitate forms in a few moments. After filtration, several washings with deionized water remove excess ions. Crystals often grow by slow evaporation at low temperature, and if higher purity is needed, repeating the precipitation process works well. Waste solutions, particularly selenium-containing filtrates, go to hazardous waste streams to avoid groundwater contamination.
Chemical Reactions & Modifications
Copper selenite brings one big feature: easy chemical modification and redox activity. Exposed to reducing agents, such as ascorbic acid or sulfur dioxide, it breaks down to elemental selenium or copper selenide. Reaction with hot, concentrated acids liberates selenium dioxide vapors. Mixing with alkali leads to complex selenate and selenite formation, changing both color and solubility. Its structure bends under some hydrothermal conditions, leading to different hydrate forms, which gives synthetic chemists flexibility. Some researchers swap copper for nickel or cobalt, adjusting color and magnetic traits. In pigment chemistry, small tweaks to the recipe govern the brightness and shade of copper selenite-based colors.
Synonyms & Product Names
In chemical catalogs and safety sheets, copper selenite runs under several names: cupric selenite, copper(II) selenite, and even selenious acid, copper(2+) salt. Older European texts occasionally call it “selenious acid, copper salt,” while modern suppliers list it simply as copper selenite. Synonyms pop up in different research papers, so anyone ordering or studying this product should cross-check registry numbers—CAS 10102-18-8 stands as the most widely accepted tag in databases and on shipping manifests.
Safety & Operational Standards
Everyone who works with copper selenite faces strict requirements because both copper and selenium bring genuine health hazards. Direct skin contact runs the risk of irritation, and just a couple of milligrams inhaled or swallowed can cause life-threatening selenium poisoning, with symptoms from stomach pain to nervous system damage. Laboratory ventilation matters more than most routine work, and chemical splash goggles stay on until every step is wrapped up. OSHA, NIOSH, and EU regulations list copper selenite among substances needing secondary containment, marked storage, and rigorous record-keeping for both educational and industrial use. Training on prompt cleanup and waste disposal sits as a part of every new employee’s safety checklist.
Application Area
Industrial uses for copper selenite stay limited, but where it goes, it matters. Glassmakers use it for coloring specialty glasses in deep blue or green hues, especially where long-term UV stability matters. Some pigment producers blend copper selenite for unique shades in glass enamels or artist supplies, although this niche has shrunk due to toxicity concerns. In electronics, chemists experiment with copper selenite-based thin films for semiconductors and photocatalysts. Academic labs lean on it for reference studies in environmental chemistry and as a precursor to copper selenide for photovoltaic materials or sensors. Agriculture and medicine steer clear because of the toxicity; the major action remains in research and art.
Research & Development
Modern R&D with copper selenite focuses on two tracks: deeper chemistry and advanced materials. Crystallographers map its lattice to spot subtle changes under pressure or chemical doping. Materials scientists look for ways to build nanoscale copper selenite films for solar cells, oxidation catalysts, and gas sensors—mainly inspired by the ease of synthesis and low temperature requirements compared to silicon-based materials. Ongoing research tries to pinpoint substitutes for more practical, less hazardous copper and selenium compounds in glass and pigment manufacturing to sidestep regulatory headaches.
Toxicity Research
Toxicology communities keep copper selenite on close watch. Animal exposure studies report severe gastrointestinal and neurological symptoms after only brief contact, which matches older human case reports from workers handling selenium compounds without protection. Chronic low-level exposure links to liver and kidney trouble. Regulatory bodies cite these risks while updating limit values in workplace air and water. In the environment, accidental releases become a problem due to the high bioavailability of selenium to plants and aquatic life—hence strict disposal protocols in all labs and factories. Work continues in trying to spot reliable biomarkers for copper selenite exposure, so both short- and long-term health effects can be tracked more effectively.
Future Prospects
Use cases for copper selenite carry a heavy caveat: the world wants less toxic materials, but in narrow spaces this old-school compound refuses to go away. In advanced electronics, researchers hunt for ways to harness its semiconducting potential without dragging in high toxicity. Pigment chemistry may circle back with stricter encapsulation or safer co-precipitates. For academic chemistry, copper selenite stays relevant as a teaching tool and redox standard. Environmental impact rules tighten year by year, pressing manufacturers and labs to reinvent safety protocols and limit release. As science pushes for greener chemistry, the challenge lies in balancing unique properties with a growing commitment to human health and ecological safety.
What are the main uses of Copper Selenite?
Understanding Copper Selenite’s Role
Copper selenite doesn’t usually land in the spotlight, but its uses underpin some vital industrial processes. Coming from both copper and selenium, it brings unique chemical properties you won’t find in every corner of the periodic table. Its real strengths show up in specialized sectors that rely on its blend of conductivity and reactivity.
Electronics: Not Just a Background Ingredient
Modern electronics rely on more than just copper wire and shiny chips. Copper selenite carries properties that serve manufacturers of semiconductors and photovoltaic cells. Its electrical characteristics, especially when used in thin films, let engineers tweak conductivity for better performance. The material doesn’t just move electricity; it helps control it. In solar power, copper selenite finds use as a precursor for other selenium-based layers in solar cells and thin-film transistors. The demand for efficient, stable energy sources pushes researchers to use materials like copper selenite to reach higher conversion efficiencies.
Chemical Synthesis and Catalysis
Laboratories and chemical plants lean on copper selenite for its catalytic talent. Chemists often use it as a trace additive in creating certain organic compounds. Its presence can make or break a reaction, especially for pharmaceuticals and nylon production. If a catalyst increases yield or reduces hazardous byproducts, it improves safety and protects the bottom line. As someone who has spent time in lab environments, there’s appreciation for the materials that quietly help complex reactions happen without drawing too much attention. Copper selenite can often be that unsung helper.
Glass and Pigments: Color With a Purpose
Artisans and industry both use metal-based pigments to produce colors that last. Adding copper selenite to glass gives products a rich red or reddish-brown hue. Pottery glazes and specialty glass windows depend on this compound for color stability during high-heat processing. I’ve seen stained glass crafters gravitate toward these pigments for their depth and resilience, which holds up in windows exposed to both city smog and shifting weather patterns.
Animal Nutrition and Fortification
Copper and selenium count as essential trace nutrients for animals. Some livestock feed producers use copper selenite—under tight regulation—to supplement feed, especially where soil lacks selenium. Farms in certain regions use it to help prevent deficiencies that can lead to health issues in cattle, sheep, or poultry. Too much of either element can be toxic, so precise dosing matters. But balanced right, it supports healthy growth and the immune system of farm animals.
Weighing Its Footprint
Handling and disposal of copper selenite must be done with care due to its environmental risks. It’s toxic in large doses and must stay out of water systems. The long-term answer lies in tighter safeguards during production, robust recycling, and further study into alternatives that carry fewer safety concerns. Investing in greener chemistry reduces worries for both factory workers and communities downstream.
Looking Forward
Copper selenite won’t headline the news, but its presence in electronics, glass, catalysis, and agriculture keeps key processes running smoother. The challenge is to use it wisely, tighten safety, and stay open to advances that let us do more with less environmental risk.
Is Copper Selenite safe to handle?
Understanding Copper Selenite
Copper selenite, with its bright blue-green hue, often grabs attention in the lab. A lot of people who work in research or industry may run into this compound, but not many outside those circles know much about its real risks. Working in labs for years and handling all sorts of metal salts shaped my view on chemical safety: don’t trust a pretty powder until you know what it really does.
Known Hazards
Copper selenite combines two things that raise eyebrows for health professionals—copper and selenium. Both play important roles in biology at trace levels, but the story shifts quickly at higher doses. Short exposures to selenite compounds can cause irritation to skin, eyes, and especially the respiratory tract. Chronic exposure to selenium dust has been linked to fatigue, joint pain, brittle hair, and stomach upset. In some cases, selenium toxicity even causes neurological symptoms.
Copper isn’t much safer on its own. Excessive contact with copper salts can lead to irritation, headaches, nausea, and in rare cases, long-term organ effects. You won’t see many people using copper selenite recklessly for good reason. The two elements together spell out a safety recipe that’s hard to ignore.
Handling in the Real World
I’ve seen newcomers brush off warnings about copper selenite, figuring it can’t be worse than other lab powders. That attitude quickly changes after the first spill or a hot summer day in a poorly ventilated workspace. Selenite dust has a way of sneaking into the air, especially during mixing or weighing. Respirators aren’t just for decoration; they make a difference between a forgettable work day and a weeks-long cough or sore throat.
Proper use of gloves and lab coats goes a long way. I learned early on that thin latex gloves can let through some copper salts with enough contact time, so nitrile or thicker options feel safer. A simple rule helps: treat anything with selenium as you’d treat something labeled toxic.
Hard Facts about Exposure
The International Chemical Safety Cards (ICSC) and Material Safety Data Sheets (MSDS) don’t mince words about copper selenite. Both acute and chronic exposure risks include serious lung and digestive symptoms. The permissible exposure limits for selenium compounds hover near the lowest mark set for workplace chemicals. That tells a story: specialists trust the compound only behind barriers or inside fume hoods.
Protecting Yourself and Others
Having safety data in hand is only part of the puzzle. Training counts. New staff members at my old lab used to run through mock spills so that real mistakes didn’t get dangerous. Good housekeeping – wiping benches with damp cloths, never dry sweeping – cut down airborne dust. Closed labeling systems meant everyone knew exactly what sat in each bottle.
Local regulations often require special disposal steps for selenium-containing materials because of the risk to water sources and living systems. Trying to skip procedures or cut corners on waste management trades small savings for big problems down the line.
The Bottom Line on Safety
Copper selenite isn’t something to handle casually. Anyone using it in research, production, or teaching owes it to themselves and their coworkers to treat it with respect. Gloves, masks, fume hoods, clear labeling, and steady training routines aren’t extra steps—they’re the basics that keep people healthy and labs in business. As someone who’s spent years surrounded by chemical jars, I’ve learned that one risky shortcut can undo months of careful work. You can explore, discover, and experiment all you want. Just don’t gamble with compounds that clearly demand caution.
What is the chemical formula of Copper Selenite?
What the Formula Means: CuSeO3
Copper selenite is written up in chemistry books as CuSeO3. It’s easy to gloss over a chemical formula like this as just a tangle of letters and numbers, but sitting behind it sits real science with real impact. Copper, selenium, and oxygen come together in these proportions to form a salt that doesn’t often get its day in the sun. Most folks know copper from wiring and coins, selenium from nutrition labels and supplement bottles, and oxygen from—well, breathing. Yet, bring these together, and you get something that can spark debate in toxicology rooms, environmental research meetings, and even discussions about what happens deep in the Earth’s crust.
Chemical Makeup and Behavior
Each unit of this compound contains one copper atom, one selenium atom, and three oxygen atoms. That’s what the formula spells out. Copper appears in the +2 oxidation state, and selenium shows up as selenite, carrying a -2 charge. These charges play a big role in how copper selenite interacts once it lands in soil, rock, or water. From an old high school lab to mining operations, this basic knowledge offers a starting point for bigger questions: where does it go, and what happens to ecosystems that have to live alongside it?
Why It Matters For Health and the Environment
People tend to forget about the rare stuff like copper selenite until it lands on their radar as a problem. In my own work on water quality, chemical forms that include selenium are often a source of trouble. Too much exposure, and animals or people can get sick; too little, and health suffers the other way. This balance is especially delicate where industrial operations or mining touch the water table.In fact, the World Health Organization and the US Environmental Protection Agency both watch selenium compounds closely. Studies by these organizations have flagged potential risks, showing what happens when substances like copper selenite become concentrated in a waterway or food chain. It’s not just another line on a periodic table—missteps with this stuff create real harm.
Addressing the Risks: Real Solutions
The first step in wrestling with the issue is understanding exactly what copper selenite does once it’s out in the wild. Research teams at universities and in government agencies dig into how this compound breaks down and moves through soil and water. High-performance liquid chromatography and mass spectrometry let scientists spot copper selenite even in complex mixtures, so cleanup isn’t done blindly.From a policy and regulatory standpoint, clarity matters. There needs to be a clear line about how much of any selenium compound is acceptable in drinking water or seafood. It helps when lawmakers work with geochemists and toxicologists rather than just writing rules from the capital. That’s not just best practice—it’s how you get standards that protect real people.Industry can play a part too. The mining and metalworking sectors sometimes overlook small-scale leaks or dumping that wind up building major environmental problems later. Keeping a strict eye on chemical handling, using storage that keeps run-off contained, and recycling where possible—these aren’t only about compliance; they’re good for business and community relations too.
The Bigger Picture
People like me, who straddle the world of lab research and public science outreach, try to turn complicated stories like copper selenite into something neighbors at a community meeting can understand. Too many folks only hear about these compounds after something goes wrong. By pulling copper selenite out of the shadows a bit more—and not just as a formula—everyone stands a better shot at making choices that don’t cost us later.
How should Copper Selenite be stored?
Understanding the Risks Behind Copper Selenite
Copper selenite might not show up in most kitchens or backyard sheds, but researchers and industry workers know this compound means serious business. Its deep roots in semiconductor tech and science labs prompt conversations about proper handling. This substance carries both chemical and toxicological hazards, with copper and selenium elements both flagged by regulatory agencies. I remember the first time I opened a new jar of copper selenite in our campus lab—training emphasized one thing: treat every granule with respect. That lesson sticks, and for good reason.
Why Air and Light Aren’t Friendly Neighbors
Copper selenite never really enjoys contact with air or sunlight. Exposed to humidity, the powder tends to clump and degrade. Sunlight kicks off reactions that break down the compound and can worsen its hazardous nature. I’ve seen bottles close with improper seals balloon out or cake up at the neck, turning a simple weighing task into a potential accident. The optimal spot favors a cool, dry, shaded cabinet, away from high-traffic work surfaces.
Best Containers: Not All Glass Is Created Equal
Plastic containers feel tempting due to price and weight, yet glass, specifically amber glass, stands out for storing copper selenite. Amber glass stops UV light, preventing chemical breakdown. Tightly sealed lids with chemical-resistant liners matter just as much—ordinary plastic lids can crack, warp, or react over weeks or months. I once witnessed a seal failure contaminate an entire batch of samples, forcing a costly cleanup. That mistake proved switching to PTFE-lined screw caps isn’t overkill—it’s insurance.
Controlling Contaminants and Cross-Contact
In labs, the risk of cross-contaminating other chemicals can’t be ignored. Powdered copper selenite loves to stick to tools, gloves, and pours easily into the air as dust. Labeling containers sharply, keeping them apart from acids and reducing agents, and dedicating utensils to this compound only takes minutes but saves hours. Simple double-bagging containers inside thick polyethylene bags gives an added layer against leaks and spills.
Health and Safety Precautions: An Ounce of Prevention
Both copper and selenium can cause acute and chronic health issues—from respiratory irritation to more serious organ impact on prolonged exposure. Long sleeves, gloves, and splash-proof goggles sit front and center at every station where copper selenite comes out. Local exhaust ventilation reduces inhalation risks. The dangers cannot be divorced from its chemical benefits, but a sound storage strategy shrinks those risks to manageable levels.
Storage Location: Away from Heat and Food
Emergency room doctors see more accidents related to chemical storage mistakes than most of us guess. Keeping copper selenite on the lowest shelf stops falls and breakage. Storing away from direct heat sources blocks accidental temperature spikes, which is key for long-term stability. Food, drink, and personal effects should stay far from any area where copper selenite is present. Strict separation keeps tragedy at bay.
Documenting and Auditing
Regularly inspecting both inventory and storage areas makes sense. Leaks, expiry, and accidents hide in plain sight if no one tracks them. Digital logs help but even a paper trail works in a pinch. Documenting every movement, checking expiry dates, and running periodic cleanup rounds support the best possible storage results.
Where can I buy Copper Selenite?
Looking for a Rare Compound
Copper selenite doesn’t show up on the shelves of the local hardware store or pop out as a top result in most e-commerce searches. I understand the curiosity here. The words themselves might even stir up thoughts of rare minerals or some exclusive chemical that could unlock new possibilities in research or industry. Only a handful of companies worldwide keep it in stock, mostly for specialized applications like laboratory work, advanced ceramics, or potentially as a precursor for creating other selenium-based materials.
Verifying the Need and Safety
Interest in copper selenite makes sense for those who work in materials science, chemical synthesis, or specific energy projects. Before seeking out a seller, it’s smart to pin down what purpose it serves. For most uses, copper selenite brings both benefits and risks. Selenium compounds deserve respect. Mishandling can harm both the user and the people around. This isn’t a simple salt you pour from a bag in the kitchen. Scientists always follow strict protocols, often guided by national safety agencies, when handling it.
Sourcing: Not Your Usual Shopping Trip
Few outlets in the United States or Europe maintain a list price for copper selenite. I’ve found that companies like Alfa Aesar and Sigma-Aldrich sometimes carry it, but not in bulk volumes. They focus on research grades and require buyers to verify intent and sometimes credentials. The same goes for international shops like ChemSpider and LGC Standards. If thinking of importing from China or other countries with looser distribution, get ready to face customs regulations and provide documentation.
Industrial suppliers might offer copper selenite under a catalog designed for other selenium products. Don’t count on big B2B chemical distributors such as Brenntag; they keep it off the shelf unless a special order comes through, and they stick to business-to-business sales with a firm paper trail. Many online retailers advertising chemicals are actually trading platforms that connect buyers with small manufacturers, not warehouse-stocked stores. Purchase only from suppliers that honor reputable documentation and SDS files—those with strong feedback and a digital footprint that stretches back several years.
Ethics and Legality Come First
It’s tempting to look for workarounds when a chemical seems rare. Some platforms might quietly offer copper selenite with vague claims or questionable origins. These products may not meet safety standards or purity requirements—sometimes they’re even mislabelled. For something with the potential toxicity of a selenium compound, ignoring these details can spell disaster. Responsible users must follow every legal requirement and best practice when buying and using such a substance.
I’ve seen people in research teams get turned away by suppliers after failing to prove their intended use. That hurts, but it’s necessary. Buying copper selenite through legitimate scientific channels doesn’t just keep buyers out of trouble—it upholds the standards that protect both the public and the scientific community. Every real supplier asks for documentation, even if it feels like a hassle. This step prevents misuse and maintains trust in the supply chain.
Seeking Safer, Official Channels
Those who genuinely need copper selenite will find the process slow at times. Reach out to well-known laboratory supply companies, get guidance from an affiliated scientist or industry expert, and gather the necessary paperwork. This careful, above-the-board approach secures what’s needed and protects both the buyer and the larger community.
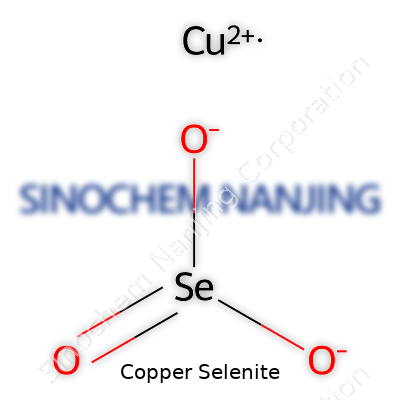
| Names | |
| Preferred IUPAC name | copper(2+) selenite |
| Other names |
Copper(II) selenite Cupric selenite |
| Pronunciation | /ˈkɒpə ˈsɛlɪnaɪt/ |
| Identifiers | |
| CAS Number | 1315-09-9 |
| Beilstein Reference | 352412 |
| ChEBI | CHEBI:86457 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 23730935 |
| DrugBank | DB11129 |
| ECHA InfoCard | ECHA InfoCard: 100.029.267 |
| EC Number | 231-006-5 |
| Gmelin Reference | 35122 |
| KEGG | C11160 |
| MeSH | D003784 |
| PubChem CID | 25157 |
| RTECS number | VS7875000 |
| UNII | 9A5R42U2L4 |
| UN number | UN3288 |
| Properties | |
| Chemical formula | CuSeO3 |
| Molar mass | 221.01 g/mol |
| Appearance | Blue crystals. |
| Odor | Odorless |
| Density | 4.39 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -2.32 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.7 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | −23.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.89 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 124.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -337.3 kJ/mol |
| Pharmacology | |
| ATC code | V03AX02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes damage to organs; may cause cancer. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-0-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 7 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 7 mg/kg |
| NIOSH | Not Listed |
| PEL (Permissible) | 0.2 mg/m³ |
| REL (Recommended) | Copper Selenite does not have a Recommended Exposure Limit (REL) established. |
| IDLH (Immediate danger) | IDLH: 1 mg Se/m³ |
| Related compounds | |
| Related compounds |
Copper(II) sulfate Copper(II) nitrate Copper(II) carbonate Copper(II) oxide Selenium dioxide Sodium selenite Copper(I) selenide |

