Copper Nitrate: More Than a Blue Crystal
Historical Development
Copper nitrate did not always get the spotlight in chemistry. Roots trace back to alchemists smelting copper with strong acids, hunting for new colors and metal forms. They did not have the luxury of pure nitric acid. They boiled saltpeter and strong acids with scrap metal, picking up blue crystals from the ash, not always recognizing the real value behind their colored scraps. By the 1800s, chemists advanced isolation and purification through better lab glassware and acid synthesis. The spread of industrial-scale nitric acid made copper nitrate as a laboratory staple. The physical blue crystals became a signal for countless reactions, a participation badge for the then wild world of inorganic chemistry.
Product Overview
Copper nitrate typically appears as a bright blue, crystalline solid. It is sold as either trihydrate or hexahydrate, the latter showing off with almost neon fluorescence under strong light. Lab supply catalogs list it for those looking to make copper salts or act as a catalyst in organic experiments. Both the technical and analytical grades do not stray much on appearance but may differ in trace impurity levels. Synthetic chemists often count on it for work-up steps, volcano “blue flame” demonstrations, or when they search for a reliable oxidizer.
Physical & Chemical Properties
These blue crystals give off a water-of-hydration scent and undergo slow efflorescence when left in open air. Hexahydrate copper nitrate sports a melting point near 114°C and slowly dries to the trihydrate at typical room humidity. It dissolves smoothly in water, giving a deep blue solution, and the salt releases noxious nitrogen dioxide if heated in open beakers. In solution, copper stays at a +2 oxidation state, making it a friendly Lewis acid for various chelation reactions. Thanks to its solubility, copper nitrate frequently acts as a source for copper ions in quantitative titrations, not just color displays.
Technical Specifications & Labeling
Chemical supply containers always print the formula — Cu(NO3)2·3H2O or ·6H2O — along with batch numbers, possible purity, and recommended storage conditions. Labels include GHS pictograms for oxidizer and acute toxicity. The technical data sheet (TDS) highlights solubility in water (125 g/100 mL, 20°C), density, and decomposition warnings for safety. Sales and shipping follow UN designation guidelines (UN 3077), which matters for those tracking hazardous substances through customs. I always glance at the moisture percent, giving clues about whether the compound will weigh out exactly or need correction for analysis-grade procedures.
Preparation Method
Making copper nitrate at scale follows a simple direct acid–metal approach. Scrap copper dissolves in hot, concentrated nitric acid, evolving toxic brown gasses and giving a blue solution. Evaporation and cooling yields the familiar salt, which is then filtered and dried. The scale-up from benchtop to industrial operation brings challenges with gas scrubbing and effluent control, especially since nitric oxide and dioxide emissions remain under heavy regulation. Some producers now source copper nitrate from recycled circuit boards or copper wire, using closed-loop acid recovery to cut down both cost and waste output.
Chemical Reactions & Modifications
Copper nitrate lends itself to a range of redox and precipitation chemistry. Adding sodium hydroxide or carbonate drops turquoise copper(II) hydroxide or basic copper carbonate out of solution. Reacting it in alcohol or with glycerol gets more exotic copper complexes, useful for catalysis or pigment production. In organic synthesis, I have used copper nitrate as an oxidant for converting primary alcohols into aldehydes, generating nitrogen oxides in situ. This reactivity stems from the nitrate group acting as a mild but versatile oxidizer, and the ease with which it swaps anions in double decomposition reactions — great for teaching fundamental inorganic patterns.
Synonyms & Product Names
Most chemical literature refers to copper nitrate as cupric nitrate, copper(II) nitrate, or simply “blue stone” among older texts. Hexahydrate and trihydrate forms must be named clearly, though most labs mean the hexahydrate unless otherwise stated. CAS number 10031-43-3 commonly appears on regulatory paperwork. For pigment uses, historic documents label the same salt as blue vitriol, but this usage now generally refers to copper(II) sulfate thanks to overlapping trade industries. In a lab, it pays to check both product code and chemistry; confusion here has spoiled more than one experiment.
Safety & Operational Standards
Copper nitrate comes with both chemical- and environmental-risk baggage. Nitrate’s ready oxidation means the salt accelerates combustion of organic or flammable materials, so lab shelves suggest segregation away from papers and reducing agents. Copper ions themselves carry acute toxicity for aquatic species and moderate risk for humans if ingested or mishandled; gloves and goggles are not just recommendations—they are the rule. Federal and state standards cover waste disposal, prohibiting travel down the drain. At proper scales, any spilled material must be collected and disposed through licensed hazardous waste handlers, and fume hoods control release of nitrogen oxides during use. I have seen older labs upgrade venting and storage as rules catch up to realizations about chronic exposure.
Application Area
Confined mostly to laboratory and industrial settings, copper nitrate acts as a precursor for other copper compounds, a catalyst, and an oxidizing agent. Electronics manufacturers use it for metal etching and finishing copper-coated surfaces. In woodworking, this blue salt assists in producing green tints on maple or beech, with careful sealing due to potential leaching. Pyrotechnicians blend it for blue fireworks, since copper compounds give a distinctive color among metal salts. Research groups deploy copper nitrate for synthesizing metal-organic frameworks (MOFs)—next-generation materials for gas separation or catalytic breakdown. Academic studies employ the salt for teaching, from the classic blue flame test to understanding ionic precipitation and solubility.
Research & Development
Chemists continually explore ways to use copper nitrate beyond classic roles. Researchers track its behavior in green chemistry, where it acts as a mild catalyst that leaves fewer dangerous byproducts than chromium or manganese oxidizers. In nanotechnology, teams use copper nitrate to synthesize copper oxide nanoparticles for sensors and antimicrobial surfaces. Environmental scientists examine copper nitrate’s breakdown in soils and watercourses, learning how it migrates through ecosystems. Interest also spikes in solar cell development, since certain copper nitrate-derived films show promise for enhancing light absorption or charge transport, especially when coupled with conductive polymers.
Toxicity Research
Both animal and cell-culture testing say copper nitrate poses risks through ingestion, inhalation, or skin exposure. Copper ions disrupt enzyme function, leading to gastrointestinal, liver, or kidney distress above certain thresholds. Regulatory agencies, like the US EPA, track maximum allowable concentrations especially in drinking water and monitor soil buildup near industrial sites. Chronic exposure, even at low levels, affects fish, invertebrates, and the microbial makeup of soils. Ongoing studies aim to uncover the compound’s long-term effects on human cells, immune response, and wildlife genes, guiding updated handling procedures and stricter factory effluent limits.
Future Prospects
Producers increasingly shift to greener synthesis and recycling, which may cut down both costs and hazards. Labs and industries seek cleaner substitutes for hazardous oxidizers, giving copper nitrate a push into more sustainable organic reactions. As electronics continue to miniaturize, copper nitrate finds new value for printed circuit repair and the development of conductive inks. The surging field of MOF research relies on copper salts for novel architecture, unlocking cheaper or more robust gas absorbers, battery materials, and slow-release fertilizers. The safety record depends heavily on handling discipline, but engineering and policy innovations promise to ease environmental footprints and expand safe use.
What is Copper Nitrate used for?
The Versatility of a Simple Chemical
People used to think of chemistry as something distant, tucked away in labs or hidden inside thick textbooks. Copper nitrate reminds me that these building blocks touch real life in countless ways, far from science classrooms. Many folks see a bottle of blue crystals and think “science experiment.” In reality, this chemical shapes a surprising number of industries and daily moments.
Color and Creativity in Industry
The first job that copper nitrate takes on involves coloring glass and ceramics. Glassmakers have long looked for ways to bring color to their pieces, whether it’s a stained-glass window or a simple vase. Copper nitrate helps create deep blues and greens in glass, and it shines in ceramic glazes. The results stand out in churches, homes, museums—wherever glass art appears. Paint makers also lean on copper nitrate for pigments that won’t easily fade.
Chemical Reactions for Practical Solutions
Copper nitrate’s real strength comes out when businesses need specific chemical reactions. In metallurgy, it helps purify copper and acts as an oxidizer during metal refining. Professionals working with metals need chemicals that deliver every time, and copper nitrate brings consistency. In the lab, it often serves as a source of copper ions during experiments—an essential ingredient for educators, researchers, and even students learning the basics of chemistry.
Environmental and Agricultural Uses
Copper compounds show up in agriculture surprisingly often. Farmers and researchers use copper nitrate solutions to treat seeds, boost soil, and control fungus. Every positive-growing season means more reliable harvests and less waste. Even so, these uses call for careful supervision. Copper can build up in soil, which can hurt helpful microbes or sneak into groundwater. Regular testing, smart application techniques, and being mindful of runoff help keep things safe.
Healthcare and Research Applications
In medicine, copper nitrate’s role feels more specialized. Scientists use it to create other copper-based drugs and catalysts during research into new treatments. Pharmacists don’t hand out copper nitrate to patients due to its toxicity, but spin-off discoveries from labs can end up saving lives. Researchers rely on copper nitrate to develop diagnostic tools, new materials, and even battery technologies. Its value stretches beyond a simple blue salt on a lab shelf.
Safety Matters and Transparency
Working with copper nitrate isn’t risk-free. Prolonged contact can irritate skin, eyes, or lungs. Swallowing it can cause severe harm. Companies train staff to handle it with gloves, goggles, and proper ventilation. For educators like myself, showing students the safety sheets and talking through the risks helps build respect for chemicals in general. Accidents drop when people know what to expect and follow smarter habits.
Responsible Use for the Future
Copper nitrate brings a lot to the table, from art to agriculture, science to manufacturing. Every benefit comes with responsibility. Improving storage, properly labeling containers, and disposing of waste through certified centers keep neighborhoods and environments safe. I’ve seen how a little planning goes a long way in classrooms and commercial labs alike.
People often take chemicals like copper nitrate for granted, but their uses cross paths with us more often than we realize. By understanding where and why copper nitrate fits in, we make choices that support both innovation and well-being.
Is Copper Nitrate hazardous to health?
Copper Nitrate: What People Should Know
Copper nitrate isn’t something you run into every day. Most folks probably remember copper pipes at home or see the green Statue of Liberty, both touched by oxidized copper. Copper nitrate, though, is a blue-green chemical that's mostly handled in labs or specialized factories. Based on my research and experience around chemical safety, the risks tied to copper nitrate often get overlooked by those who don’t deal with chemicals daily.
The Risks of Exposure
Copper nitrate, like a lot of laboratory salts, should not be brushed off as just another harmless powder. Its toxicity lies in both the copper and the nitrate. Skin or eye contact can produce burns, redness, and pain. Breathing the dust or fumes irritates the throat and lungs. Swallowing a small amount can trigger stomach cramps, vomiting, and diarrhea. In severe cases, it can disrupt key organs like the kidneys or liver. Chronic or repeated exposure may even lead to copper buildup, damaging nerves or brain tissue. It’s not rare to see cases where poor storage or broken safety routines send people to emergency rooms—problems that could easily have been avoided with some respect for the material.
Real-World Consequences
I’ve worked with students who handle all sorts of chemicals while learning basic experimental methods. Sometimes, I see folks grab copper nitrate with bare hands, thinking gloves slow them down. In a few cases, those small lapses have led to burns or inhalation incidents. That’s often a wake-up call: things can go wrong even during everyday activities. Hospitals have documented cases of accidental poisonings tied to this chemical, sometimes leading to severe complications or prolonged recovery.
Supporting Facts
Regulatory agencies like the US Occupational Safety and Health Administration warn about the hazards. They’ve set exposure limits for copper compounds in workplace air. The Centers for Disease Control points to animal studies showing nerve and muscle problems after copper nitrate gets into the bloodstream. These aren’t just theoretical problems; industrial spills and improper disposal have sometimes contaminated water or soil in certain locations, affecting both workers and communities nearby.
What Safer Practices Look Like
Respecting copper nitrate starts with solid routines. I teach that gloves and goggles aren’t optional—they're essential, even for short experiments. Good ventilation keeps dust out of the air. Labels show what’s actually inside each bottle, not just a shorthand label that only old-timers recognize. People working with copper nitrate get regular safety reminders, and everyone knows the emergency procedures by heart. That approach lets us avoid stories about accidents that should never have happened.
Reducing Risks for Everyone
Most households never need to worry about copper nitrate. For labs and industry, strong safety culture keeps people safe. Spills get cleaned up right away, and waste receives safe handling. Schools put strong safety policies in place, reinforcing respect for all chemicals, not just the ones with scary labels. Open communication about possible health hazards beats keeping things quiet or sweeping them under the rug.
Looking Ahead
People involved with copper nitrate must understand that small lapses can have big consequences. Care, education, and clear rules stand between routine work and unexpected emergencies. Just as we don't ignore the sharp edge on a kitchen knife, it makes sense to handle this chemical with focus and respect—protecting not only ourselves but also those around us.
How should Copper Nitrate be stored?
Why Safe Storage Matters
Anyone who’s spent time around a chemical supply room or worked in a lab knows the mess a simple mistake can cause, especially with compounds like copper nitrate. Blue crystals might not look dangerous sitting in a jar, but once moisture seeps in, or the wrong cap is used, you’re opening the door to leaks, spills, and even chemical reactions that nobody wants to deal with. Years back, I learned the hard way that even small storage errors can lead to big headaches—missing labels, corroded lids, or powder clumping together because someone left the jar unsealed.
Chemical Risks and Practicalities
Copper nitrate draws water out of the air. That means in a humid spot, it dissolves into a sticky mess, sometimes dripping out of the container and leaving behind blue-green stains that are maddening to clean. The compound’s oxidizing properties also raise the stakes: it can accelerate fires if it comes into contact with flammable materials. The risk grows in places where people don’t think twice about keeping lab chemicals beside paper boxes. So step one—move those containers somewhere cool, dry, and far from anything you’d throw in a bonfire.
Best Storage Choices
In every lab I’ve worked in, the safest bet has been clear labeling and rigid separation. Use strong glass or polyethylene bottles fitted with tight, chemical-resistant lids. Skip metal containers entirely, since copper nitrate’s corrosive nature pushes reactions you can’t always see coming. Years ago, a colleague stashed copper nitrate in a cheap metal tin to “save space”; a week later, the bottom rusted through, leaving a puddle to mop up and hazardous waste to dispose of.
Shelf placement makes a difference as well. Keep it eye-level—this isn’t something to toss on an upper shelf where jars can topple or bump into each other. If the container falls, it risks breaking open and mixing with whatever’s stored nearby. In one university storeroom, chemicals ended up jumbled together and workers dealt with a fire after a spillage mixed with paper towels and spilled ethanol.
Additional Layers of Safety
Make life easier by practicing sharp organization. Assign dedicated cupboards for oxidizers and double-check that ventilation holds up on muggy days. Employers sometimes cut corners on keeping storerooms climate-controlled, but condensation turns into a nightmare for any chemical that pulls in moisture. It isn’t being too careful—these steps become routine after a few scares, or worse, after health inspectors threaten fines.
Always set aside a spot nearby for materials to soak up spills—think chemical absorbent granules, not kitchen rags. Keep eye protection close, and train everyone new how to read the hazard codes. It doesn’t take a degree in chemistry to screw this up; I’ve watched new hires shovel things into storerooms with zero sense of danger or care.
How Rules and Records Help
Local authorities and the Occupational Safety and Health Administration (OSHA) lay out clear schedules and guidelines for safe chemical storage, and for a good reason. Solid record-keeping saves time in emergencies and during inspections. Make it crystal clear with updated safety data sheets, quick access to emergency plans, and by encouraging coworkers to speak up when they see poor storage practices slipping in.
In my own experience, open conversations about past mistakes—and not just quoting rules—help everyone get on board. Sharing these lessons, supporting ongoing training, and taking regular inventory checks keep the workplace safer for everyone involved. That level of vigilance makes the difference between a tidy shelf and a hazardous mess waiting to happen.
What is the chemical formula of Copper Nitrate?
Getting to the Basics: What Makes Up Copper Nitrate
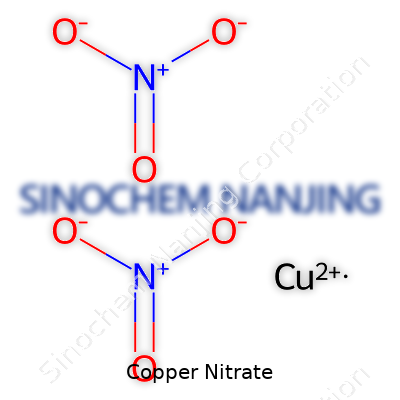
Copper nitrate doesn’t show up in many homes, but anybody who’s handled chemistry, agriculture, or even metalwork probably bumped into it sooner or later. The key fact: copper nitrate usually refers to copper(II) nitrate, which chemists write as Cu(NO3)2. That subscript of 2 on the nitrate part really just means two nitrate ions latch onto each copper ion.
Real-World Importance and My Experience With Copper Compounds
Working in a high school chemistry lab, I watched copper nitrate crystals form when dropping copper metal into nitric acid. Blue crystals mean the job is done—copper atoms team up with nitrate ions in the right ratio, which boils down to Cu(NO3)2. It’s not just a random detail. In fields like fertilizers, metal treatment, or pigments, precision in chemical formulas drives results. If you use the wrong ratio, you get the wrong product or worse, dangerous reactions.
Understanding the Facts Behind the Formula
The formula comes straight from copper’s charge in its common “+2” state. Nitrate, the NO3- part, carries a single negative charge. Balancing these charges (two nitrates for every copper ion) gives you a stable compound. Scientists spent centuries figuring out these ratios not through abstract theory, but through trial, error, and sometimes accidents. Today, lab technicians and industry pros rely on this kind of formula to scale up reactions safely.
Importance in Safety and Sustainable Industry
Mishandling copper nitrate has real-world consequences. Its vivid blue hue might look inviting, but the compound can irritate skin, stain surfaces, and pollute groundwater if tossed casually down a drain. In one college lab cleanup, a colleague nearly caused a minor hazmat scare—he assumed blue water was harmless rinse-off. A quick consult with the chemical safety officer set him straight. Rigid attention to the formula keeps everyone clear about what’s in that beaker or bottle.
Industry workers and plant operators pay attention to chemical formulas to keep waste streams safe, recycling efforts efficient, and compliance with environmental regulations intact. Every batch sheet and storage label features these formulas front and center, because errors become costly fast.
Finding Practical Solutions
Science classes, industry training sessions, and digital fact sheets can all stay a step ahead by focusing less on dry memorization, more on hands-on demonstrations and clear labeling. Reminders of the charge-and-ratio story behind Cu(NO3)2 help newcomers learn faster and encourage seasoned workers to double-check. Good practice means clear, bold labeling, dedicated spill kits, and up-to-date instructions hanging right above chemical storage.
Teachers, safety officers, and managers should push for active, scenario-based drills where staff and students actually handle mock spills and segregate compounds for disposal. Real understanding grows out of repetition and attention to detail, not just a printout with the formula.
How do you safely dispose of Copper Nitrate?
A Closer Look at Copper Nitrate
Copper nitrate doesn’t earn a lot of headlines, but folks in labs and workshops often deal with it. This blue, crystalline salt finds its way into school chemistry classrooms, hobbyist spaces, and even art studios. Many people ask how to get rid of the leftovers once an experiment or project wraps up. I remember the first time I looked at a beaker full of the stuff and wondered, "Can I just pour this down the drain?" It turns out that’s a fast way to create a much bigger environmental headache.
Copper compounds, including copper nitrate, bring real risks if tossed carelessly. Dumping it in a sink puts that copper straight into our water system. Scientific studies show excess copper can poison fish, harm soil microbes, and eventually return to our own tap water. This isn’t just about following rules; it’s about protecting your neighbors, your family, and yourself.
Health, Local Guidelines, and My Experience
I used to teach high school chemistry. Before any disposal, I checked local regulations—every town has its own rules, and getting it wrong brings fines. In my city, copper nitrate counts as hazardous waste. The Environmental Protection Agency (EPA) and local authorities agree on this point. If your area’s rules aren’t clear, call the nearest hazardous waste facility and ask directly. They usually give advice that’s simple to follow, and I’ve never had a problem dropping off small amounts for proper handling.
Direct contact with copper nitrate irritates skin, eyes, and even the lungs if inhaled. I learned my lesson the hard way: long sleeves, goggles, and gloves every time. Those few extra minutes could save you from a trip to the nurse or doctor. Remember, it doesn’t just hurt people; it damages plumbing and wildlife downstream.
What You Can Do to Minimize Risk
Storing the solution correctly is boring, but it’s crucial. Use clearly labeled containers and keep lids tight. Stray droplets or spills eat through steel, so stick to glass or acid-resistant plastic. I find labeling sharpies and painter’s tape work fine for temporary storage, and I’ve always kept any copper nitrate mix out of reach from kids or pets at home.
Most waste facilities ask for details about the waste: how much you have, if it’s mixed, and how concentrated it is. Never mix copper nitrate with household chemicals to "neutralize" it unless you are sure of the chemistry behind it. Trying to shortcut this step risks mixing up toxic gases. Many schools coordinate yearly hazardous waste pickups. If you’re on your own, reach out to nearby colleges or even fire departments; they often know what to do or can point you to a local drop-off site.
Thinking Ahead: Reducing Leftovers
Over the years, I found the smartest move was to buy or make only what I actually used. Small batches cut down on leftovers, and if an experiment flopped, the clean-up was simpler. Working in a team or sharing supplies in a class or local maker space goes a long way to prevent excess in the first place. A little planning and good communication matter more than any disposal cheat sheet.
Safe disposal isn’t flashy, but it pays off all around—cleaner water, less damage to wildlife, and a healthier community. Learning how to handle stuff like copper nitrate builds trust in science, schools, and ourselves. It’s worth doing right every time.
| Names | |
| Preferred IUPAC name | copper(II) nitrate |
| Other names |
Cupric nitrate Copper(II) nitrate Nitric acid, copper(2+) salt Blue copperas Cu(NO3)2 |
| Pronunciation | /ˈkɒpər ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 10031-43-3 |
| Beilstein Reference | 358706 |
| ChEBI | CHEBI:78027 |
| ChEMBL | CHEMBL3407282 |
| ChemSpider | 32554 |
| DrugBank | DB14577 |
| ECHA InfoCard | ECHA InfoCard: 029-014-00-6 |
| EC Number | 215-204-7 |
| Gmelin Reference | Gmelin Reference: **1314** |
| KEGG | C01778 |
| MeSH | D003827 |
| PubChem CID | 10197986 |
| RTECS number | QL5950000 |
| UNII | CF1L11K5RQ |
| UN number | UN1477 |
| Properties | |
| Chemical formula | Cu(NO3)2 |
| Molar mass | 187.56 g/mol |
| Appearance | Blue crystals |
| Odor | Odorless |
| Density | 2.32 g/cm³ |
| Solubility in water | 1250 g/L (20 °C) |
| log P | -2.13 |
| Acidity (pKa) | 3.7 |
| Basicity (pKb) | 7.87 |
| Magnetic susceptibility (χ) | +48.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.885 |
| Viscosity | 1.096 cP (25°C) |
| Dipole moment | 2.19 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 112.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -451 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | No data |
| Pharmacology | |
| ATC code | V07AY04 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes severe skin burns and eye damage, toxic to aquatic life |
| GHS labelling | GHS07, GHS09, Warning, H302, H315, H319, H410 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| NFPA 704 (fire diamond) | 2-3-0-OX |
| Autoignition temperature | 200°C |
| Lethal dose or concentration | LD50 oral rat 940 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Copper Nitrate: 940 mg/kg (oral, rat) |
| NIOSH | Q988 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | 100 mg |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Copper(II) chloride Copper(II) sulfate Copper(II) carbonate Copper(II) oxide Copper(I) nitrate |

