Copper Dichromate: A Deep Dive
Historical Development
Copper dichromate, a compound that never gets the spotlight in hobbyist chemistry kits, carries a story that stretches back to the roots of industrial chemistry. Research and early descriptions in the 19th century placed it among a string of discoveries that rode the wave of fascination with transition metal salts. Back then, scientists tinkered with all sorts of metal-chromate combinations, searching for colorants, pigments, and catalysts. In those days, laboratories poured resources into figuring out why some of these compounds offered wild oxidative properties and others simply corroded away. As early as 1850, copper’s relationship with chromium piqued curiosity, especially since chromates and dichromates soon earned notorious reputations as both useful workhorses and toxic agents.
Product Overview
Copper dichromate forms when copper ions and dichromate ions meet in solution and fall out together as an orange-red powder. Industry folk rarely work hands-on with it compared to common copper sulfate or potassium dichromate, but it fills a crucial niche. Its unique color invites use in ceramics and glassmaking and sees niche interest as an oxidant in organic chemistry. On the inventory sheet, most suppliers list copper dichromate as a specialty item—not your everyday chemical, but a staple for those who need its special blend of reactivity and stain potential. You’ll come across it in research labs, specialty pigments, and sometimes as a stopgap for corrosion studies.
Physical & Chemical Properties
Copper dichromate has a striking orange-red appearance, usually sold as a fine powder. The formula, CuCr2O7, puts copper (II) ions in play alongside the powerful chromate ions. Solubility sits at a low level in water, but it’s enough to release chromate ions slowly, driving the chemistry that makes this compound interesting. It decomposes on heating above 300°C without melting, throwing out toxic fumes like chromium(VI) oxides and, occasionally, copper oxides. It fits the classic mold for strong oxidizing agents—corrosive, reactive with many organics, and with a tendency to stain skin and surfaces permanently.
Technical Specifications & Labeling
Shelf stock from reputable suppliers often arrives with purity numbers above 98%, sometimes specified by ICP or AAS for both copper and chromium content. Packaging gets labeled with proper hazard pictograms for oxidizers and carcinogens, given the chromium(VI) involved. A decent supplier staggers batch records, offering traceability for both academic and industrial buyers. Labels don’t just list the CAS number (usually 13765-19-0), but point out hazard classes, storage guidance—keep out of sunlight, away from heat, locked and properly segregated from fuels and strong acids.
Preparation Method
Chemists have relied on a straightforward double displacement reaction to prepare copper dichromate for more than a century. Adding a solution of copper(II) sulfate to one of sodium dichromate under acidic conditions gets the job done. The orange-red precipitate forms quickly, ready for filtration and careful washing. Many labs dry it under vacuum to minimize exposure to air and lower the risk of losing chromate ions through unwanted side reactions. The process is finicky, and proper ventilation remains essential—the fumes are no joke, and spillage stains lab benches for months. You often find that commercial-scale production sticks to small batches, not just to control waste but because of the strict regulations stitched into chromium chemistry.
Chemical Reactions & Modifications
Once in hand, copper dichromate doesn’t play nice with many reducing agents; throw in a dash of ethanol or sulfur dioxide, and you’ll see rapid reduction of chromium(VI) down to green chromium(III) oxides, with plenty of heat and sometimes a pop of flame if conditions run dry. This strong oxidative talent explains why organic chemists keep a bottle of it close when working out new routes to carboxylic acids from alcohols. Another reaction line comes when copper dichromate meets heat or strong acids; you can split it up, extracting either copper or chromate, letting chemists recover ingredients for different experiments. Creative minds always find a new way to harness the reactivity, but slip-ups cost dearly in terms of exposure to toxic dust or risk of fire.
Synonyms & Product Names
You’ll see copper dichromate go by other names, especially in older literature and in company catalogs. The IUPAC calls it copper(II) dichromate, pointing to the copper oxidation state, but other tags include cupric dichromate, copper chromate(VI), or copper bichromate. Each name signals its historical journey, and in some regional markets, suppliers print trade names based on the manufacturer’s codes, adding a layer of confusion. The synonyms may look similar, but strict chemists always check the formula before mixing it into any reaction.
Safety & Operational Standards
Workplace safety around copper dichromate goes far beyond goggles and gloves—occupational standards have tightened sharply in recent years. Any handling calls for a certified fume hood, and many labs insist on full respirators. The dust risks inhalation and skin absorption, and the chromium(VI) portion brings carcinogenic hazards that regulators highlight as a red flag. Most training programs teach emergency decontamination: calcium gluconate gel for copper exposure, immediate washing for any splash, and isolation and neutralization for even tiny spills. Labs keep MSDS sheets handy, and disposal only runs through approved waste channels since chromium(VI) lands on almost every environmental blacklist worldwide. Secure storage must be away from combustible materials. At my old lab, we kept a dedicated chromate safety bin, triple-lidded and checked weekly by safety staff, just to keep regulatory trouble far from our doors.
Application Area
Despite all the safety headaches, copper dichromate holds real value across several technical fields. Artisans in glass and ceramics prize it for colors nobody else can match, especially vivid oranges and reds that withstand high kiln temperatures. Industrial engineers eye it for specialized corrosion testing, since the compound stresses coatings and paints like few others. Research chemists once leaned heavily on its oxidative muscle for organic reactions, especially oxidation of alcohols, though greener alternatives slowly take over each year. The pigment industry once used it for deep stains, but environmental rules pared back that use. Yet, in glass, specialty pigment, and the odd catalyst formulation, chemists still pull copper dichromate off the shelf for jobs that nothing else can quite finish.
Research & Development
Lab teams still tinker with copper dichromate, more in the context of curiosity-driven research than mass-market development. Recent years brought renewed interest in new oxidation mechanisms and safer handling practices. Some green chemistry labs test ways to recycle or neutralize the compound, capturing chromium in safer oxidation states for reuse. Studies from top research institutes test novel encapsulation techniques to cut down on dust exposure during pigment manufacturing or catalysis. You won’t find flashy patents, but small improvements and clever workarounds keep copper dichromate relevant. Some groups also study composite materials, embedding the dichromate in stable, inert matrices to eke out reactivity without exposing workers to raw powder—usually pulling inspiration from nanotech and materials science.
Toxicity Research
The danger from copper dichromate always comes back to its chromium(VI) content. Extensive toxicology work, both from industrial labs and public health organizations, shows that airborne dust and skin contact allow chromium(VI) ions into the body, leading to skin allergies, ulcerations, and sharply increased cancer risk with chronic exposure. Animal studies point out acute toxicity through ingestion or inhalation, with damage showing up in kidney and liver cells. Regulatory agencies like OSHA and the EPA have set very low workplace exposure limits and listed it as a confirmed carcinogen. Research has now turned toward not just monitoring worker exposure, but figuring out reliable detection methods for trace amounts in workplace air and run-off water. Medical research keeps a close watch for biomarkers of early exposure among at-risk worker groups, and studies push for strong substitution in industrial uses wherever possible.
Future Prospects
Most forecasts suggest the era of routine copper dichromate use has passed, at least in large-scale industrial environments. Green chemistry and strict regulations on hexavalent chromium shoved the compound from mainstream production into niche and specialty applications. Still, research labs find that nothing else quite matches its oxidation power or pigment shade in certain artistic and analytic projects. The future likely holds more work on containment, recycling, and substitution. Where science circles need the compound, engineers will continue to refine encapsulation techniques and worker monitoring, striving to keep the chemical’s hazards in check. Countries plan even stricter controls, while forward-thinking researchers keep searching for safer, renewable alternatives that match the robust properties of copper dichromate—ensuring that the compound’s legacy will outlast its mass-market run.
What is Copper Dichromate used for?
What You Get With Copper Dichromate
Copper dichromate stands out as one of those chemical compounds with a distinct, almost iconic, bright color—often a vivid greenish-blue. As a writer who’s seen plenty of substances pass from bench to industry floor, I pay attention when something gets used across different fields. This compound isn’t a household name, but chemists and industry veterans recognize it right away.
Wood Preservation That Lasts
In the real world, old fences, telephone poles, or deck planks face one major enemy: decay. Rain, humidity, and insects make short work of untreated wood. Copper dichromate came onto the scene offering a tough, water-based treatment. Once absorbed, it not only slows down the spread of fungi and mold but also keeps termites at bay. Some might say wood preservatives sound boring. They're not, especially for anyone relying on sturdy framing in wet climates. By prolonging the life of wooden structures, this substance cuts down on wasted resources and unnecessary rebuilds.
Pigments and Dyes Seen in Art and Industry
Take a stroll through an art supply shop. Hidden somewhere between the aisles, copper-based pigments show up in things like watercolors and oil paints. The greenish tinge of copper dichromate appears here, especially when artists seek bold, lasting shades. The compound gets mixed into varnishes, ceramics, and even dyes for textiles. Its properties don’t just benefit professional artists—schoolchildren pressing watercolor brushes into paper have probably used colors containing traces of this very compound.
Chemistry in the Lab
Curiosity drives a lot of scientific innovation. In labs, copper dichromate offers more than eye-catching color. It acts as an oxidizing agent. Chemists working on organic reactions appreciate its ability to speed up the transformation of alcohols to carbonyl compounds—essential in research and manufacturing. Plenty of publications lay out examples of successful syntheses where this greenish compound plays a critical role.
Concerns With Toxicity
Shelves of chemistry storerooms come with warning signs for a reason. Copper dichromate carries both copper and chromium, both of which can become hazardous at certain exposures. Chromium(VI), in particular, is known for posing health risks including skin problems and, after long-term contact, higher cancer risk. Inhaling dust or having unprotected skin contact isn’t safe. This matters for more than just lab workers—communities near large wood treatment facilities pay attention to local regulations and cleanup after leaks. Personal experience working around toxic compounds teaches respect for gloves, goggles, and well-ventilated spaces.
Moving Toward Safer Alternatives
In recent decades, demand for safer solutions has intensified. More companies now look for preservatives that work just as well without the chromium footprint. Copper-based treatments persist, but new formulas exclude dichromates and rely on more stable, less harmful ingredients. Stricter rules forced this transition, and for good reason. Preserving wood matters, but doing so with the community’s safety at the forefront matters even more.
The Big Picture
Copper dichromate serves a unique niche: effective in preservation, color, and chemical reactions. It shows what happens when deep scientific knowledge meets practical demand. But each use comes with accountability—to workers, the environment, families nearby. Now, with new methods and materials available, it’s possible to keep the benefits without ignoring the risks.
What is the chemical formula of Copper Dichromate?
Understanding Copper Dichromate
Stepping into a chemistry lab, the names on containers sometimes blur together. Copper dichromate stands out for its striking color and unique behavior. Many remember it from school or college labs—a reddish or greenish powdery substance that can stain both gloves and benches. The formula itself, CuCr2O7, reveals a lot about its identity. Each part signals one copper atom and two chromium atoms paired with seven oxygen atoms. The chromium comes in its +6 oxidation state, which marks it as a strong oxidizer with environmental drawbacks.
Importance in Industry and Everyday Life
Copper dichromate plays a modest yet important role in industries with needs for pigmentation, oxidation, or coloring. Some artisans and manufacturers once relied on it for pigmentation in ceramics and glass. Its color, a brilliant reddish-orange, brings vibrance to works—and memories for anyone who's glazed pottery or glass. The chemical does not enjoy the same spotlight as copper sulfate or potassium dichromate, but it has a seat among those important compounds that quietly shape material properties.
Handling Hazards and Environmental Concerns
Working with copper dichromate taught me respect for chemical hazards. Chromium(VI) compounds sit on the list of substances of concern for their toxicity and negative impact on health and the environment. Many chemists remember cracked hands from careless handling of chromates, which can irritate skin and even trigger allergic reactions. In labs, strict guidelines instruct users to wear gloves, goggles, and sometimes even dedicated lab coats. Waste is never poured down the drain but collected to keep streams and soil free of hexavalent chromium.
The environmental risks attached to copper dichromate force a deeper conversation about chemical stewardship. Industrial accidents and improper disposal can harm aquatic life, poison groundwater, and hurt plants. These outcomes prompt regulations in many countries. Industrial users now look for alternatives that achieve similar coloring or oxidation without the toxicity. Over the last few decades, green chemistry approaches have spawned safer pigment blends, reducing the reliance on hazardous legacy chemicals like copper dichromate.
Moving Toward Safer Solutions
Acknowledging the dangers doesn't mean compelling panic or dropping such compounds without a plan. Scientists and companies explore substitute materials—blends or single compounds with similar properties but fewer health and safety risks—to keep both people and ecosystems safe. Labs everywhere train younger chemists not only to use protective equipment but also to question whether using certain chemicals remains necessary. Substitution sometimes involves a trade-off in color or strength, but the payoff in reduced environmental risk benefits communities in the long run.
Society asks more from industries than ever before. Responsible chemical management now ranks up with quality and efficiency. Knowing the formula CuCr2O7 lets us see the chemical on more than just a molecular level—it’s a reminder of the choices made by industries and researchers every day, and how these choices shape both safety and sustainability for future generations.
Is Copper Dichromate hazardous to health?
Understanding the Risks
The deep orange-red color of copper dichromate might catch the eye in a lab, but the risks tied to this compound are no secret in the chemical community. Working around copper dichromate means you’re dealing with both hexavalent chromium and copper ions, two elements that carry their own baggage.
Personal Experience from the Lab Bench
My years working around industrial chemicals taught me early: keep your masks tight and your gloves clean. During my grad study days, we had a small chromium-related incident. It wasn’t copper dichromate exactly, but clean-up protocols were similar. A few careless moves, a broken vial, and we had hazardous dust on hands, floors, and even a lab bench sandwich someone had left out. It hammered home the point: these compounds don’t belong near bare skin or uncovered food.
Chromium Compounds – What the Science Says
Let’s dig into the facts. Hexavalent chromium—the form found in copper dichromate—doesn’t just cause skin rashes or eye irritation. According to CDC and the International Agency for Research on Cancer, hexavalent chromium compounds are proven carcinogens. Breathing dust or vapors over time links to lung cancer, nasal irritation, and even kidney or liver damage. Even a small dusting can spark trouble for those with asthma or sensitive airways.
Copper sounds less scary, but don’t brush it off. Swallowing copper dichromate or inhaling enough particles leads to metal toxicity. We’re talking cramps, vomiting, and liver strain. Some people develop a sensitivity, with rashes or breathing problems cropping up after repeated exposure.
Environmental and Workplace Realities
Many places still use chromium compounds for pigments, wood treatment, or corrosion resistance. The stories out of old factories and workshops still cause concern. Rates of cancer and chronic illness tick higher in neighborhoods that handled these substances without strict controls. The environment also pays the price. Runoff carrying chromium finds its way into drinking water. I’ve walked the banks of old industrial rivers where nothing but weeds grow and fish are long gone.
Solutions and Safer Practices
A solution starts with respect for the danger and real training. Newer labs now run air monitoring, keep locked chemical storage, and require fit-tested respirators around these substances. Spills call for more than a mop. Absorbent pads, acid-resistant gloves, and proper waste disposal keep accidents contained. Good ventilation, HEPA filters, and strict labeling rules help. I’ve known workplaces that let training slip, and it always leads to close calls. That risk just isn’t worth it.
Regulation has teeth, too. The United States Occupational Safety and Health Administration (OSHA) requires employers to limit worker exposure to chromium (VI). EPA tracks use and makes sure companies clean up sites where materials like copper dichromate were handled. Countries with strict chemical oversight protect both workers and neighbors. Taking shortcuts means gambling with long-term health. If possible, switching to less toxic substances avoids chronic health worries. Some companies now turn to alternative pigments and inhibitors wherever science allows.
Copper dichromate’s vivid color can dazzle, but nobody wants a glowing pigment that glows with danger. Experience, research, and common sense all say: treat this chemical like trouble, and you’ll avoid a world of regret.
How should Copper Dichromate be stored?
Why Safe Storage Matters
Copper dichromate looks harmless—a bright, powdery compound often used in labs for chemical synthesis and some industrial processes. Judging by the name alone, it’s easy to assume it’s just another basic chemical, but this stuff packs a punch. Known to be toxic and highly oxidizing, copper dichromate demands serious care. I’ve seen far too many stories where lab workers brushed aside safety rules and paid the price, with spills or health problems down the road. Responsible storage isn’t only about ticking off a regulatory box; it’s about protecting your lungs, skin, and the wider environment around your workspace.
What Science Tells Us About the Hazards
Here’s how things play out with copper dichromate: One whiff or a bit on the skin leaves you courting trouble. It releases toxic chromium(VI) compounds, known to cause cancer and damage internal organs. Splashes or puffs in the air seem minor at first, but chromium-based dust has been linked to lung problems, nosebleeds, and skin burns. I once knew a tech who underestimated a small container and landed in the emergency room with chemical burns. Nobody needs that reminder twice.
Practical Storage Steps in the Real World
Let’s face it, most chemical storage advice sounds like legalese. From my time in university and in industrial settings, the best protection always followed common-sense steps, not memorized rules. Keep copper dichromate in tightly-sealed, clearly labeled containers made of glass or specialized plastic. Store it away from incompatible materials—no acids, no organic materials, and never near things that burn easily. Put it in a cool, dry cabinet that stays locked, preferably a cabinet meant for oxidizers. I’ve seen labs skimp on this and stick oxidizers next to solvents—all it takes is one leaky bottle for a bad day to turn disastrous.
No open shelving, no storing above eye-level, and forget putting it anywhere in direct sunlight or near sources of heat. That’s a recipe for decomposition and risky fumes. Ventilation matters. Even with a sealed container, storing it in a non-ventilated space can increase danger from leaks or accidental spills. Every reputable safety data sheet recommends a space equipped with exhaust or at least a chemical fume hood nearby for safer air.
The People Factor: Training Equals Safety
There’s no point in top-shelf cabinets or warning signs if nobody reads or follows them. In every lab I’ve worked, accidents always came from lapses—forgetting gloves, missing labels, or new people left to guess at the rules. Real safety starts with hands-on training, not just reading protocols once a year. Workers handling copper dichromate need to wear chemical-resistant gloves, goggles, and proper lab coats. Sounds obvious, but after a while, even smart folks get lazy or rushed. Regular drills—how to handle a spill, how to wash down exposed skin—make all the difference.
Waste: Not Just Another Trash
Disposing of copper dichromate means following strict hazardous waste rules, plain and simple. Pouring it down the drain guarantees it enters water systems, harming wildlife and potentially local communities. Every lab or plant I’ve worked at had a dedicated hazardous waste container, with clear logbooks. Never mix with other chemicals, and always tell new team members how disposal works. The EPA and OSHA publish clear guidelines; following them keeps you and the people around safe, and avoids massive fines or legal drama.
Looking Ahead: Smarter Chemical Management
Being around copper dichromate or similar compounds isn’t about fearmongering. With strong policies, clear labeling, real training, and practical storage plans, people can use these chemicals without putting themselves or the environment at risk. Safety depends on daily habits, as much as any technical standard. Anybody storing copper dichromate should treat it as a top priority—not because it’s the law, but because the risk is real and preventable.
What is the appearance and color of Copper Dichromate?
Distinctive Hue and Physical Traits
As a seasoned science writer, I’ve spent years in university labs, often up close with a variety of salts and pigments. A standout among transition metal compounds, copper dichromate presents a distinct visual impression. In hand, it doesn’t look dull, nor does it shy away from attention. Samples of copper dichromate often reveal a robust green coloring—sometimes pushing into shades of blue-green. That color reflects the interplay between copper’s natural blue-green lean and the vibrant orange tones from the dichromate ion. It’s not the bright, flaming orange of potassium dichromate, nor does it mimic the familiar patina of weathered copper pipes. It combines both influences to produce a striking emerald appearance with a touch of yellow undertone that’s easily spotted on a lab bench.
What Makes This Color Unique?
The color of copper dichromate comes straight from its molecular structure. I’ve watched countless reactions and always notice how the exact shade shifts with particle size and moisture content. Freshly prepared, fine copper dichromate powder shows a deep blue-green, especially under good lighting. If the sample takes on moisture from the air, it shifts toward olive green or gains a muddy cast. This change matters in analytical chemistry, where the right color signals a pure, well-made sample. Copper ions deliver the base green, while the addition of dichromate pushes the mix into a rich, jewel-toned shade, setting it apart from similar compounds.
Practical Work and Laboratory Experiences
In classrooms and companies, quality control often starts with visual checks. Early in my career, I learned to trust my eyes before any spectrometer could verify results. Pouring copper dichromate into a beaker, I’d look for consistency—a clean, vivid green always signaled that the initial synthesis went smoothly. Mixed colors, white patches, or visible chunks meant a mistake somewhere down the line. Lab workers rely on this visual test every day because it’s quick and saves money during early screening.
Why Color Consistency Matters
Getting the appearance right isn’t just a cosmetic issue. Research from the Royal Society of Chemistry shows that batch color tells the story of both chemical purity and the effectiveness of reaction conditions. In pigment manufacturing, off-color material can mean lost money, wasted product, or even safety risks if contaminants sneak in. Color variation can signal impurities like leftover reactants or unexpected by-products, both of which affect a compound’s reliability in downstream applications.
Solutions to Quality and Purity Challenges
Chemists control copper dichromate color by tightening up reaction conditions. I’ve improved outcomes by carefully adjusting temperature and using freshly distilled water, which keeps unwanted ions out of the mix. Journals detail that precise pH adjustment ensures a true green, free of brown or blue-gray hints. Storage in airtight containers limits moisture absorption, stopping gradual color drift—a lesson I learned after watching bright green powder turn olive on a humid day.
Trust Built from Evidence and Observation
Years of handling copper dichromate teach that a vivid green signals not just a successful synthesis, but a compound ready for use in chemical analysis or pigment work. That color ties together research, experience, and careful preparation—qualities at the core of good lab practice and trusted scientific work. If the powder looks right, chances are the chemistry behind it stands strong.
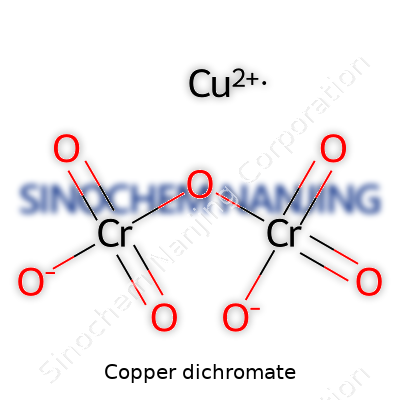
| Names | |
| Preferred IUPAC name | Copper(II) dichromate |
| Other names |
Copper bichromate Cupric dichromate Copper(II) dichromate |
| Pronunciation | /ˈkɒpər daɪˈkrəʊmeɪt/ |
| Identifiers | |
| CAS Number | 13765-19-0 |
| 3D model (JSmol) | CU(Cr2O7) JSmol string: ``` CU + [O-] [Cr](=O)(=O)O [Cr](=O)(=O)O ``` |
| Beilstein Reference | 31458 |
| ChEBI | CHEBI:91241 |
| ChEMBL | CHEMBL3286941 |
| ChemSpider | 187410 |
| DrugBank | DB14606 |
| ECHA InfoCard | 200-838-7 |
| EC Number | 232-192-7 |
| Gmelin Reference | Gmelin Reference: **1454** |
| KEGG | C16296 |
| MeSH | D003797 |
| PubChem CID | 25021 |
| RTECS number | GB6300000 |
| UNII | NF8U3I6YWG |
| UN number | UN3288 |
| CompTox Dashboard (EPA) | F1909KQ0UZ |
| Properties | |
| Chemical formula | CuCr₂O₇ |
| Molar mass | 341.52 g/mol |
| Appearance | Orange crystals |
| Odor | Odorless |
| Density | 2.97 g/cm³ |
| Solubility in water | Insoluble |
| log P | -2.625 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 7.7 |
| Magnetic susceptibility (χ) | +3090.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1015 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause cancer, may cause genetic defects, very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H272, H301, H314, H317, H334, H341, H350, H360, H400, H410 |
| Precautionary statements | P260, P264, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P337+P313, P362+P364, P391, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-0-2-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 65 mg/kg |
| NIOSH | GB5950000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Copper Dichromate: "0.01 mg/m³ (as Cr VI) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 15 mg Cr(VI)/m³ |
| Related compounds | |
| Related compounds |
Ammonium dichromate Potassium dichromate Sodium dichromate Chromic acid |

