Copper Chlorate: Chemical Curiosity with a Dangerous Edge
Historical Development
Copper chlorate has a history woven into the fabric of classical chemistry. Early experimenters, working in the candle-lit laboratories of the nineteenth century, first synthesized it as part of broader exploration into copper salts. For a while, the compound sparked excitement among chemists who hunted for new oxidizers. Laboratory workers, often unaware of modern safety standards, prepared copper chlorate to explore its vivid blue-green crystals and reactive chemistry. The most noticeable period of interest coincided with the golden age of pyrotechnics research. You can find scattered references to copper chlorate in old chemical treatises and fireworks manuals, often with warnings handwritten in the margins. Unlike copper sulfate, which found its footing in agriculture and antifungal treatments, copper chlorate raised eyebrows because its unstable chlorine-oxygen bonds brought volatility along with bright, almost mesmerizing coloration.
Product Overview
Chemists know copper chlorate for its strength as an oxidizer, typically prepared as either a blue or green crystalline powder. The material draws attention both for its striking appearance and for its hazards. Unlike household copper compounds, this one doesn’t belong in casual settings. Laboratory use calls for care, skill, and a sharp eye on chemical compatibility. Distributors rarely offer it for regular purchase, and most of what exists gets prepared in academic chem labs or specialty research outfits. Part of its intrigue lies in its scarcity—few researchers actively select it today due to its risks compared to safer alternatives.
Physical & Chemical Properties
Copper chlorate stands out with intense color, generally manifesting as deep blue-green crystals. Its solubility in water sets the stage for a wide range of chemical demonstrations, though even small samples bring substantial reactivity. The compound’s odorless nature means no warning for the nose, but the oxidizing power betrays itself fast in reaction with organic materials. Left exposed, it attracts moisture, and most samples quickly take on a damp sheen under humid conditions. Its melting point hangs lower than many comparable inorganic salts, hinting at instability. When heated, copper chlorate decomposes, releasing toxic chlorine compounds and oxygen—turning it into a dangerous ignition source rather than a bystander.
Technical Specifications & Labeling
Regulatory agencies insist on clear labeling due to the compound’s risks. All containers must indicate its strong oxidizing properties, the dangers from thermal decomposition, and compatibility limits with reducing agents and organics. Given the toxicity of both copper and chlorate ions, labels always warn against ingestion, inhalation, and direct skin contact. Unlike common salts, this one brings strong environmental risks if released, especially in aquatic systems where copper toxicity devastates invertebrate life. Laboratory vials, if one ever comes across them, usually bear secondary labels reminding handlers to keep the chemical locked and away from organics or anything flammable.
Preparation Method
Making copper chlorate calls for careful handling and well-controlled conditions. The classic approach uses copper sulfate and a soluble chlorate, such as sodium chlorate, in solution. Both solutions mix, leading to precipitation as the two salts swap ions and form the copper salt of chloric acid. Filtering and slow evaporation help crystals form, a step that often exposes the product to accidental friction or shock—each posing risk of initiation. Over the years, chemists phased out industrial production almost entirely because yields rarely justify the danger. In old manuals, warnings dominate the preparation section, and modern procedures relegate the synthesis to demonstrations for advanced instructional settings.
Chemical Reactions & Modifications
Among chlorates, copper chlorate’s burning interest lies in its ability to transfer oxygen. It reacts eagerly with many organics, sometimes too eagerly for safety. Simple mishandling during weighing or blending has caused small-scale explosions in poorly equipped labs, driving home the need for deliberate, methodical work. The compound oxidizes common reducing agents and metals, and its reactivity extends through a wide range of temperatures. Over time, exposure to acidic or basic conditions can shift the copper oxidation state or decompose the chlorate, sometimes giving rise to copper oxide or chloride as yellow or brown byproducts. Little research goes into altering copper chlorate’s structure, as safer oxidizers now fill most roles in labs and industry.
Synonyms & Product Names
Chemistry literature sometimes lists copper chlorate under names such as cupric chlorate or copper(II) chlorate, and references often shorten it to its molecular formula, Cu(ClO3)2. Beyond technical sources, the compound rarely appears under any commercial brand. Its instability and risk profile keep it out of everyday channels, and you’d be hard-pressed to find it on the open market, even in niche pyrotechnic catalogs.
Safety & Operational Standards
In my time working with oxidizers and energetic materials, few substances get the level of caution that copper chlorate deserves. Proper labs follow strict protocols: only small quantities, isolated workspaces, and use of non-sparking tools. Chemical safety regulations require personal protective equipment including gloves, goggles, and, ideally, a full face shield. People handling the material must maintain immaculate workspace cleanliness; even a trace of organic powder or dust brings serious risk. All operations take place far from heat sources or sunlight, and waste gets neutralized at the earliest stage. Regulatory bodies discourage amateur or industrial use except under tightly regulated research settings, as the temptation for pyrotechnic experimentation too often turns tragic.
Application Area
Despite its historic fame among early fireworks experimenters, copper chlorate lost its favor as safer green colorants hit the scene. In the height of the fireworks age, formula books often suggested copper chlorate for green flames or torch mixes. Still, the compound’s hazard profile and unpredictable burning behavior led most artisans to drop it in favor of barium, boron, or more benign copper salts. Beyond pyrotechnics, it sees minor use as a laboratory oxidant for specialized organic reactions—though most chemists steer toward less volatile options. In modern research, experiments with copper chlorate serve more as demonstrations of historical chemical practices or as cautionary tales than as practical process additives.
Research & Development
Any new work on copper chlorate focuses on understanding its decomposition pathways or on quantifying its energetic hazards. Academic chemists sometimes revisit old reactions to map out exactly how the compound breaks down under various stimuli. Interest in green-flame technology occasionally brings up copper chlorate, but regulatory pushback and advances in material science have narrowed its role. Environmental chemists sometimes look at its behavior in contaminated sites, tracking copper and chlorate mobility through soils. No one seeks commercial expansion of copper chlorate’s applications given its risk and regulatory scrutiny; the future of research centers on hazard reduction, improved waste management, and historical study rather than product development.
Toxicity Research
Both copper and chlorate ions show strong toxicity to living systems. In mammals, ingestion or long-term exposure produces symptoms from gastrointestinal distress to serious metabolic disruption. Copper accumulates in the liver and kidneys, disrupting enzyme systems and leading to copper poisoning with enough exposure. Chlorates interfere with oxygen transport in blood, leading to methemoglobinemia—a condition where blood loses its ability to deliver oxygen. In the environment, copper’s aquatic toxicity remains well-documented; releases harm fish and invertebrates even at low concentrations. Regulations treat the compound as toxic waste, and laboratories work to ensure no run-off or accidental spills. Most research in this area tries to map the exact thresholds and to develop new ways of rapidly neutralizing copper chlorate before it enters ecosystems.
Future Prospects
Forward-thinking chemists see copper chlorate mostly as a relic of chemical history, a cautionary example from a time before modern safety practices. The combination of acute toxicity, instability, and environmental hazard has pushed the compound far to the margins of industrial chemistry. Better oxidizers, safer colorants, and improved laboratory practices stand between copper chlorate and any renewed interest. Future work targets greener chemistry, and discussions about copper chlorate usually lean toward phasing it out of any remaining instructional or specialty uses. The ongoing challenge lies in teaching its lessons, training future chemists to respect energetic material hazards long before curiosity turns into calamity. My hope is that the only role left for copper chlorate will be as an object lesson—how chemistry’s early achievements sometimes left behind more risk than reward, and how today’s standards keep labs, workers, and the environment safer than ever.
What is Copper Chlorate used for?
An Unstable Ingredient That Draws Attention
If you poke around behind the scenes in a chemistry lab, you find all sorts of interesting substances, but copper chlorate stands out for both its striking green-blue crystals and its volatility. Anyone who has spent time in a chemistry classroom has run into demos where copper heated with other chemicals burns with a blue-green flame—copper ions at work. But copper chlorate isn’t just a pretty show. It brings together copper, a useful metal, and chlorate, a powerful oxidizer. That makes it both useful and risky.
Explosives and Pyrotechnics: Where Copper Chlorate Makes Itself Known
The main place copper chlorate turns up is in pyrotechnics and explosives. Its color-forming properties help fireworks makers get that special turquoise flash—a color that isn’t so easy to pull off. The copper in the compound handles the color, and the chlorate jumps in as an oxidizer, feeding much needed oxygen to the reaction. You’ll rarely find it in store-bought fireworks because even folks in the industry worry about how sensitive it is to heat, friction, and even sunlight. In homemade mixes, often among hobbyists, it sometimes gets used despite the danger. Misuse can easily lead to accidents and injuries, which is why strict controls surround its sale and handling in most countries.
The Lab and the Classroom: Teaching Caution
Up in higher education labs, teachers and students look at copper chlorate as a teaching tool. It helps show chemical reactions, color emission, and oxidation-reduction processes in action. I remember struggling to get that perfect blue-green solution for a flame test, battling nerves knowing how quickly things could go wrong with an unstable compound. Yet, its risks mean most teachers avoid using it unless there’s no safer substitute. The lesson? Even useful chemicals have tradeoffs, and safety always trumps spectacle.
Why Agriculture and Industry Leave It Alone
Chlorate compounds pop up in herbicides. But copper chlorate rarely gets the call. Other chlorates manage weeds more predictably and don’t combine a heavy metal with a strong oxidizer. The environmental risks are high enough that copper chlorate stays off the agricultural menu in most places. Its impact on soil, water, and animals brings along another headache, especially in regions trying to protect pollinators and water tables already facing stress from other chemicals.
Health and Environmental Risks Won’t Go Away
Even in small amounts, copper compounds can cause trouble for people and wildlife. Chronic exposure leads to headaches, nausea, and, over time, organ damage. Chlorates create their own set of issues, interrupting how red blood cells carry oxygen. Combined, they spell trouble for anyone handling them without care. I think about the times in the lab where just cleaning up copper spills left green stains on my skin for hours. Harder to see are the long-term effects on waterways and urban soils, which don’t bounce back easily after contamination.
A Search for Safer Alternatives
Most pyrotechnic artists and scientists know other chemicals often fill in the roles originally played by copper chlorate. Copper sulfate and other copper salts step in for color without the same instability. Safer oxidizers offer more control and less risk to both workers and the environment. Whenever a process involves a hair-trigger compound, the clock ticks toward the next accident or spill. Industry moves toward alternatives because employee safety, environmental cleanup costs, and stricter chemical rules demand it. The stories behind those changes—scarred fingers, scorched benches in school labs—remind us that “interesting chemistry” can’t come at the expense of a safe, livable world for everyone.
Is Copper Chlorate dangerous or toxic?
The Real Dangers Behind the Chemical Name
Some chemicals simply don’t belong outside specialized labs. Copper chlorate is one of them. You won’t see this blue-green compound lying around at a garden center or a typical classroom. There are good reasons for this. For starters, copper chlorate contains both copper and chlorate ions, each notorious for their own hazards. Together, they make a risky combination that deserves more caution than curiosity.
Why Copper Chlorate Can’t Be Taken Lightly
I remember once helping a friend clear out a garage stacked with old bottles and bags from a grandparent’s chemistry hobby. Names like potassium nitrate made us pause, but copper chlorate was the one that sent us searching for gloves and a face mask. Its chemical makeup lends itself to both toxicity and potential for violent reactions.
The chlorate part poses a serious threat. Chlorates react strongly with combustible materials—even something as simple as a drop of oil or a bit of paper can trigger an explosion under the right circumstances. At school, we heard stories in the chemistry lab about fiery mishaps and why gloves and goggles existed for a reason. But this compound can introduce trouble far outside the classroom. Street-level fireworks recipes and home chemistry blogs sometimes mention copper chlorate, as if it’s just another reactant. That’s playing with fire, literally. The explosive power of copper chlorate remains one of its biggest threats.
Toxicity and Health Risks
The toxicity of copper compounds has long been known. They can damage organs if swallowed or inhaled. Symptoms range from vomiting and stomach pain to more severe impacts like kidney and liver damage. Add the chlorate part, and things get worse. Chlorates can disrupt the blood’s ability to carry oxygen. Methemoglobinemia, a condition where oxygen transport fails, can put people in serious danger fast. Even skin or eye contact sometimes causes burning, redness, or worse. Home mishaps might send someone to the hospital, and agricultural exposure can mean a toxic threat to wildlife as well.
Environmental Impact Beyond Human Risk
No talk about a chemical’s danger stops at personal health. Once copper chlorate spills or leaches into soil or water, the copper itself builds up in the environment. Too much copper poisons fish and disrupts microorganisms. Chlorates, on the other hand, persist and affect plants, stunting growth. Cleanup isn’t easy or cheap. Local governments end up footing the bill for cleaning up illegal chemical dumping, and in the end, the environment has no say in how it is treated.
Things That Actually Work to Reduce Danger
Safer labs and stricter controls protect people where copper chlorate is used. Proper labeling and clear guidelines for storage can prevent some accidents. Education plays a key part—every level from high schools to small businesses needs fact-based information that prevents risky handling. Never underestimate the value of simple steps: store away from flammables, keep in sealed containers, and dispose of it through professional hazardous waste services, not in household trash or drains.
Personal experience isn’t everything, but it does teach respect. Chemists, teachers, and even curious hobbyists can agree—respect for these mixtures saves lives. If safety gear ever feels like an inconvenience or warning labels look overdone, copper chlorate proves both are necessary. Understanding what makes chemicals dangerous, and learning how to handle or avoid them, highlights why public health rules deserve our attention.
What is the chemical formula of Copper Chlorate?
A Closer Look at Copper Chlorate
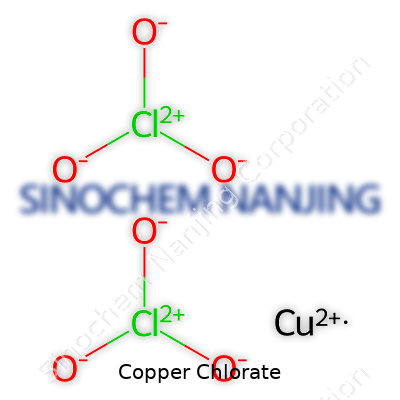
Chemical formulas might not seem like the sort of thing you’d think about outside of a science lab, but they show up in real life more often than some folks might expect. Copper chlorate pulls double duty as both a chemical curiosity and a practical compound. Its formula, Cu(ClO3)2, looks a bit like something straight out of old chemistry textbooks, but this arrangement—one copper atom paired with two chlorate ions—actually matters in a few hands-on settings.
Why Formula Details Matter
Chemistry habits grow from time in the lab—some of my earliest experiments featured various copper compounds, from bright blue copper(II) sulfate to the more exotic chlorates. Understanding the bonds between copper and chlorate changes how the compound behaves. More than just book knowledge, it shapes how we handle, store, and even dispose of chemicals like copper chlorate.
Copper carries a +2 charge here, and chlorate anion brings -1 each. Scientific notation might fade into the background for some people, but mix-ups have real consequences. Labs across the globe put effort into tracking these formulas to avoid errors, which could result in unstable reactions or safety hazards. The right ratio of elements is the backbone of safe chemical handling, whether you’re in a high school chemistry class or running quality checks in an industrial plant.
Where Copper Chlorate Turns Up
Copper chlorate doesn’t grab headlines like sodium chloride or household bleach, but its uses stretch far beyond plain theory. In the world of pyrotechnics, Cu(ClO3)2 brings vibrant blue hues to fireworks displays. Pyrotechnics experts rely on its formula to get the colors bright and consistent, substituting it in formulas only when it will safely deliver the effect they’re after. Some folks explore copper chlorate in chemical demonstrations for teaching oxidation-reduction reactions and decomposition science. These experiments demand accuracy in chemistry, not just because it’s science tradition, but because it keeps teachers and students safe from mishaps like unexpected explosions or toxic fumes.
The E-E-A-T Angle
Trust matters more than ever with chemical compounds. With so many articles floating around, folks need assurance that they’re reading dependable details based on both evidence and experience. Studies on copper compounds have catalogued copper’s variety of roles, linking specific formulas to both chemical reactivity and safety protocols. Years of lab work have taught me to trust formulas like Cu(ClO3)2 because the science tracks from textbook to real-world practice. Communities of chemists, teachers, and engineers carry out checks and exchanges of information through professional groups, keeping details transparent and advice up-to-date.
Better Safety and Solutions
Risks go up when chemical recipes get cloudy. Accidental substitutions can cause property damage, environmental issues, and even health risks, especially with compounds prone to instability. The answer sits in better education—not just for students but for anyone handling or discussing these materials. Hands-on demonstrations help reinforce what the copper chlorate formula means. Digital resources and safety guides, published by verified science organizations, add plenty of up-to-date information for pros and amateurs alike, lowering the odds of mistakes. Labs and classrooms with clear labeling, helpful instructors, and safety-first practices set the right example for what responsible chemistry looks like in action.
How should Copper Chlorate be stored safely?
Understanding What Makes Copper Chlorate Tricky
Copper chlorate draws attention because it's powerful stuff. It’s not something that sits harmlessly on a shelf. At room temperature, it carries a real risk of breaking down if mixed with the wrong material. Known for strong oxidizing properties, it can feed a fire and even trigger explosions if there’s contamination or careless handling. I’ve seen cases in school labs and hobbyist shops where one mistake left people shocked by just how quickly things can go wrong.
Picking the Right Spot
The safest spot for this chemical is in a cool, dry, and dark place. Sunlight or warmth can accelerate decomposition. Moisture invites clumping and unintended reactions, so a storage spot kept safely away from sinks, radiators, or south-facing windows makes a difference. Some professionals use climate-controlled cabinets with humidity alarms, which consistently give early warnings.
Only Non-Combustible Containers Work
Glass and high-quality polyethylene containers have proven effective for copper chlorate. Metal and wood never belong in this equation. I’ve seen people reach for metal cans because they are handy, but copper chlorate can corrode some metals and start a chain reaction. Always label the container clearly, including the date—old samples tend to grow more unstable.
Avoid Cross-Contamination Like the Plague
Copper chlorate comes alive in the presence of organic stuff. Bits of paper, sawdust, or even leftover powder from sweepings can ignite with almost no warning. Portable work surfaces and disposable gloves make direct contact less likely. Handle everything on trays that get cleaned before and after every use, and store away from anything that might catch fire easily, including cardboard and cleaning rags.
Emergency Measures Should Never Be Afterthoughts
Having emergency gear nearby can truly save the day if something unexpected happens. Fire extinguishers rated for oxidizers, spill acid neutralizer, and safety goggles belong within arm’s length. Staff and frequent users need to run through practice drills, not just read the safety poster pinned to the wall. In a real emergency, muscle memory always beats a quick search for the manual.
Regulated Storage and Disposal
States and many countries have strict rules about how chemicals are stored and disposed of. There’s a good reason: improper handling turns a safe compound into a public hazard. It’s best to work with certified hazardous waste contractors who understand every detail, from sealed containers to paperwork. Some local and school authorities offer safety courses or consultations, a valuable resource for smaller organizations that may not have a full-time safety officer.
The Value of Reliable Information and Training
Even though instructions come with every shipment, they never replace the value of steady, updated training. Online databases from respected safety organizations provide real-life case studies, best practice checklists, and updates about material safety data sheets. Community colleges often offer annual refreshers, which help reinforce good habits. Relying on word-of-mouth or DIY videos often leads to overlooked steps that multiply risks.
Safer Science and Smarter Practice
Working with copper chlorate should build respect for chemical safety into every routine. By choosing strong containers, keeping a clean workspace, using the right labeling, and always expecting the unexpected, accidents become rare. It’s not about fear; it’s about building habits that last and protect lives, property, and the wider community.
Can Copper Chlorate be purchased for laboratory use?
The Realities Behind Buying Copper Chlorate
Walking into a well-equipped lab, I’ve always noticed shelves lined with reagents like copper sulfate, copper(II) chloride, and other common chemicals. Even in specialist academic labs, copper chlorate rarely appears among the stock bottles. There’s a solid reason for that—copper chlorate brings a mix of attractive chemistry and heavy regulation.
Ground Rules: Regulations and Hazards
Copper chlorate isn’t your everyday copper salt. It is an oxidizer, reactive and unpredictable in the wrong setting. Years ago, I watched a demonstration where a small pinch spilled and dried—its sensitivity shocked me. These risky properties catch the eye of regulators everywhere. In most countries, strong oxidizers face restrictions aimed at reducing accidents and deterring misuse, especially since chlorates can play a role in making explosives.
These controls aren’t loose. In the United States, the DEA and the Department of Homeland Security carefully watch the sale and distribution of oxidizers with profiles like copper chlorate. Retailers hold back unless orders come from traceable, authorized users—think legitimate research labs, not hobbyists or teachers. Licenses, proof of purpose, and tight documentation stay part of the process. Europe keeps similar stances. REACH regulations and specific national measures ask for paperwork that goes beyond a mere invoice.
Availability for Research: Who Gets Access?
Most scientific suppliers, like Sigma-Aldrich or Fisher Scientific, won’t stock copper chlorate for open purchase. Researchers working on oxidation reactions or studying metal salts often improvise with more stable compounds. If your lab project needs a strong oxidizer, alternatives like potassium chlorate or even peroxides often step in where copper chlorate would otherwise serve. Institutions that really need copper chlorate, for example to investigate thermal decomposition pathways, almost always synthesize it for immediate use rather than order it.
In my own research experience, requests for restricted chemicals meant weeks of paperwork and sometimes outright rejection. Some chemicals add layers of liability many universities or labs don't want. That reality slows down progress in some areas but keeps people safe.
Looking for Solutions: Safer Substitutes and Lab Protocols
Chemistry doesn’t hit a brick wall here. Colleagues working in energetic materials have developed a knack for using less hazardous chemicals, thanks to decades of tough regulations. Safer oxidizers, milder reaction setups, and more robust lab training all keep science moving ahead. Online communities—even those from open science backgrounds—talk more about alternatives and safer synthesis pathways than about purchasing the original material.
In classrooms and teaching setups, teachers demonstrate principles with household items or low-risk reagents, steering clear of chemicals like copper chlorate entirely. That approach pays off, lowering accident rates without dulling the spark of curiosity for young scientists.
The Takeaway: Weighing Safety Against Curiosity
Strict controls on copper chlorate draw a clear line between curiosity and safety. Labs that want a chemistry set from the golden age of research won’t find this compound lying around. The focus moves from chasing the purest reaction to building a responsible, safety-minded lab culture—one that can meet tomorrow’s scientific problems without unnecessary risk.
| Names | |
| Preferred IUPAC name | Copper(II) chlorate |
| Other names |
Chloric acid, copper(2+) salt Copper(II) chlorate Cupric chlorate |
| Pronunciation | /ˈkɒpər ˈklɔːreɪt/ |
| Identifiers | |
| CAS Number | 13477-10-4 |
| Beilstein Reference | 415872 |
| ChEBI | CHEBI:85155 |
| ChEMBL | CHEMBL501197 |
| ChemSpider | 159411 |
| DrugBank | DB11000 |
| ECHA InfoCard | 100.025.428 |
| EC Number | 231-847-6 |
| Gmelin Reference | Gmelin Reference: **1178** |
| KEGG | C18703 |
| MeSH | D003985 |
| PubChem CID | 24565 |
| RTECS number | GB9625000 |
| UNII | 12MVR71K9Q |
| UN number | UN1468 |
| Properties | |
| Chemical formula | Cu(ClO3)2 |
| Molar mass | 247.00 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | Density: 2.28 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -2.45 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.98 |
| Basicity (pKb) | <0.5 (strong base) |
| Magnetic susceptibility (χ) | +76.0·10⁻⁶ |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -534.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB47 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes burns, environmentally hazardous |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | H271, H302, H332, H410 |
| Precautionary statements | P210, P220, P221, P260, P264, P273, P280, P283, P305+P351+P338, P306+P360, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-0-3-OX |
| Flash point | No flash point |
| Lethal dose or concentration | LD50 (oral, rat): 105 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 782 mg/kg |
| NIOSH | GL8575000 |
| PEL (Permissible) | PEL for Copper Chlorate: Not established |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Copper(I) chlorate Copper(II) sulfate Copper(II) nitrate Copper(II) chloride Chloric acid |

