Copper Arsenate: A Closer Look at an Old and Controversial Compound
Historical Development
Copper arsenate tells a story that stretches back centuries. Early chemists reached for minerals laced with copper and arsenic, noticing their vibrant colors and strong effects on fungi and pests. By the late 1800s, copper arsenate emerged in orchard management as one of the first chemical answers to insect infestations plaguing apple and pear growers. Evidence from agricultural bulletins of the early 1900s shows that specialists in the United States Department of Agriculture distributed guidance on safe and effective use. In my own research digging through old county extension reports, references to copper arsenate often appear beside Bordeaux mixture and Paris green as part of a trio of hard-hitting agents in orchard and vineyard care. Pesticide historians point to these compounds as the birth of industrial-scale agriculture’s chemical era, for better or worse.
Product Overview
Many years ago, suppliers sold copper arsenate mainly as a fine powder or in coarse granules, packaged in drums or sturdy paper sacks. Its main draw wasn’t just the ability to kill pests notoriously resistant to other measures, but also its resilience to rain and sun. Applicators found it stubbornly stuck around, which could be a blessing or a curse depending on your view of chemical residues. Convenience and price kept demand steady until environmental and health questions mounted.
Physical & Chemical Properties
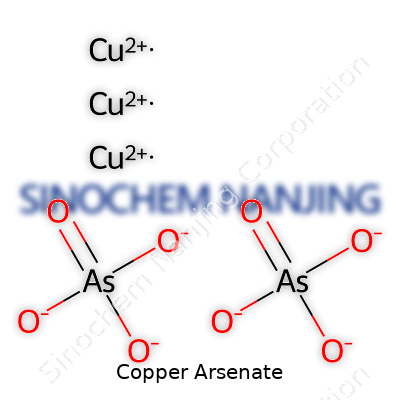
Commercial copper arsenate usually comes as a blue-green solid. Chemically, it most often takes the form Cu3(AsO4)2, though mixtures contain tweaks and impurities. It doesn’t dissolve easily in water at room temperature, which means it tends to stay where it’s applied and avoids quick runoff, a feature growers once liked. Its density sits around 4 grams per cubic centimeter. The color earned it a popular spot as a pigment in ceramics and glass before its true toxicity became more widely recognized. In the lab, heating copper arsenate produces arsenic trioxide and copper oxide, both hazardous. Exposure to acids drives off dangerous arsine gas, meaning storage and handling call for care and experience.
Technical Specifications & Labeling
Specifications for copper arsenate in industry focus on copper content, arsenic purity, moisture levels, and potential trace contaminants such as lead or cadmium. Packaging always includes strong warnings—skull-and-crossbones icons, statements about acute toxicity, and precise information about required personal protective equipment. I’ve handled safety data sheets labeling it as an acute oral and inhalation hazard, with guidelines on safe storage, spill response, and first aid. Any facility handling this material posts detailed instructions on wall placards and keeps antidotes and emergency wash stations at hand.
Preparation Method
Manufacture involves reacting a soluble copper salt like copper sulfate with sodium arsenate or arsenic acid in water. Under careful control of temperature and pH, copper arsenate precipitates out, and operators filter, wash, and dry the solid compound. This process, whether adjusted for batch size or industrial flow, calls for strict engineering controls—ventilation, dust suppression, and real-time monitoring of arsenic vapors. I’ve seen processes where even a slight drift in pH changes product purity, so technicians keep close tabs on every stage.
Chemical Reactions & Modifications
Copper arsenate reacts with acids to release arsenic compounds, some of which become airborne and highly toxic. In the environment, soil microorganisms eventually break down the compound, turning arsenic into less or more mobile forms depending on conditions. Over time, researchers found ways to change its crystal form to tweak how long it persists and how effectively it sticks to leaves or wood. Some modified products swap out one copper salt for another or add stabilizers, hoping for less leaching and stronger biological action.
Synonyms & Product Names
This compound goes by several names—tribasic copper arsenate, copper(II) arsenate, and basic copper arsenate mainly. Historical trade names pop up in pesticide catalogs and archives, such as “Paris Green,” though purists separate that pigment from pure copper arsenates used as insecticides. In industrial supply lists, I’ve found references to “CCA” when copper arsenate combines with chromate in wood preservatives.
Safety & Operational Standards
Handling copper arsenate brings tight regulation. Workplaces keep it locked behind secured storage, with authorized access only for trained staff. Regulations from organizations like OSHA and the EPA in the US lay out required protective gear, air quality testing, and disposal protocols for both waste and contaminated equipment. Workers wear respirators, gloves, and full-body suits. Regular medical screenings form part of occupational health programs. Community health studies from orchard regions in the 1960s drove calls for stricter enforcement, especially where groundwater arsenic contamination appeared.
Application Area
Copper arsenate found a niche in pest management and wood preservation. Orchards relied on it for decades against caterpillars, leafrollers, and beetles that scoffed at less potent sprays. Pressure-treated lumber soaked in copper arsenate, especially when combined with other chemicals in “CCA,” protected fenceposts and railroad ties from rot and termites, making wooden structures last decades longer in rain and sun. Its effectiveness earned it wide adoption until concerns about persistence and leaching led regulators to phase out many uses. All the while, home gardeners, municipal groundskeepers, and railroad companies counted on its potency, often without realizing the long-term environmental fingerprints.
Research & Development
Pressure to find safer pest control led chemists and environmental scientists to compare copper arsenate with newer organophosphates and pyrethroids. Studies tracked residue breakdown under different soil conditions and the movement in water tables. Modifications to the formula, sometimes through microencapsulation or binding agents, attempted to trap arsenic close to application zones. Testing for improved uptake by insects versus leaching to groundwater cropped up in academic journals, with mixed results. A fair bit of research explored biological alternatives, but even now, no simple, universally effective organic solution replaces the spectrum of pests copper arsenate once managed.
Toxicity Research
Copper arsenate spells serious risk. Chronic arsenic exposure links to skin, lung, and bladder cancers, along with vascular disease and neurodevelopmental impacts. Studies in the mid-1900s saw children in agricultural towns with elevated urinary arsenic. Wildlife researchers found bird and fish populations suffered near treated sites, accumulating arsenic well above safe levels. Copper on its own tends to accumulate in river sediments, adding further complexity. Medical professionals stress that ingestion, inhalation, or skin contact can provoke acute reactions—vomiting, diarrhea, circulatory collapse—while slow, low-level exposure builds up trouble silently. Regulatory science, tasked with weighing agricultural benefit versus public risk, swung away from tolerance toward outright bans and strict controls as evidence piled up.
Future Prospects
Most countries sharply restrict copper arsenate use, but the search for less hazardous alternatives remains tough in regions battling wood rot, termites, and resistant pests. Research continues into controlled-release formulations, recycling or detoxifying contaminated soils, and closing the loop on wood waste from old structures. Scientists look at plant-derived pesticides, breeding disease-resistant crops, and bio-based wood treatments. The challenge isn’t just efficacy—it’s about replacing copper arsenate without trading one hazard for another. I’ve seen some hope in bioremediation, with engineered bacteria breaking down arsenic pollution, though scaling up remains daunting. Many public health advocates push for continued monitoring at old use sites and investment in cleanup technology to address the compound’s stubborn legacy.
What is copper arsenate used for?
Why People Still Talk About Copper Arsenate
Copper arsenate never pops up in the news without raising eyebrows. Anyone growing up near farms or in neighborhoods bordered by wooden playgrounds probably heard about it. This chemical doesn’t sound like something you’d toss around the garden, but it played a big role in farming and home construction for decades. People trusted it to protect wood from bugs, rot, and fungus. Walk through any older American park or examine vintage fencing, and you might spot wood treated with this powerful mixture.
From Orchards to Fences: Real-Life Uses
I remember seeing labels on old green-shaded decks that warned against burning or sawing treated wood—the culprit was often copper arsenate or its cousin, chromated copper arsenate (CCA). Lumberyards liked it because the mix kept termites and mold away. In the mid-1900s, copper arsenate earned a spot in apple orchards and grapevines, sprayed to fight leaf munchers and tree diseases. Schoolyards and farm supply sheds used it for the same reason. Its staying power in wood products made every nail and board in playgrounds a little less tasty to nature’s destroyers.
Copper Arsenate’s Risky Side
Some facts can’t be ignored. Arsenic, part of the mix, builds up in soil and water. Years ago, folks didn’t worry as much about long-term health. Today, studies point to real dangers. Arsenic leaches from treated wood into the ground. It lingers in dust and even in vegetables grown nearby. The Environmental Protection Agency (EPA) stepped in after reports linked treated wood to higher cancer risk and nervous system issues, especially in kids. It’s easy to forget about a chemical after it falls out of favor, but it doesn’t just disappear when regulations change. I’ve seen community boards debate whether to tear out old playgrounds for this reason.
What We Do Now Matters
Copper arsenate isn’t banned everywhere. Some places use it for industrial purposes, though it has left most residential hardware stores. As someone who has handled both new and salvaged wood, I know those reminders about gloves and masks aren’t just legal talk—they matter. Washing hands after working with old decks or removing treated posts isn’t being overly cautious; it’s just good sense.
Looking for Alternatives—and Safer Future Choices
Safer wood treatments exist. The rise of ACQ (alkaline copper quaternary) and other copper-based preservatives points to big changes in the building industry. Growers and homeowners now tap into natural oils and biological options to guard against pests. These newer products, while not perfect, reduce risk for families and help keep soil cleaner. Switching to safer alternatives costs a little more, but no one wants to trade quick fixes for years of health worries. People want to enjoy their gardens, yards, and parks without thinking twice about what’s under their feet.
Building Trust with Clear Information
Knowing the history of copper arsenate makes it easier to ask good questions before picking up any “pressure treated” wood or pest control product. Reading up, asking suppliers about safer picks, and sharing what you learn keeps everyone in the loop. Trust grows from straight talk and the willingness to look out for neighbors. Copper arsenate’s story isn’t just about chemistry; it’s about the ways communities protect themselves by making thoughtful choices.
Is copper arsenate toxic to humans or animals?
Why People Still Talk About Copper Arsenate
Copper arsenate usually pops up in conversations about pesticides or wood preservatives. It’s been around for decades. Its vivid green color once made it the darling of fence posts, decks, and even some farm fields. I remember, as a kid, seeing old lumber in the backyard painted that telltale green, my parents reminding me not to touch it too much. Back then, nobody really seemed to worry about what was inside the wood.
The Real Risks to Humans and Animals
Copper arsenate deserves a tough look. Both copper and arsenic can hurt people, but arsenic steals the spotlight in terms of risk. This compound acts as a poison for insects and fungi, but it also threatens humans and animals who get too close. Breathing in sawdust from treated wood, touching the surface with bare skin, or burning it in a backyard bonfire can all cause problems. There’s plenty of proof: arsenic leaches from wood, it contaminates soil and water nearby, and it works its way up the food chain.
The body doesn’t recognize arsenic, and it doesn’t have a way to handle big doses. Once inside, arsenic latches onto proteins and enzymes, slowly grinding down organs like the liver and kidneys. Over months or years, small exposures might show up as nausea, skin rashes, or brain fog. Stronger, more direct exposures can land someone in a hospital bed within hours. Animals roam outside, digging or chewing and sometimes pick up the dust or chips, putting them at risk, too.
What Science Says About Long-Term Exposure
Look at studies from the World Health Organization, Centers for Disease Control, and cancer researchers: they point to real problems. Chronic exposure ties in with skin problems, heart issues, and several types of cancer. Some studies link low doses over the years to developmental problems in children. Animals exposed regularly start to show changes in behavior, clouded eyes, and trouble breathing. These aren’t just headlines—these are real families, farmers, and pet owners.
Turning Concern into Action
We need tougher rules for safe disposal and handling. Many countries already block new use of copper arsenate, but there’s still plenty of old lumber out there in workshops and barns. Anyone working around it—home renovators, landscapers, parents building playhouses—deserves to know the risks and take precautions. Wearing gloves, masks, and keeping scraps away from pets can cut down on accidents.
Old habits die hard. Some folks argue that treated wood lasts longer and keeps termites away. The problem is that the price for longevity shouldn’t come at the expense of future health. Alternatives exist now: safer wood treatments or composite materials that don’t need harsh chemicals to stay strong. Building codes have started to change, nudged by research and a growing pile of evidence from real communities who have seen copper arsenate’s harm up close.
Clear labeling, honest education, and better disposal sites should top any action plan. Local governments could offer safe collections for old treated wood. Hardware stores might train staff to talk about safe alternatives. Everyone who might deal with this compound deserves more than a warning sign—they deserve a safer environment for families and animals.
What safety precautions should be taken when handling copper arsenate?
Understanding the Risks
Copper arsenate isn’t the kind of thing anybody should treat lightly. It’s a chemical that has played a role in wood preservation and pest control, but it carries high risks. Exposure brings health dangers, from skin irritation and respiratory trouble to long-term issues like cancer and organ damage. Having studied and written about chemical hazards, I’ve seen how even a small slip with a toxin like this can turn into a life-changing event. That reality makes safety non-negotiable.
Personal Protective Gear
Working with copper arsenate demands gear that actually stands up to the job. Gloves made from nitrile or neoprene shield skin from contact. A splash apron or heavy-duty lab coat keeps clothes from getting contaminated. Safety goggles, not regular glasses, keep particles and splashes away from your eyes. A proper mask, such as an NIOSH-approved P100 respirator, protects your respiratory system, especially if there’s a chance of dust. Based on experience, cutting corners with gear lands people in emergency rooms more often than most realize.
Planning the Workspace
Never use copper arsenate in a closed-off, poorly ventilated spot. An outdoor area with moving air or a laboratory setting with a fume hood will help. Keeping kids, pets, and food far from the work area seems basic, but accidents tend to happen when these steps get skipped. With arsenic compounds, no one wants to find out after the fact that a little dust or a contaminated glove found its way to the wrong place.
Handling and Storage
Keeping copper arsenate in original, clearly labeled, tightly sealed containers makes tracking easy and reduces the risk of accidental mix-ups. I’ve seen what happens when someone puts chemicals into old drink bottles—confusion, danger, and sometimes tragedy. Store these chemicals in a locked cabinet, away from heat, water sources, and living spaces. Only trained people should have access—keeping keys out of reach can stop a serious accident before it starts.
Cleaning and Disposal
Any spill should get handled on the spot using a HEPA-filter vacuum or wet methods, never sweeping or dry brushing. Wet paper towels can stop dust from going airborne. Always discard waste in hazardous material containers, following state or local hazardous waste guidelines. Never wash spills down the drain. In the past, I’ve seen costly environmental cleanups caused by shortcuts during disposal. Responsible handling also protects the water supply and wildlife.
Personal Hygiene Practices
Washing hands and forearms with soap and warm water right after handling copper arsenate makes a big difference. Waiting until later, or skipping a good wash, tends to transfer toxins onto doorknobs, phones, and eventually food. Change clothes before heading home and keep work clothes separate from everyday laundry. These small habits protect families from invisible harm.
Education and Training
No one learns how to use dangerous chemicals safely by accident. Regular training refreshes good habits and builds respect for the risks. Posters, quick safety talks, and real stories from people who’ve been through incidents keep information sticky. Even pros sometimes forget, so updated training saves lives and health.
Smart Solutions in Policy and Technology
Push for safer alternatives when possible. Many organizations have replaced copper arsenate with less harmful options. Support for clear labeling, better storage, and stricter regulation keeps everyone safer. Advocacy from scientists, health professionals, and workers matters—better rules lead to fewer emergencies and healthier communities.
How should copper arsenate be stored and disposed of?
Why Copper Arsenate Matters in Safety
Copper arsenate sits among those chemicals you can't just toss aside without some thought. It’s often used in pesticides and wood preservatives, but this combination of copper and arsenic spells high toxicity for people, animals, and water systems. Growing up near an orchard, I saw barrels of old chemicals stored in forgotten sheds—an unsafe habit, especially for something as dangerous as copper arsenate.
Storage: Think Clean, Dry, and Secured
Keeping copper arsenate in check starts with solid storage habits. Forget the idea of just stacking it on open shelves in the barn or basement. You want a storage space that stays dry year-round. Moisture makes the chemical more likely to leak or break down, and those leaks can mean serious health risks.
People sometimes think a locked toolbox or rusty cabinet is good enough. That’s risky and short-sighted. Secure storage means a locked cabinet or building designed for hazardous materials. This should sit away from the living spaces and far from food, animal feed, and water sources. Signage really matters—bold “Danger: Toxic” warnings keep accidental visitors from harm and reinforce safety routines. Growing up, I watched how even clear signals could stop a curious child or neighbor from making a dangerous mistake.
Keep Track: Inventory and Label Everything
To stay organized, a simple log of how much copper arsenate you keep in storage—and where—can save headaches. Date every container. Check often for rust, cracks, or leaks. It’s tempting to ignore this, but a small crack today can lead to a costly spill tomorrow. Clear labeling prevents mix-ups, especially if you ever have employees or helpers around.
Real Risks: Exposure and Environmental Impact
Arsenic turns up in drinking water, blood, and soil after spills—no one wants that in their garden or community. Long exposure increases cancer risk, harms nerves, and damages the liver. It also sticks around in the environment for years, so mistakes last beyond your own lifetime.
Disposal: Don’t Cut Corners
I’ve seen local stories where leftover pesticides got dumped in ditches or burned. That’s illegal and dangerous; arsenic doesn’t just vanish with heat or sunlight. The Environmental Protection Agency and state health departments list copper arsenate as hazardous waste. Dumping in the trash or pouring down the drain isn’t safe.
The right approach involves working directly with certified hazardous waste disposal services. Most counties host occasional collection days where you drop off chemicals for professional handling. You’ll need to transport it in sealed, original (or compatible) containers—no homemade fixes. Driving with copper arsenate in the trunk means planning the route, tying everything down tightly, and wearing gloves and goggles.
Reducing Waste for the Future
If storing copper arsenate, buy the smallest amount you need. Avoid stockpiling. Look for alternatives where possible. Many wood treatments and pest controls use less toxic ingredients—these can tackle the job without as much hassle. Less hazard on-site means less risk for everyone, from workers to neighbors.
Community Responsibility
Too many communities suffer after someone carelessly stores or dumps toxic chemicals. If you use copper arsenate, your choices ripple outward. Take time to read official guidance and talk to professionals. Local extension offices, poison control, and environmental agencies offer advice anytime. Teaching one more person about proper storage and disposal helps another family avoid the costs of a spill or exposure.
Are there any regulations or restrictions for using copper arsenate?
Looking at the Facts
Copper arsenate popped up years back as a powerful wood preservative. It keeps termites, fungi, and rot at bay. Folks found it in fence posts, decks, even children’s playgrounds before anyone realized the health problems arsenic could stir up. I remember when everyone just wanted sturdy wood, not thinking twice about what chemicals soaked into it. But that’s the thing with chemicals—eventually, the toll on people and on the backyard environment shows up.
Why Regulations Got Tight
Copper arsenate shifts from a helpful barrier against decay into a hazard for families and wildlife because arsenic doesn’t stay put. Rain or direct touch exposes people to toxic dust or splinters. Studies linked arsenic to cancers and neurological issues. US Environmental Protection Agency (EPA) and similar groups overseas couldn’t just look away. In 2003, the US sharply limited its use, blocking its sale for most residential purposes, especially on anything kids might touch. Canada, Australia, and countries across Europe rolled out their own bans or strict rules. Trying to avoid playground nightmares, regulators demanded safer alternatives, and companies made the switch to less risky wood treatments.
Who Still Handles Copper Arsenate?
Copper arsenate hasn’t vanished from every industry. Railroads and utilities still buy treated wood for poles and cross ties buried out in isolated spots, where maybe an old fence needs to last fifty years and no kids are climbing on it. The difference these days is rules call for heavy gloves, face protection, sealed containers—the works. Workers have to handle every stage with safety plans. Labels on the product spell out don’t-breathe-it, don’t-burn-it, don’t-dump-it instructions. If someone wants to get rid of treated lumber, the town dump won’t take it with household trash. Landfills that take hazardous waste have learned to treat it as a toxic material.
Why These Limits Matter
Trouble comes easy with something as potent as arsenic. Chronic exposure doesn’t always show up right away. It sits quietly in soil or leaks into groundwater, becoming a bigger worry for children and pets digging around in yards. I’ve seen public outcry push local lawmakers into action after high arsenic levels cropped up near schools or parks. Responsible stewardship means thinking ahead about what’s left behind for future generations. Science keeps finding new connections between chemical exposure and long-term ailments.
What Can Be Done?
Communities work best when folks know the risks and speak up. Building supply stores now carry alternatives like borate-treated lumber or composite materials that don’t leach heavy metals. Homeowners still holding onto old railroad ties for gardens may find it worthwhile to swap them out and get soil tested. Municipalities can educate on safe disposal, post clear signs about avoiding burning or chipping treated wood, and enforce rules at the landfill. Builders and contractors can choose green-certified products. All these changes add up, preventing a quiet killer from working its way into daily life.
Staying Informed Gives Everyone an Edge
Regulations around copper arsenate reflect a hard lesson learned over decades. Good science and strong voices in public health worked together for these changes. The story isn’t just about stopping a dangerous chemical; it’s about paying attention to what gets used in communities and making sure the cure never turns into a new problem.
| Names | |
| Preferred IUPAC name | tricupric arsenate |
| Other names |
Copper(II) arsenate Cupric arsenate Arsenous acid, copper(2+) salt Copper diammonium arsenate |
| Pronunciation | /ˈkɒpər ɑːˈsɪneɪt/ |
| Identifiers | |
| CAS Number | 12002-67-6 |
| Beilstein Reference | 35874 |
| ChEBI | CHEBI:78744 |
| ChEMBL | CHEMBL1201642 |
| ChemSpider | 20538 |
| DrugBank | DB11369 |
| ECHA InfoCard | 100.961.226 |
| EC Number | 266-898-5 |
| Gmelin Reference | 11337 |
| KEGG | C18797 |
| MeSH | D003822 |
| PubChem CID | 25157 |
| RTECS number | GB6475000 |
| UNII | 96H8NH5S3T |
| UN number | UN3077 |
| Properties | |
| Chemical formula | Cu3(AsO4)2 |
| Molar mass | As2Cu3O8: 453.63 g/mol |
| Appearance | green powder |
| Odor | Odorless |
| Density | 3.74 g/cm³ |
| Solubility in water | Insoluble |
| log P | -2.34 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.1 |
| Basicity (pKb) | 11.54 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Dipole moment | Zero Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 180.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -836.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB44 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or absorbed through skin; causes skin and eye irritation; may cause cancer; very toxic to aquatic life. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H331: Toxic if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P201, P202, P260, P264, P270, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P314, P330, P362+P364, P391, P405, P501 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD₅₀ (oral, rat): 8 mg/kg |
| LD50 (median dose) | LD50: 40 mg/kg |
| NIOSH | AS8700000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Copper Arsenate: "0.01 mg/m³ (as As) |
| REL (Recommended) | 0.01 mg/m3 |
| IDLH (Immediate danger) | 40 mg As/m3 |
| Related compounds | |
| Related compounds |
Copper(II) arsenite Copper(II) sulfate Copper(II) phosphate Lead arsenate Zinc arsenate |

