Copper Acetylarsenite: A Closer Look at Scheele’s Green
Historical Development
Few chemical compounds have a story as tangled as copper acetylarsenite, better known by its trade name, Scheele’s Green. Back in the late 18th century, Carl Wilhelm Scheele developed this distinctive green pigment almost by accident. Artists, wallpaper makers, and textile dyers flocked to it because no other green produced quite the same pop or stayed as vibrant. In those days, few people worried about what went into dyes and paints, as long as they looked good and lasted. Demand surged, and manufacturers churned out boats of the stuff. But grim fates often lingered behind the colors people craved. By the late 19th century, as more folks fell ill from arsenic poisoning—sometimes just from living with wallpaper or curtains—the pigment’s fame turned to notoriety. Folks started asking questions, and gradually, the hazards this chemical carried into homes and workplaces came into focus.
Product Overview
The color of copper acetylarsenite lands somewhere between emerald and apple, with a slight blue tinge that stands out in both thick paint and thin glazes. At its peak, decorators and artists couldn’t get enough. From fancy carriages to theater sets, anything meant to impress looked better with a coat of this green. Paint makers blended it into oil and water-based paints. Because it cost less than some other pigments, penny-pinchers and luxury buyers alike found places to use it. As people caught on to the dangers, demand dried up, although a niche crowd of restoration artists, chemical historians, and toxicologists still need the real thing for their work.
Physical & Chemical Properties
Copper acetylarsenite comes as a fine, almost velvety powder. The pigment feels substantial in your hand, heavier than you would expect from such a delicate appearance. Its chemical formula, CuC2H3AsO3, tells you that copper, arsenic, carbon, hydrogen, and oxygen combine in an unusual way. In water, it does not dissolve easily, helping it stick around on painted surfaces. Long sunlight exposure fades its intensity, especially in outdoor murals or poorly protected objects. Heat, strong acids, or bases can break it down, often releasing sharp-smelling arsenic fumes that threaten anyone nearby.
Technical Specifications & Labeling
On labels and technical documents, copper acetylarsenite usually turns up as Scheele’s Green, Paris Green in some trade circles, or by its chemical make-up. Regulations enforce clear labeling due to the severe toxicity. Transport trucks need placards for hazardous materials. Chemical catalogues specify minimum purity standards, batch numbers, and recommended storage temperatures—usually cool and dry. Internationally, the label bears the skull-and-crossbones pictogram and dire warnings, not just for handlers but for anyone in the supply chain. For research or historic purposes, buyers must show permits and meet strict requirements before receiving a shipment.
Preparation Method
Manufacturing the pigment in its original method involves reacting copper(II) sulfate with a mixture of sodium arsenite and acetic acid. Sometimes, labs swap in slightly different starting materials, but the key is always to coax out the bright, crystalline green solid. The process pumps out a significant amount of toxic waste. Modern labs that make it—for museum pieces, forensic tests, or archival research—work behind thick barriers with robust ventilation. Waste byproducts are neutralized and disposed of as hazardous material. Technicians keep records accounting for every gram of arsenic, and by law, must wear protective gear, gloves, and filtered masks.
Chemical Reactions & Modifications
Introduce acids to copper acetylarsenite, and you set off a reaction that splits it apart. The pigment starts to brown and releases arsenic-containing gases, sometimes invisible and always dangerous. Mixing with bases alkalizes the copper, destabilizing the compound even further. In my own undergraduate chemistry days, just a stray whiff from a sample ruined an afternoon and left the lab smelling like garlic for hours. For those who experiment with trying to tweak its properties, substitutions with similar transition metals or slight changes in the salt components produce a range of green hues, but none has ever matched the original’s odd brilliance—or its level of risk.
Synonyms & Product Names
Scheele’s Green and copper acetylarsenite are not the only names in circulation. Through history, it has shown up as Swedish Green, Schloss Green, and Imperial Green. Chemically, its registry number is recognized across international chemical indices, and older shipping manifests still call it “arsenite of copper.” For folks researching the pigment’s past, it pays to know a half-dozen aliases to track down old studies, patent filings, and safety alerts.
Safety & Operational Standards
Safety standards around copper acetylarsenite operate with a zero-tolerance mindset for shortcuts. In workplaces, engineers retrofitted storage areas with negative air pressure and sealed-off transfer points long before digital controls were the norm. Chemical suits keep particles off skin. Emergency showers and eye-wash stations are never out of arm's reach. Regulatory agencies like OSHA and the European Chemicals Agency bar the pigment from nearly all consumer uses. Where handling persists, air and surface monitoring runs in the background, and periodic health checks for arsenic build-up in workers catch trouble early. Disposal laws treat waste from copper acetylarsenite as acutely toxic—containers go to special incinerators, and liquids get chemically neutralized before release.
Application Area
The heyday of Scheele’s Green stretched from wallpaper factories and print shops to art studios. Glorious Victorian patterns relied on it, as did some confectioners before the dangers were clear. Only after repeated poisonings—documented in coroner’s reports and stories of mysterious illnesses—did governments intervene. These days, you’re not going to find copper acetylarsenite in your home or local crafts store. Its domain has narrowed to the world of art restoration, scientific specimen work, and forensic research. Any commercial application faces so many barriers, both from a supply chain and safety perspective, that safer pigments win out almost every time.
Research & Development
Even now, researchers dig into the history and chemistry of copper acetylarsenite. Museums depend on forensic chemical tests to authenticate old paintings and textiles. Chemists look for subtle markers indicating if a green pigment is the original Scheele’s mix or a low-arsenic modern substitute. Toxicologists still cite it as a case study for regulatory reform. Occasionally, academic groups experiment with encapsulating the pigment, trapping the powder in microbeads to allow safe handling for teaching or exhibition. These projects challenge clever minds to make the stuff less dangerous, to learn from the past and protect people who still need to work with it.
Toxicity Research
Copper acetylarsenite’s toxicity is no secret. Even in the old days, folks suspected something foul when children, pets, and even family breadwinners took sick for no obvious reason. Modern toxicology leaves little to mystery—arsenic disrupts cellular function, damages nerves, and in high enough doses, stops vital organs. Inhaled dust, skin contact, or accidental ingestion can trigger symptoms ranging from nausea to organ failure. Chronic exposure, even at low levels, raises cancer risk and damages tissues throughout the body. Countless rat, mouse, and even plant studies over two centuries confirm the risks don’t fade with age. Emergency response teams drill on decontamination and rapid treatment—in my own fieldwork, I have watched doctors act quick on arsenic cases, knowing every minute wasted means longer recovery or worse.
Future Prospects
Scheele’s Green has lost its place on the shelves, but its legacy shapes how industry manages and bans hazardous substances. The pigment serves as a cautionary tale for new materials science—beauty does not outweigh safety. Chemists and regulators take lessons from its story, building stronger safety nets for other toxic substances before they slip into mass use. In specialty circles, folks keep copper acetylarsenite alive for training, research, and historical conservation, always under strict control. The compound’s future lies in teaching, not in trade: showing why due diligence and clear-eyed risk assessments save lives. So, even if the paint cans stay closed, its place in chemical history is secure—and its lessons won’t fade, even if the color finally does.
What is Copper Acetylarsenite used for?
Origins and Recognition
Copper acetylarsenite, people might recognize it by another name: Paris Green. Artists and day laborers alike once mixed or brushed this powder with little thought about what it could do. It’s a green pigment, bright and misleadingly beautiful. In paint, it offers intensity that few natural colors match. Its tone lights up museum canvases and 19th-century Victorian wallpaper, drawing eyes before any warning bells chime.
Uses in Art and Beyond
For painters, Paris Green carried status and elegance. It made landscapes lush and clothing vibrant. You can find it on the delicate textures of Cézanne or Monet. Artists say pigment choice shapes mood, and this one draws attention through its punchy green. Collectors might marvel at how some works retain their glow centuries later, thanks in part to copper acetylarsenite’s chemical stability—at least when left undisturbed behind glass.
From Paints to Pests
Art history is just the start. Early chemists and farmers latched onto Paris Green for another reason: its effectiveness as a pesticide. Malaria’s threat loomed heavy in hot, stagnant summers, so people dusted fields and swamps with copper acetylarsenite to keep mosquitoes away. Orchards, in particular, saw heavy use. Grape growers in France spread the powder, hoping to save crops from insects. The green would coat vines—a line of defense strong enough to bring hope for big harvests.
Dangers and Realizations
It’s impossible to talk about copper acetylarsenite without bringing up the shadows. As a compound laced with arsenic, its toxicity affects both the environment and human health. Factory workers and artists struggled with chronic illness, or worse. Victorian families hung wallpaper colored by Paris Green and didn’t realize damp air and warmth would send arsenic gas leaking from the walls. Some blame acute poisonings and mysterious deaths on this chemical, especially in homes where ventilation failed or dust crept into teacups and food.
Those experiences taught a hard lesson about the gap between beauty and safety. The dangers of copper acetylarsenite also show up in local water contamination when used as a pesticide, as arsenic compounds linger in soil and pose risks to food chains. Medical science now connects chronic low-level exposure to various cancers, nerve damage, and other serious health conditions.
Accountability and Solutions
It’s clearer today how important transparency and regulation are. Governments now restrict or outright ban copper acetylarsenite for consumer goods and pesticides in most countries. Artists and preservationists use substitutes that avoid arsenic’s risks, protecting creators and those who appreciate art. On farms, attention shifts to biological controls and integrated pest management, relying on knowledge drawn from decades of cautionary stories.
When museums and restorers handle historic pieces, strict procedures kick in, including filtered ventilation and protective equipment. Expert conservators raise awareness among the public who visit galleries, making health education part of the experience. On a personal note, seeing the shimmering green of these old works always leaves me conflicted—awed by human ingenuity, but sobered by the history behind the pigment.
Is Copper Acetylarsenite toxic or dangerous?
What is Copper Acetylarsenite, and Why Do People Talk About Its Dangers?
Copper acetylarsenite, known to many as Paris Green, carries a reputation that still echoes through history books and science textbooks. In the 19th and early 20th centuries, folks turned to it for everything from vibrant green paint to insecticide. Homes and gardens sported its trademark hue. What they didn’t always realize: this compound packs a heavy dose of poison.
The Risks Hidden in that Bright Green Powder
Copper acetylarsenite combines copper with arsenic—two elements you don’t want in your body. Arsenic is notorious for its ability to damage nearly every system in the body. Swallow a small amount, inhale the dust, or even let it sit against your skin, and trouble isn’t far behind. People started learning about its dangers the hard way. Chronic arsenic exposure links to skin lesions, cancer, and organ damage. Even a modest exposure can trigger nausea, skin irritation, and more serious symptoms over time.
The copper part only adds another layer. In healthy amounts, copper keeps nerves and blood vessels working. Too much, though, and it turns into a toxin, especially for the liver and kidneys. Combine these two, and you get a chemical that doesn’t just color your walls—it risks your health in ways nobody can afford to ignore.
The Science Behind Its Toxicity
Old laboratory experiments and reports from poison centers draw a clear picture. Ingesting copper acetylarsenite—even in tiny quantities—causes headache, vomiting, and stomach cramps. Repeated exposure, just touching powder or breathing its dust, ramps up the risk of chronic poisoning. Chronic arsenic poisoning still affects people around the world, especially in places where regulations fall short. Both the World Health Organization and the US Environmental Protection Agency list substances like copper acetylarsenite among those with no safe exposure threshold.
Why This Compound Still Matters Today
Sometimes, antiques, paintings, and old pesticides show up in homes or at markets. They might look harmless, even pretty, but that nostalgic value often hides a risk. Antique wallpaper scraps or old toy paint cans sometimes test positive for copper acetylarsenite. Unsuspecting homeowners and collectors can disturb and inhale dust, not realizing the potential harm.
Practical Solutions and Cautionary Steps
Education forms the first line of defense. Knowing the names, the history, and the look of this compound helps people recognize it before an accident happens. Professional testing—using arsenic and heavy metal test kits—matters for folks dealing with antique paints or fishing weights, even old rodent poisons. Personal protective equipment like masks and gloves provide an extra layer of protection during cleanup or restoration.
Safe disposal comes next. Most communities treat arsenic compounds as hazardous waste, offering designated drop-off sites. Washing hands thoroughly after any contact with suspicious powders, and keeping curious kids away from old pigments, helps break the cycle of accidental poisonings.
Avoiding Old Mistakes, Keeping Safety Front-and-Center
Copper acetylarsenite tells a bigger story: scientific progress sometimes comes with a steep learning curve. With the facts available today, people have the tools to treat this chemical with the respect it demands—protecting homes, families, and curious hands from the dangers hidden behind a bright green color.
What is the chemical formula of Copper Acetylarsenite?
A Toxic Legacy with a Precise Makeup
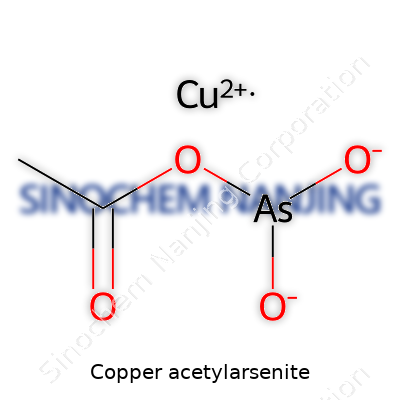
Copper acetylarsenite carries a punch both in chemistry and history. Known as Paris Green, this substance landed its formula in textbooks and laboratories as Cu(C2H3O2)2·3Cu(AsO2)2. Picture this: a blend of copper(II) acetate and copper(II) arsenite, fused in a deep green pigment that once brightened walls and killed pests.
Experience and Risk: What’s at Stake
Growing up, I marveled at old wallpaper with colors that stood out differently. Parents used to choose Paris Green for that pop in children’s rooms or gardens. Nobody wanted to think about toxicity, but skin rashes and breathing problems made families ask if beautiful pigments deserve a place in the home. The compound didn’t just color—it poisoned, silently leaching arsenic into the air or soil.
Scientists later found how the formula spelled trouble. Its copper acetate core carried vivid color, but it was the arsenite groups that packed the real hazard. Each molecule could break apart under humidity, spreading arsenic dust. Historical records show factory workers and artists became ill, as the powder got everywhere—on lunch sandwiches, under fingernails.
An Environmental and Health Red Flag
Paris Green’s formula is not just a curiosity for chemists. Environmental agencies link arsenic-based pigments with serious illnesses, including cancers and organ damage. A garden sprayed against potato beetles might look lush for a season, then poison the food that grows from it. Elevated copper and arsenic in groundwater near old industrial sites still test above safe limits. People living in those regions, just like anyone anywhere, deserve to know what lurks beneath the green surface. Parents today refuse wall colors laced with copper acetylarsenite, making informed choices to keep homes safe.
Learning from the Past, Building for the Future
Recognizing the risks changes the way we view the chemical formula. It’s not just about memorizing Cu(C2H3O2)2·3Cu(AsO2)2; it’s about knowing what each symbol means in the real world. Education campaigns help people identify and avoid leftover paints and pesticides. Environmental cleanup programs remove old supplies and contaminated soils. National regulations now ban the use of arsenic-based pigments in consumer products, reflecting decades of lessons learned—lessons rooted in chemistry as much as human experience.
Chemists keep exploring safer alternatives. Schools teach about molecules not as sterile symbols, but as something that can shape—sometimes endanger—lives. The history and formula of copper acetylarsenite serve as a warning and a spark for responsible science. The numbers tell a story, and everyone gets a happier ending when that story shapes safer choices.
How should Copper Acetylarsenite be stored and handled safely?
Chemistry Doesn’t Forgive Mistakes
Copper acetylarsenite has a reputation, and not the good kind. Also called “Paris Green,” this compound has a story full of cautionary lessons. Years ago, folks didn’t think twice about handling it, and many paid a high price for that. These days, with everything we know, safety must come first. No shortcuts. No room for arrogance. That stuff mixes copper, arsenic, and acetic acid—a lineup with serious health risks. It was once famous as a pigment and an insecticide, but its toxic legacy lingers. Accidents with copper acetylarsenite rarely end well.
Storage: No Such Thing as Overkill
If “locked up” sounds extreme, that’s only because risks get ignored too easily. Copper acetylarsenite belongs in a locked, labeled, and well-ventilated cabinet—one built for poisons, away from heat, sunlight, and humidity. Forget about keeping it near food, drink, or any path where people might wander unknowingly. Evidence shows improper storage brings higher rates of poisoning, especially in labs without chemistry-trained staff.
Temperature swings let moisture in, which can turn solid powder into a clumpy mess and raise inhalation risks. Precaution pays off: every chemical should have a clear label, but this one deserves a bright, loud warning. Never transfer it to another container without a matching hazard sign.
Every Exposure Counts
People sometimes underestimate inhalation and skin contact. I’ve spent enough hours in college lab classes to see the difference between following protocol and improvising. One small cut, or a wave of dust, is all it takes for copper acetylarsenite to leave its mark. Anyone handling this material should wear chemical-resistant gloves (not just thin latex), a full lab coat, and safety goggles. Many safety data sheets recommend a fit-tested respirator rated for particulates. Skip these protections and chances of arsenic poisoning jump fast.
Any accidental dust spill calls for a wet cleaning method—no dry sweeping. Use damp towels or wipes designed for hazardous chemicals, then store the waste in sealed, labeled containers. Don’t trust regular trash bins or household cleaning supplies.
No Room for Improvisation
Some places treat chemical accidents like rare events. That’s exactly backwards. The Environmental Protection Agency ties many laboratory accidents to “routine” work done without full attention. Just because copper acetylarsenite is a solid powder doesn’t mean it’s less dangerous than a caustic liquid. It’s easy to get comfortable, but facts prove discipline is the only friend in the room. Stores and labs should always have emergency showers, eyewash stations, and spill kits close at hand. No excuses if they don’t. If you’re unsure about cleanup, call the professionals, not a neighbor.
Knowledge Saves More Than Rules
Education does more than rules for keeping people safe. Every handler, from researchers to teachers, needs solid training—CDC stats highlight that most mishandling comes from gaps in hands-on understanding. Reading Material Safety Data Sheets closely, attending safety drills, and asking “what could go wrong?” each day shifts a work culture from careless to careful.
Better to Overprepare Than Underestimate
Stories in chemistry aren’t just about science—they’re about decisions. Copper acetylarsenite won’t make exceptions for overconfidence. Give it isolation, clear warning signs, good gear, and a workplace attitude that takes risk seriously. Trust experience, not luck.
What are the physical properties of Copper Acetylarsenite?
A Closer Look at Paris Green
Folks often call copper acetylarsenite by its old nickname—Paris Green. That name comes from the color it brought to paints and wallpapers in the 19th and early 20th centuries. Today, most people recognize Paris Green for what it is: a deeply toxic compound made from copper, arsenic, and acetic acid. The vivid green powder draws attention long before anyone picks up a microscope or a chemistry book.
Color and Structure You Can’t Miss
Copper acetylarsenite stands out as a bright emerald or blue-green powder. This is not your average household colorant—it carries a certain shimmer. Under light, it gives off an almost metallic luster, which explained its wide use in art and decoration back in the day. The compound forms tiny crystalline particles. You can spot shards or specks with the naked eye in a well-lit room, though a hand lens brings out the jagged shapes.
Solubility and Stability
The old wallpapers painted in Paris Green lasted for years because this compound barely dissolves in water. Rain or high humidity won’t wash it off. In oil or organic solvents, it also stays put. The stuff is more brittle than malleable, and it’s known to break down slowly in the presence of acids, especially if the air is damp or polluted. That’s why old painted walls sometimes show worn green spots after decades, but never dissolve away overnight.
Toxicity You Don’t Ignore
Paris Green built itself a dark reputation for its toxicity. Both copper and arsenic come from elements nobody wants on the dinner plate. Just handling this green powder meant real risk. Historical accounts link it to poisoning not just for bugs and rodents, but also to people who lived around walls and curtains treated with the pigment. Dust in the air, powder on the hands, and flakes brushing onto food led to illness. That reputation forced a sharp decline in its use after better health standards took hold around the world.
Melting and Reactivity
Copper acetylarsenite won’t melt easily. It decomposes as the temperature rises, giving off fumes that hazard any unprotected lungs nearby. Firefighters and museum workers keep this in mind when dealing with antique objects. Chemists found the material starts to break down well before reaching 200 degrees Celsius. The breakdown releases not just vapor, but highly toxic arsenic compounds. In a lab, safe handling comes before any curiosity about melting points or reactions.
Density, Feel, and Everyday Experience
The powder feels lightly gritty between the fingers—though nobody should try this for obvious reasons. I’ve seen conservators work with gloves and vacuum systems just to touch restoration pieces with Paris Green. Its density helps it settle quickly in water, forming a green sediment. This property helped 19th-century farmers spread it as a pesticide, knowing it would stay put instead of blowing away with the wind.
Solutions and Safe Handling
Treatment routines for Paris Green combine knowledge and technology. Anyone restoring old art or investigating historic poisoning cases relies on layers of plastic, masks, and specialized waste disposal. Awareness among homeowners, hobbyists, and workers prevents the kind of tragedy that old stories described. Even school textbooks now stress how chemistry's bright past brings responsibility today. Knowing physical properties aren't just science—they guide us in protecting ourselves and the wider community.
| Names | |
| Preferred IUPAC name | Copper(II) acetate arsenite |
| Other names |
Paris Green Acetoarsenite of copper Copper(II) acetoarsenite Schweinfurter Green Vienna Green Mineral Green |
| Pronunciation | /ˈkɒpər əˌsɛtɪlɑːˈsiːnaɪt/ |
| Identifiers | |
| CAS Number | 12017-29-5 |
| Beilstein Reference | 1858731 |
| ChEBI | CHEBI:85173 |
| ChEMBL | CHEMBL510867 |
| ChemSpider | 21567612 |
| DrugBank | DB14076 |
| ECHA InfoCard | 03d3f17e-ba2a-44fa-b26f-8862b579c88c |
| EC Number | 215-572-9 |
| Gmelin Reference | 31994 |
| KEGG | C18600 |
| MeSH | D003822 |
| PubChem CID | 16682901 |
| RTECS number | GL6475000 |
| UNII | 9G1ON3801C |
| UN number | UN1585 |
| Properties | |
| Chemical formula | Cu(C2H3O2)2·3Cu(AsO2)2 |
| Molar mass | 468.63 g/mol |
| Appearance | green crystalline powder |
| Odor | Odorless |
| Density | 6.01 g/cm³ |
| Solubility in water | insoluble |
| log P | 0.88 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 25.1 |
| Basicity (pKb) | 11.27 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.732 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Pharmacology | |
| ATC code | S01AX08 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P202, P261, P264, P270, P272, P273, P280, P284, P302+P352, P304+P340, P305+P351+P338, P310, P314, P320, P330, P361, P363, P405, P501 |
| NFPA 704 (fire diamond) | 4-2-2-☠ |
| Lethal dose or concentration | LD50 (mouse, oral): 10 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1 mg/kg (oral, human) |
| NIOSH | GY9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Copper Acetylarsenite: "0.01 mg/m³ (as arsenic) |
| REL (Recommended) | 0.15 mg/m³ |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Paris Green Copper(II) acetate Copper(II) arsenate Copper(II) arsenite |

