Cobalt Nitrate: A Deep Dive into Its Journey, Chemistry, and Future
Historical Development
Cobalt nitrate first drew the attention of scientists chasing colors for pottery and glass in Europe centuries ago. By the late 18th century, people in chemistry labs began isolating cobalt compounds while hunting for alternatives to expensive coloring agents. Miners stumbled upon cobalt ores while searching for silver, and the smelly fumes from roasting these ores clued researchers into the special nature of cobalt salts, especially as vibrant pigments. Cobalt nitrate entered the scientific limelight once manufacturing processes allowed for consistent quality during the industrial boom. Labs had something reproducible, which meant pigment makers, metallurgists, and researchers could count on a fairly uniform product in their work. Chemical suppliers ramped up production by reacting cobalt metal or cobalt oxide with nitric acid, setting a reliable supply line that has kept the compound accessible for more than a century.
Product Overview
Cobalt nitrate is a bright red, crystalline solid. It’s usually supplied as a hexahydrate—scientists know it as Co(NO3)2·6H2O—but anhydrous forms exist for specialized applications. Its color makes it a favorite in teaching labs for demonstrating cobalt reactions, and its water solubility keeps it relevant in many areas of research and industry. Manufacturers package it as granules, flakes, or powders, each form chosen based on how easily it dissolves or blends in with other ingredients. Packaging includes warnings for toxicity, water reactivity, and storage away from direct heat or sunlight—common sense for any material containing both heavy metals and strong oxidizers.
Physical & Chemical Properties
You can’t mistake cobalt nitrate for anything else once you’ve seen its ruby red crystals. It has a melting point around 55°C for the hexahydrate form, which turns to a blue solution once dissolved in water and gives off a distinctly metallic, faintly sweet odor. Absorbing water from the air, it dissolves easily in most polar solvents, including alcohols. As a source of both cobalt ions and nitrate ions, it behaves as a strong oxidizer—making it incompatible with organic materials, powders, and acids like sulfuric acid. On exposure to high temperatures, it gives off toxic nitrogen oxides and decomposes, leaving behind cobalt oxide. Its density (around 1.88 g/cm³ for hexahydrate) and crystalline texture affect how it’s handled in industrial settings, especially when workers weigh, blend, or store large quantities.
Technical Specifications & Labeling
Regulations around cobalt nitrate labeling differ worldwide, but every drum or bottle ought to show its chemical name, hydration state, purity percentage, date of manufacture, hazard pictograms related to toxicity and oxidizing power, and a UN number (1477 for inorganic nitrates not otherwise specified). For laboratory applications, purity runs above 98%, with traces of iron and copper tightly limited. Calibration standards and reference grades push for even higher purity for analytical chemistry. Container labels flag the need for chemical goggles and gloves, and storage requirements warn against mixing with organics, acids, or anything reactive. Manufacturers add batch numbers for traceability and quality assurance in case of spills or safety incidents.
Preparation Method
Cobalt nitrate production isn’t complicated but must follow proper safety routines. Most large-scale batches start with pure cobalt metal, cobalt carbonate, or cobalt(II) oxide, slowly added to concentrated nitric acid. As the reaction proceeds, nitrogen dioxide fumes bubble out (noxious and toxic), and deep red solution indicates the formation of cobalt nitrate. Removal of excess nitric acid usually means gentle heating to evaporate water, followed by controlled crystallization via cooling. For the hexahydrate, crystals are separated and dried in cool, moisture-free rooms. Industrial producers monitor pH, temperature, and contaminant ions, since impurities in the starting material can undermine quality, especially for electronics-grade batches. Waste gases are scrubbed to prevent atmospheric release, and spent acids are neutralized for safe disposal.
Chemical Reactions & Modifications
Cobalt nitrate acts as both a source of nitrate ions and as a cobalt reservoir. In nitrate’s role as an oxidizer, it reacts violently with flammable materials—an incident with sawdust or sugar is enough to start a fire. When added to basic solutions, it forms insoluble cobalt(II) hydroxide. In reducing atmospheres or under intense heat, nitrate breaks down, releasing nitrogen oxides and converting to black cobalt oxide. Some labs convert cobalt nitrate to other cobalt salts by reacting it with halide acids, acetic acid, or sulfuric acid, producing corresponding cobalt halides, acetates, or sulfates. For catalyst makers, cobalt nitrate becomes a precursor for controlled precipitation of cobalt oxides on ceramic substrates. Researchers have tweaked the nitrate using organic ligands, finding uses in molecular catalysis and as a dopant in magnetic and optical materials.
Synonyms & Product Names
Cobalt nitrate wears several labels—Cobaltous nitrate, Cobalt(II) nitrate, Nitric acid cobalt(2+) salt, and red cobalt salt appear on safety data sheets around the globe. Some specialty suppliers refer to it according to hydration state, like “Cobalt Nitrate Hexahydrate” or simply “Co(NO3)2·6H2O.” On shipping manifests, it goes by its UN code and hazardous classification. In pigment and ceramics catalogs, it’s “Cobalt Red” or “Cobalt Nitrate Crystals.” University teaching labs may just call it “Cobalt Nitrate” or “Lab-Grade Cobalt Nitrate.” Whichever name appears, the hazard labeling always stays—no supplier skips the oxidizer and heavy metal warnings.
Safety & Operational Standards
Handling cobalt nitrate knocks home the importance of strong safety culture. This isn’t a material to treat lightly: skin exposure can trigger rashes, and inhalation of dust or vapors creates risk of lung and systemic toxicity. People wear gloves, goggles, and face shields when measuring or transferring the compound. Ventilation systems and fume hoods reduce the danger of breathing in tiny particles, especially important in research and pigment labs. Occupational guidelines call for regular monitoring of airborne particles, with limits usually under 0.1 mg/m³ for long-term exposure. Spills get handled by wet methods to keep dust out of the air—dry sweeping is a definite no-go. Regulatory bodies—OSHA in the US, REACH in Europe—raise the bar on worker protections each year. Training covers not just cobalt nitrate, but the whole family of cobalt and nitrate risks. Waste gets collected for special disposal, because you can’t dump anything containing cobalt down a regular drain; it persists in the environment and harms aquatic life.
Application Area
Cobalt nitrate finds its way into many areas, even though most people never see it directly. In ceramics, it gives rich, vivid blues to glazes and glass—tests show that the depth and brilliance can’t be matched with cheaper alternatives. Metallurgists use cobalt nitrate as a heat-treatment additive and during surface hardening. It’s a go-to catalyst precursor in the petrochemical industry, where cobalt-based catalytic systems boost the efficiency of hydrocracking and Fischer-Tropsch synthesis. In batteries and electronics, it works as a dopant for layered cathode materials. Chemical researchers count on it as a colorimetric standard and reagent during tests for alkalis and organic molecules. In medicine, the days of experimenting with cobalt salts for anemia have passed, but researchers still use the compound for staining and imaging. Scrutiny over its environmental and health risks increases yearly, yet functional needs in high-performance technologies and specialty ceramics continue to drive demand.
Research & Development
Scientists keep looking at cobalt nitrate with fresh eyes. Developments in nanotechnology see researchers using it for uniform deposition of cobalt oxides on nanoscale supports for catalysis and magnetic storage. Materials engineers test new formulations for use in advanced ceramics, where cobalt’s ability to change oxidation states unlocks new color and functional effects. Academic labs experiment with hybrid organic-inorganic complexes built around cobalt nitrate, sometimes finding breakthroughs in solar cell and sensor technology. Environmental studies center on greener synthesis methods—environmental chemists want to cut down on nitric acid waste and start with recycled metals. R&D into personal protective equipment, ventilation upgrades, and real-time exposure sensors addresses safety inside labs and on the factory floor. Those priorities shape how universities and industries work with this compound in the future.
Toxicity Research
Cobalt nitrate stands out in toxicology literature. Exposure in animals causes a range of issues—respiratory irritation, skin problems, and in high doses, effects on the heart, thyroid, and red blood cells. Chronic human contact links to allergic reactions, and new work looks for links to long-term organ damage and possible carcinogenic effects of cobalt compounds. Regulatory agencies recommend extra attention for pregnant women and people with existing heart or thyroid conditions. Toxicity studies also track cobalt release in soil and water, following its fate through food chains. Laboratory safety manuals have entire pages dedicated to the compound’s risk profile, and every year, environmental chemists publish reports on the presence of cobalt nitrate residues in industrial effluent. In my own lab experience, air quality monitors flashing red signals remind everyone that cutting corners with safety puts real people at risk.
Future Prospects
Demand for cobalt nitrate won’t fade soon, since industries depend on it for catalysts, energy storage, and high-performance ceramics. At the same time, pressure from regulators and advocacy groups will likely drive change in sourcing, processing, and disposal. Future prospects hinge on cleaner, closed-loop manufacturing and the development of cleaner routes—possibly with less dangerous byproducts. Some see opportunities for cobalt recycling from batteries and industrial waste, which could reduce the environmental burden of new extraction. Scientists will likely keep hunting for substitute compounds with lower toxicity, although matching cobalt’s performance is a tall order. Efforts now aim to balance safety, environmental protection, and industrial need. Innovations in exposure monitoring, advanced personal protection gear, and waste remediation show the industry is adapting, but everyone—from researchers to factory workers—faces an ongoing responsibility. Responsible stewardship, not just new chemistry, will shape the way ahead for cobalt nitrate.
What is Cobalt Nitrate used for?
Digging Deeper into Cobalt Nitrate’s Uses
The deep red crystals of cobalt nitrate stay tucked away in labs, factories, and workshops most days. Ask anyone who works with pigments, batteries, ceramics, or lab chemicals, and they can probably tell stories about this striking chemical. Cobalt nitrate crops up in fields that touch everyday life – not just in science textbooks.
Pigments and Coloring: Going Beyond Paint
Artists have leaned on cobalt compounds for centuries thanks to their vibrant blues. Cobalt nitrate doesn’t show up in oil tubes directly, but factories use it as a key starting point for making cobalt blue and other vivid pigments. It brings out permanent color that doesn’t fade fast, even on glass or ceramics. Walk through a kitchenware store or look at fine porcelain, you’ll spot the results – resilient, beautiful blues and purples, fired right into the surface. Without chemicals like cobalt nitrate, many of those colors would be impossible to produce and keep stable over time.
Batteries Rely on Cobalt, Too
Cobalt’s reached household name level in recent years because of electric car batteries and smartphones. Cobalt nitrate plays a role in making battery-grade cobalt compounds. By feeding the manufacturing process, it helps create materials used for lithium-ion batteries. These batteries power the stuff that most people carry all the time – phones, laptops, and, increasingly, electric cars. The supply chain for batteries starts long before minerals reach an assembly line, and it often relies on intermediary chemicals like cobalt nitrate to refine raw cobalt ore into something usable.
Ceramics and Enamel: Not Just Decorative
Factories that craft tiles, sinks, and sanitaryware use glazes to seal and color their products. Cobalt nitrate helps give these glazes hardness, brightness, and durability. Next time you visit someone’s house and see cobalt-blue tile, think about the steps – all the way from a sack of reddish powder in a warehouse to a smooth, finished surface under your hand.
Not Just for Industry—Lab Work Counts, Too
Colleges and research labs stock cobalt nitrate to run chemical tests, especially to detect potassium or as a humidity indicator. I remember working in a college lab and needing reliable reagents for mineral analysis. Cobalt nitrate always stood out because it worked every time and signaled changes through those unmistakable color shifts.
Safety and Environmental Concerns
Cobalt nitrate isn’t like table salt—it’s hazardous to handle and can hurt people if they breathe it in or get it on their skin. Studies link repeated exposure to serious health problems. Factory workers and researchers have to handle it with gloves, masks, and dedicated ventilation. Regulators across the world keep an eye on cobalt compounds to protect both workers and the environment from accidents. In recent years, companies keep trying to cut waste, recycle leftover material, and develop safer processes. Tools like closed-loop recycling help recover cobalt rather than dumping it—critical for an increasingly scarce and expensive resource.
Keeping the Cycle Moving
Cobalt nitrate won’t pop up in a grocery aisle. It’s one of those building blocks that barely gets public attention, but it supports industries most people rely on every single day. Advances in recycling, better worker protection, and ongoing research into alternatives are making its impact safer and more efficient. People who work with cobalt nitrate—whether in pigment factories, ceramic shops, or high-tech battery labs—see it as more than just a chemical; it’s a bridge between minerals and finished goods, one that deserves a closer look the next time cobalt colors or batteries pop up in conversation.
Is Cobalt Nitrate hazardous or toxic?
The Risks Lurking in Industrial Materials
Cobalt nitrate might sound like something that belongs deep in a chemistry lab or tucked away in a corner of a warehouse, but plenty of places use it. This compound goes into ceramics, dyes, and even battery production, which means regular workers, students, technicians, and sometimes entire communities can cross paths with it. Handling cobalt compounds has always demanded respect because the risks involved stretch from immediate health problems to long-term complications.
How Exposure Happens and What That Means
The most direct way people experience cobalt nitrate is through dust or mist in the air. Breathing in this stuff—even in tiny doses for a long stretch—doesn’t just irritate the nasal passages and throat. People report chronic coughing, wheezing, and tightness in the chest. Some get skin rashes or itching after touching it. More concerning: scientists have linked prolonged exposure to kidney, liver, and heart issues. International agencies, such as the International Agency for Research on Cancer (IARC), label cobalt compounds as potential carcinogens—meaning there’s real evidence they may cause cancer in humans.
What the Data Tells Us
Studies collected over the years show that people working closely with cobalt nitrates can wind up with higher rates of asthma-like symptoms and occupational lung diseases. A 2017 research paper traced chronic occupational exposure to damaged lung tissue among battery factory workers. In Norway’s hard metal industry, health reports tracked allergies and eczema tied directly to cobalt’s interaction with skin.
Environmental experts track runoff and spills as well. In water, cobalt nitrate breaks down, releasing ions that harm aquatic life. Fish and other water creatures don’t stand much chance if the concentration climbs high enough, risking entire ecosystems.
Why Attention to Safety Has Teeth
There’s plenty of evidence supporting control and regulations for this stuff. Just a few years back, American and European agencies updated recommended exposure limits for workplace air quality. The Occupational Safety and Health Administration (OSHA) caps the legal limit for cobalt compounds at a low threshold for a reason. No one benefits from a system where basic protection gets skipped. I’ve spoken with plant safety managers who swear by comprehensive training—knowing how to safely handle, store, and dispose of hazardous materials lowers accidents and keeps more workers out of the emergency room.
Building Practical Protections—Steps That Actually Help
Solutions show up in policies, but also in daily habits. Smart workplaces swap open handling for closed systems that limit spills and airborne particles. Everyone near cobalt nitrate should wear protective clothing, gloves, and good masks. Seeking out regular health checks matters, too. These might pick up trouble before someone ends up with permanent health loss. On the bigger stage, fire departments and emergency responders hold drills so they can handle spills fast—so the stuff doesn’t end up in water supplies or linger in the air.
Alternatives exist in some applications, though not every process accepts a simple swap. Finding and developing safer chemicals for industry, or improving the engineering controls inside factories, looks like the clear path forward. Community awareness shapes regulations and pushes companies to do better. By demanding accountability and smarter design, ordinary people keep factories honest and reduce the chance of catastrophe.
The story of cobalt nitrate plays out every day in the background of industry, science labs, and trade schools. Where safety rules get ignored, the risks can come home to roost—through illness, environmental harm, and lost livelihoods. Paying attention, asking tough questions, and following through on basic protections helps everyone breathe easier—literally and figuratively.
What is the chemical formula of Cobalt Nitrate?
The Basics: What’s in Cobalt Nitrate?
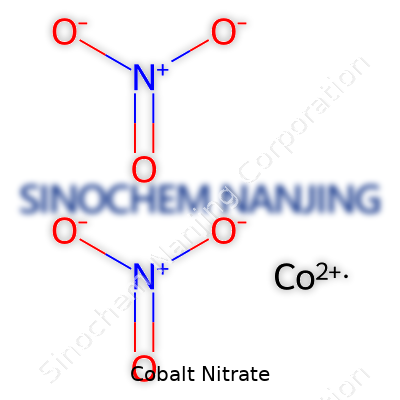
Cobalt nitrate may sound like a mouthful, but it’s just a salt made from cobalt, nitrogen, and oxygen. The formula is Co(NO3)2. If you see the “hexahydrate” version, you’ll run into a more complicated formula: Co(NO3)2·6H2O. That just means six water molecules tag along. The dry version and its watery cousin both play a big role in chemistry labs and factories.
Why Co(NO3)2 Matters in Everyday Work
Cobalt nitrate isn’t just a line in a textbook. Take it from anyone who has ever worked in a ceramics studio or dabbled with catalysts in a research lab. This salt lets scientists and makers add cobalt into other mixtures. Drop this red, easily dissolvable stuff into water and you’re set up for all kinds of chemical reactions. Factories use it to prep surfaces for electroplating. Potters use it for coloring glass and ceramics. The simplicity of the formula means workers can calculate exact doses, keeping costs predictable and results consistent.
Risks that Can’t Be Ignored
There’s a less charming side to cobalt nitrate. Just because it mixes into water smoothly doesn’t mean it’s harmless. Inhaling or ingesting it can pose real health risks. People handling this compound in industrial settings should never skip gloves or ignore ventilation, since repeated exposure can cause skin irritation, respiratory problems, and even more serious health effects over time. Agencies like OSHA and the CDC have clear limits on how much exposure is safe. Relying on safety sheets and best practices isn’t a nice-to-have. I remember one old lab tech who never bothered with proper protection. Over time, he started suffering from breathing issues that might have been avoided by sticking to basic safety.
How Quality and Purity Make a Difference
In research and manufacturing, reliable results start with a consistent product. A batch of cobalt nitrate that contains hidden impurities or shows the wrong color can throw off a whole production run. High-purity versions, certified by respected suppliers, cut out a lot of headaches. Back in my own grad school days, a simple mix-up between anhydrous and hexahydrate versions led to days of wasted work and a very confused research group. Practically every team I’ve ever talked to keeps a close eye on lot numbers and certificates of analysis for this reason.
Improving Safety and Sustainability
Public awareness only matters if companies take it seriously from the top down. Training workers, providing proper gear, and sticking with suppliers who document the traceability of their products creates safer workplaces. Waste disposal is another piece people overlook. Dumping leftover cobalt nitrate down the drain can contaminate water sources. Responsible labs and factories collect waste solutions and send them to certified processors. Using smaller amounts where possible, and recycling spent solutions, also limits environmental impact.
Better Practices Build Trust
Basic chemistry, done right, drives progress in research, manufacturing, and even art. Sticking to recognized safety standards and sourcing from trusted suppliers keeps everyone safer and keeps projects on course. The cobalt nitrate formula, Co(NO3)2, is more than a few letters and numbers—it's a tool for those who respect both science and responsibility.
How should Cobalt Nitrate be stored?
Why Proper Storage Matters
Sometimes you hear about chemical safety standards and think, “Is this really necessary?” Cobalt nitrate tells a different story. It brings risks you can’t ignore. Inhaling dust or fumes can harm your lungs. Letting it contact your skin can cause irritation. Crystals might look harmless, but once they spill, you’re looking at a tough cleanup job and possible health problems.
Choosing the Right Spot
Experience in the lab makes you respect the real dangers of moisture. Cobalt nitrate loves water. Any humidity creeping in can wreck the original substance by causing clumping or dissolving it partially, which makes it harder to handle and measure. You don’t want to discover a cake of ruined chemical when you have an experiment to run. I always pick a dry, cool, well-ventilated cabinet. It sounds simple, but the urge to grab the nearest shelf has to be resisted every time. Chemical storage only works in dedicated spaces away from anything you eat or drink.
Container Counts for a Lot
People tend to think the bottle from the supplier does the job well enough forever. That’s not the case if the seal has been opened more than a couple of times—the risk of leaks rises. Thick-walled glass or HDPE containers that actually close tightly help avoid spills or evaporation. Even so, that container still sits in a tray that catches any leaks, instead of being lined up on a bare shelf. From past mistakes, I can vouch for the importance of replacing worn lids—nobody wants a slow leak eating into the shelf or mingling with other chemicals.
Looking at Compatibility and Security
Mixing cobalt nitrate near acids, reducing agents, or combustibles makes for a bigger problem than many realize. A small spill next to something reactive turns an inconvenience into an emergency. Keeping incompatible materials apart is not just a good suggestion; it saves you a visit from the fire department—or worse. I label the container with bold, clear writing so anyone passing by knows exactly what's inside. It's a routine you never skip, and it helps in emergencies.
Extra Steps That Make the Difference
PPE sounds like overkill until that first exposure, then you won't handle chemicals without gloves, goggles, and a lab coat again. For cobalt nitrate specifically, I’ve learned not to overlook ventilation. Airflow helps scatter accidental fumes and dust before anyone breathes them in. If your workspace gets a bit stuffy or overcrowded, store cobalt nitrate separately in a locked cabinet—not just for safety, but also because theft and misuse are always real-life concerns in any shared facility.
What the Guidelines Say—and Why They Matter
Health regulations like OSHA, the EPA, and similar bodies in other countries publish detailed guidelines, but skipping them is not an option for anyone wanting to stay safe. Regular checks on dates and condition of containers can catch early signs of trouble, such as crusting around lids or colored stains inside cabinets. These checks have saved more than one lab from a serious situation when a leaking jar could have turned the work area into a hazard zone.
Moving Forward: A Culture of Care
Strict habits save time and trouble. Good storage of cobalt nitrate, with constant attention to detail and proper equipment, supports not just your health, but everyone working around you. No lab or facility functions well when basic safety is an afterthought. Building a routine—solid containers, dry spots, clear labels, and regular checks—brings peace of mind that no warning label ever could.
What are the safety precautions when handling Cobalt Nitrate?
Why Handling Cobalt Nitrate Matters
Cobalt nitrate grabs attention in science labs for good reason. Its bright red crystals look harmless, but this compound brings plenty of risks, both to health and the environment. I've worked in spaces where a slip with this stuff could mean costly medical bills or a long conversation with the environmental coordinator. That kind of experience makes you respect the rules a lot more.
Everyday Exposure Risks
Cobalt nitrate packs a punch that goes far beyond stained lab coats. It irritates skin, eyes, and lungs fast—one errant crystal or missed glove can set off days of discomfort or hospital trips. Inhaling the dust brings headaches, nausea, and can spark an allergic reaction. Repeated exposure ramps up the dangers, sometimes leading to lasting lung or heart issues. Cobalt has another nasty habit: it's considered a possible carcinogen, so messing around with it bare-handed hurts more than your pride.
Personal Protection Isn’t Optional
You learn early in chemistry that splurging on good gloves and safety goggles isn’t just about ticking boxes. Not once did I see someone regret wearing a lab coat around cobalt nitrate. Always suit up with nitrile gloves and a buttoned-up coat. Face shields and goggles add a critical layer of protection—dust has a sneaky way of ending up in your eyes, even if you think you’re careful. Closed shoes and long pants keep any spill from hitting skin. Trust me, short sleeves are a shortcut to chemical burns.
Never Underestimate Ventilation
Cobalt nitrate makes air quality a big concern. Any open container releases dust or vapors that hang in the air, especially in tight spaces. Labs and factories invest in fume hoods and ventilation—these aren't luxuries. I found even a small home setup can benefit from a sturdy fan and plenty of airflow. Without a hood, the risk of breathing in toxic particles jumps fast, and the effects can last far longer than the smell.
Smart Handling and Storage
Spills happen to everyone, but dealing with cobalt nitrate means thinking two steps ahead. Store containers firmly sealed, out of sunlight, and away from acids or other reactive chemicals. Keeping materials labeled isn’t bureaucracy—it saves lives when someone else picks up your bottle or a spill needs cleaning. I keep mine in a locked cabinet and run regular shelf checks for leaks or crusty caps. If you’re measuring or transferring, never improvise with makeshift tools—use properly labeled scoops and pour slowly to control dust.
After the Work is Done
Disposal causes headaches for plenty of scientists and safety teams. Cobalt nitrate isn’t fit for the trash or sink. Treat waste like hazardous material every time—package it as your local rules suggest, label everything, and call waste services for pickup. Contaminated gloves, wipes, or labware must head to hazardous bins, not regular garbage. Even tiny amounts can leach into the ground or water, harming animals and people.
Respect Builds Safe Habits
Being lax with cobalt nitrate comes back to bite you. From the first time I nearly touched my face before washing up, I learned routines can save your health. Wash hands thoroughly, double-check the area, and never let food or drink nearby. Forgetting simple steps—like removing gloves after handling—brings accidents closer. Building habits that protect you helps everyone else in the lab or factory, and prevention always beats treating chemical burns.
| Names | |
| Preferred IUPAC name | Cobalt(2+) dinitrate |
| Other names |
Nitric acid cobalt(2+) salt Cobalt dinitrate Cobalt(II) nitrate Cobaltous nitrate |
| Pronunciation | /kəʊˌbælt ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10141-05-6 |
| Beilstein Reference | 358990 |
| ChEBI | CHEBI:78091 |
| ChEMBL | CHEMBL1200932 |
| ChemSpider | 53612 |
| DrugBank | DB11361 |
| ECHA InfoCard | echa.infocard.100.028.330 |
| EC Number | 233-402-1 |
| Gmelin Reference | 72376 |
| KEGG | C01082 |
| MeSH | D003054 |
| PubChem CID | 24437 |
| RTECS number | GF9590000 |
| UNII | OU728OED1Z |
| UN number | UN1477 |
| Properties | |
| Chemical formula | Co(NO3)2 |
| Molar mass | 182.943 g/mol |
| Appearance | Red crystalline solid |
| Odor | Odorless |
| Density | 2.49 g/cm³ |
| Solubility in water | Very soluble |
| log P | -1.37 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 5.3 |
| Basicity (pKb) | pKb: 9.15 |
| Magnetic susceptibility (χ) | +2220·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.682 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 136.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -237.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -373.5 kJ/mol |
| Pharmacology | |
| ATC code | V09DX04 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, toxic if inhaled, causes skin and eye irritation, may cause allergic skin reaction, suspected of causing cancer, may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS03,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H332, H350, H360F, H410 |
| Precautionary statements | P210, P220, P221, P264, P270, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P337+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 400 °C (752 °F; 673 K) |
| Lethal dose or concentration | LD50 oral rat 691 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 691 mg/kg |
| NIOSH | QN2400000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.02 |
| IDLH (Immediate danger) | 25 mg/m3 |
| Related compounds | |
| Related compounds |
Cobalt(II) chloride Cobalt(II) sulfate Nickel(II) nitrate Iron(III) nitrate Copper(II) nitrate |

