Citric Acid: More Than a Sour Taste
Historical Development
Citric acid traces its story back to citrus fruit, where its role gave lemons and limes their sharp, unmistakable tang. Early chemists worked with whole fruit before they could get their hands on purified crystals. In the 18th century, Swedish researcher Carl Wilhelm Scheele separated crude citric acid, learning how to extract it using calcium citrate. Citric acid’s use in beverages, preserves, and confections exploded during the Industrial Revolution. By the early 1900s, people stopped relying on mountains of imported lemons once the discovery emerged that certain strains of Aspergillus niger could churn out the acid by feeding on cheap sugar. Suddenly, the world opened up to citric acid—from foods to pharmaceuticals—thanks to low cost and easy access.
Product Overview
Citric acid appears as white, needle-like crystals, water-soluble and sour. Food industry depends on it as a flavor enhancer and preservative. It keeps jams and canned tomatoes bright, and many soft drinks taste crisp thanks to its consistent acid bite. Pharmaceutical industries value it both to balance pH and help active ingredients dissolve. Cleaning products use citric acid to break down scale and soap scum where hard water creates stubborn deposits.
Physical & Chemical Properties
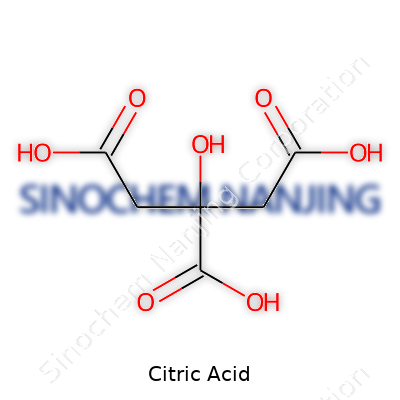
A close look at a bottle of citric acid powder reveals a solid that flows easily, dissolves quickly, and leaves tastes unmistakable—more tart than vinegar, yet without lingering bitterness. Chemically, it carries three carboxylic acid groups, making it a tricarboxylic acid (C6H8O7). This lets it bind metals easily, both softening the harshness of some foods and grabbing hold of minerals to clean industrial boilers. Citric acid melts at 153 °C, and when heated strongly, it breaks down, leaving behind a caramel-like residue and emitting a slight burnt sugar smell in the process. Its pKa values—3.13, 4.76, and 6.40—mean it works well for precise acidification in laboratories and food processing.
Technical Specifications & Labeling
Quality manufacturers consistently report moisture content below 0.5% and identify compliance with standards like FCC (Food Chemicals Codex) and USP (United States Pharmacopeia). Bulk shipments arrive in lined drums or kraft sacks to keep powder dry and prevent caking. Each shipment includes certificates documenting heavy metal screening, microbial counts, and absence of allergens. Labeling requires clear declaration of E-number (E330), net weight, production batch, and recommended shelf life. The push for traceability responds to more rigorous food safety standards and transparency demands from both food processors and consumers.
Preparation Method
Centuries of squeezing lemons have been replaced by fermentation tanks, where Aspergillus niger fungus grows on molasses or corn syrup. The process goes in stages: feedstocks get sterilized, microbes get pitched, and the sugar feed gets controlled with careful aeration. After a week or so of fermentation, the broth contains enough citric acid for extraction. Citric acid finally separates by filtering fungal matter and using lime to form insoluble calcium citrate, which then gets acidified with sulfuric acid to release pure citric acid crystals. These steps squeeze out every drop of profit, so some plants choose special strains bred for higher production or tweak fermentation with nutrients to speed up the yield.
Chemical Reactions & Modifications
Strong chelating ability allows citric acid to bind magnesium and calcium in detergents and water softeners, leading to less soap scum and longer-lasting appliances. In laboratories, it becomes a buffer, keeping pH steady for enzyme reactions. Chemical engineers modify citric acid to create esters like triethyl citrate—useful as a plasticizer for biodegradable films—or react it with bases and alcohols for surfactants and pharmaceutical excipients. Sometimes, it acts as a precursor for synthesizing citric acid derivatives that deliver nutrients more effectively in agriculture. Its threefold acidity lends itself to layering flavors and reactions in processed foods, where sugar, salt, and fat all play off tartness for a fresher taste profile or longer shelf life.
Synonyms & Product Names
Most people see citric acid listed as “E330” on food packages in Europe. Pharmaceutical and laboratory markets refer to it as 2-hydroxypropane-1,2,3-tricarboxylic acid or Hydrogen Citrate. Beyond food, cleaning brands might call it “natural descaler” or “sour salt.” In North America and East Asia, “citric acid monohydrate” and “citric acid anhydrous” appear on ingredient lists, which only differ by water content in the crystal structure. These synonyms help suppliers reach specialized needs in food, supplements, and technical industries alike.
Safety & Operational Standards
Citric acid typically carries a “Generally Regarded As Safe” (GRAS) status from regulatory bodies like the FDA and EFSA, but safety measures in industrial settings focus on dust generation. Workers wear dust masks and goggles since citric acid powder can irritate eyes and respiratory tracts, especially in poorly ventilated spaces. Facilities tune their cleaning schedules and exhaust systems to avoid hard-to-scrub buildup and airborne concentrations above recommended workplace limits. Labels spell out storage guidelines—cool, dry environments, sealed containers—and include all hazard icons according to GHS standards, stressing the need for immediate flushing with water in case of accidental contact.
Application Area
My earliest memory of citric acid comes from tinkering with canning recipes alongside family, where it replaced lemon juice to maintain bright color in sliced peaches and pears. The bigger picture covers a remarkable range—preserving jams, creating shelf-stable sodas, balancing pH in hard candies, and serving as a critical pharmaceutical solubilizer. Chefs trust citric acid to keep guacamole green; cleaning companies tout its descaling action in eco-friendly detergents; brewers add it for acidity in sours. Complex industrial processes depend on citric acid to remove rust before painting shipping containers or prepping airplane parts for coating. A decade spent in R&D taught me that new uses keep emerging, especially as industries react to consumer pressure for “clean label” and “natural” claims.
Research & Development
Recent research tackles “green chemistry”—replacing older, harsher chemicals with renewably sourced citric acid. Biotechnologists have engineered new fungal strains to squeeze out more product per ton of sugar, cutting waste and energy use. The surge in plant-based foods brought forth new hurdles: adjusting sourness in dairy-free yogurt or building plant protein beverages that mimic the real thing. Scientists keep fine-tuning industrial fermentation to cut the carbon footprint, exploring agricultural scraps as feedstock instead of corn or cane sugar. Combining citric acid with tailor-made enzymes or encapsulating it in food matrices opens doors to targeted, slow-release acidification—think shelf-stable salad dressings or sour candies with a longer punch.
Toxicity Research
The decades-old safety profile got its strongest backing from repeated animal studies showing no evidence of cancer, birth defects, or DNA damage, even at doses much higher than daily exposure. Reports do surface of some people reacting to processed foods containing citric acid—not allergic reactions per se, but sometimes oral irritations or exacerbation of pre-existing gut problems, reminded me how individualized nutritional response can be. The concern about “natural” vs. “fermented” source rarely holds up in chemical terms, since the molecule does not change. Still, regulatory agencies demand regular reviews, keeping an eye on emerging toxicology or environmental impacts, especially as global production scales up.
Future Prospects
Growth in citric acid looks unstoppable, as food trends continue to swing toward fresh flavors, shelf stability, and plant-based claims. Industry groups invest in smarter fungi and better fermentation controls, trying to keep cost and ecological impact as low as possible, tapping agricultural waste and byproducts as new feedstocks. Researchers probe its role in pharmaceuticals beyond buffering—such as targeted delivery of micronutrients and minerals in supplements or transdermal skin treatments. Markets for sustainable cleaners, fertilizers, and water treatments open wide possibilities, especially where consumers want non-toxic, biodegradable solutions. Looking ahead, the molecule’s ability to chelate, react, and flavor keeps citric acid central in new product development and greener manufacturing strategies, driving companies to think harder about refreshment, preservation, and cleaner chemistry.
What is citric acid used for?
What’s On Your Plate and in Your Cleaning Cabinet
If you’ve ever sliced open a lemon, you know that tangy bite—the sour spark that wakes up a glass of water or a piece of cake. Citric acid is the main reason for that zing. It’s found in citrus fruits, but it shows up in so much more, from fizzy sodas to bathroom cleaners. Most shoppers probably don’t think twice about this ingredient, printed in small type on food labels. At home, I’ve seen it listed on everything from canned tomatoes to bottles of flavored seltzer, and I’ve sprinkled it on berries to sharpen their taste.
The food industry leans on citric acid for its sharp flavor, but its job isn’t just about taste. It keeps food fresh by holding back mold and bacteria. That ability to preserve is no accident. Companies add it to fruit drinks, jarred salsa, and snacks because a sprinkle keeps things safer on store shelves and in pantries. A packet of beef jerky or a sealed jar of pickles can stay good for a long stretch because of how citric acid controls pH, making it harder for harmful things to grow.
Behind the Scenes in Factories and Home Kitchens
Grimy coffee pots and cloudy shower doors benefit from citric acid’s power. It breaks down mineral deposits and clears away stubborn stains. I’ve scrubbed out old kettles with a simple mix of citric acid and hot water, amazed at the shine left behind. Unlike harsh cleaners, this ingredient does the job without raising concerns about dangerous fumes. Plenty of eco-friendly cleaning brands rely on it for those same reasons.
Wander over to the medicine cabinet and you’ll see it on the back of effervescent tablets or as an ingredient in vitamin C supplements. It helps medicines dissolve quickly in water, making them easier to swallow and absorb. Pharmacies use it to balance the acidity in creams and syrups, a quiet helper that makes everyday products safer and more comfortable to use.
Why Its Use Matters
A lot of citric acid is made using a fermentation process with certain molds and sugars from corn or beets. This keeps costs in check and production steady but raises some questions for people with allergies or those curious about GMOs. There’s little direct risk, but labels rarely mention where it comes from. Transparency would help those with specific allergies or concerns feel more comfortable about what’s in their food.
Citric acid stands out as a safer alternative to harsher chemicals in food and home care but even safe things are not perfect for everybody. In rare cases, people with certain health conditions may need to moderate their intake. It doesn’t mean folks should shy away from the ingredient, but it pays to keep reading labels and understand where ingredients come from.
Looking Toward Healthier Choices
Trust in food and cleaning products connects to what we know about them. Brands can support better decisions by being clear about how they make their products. Adding more detail on packaging builds trust and avoids confusion. Citric acid shows what a common kitchen ingredient can do, making food tastier and homes cleaner. For folks interested in simple, less processed options, there’s always the original: a slice of lemon, squeezed fresh.
Is citric acid safe for consumption?
A Look at Citric Acid in Food
Citric acid ends up in more packaged foods and drinks than many folks realize. As someone who enjoys reading ingredient labels, I spot it all the time in sodas, candies, jams, yogurts, and even pre-cut fruit salads from the grocery store. The tangy, sour kick comes from this organic acid, first discovered in lemon juice centuries ago but today mostly produced through fermentation using certain yeast or mold strains.
Widespread Use—And Why
Food makers like to use citric acid for its sour flavor, but also for what it does behind the scenes. It helps keep foods shelf-stable by lowering the pH, which slows down the growth of bacteria and fungi. That’s how fruit cups or salad dressings last weeks in the fridge.
Citric acid also keeps produce looking fresh. Sliced apples or avocados last a bit longer before browning, thanks to this simple compound. For home cooks and commercial kitchens, this can mean less food waste—and that’s something I appreciate every day when packing lunches.
Is It Safe to Eat?
Most citric acid in foods today comes from a fermentation process, usually using Aspergillus niger, a kind of mold. Some people raise concerns about that. Current research by food safety authorities suggests there’s no danger at the low levels found in foods. The U.S. Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) both list it as generally recognized as safe. These agencies monitor new research, keeping an eye out for any adverse effects.
Many fruits naturally contain citric acid in higher concentrations. A glass of lemon juice provides more citric acid than a typical can of soda. As someone with a sensitive stomach, I sometimes feel a twinge after eating very sour candy or drinking straight lemon juice, but it usually takes a hefty dose to notice any discomfort.
Concerns and Reactions
A few people who consume foods with added citric acid may experience mouth irritation or digestive upset. Anyone with mold allergies might wonder if citric acid triggers reactions, but purified forms used in food contain no actual mold or spores. Still, if you tend to get canker sores after eating certain sweets or tomatoes, moderation helps.
In rare cases, people living with chronic inflammatory diseases report sensitivity to fermented additives. If that’s your experience, avoiding highly processed foods usually makes more sense than targeting citric acid alone. If you’re worried about additives in general, stick to whole foods as much as possible—fresh fruit, vegetables, and minimally processed ingredients. That’s the approach I take at home.
Looking for Solutions
People want honest answers about what they’re eating. Food producers can be clearer about their ingredients, especially as more consumers ask questions about sources and processing methods. For anyone concerned, public health agencies provide clear resources—don’t be shy about using tools like the FDA’s food database or consulting a dietitian.
As more research appears, staying informed helps. If you find yourself reacting to specific foods, take note and talk to a healthcare provider. Knowledge makes it easier to keep your family’s diet both safe and enjoyable.
Is citric acid natural or synthetic?
The Origin Story Matters
Ask anyone about lemons and oranges, and most people point to sourness as the taste of citric acid. Nature gives us this weak organic acid in fruits, and folks have used it for flavor and preservation for a long time. But walk down the grocery aisle, pick up a bottle of soda or even a bag of chips, and the label lists “citric acid.” At that moment, most shoppers imagine squeeze bottles lined up in a food factory. The truth comes with a twist. Today’s commercial citric acid does not get squeezed from lemons or limes. Instead, production relies on a fermentation process using a mold known as Aspergillus niger. Manufacturers pour glucose syrup into giant vats, feed it to the fungus, and collect the acid that forms during fermentation.
How Nature and Science Blend
Over a century ago, factories needed a better source for food and medicine. Extracting acid from citrus cost way too much, so chemists searched for a shortcut. An American food chemist discovered that mold turning sugar into acid worked perfectly, and from there, industry never looked back. These fermentation vats create the same molecule found in fruit. That fact tricks most people into thinking nothing has changed. But growing fungus in a controlled environment looks quite different from picking fresh oranges. Even though the origin now traces to a lab, the end product remains identical to its fruit-born counterpart.
So Why Should Anyone Care?
The question gets personal for some shoppers. People with allergies or those who want to eat closer to the earth want to know where their food comes from. Friends in the restaurant business tell me more customers ask if “citric acid is natural.” Technically, it is, because the molecule matches the one in fresh lemons. But since factories use fermentation, many sources call it “nature-identical” or “synthetic.” This affects those who follow strict diets, avoid processed foods, or have concerns about mold-based ingredients.
Health agencies include citric acid on safe food additive lists. The World Health Organization and FDA confirm its record of safety, especially since it breaks down quickly in the body. Still, not every eater feels satisfied with that answer. Rare cases exist where people with mold allergies may react. Some believe that food should only use ingredients pressed or picked from plants without heavy industrial steps. Consumers also want full transparency on sourcing because nature and lab-grown can mean something different, depending on individual values.
Clearer Labels and Better Choices
As a cook who reads way too many ingredient lists, transparency matters more than ever. Shoppers deserve labels that make it clear whether something comes from fruit or fermentation. Current food laws permit “citric acid” without any extra description, which leaves people guessing. Grocery chains and food brands could go further—offering choices that specify origin gives power back to the shopper. Organic foods sometimes choose fruit-derived acid or specify the fermentation source, meeting demand for simple, clearly sourced ingredients.
Citric acid shows up everywhere in kitchens, restaurants, and manufacturing. People want straight answers about what goes into their food and where it starts. Industry can take the next step—sharing the full process with the public and letting eaters decide what belongs on their plates.
Can citric acid be used for cleaning?
A Cleaner Pantry Staple
Walk through just about any grocery store and a bag of white citric acid crystals sits quietly on the shelves near canning jars or the baking aisle. Most people see it listed in homemade lemonade recipes or as a tangy booster for jams. Fewer folks know those same tart crystals tackle some of life’s stubborn messes better than a bottle loaded with synthetic chemicals.
Limescale and Mineral Deposits: Bye-Bye Hard Water Stains
Ever looked inside a kettle that has seen years of service? Scaly white lines hug the heating element, and no regular dish soap will budge them. I grew up in a city with hard water, and every few months my mom would boil a tablespoon or two of citric acid with water in the kettle. After a short soak, a gentle rinse left the inside gleaming. That kitchen wisdom comes from a simple truth: citric acid breaks down calcium and magnesium deposits naturally. No harsh fumes hang in the air, and you don’t risk scratching your appliances.
No-Nonsense Bathroom Cleaner
Cutting through soap scum often means scrubbing and fighting with clinging, filmy residue. Citric acid offers a shortcut. Mix it up with hot water and spray it on shower doors or around faucets. Left for ten or fifteen minutes, that stubborn buildup turns soft enough to wipe away. For grout, I sprinkle a damp sponge with a pinch of powder and scrub—it lifts stains without scratching tiles or leaving behind a chemical smell. That’s a big plus for families with kids or pets roaming around.
Laundry and Stain Removal
Laundry, especially in older houses with old plumbing, brings its own aches and pains. Towels may feel rough and faded clothes don’t quite bounce back in the wash. Citric acid, a natural fabric softener and mild bleach alternative, works here too. Tossing a spoonful in with the detergent can soften textiles and help pull mineral deposits away from fabrics. It works well on sweat stains, grass marks, and safe on most colors when used in moderation.
Why it Matters: Health, Environment, and Budget
A lot of commercial cleaners promise rapid results but fill the air and water with harsh chemicals. In my home, a bottle of citric acid means less clutter and less worry about what’s getting left behind after cleaning. Studies from the Environmental Working Group suggest that many conventional cleaners release volatile organic compounds. Citric acid delivers the punch without hurting indoor air or leaving chemical residues on surfaces where kids eat or play.
Easy on the Pocket, Easy on Conscience
Buying citric acid in bulk costs less over time. A single container makes dozens of cleaning solutions. No fancy branding, no unnecessary fragrances masking real cleanliness. For food surfaces or things like baby toys, I’d trust a simple acid that’s already safe enough for the kitchen jar.
How to Use: Keep It Simple
Mix one or two teaspoons with a cup of warm water for light cleaning. Double or triple it for tough stains or hard scale. Always rinse with plain water after use, not because citric acid will hurt surfaces, but to keep things tasting neutral and free of residue.
Some Cautions and Responsible Use
Citric acid isn’t a miracle worker for everything. It can dull marble and etch natural stone, so never pour it on countertops made of those materials. Store it dry and sealed; once moisture gets in, the powder clumps and dissolves.
Real Change Starts Small
Switching to citric acid for cleaning is a small act, but it piles up to safer homes, less waste, and more control over what touches our food, hands, and clothes. For me, it’s one of those little life upgrades that keeps paying off year after year.
Does citric acid have any side effects?
Citric Acid: Found in More Than Just Lemons
Most people know citric acid as the sour kick in lemons, oranges, or limes. The thing is, you find it tucked into ingredient lists everywhere: sodas, salad dressings, even cleaning sprays. Food makers love using it to keep flavors bright and to help products last longer. It’s also not just squeezed from fruit anymore—most industrial citric acid comes from corn processed by certain molds. With that much exposure, people start to wonder if it comes without baggage.
Does Citric Acid Really Cause Side Effects?
For most healthy folks, tossing back a lemonade or eating foods with a hit of citric acid won’t seem like anything major. Still, a handful of people run into problems. From what I’ve seen and read, those problems usually crop up for folks with allergies, sensitive mouths, or stomach troubles.
If you deal with canker sores or sensitive teeth, you might notice mouth pain after drinking lots of citrus drinks or sucking on sour candies loaded with it. Dentists say citric acid can soften tooth enamel, wearing it down with constant exposure. I’ve seen people reach for water right after eating sour snacks, trying to rinse away the sting and cut back on damage.
Gut Discomfort and Allergic Reactions
The stomach has its own thoughts about acid. For anyone battling acid reflux or gastroesophageal reflux disease, higher acidity can spark heartburn and discomfort after meals. Some people say that processed citric acid, especially the mass-produced kind, triggers gut issues or headaches—though solid studies don’t confirm this reaction for most. Some experts chalk it up to the body’s individual sensitivities rather than the acid itself.
Over the years, some folks have come forward saying they break out in hives, feel itchy, or get other allergy-like reactions after eating foods with commercial citric acid. The kicker is, the actual allergy isn’t to the acid but to leftover proteins from mold or corn used in factories. These reports stay rare; the FDA and similar agencies still list citric acid as safe for most people. Still, anyone with an allergy to mold or corn should keep an eye on labels.
Doesn’t Sound Serious—But, for Some People, It Can Be
I’ve talked with family and friends struggling with migraines who swear off processed foods that list citric acid, believing it ramps up their symptoms. The evidence isn’t airtight, but the lesson sticks: if something in your diet bothers you, cut it for a bit and see how you feel. People with kidney problems get stricter advice, since citrus and citric acid in high doses can sometimes mess with potassium levels.
Look, citric acid doesn’t top the list for most dangerous additives. Still, smart eating means paying attention to what triggers your symptoms. If you worry about acids wearing down your teeth, stick to water after acidic meals and talk with your dentist. Concerned about food allergies or suspect processed citric acid makes you itch? Try a homemade approach or stick with natural fruits for your sour fix.
Food Labels and Smart Choices
Most health authorities say citric acid is safe. People with specific sensitivities might want to check labels and experiment with what works. More research can help pin down who really stands at risk and how processed citric acid could affect certain groups. Until then, the best bet sits in listening to your body, reading those long ingredient lists, and keeping your doctor in the loop if reactions pop up.
| Names | |
| Preferred IUPAC name | 2-hydroxypropane-1,2,3-tricarboxylic acid |
| Other names |
2-Hydroxy-1,2,3-propanetricarboxylic acid Citrate Lemon salt E330 |
| Pronunciation | /ˈsɪtrɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 77-92-9 |
| Beilstein Reference | 1720243 |
| ChEBI | CHEBI:30769 |
| ChEMBL | CHEMBL1131 |
| ChemSpider | 530 |
| DrugBank | DB04272 |
| ECHA InfoCard | 03f83a68-7a01-4542-bc6a-6e7da9b3a14a |
| EC Number | 211-529-5 |
| Gmelin Reference | 725 |
| KEGG | C00158 |
| MeSH | D002244 |
| PubChem CID | 311 |
| RTECS number | GE7350000 |
| UNII | XF417D3PSL |
| UN number | UN0775 |
| CompTox Dashboard (EPA) | DTXSID2020174 |
| Properties | |
| Chemical formula | C6H8O7 |
| Molar mass | 192.12 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.665 g/cm³ |
| Solubility in water | 1600 g/L (20 °C) |
| log P | -1.64 |
| Vapor pressure | Vapor pressure: <0.01 mmHg (20°C) |
| Acidity (pKa) | 3.13, 4.76, 6.40 |
| Basicity (pKb) | 3.13 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.493 |
| Viscosity | 1.7 mPa·s (20 °C) |
| Dipole moment | 3.69 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 189.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -947.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1986 kJ/mol |
| Pharmacology | |
| ATC code | A09AB13 |
| Hazards | |
| Main hazards | Irritating to eyes and skin. |
| GHS labelling | GHS07, exclamation mark |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | **"May cause respiratory irritation. Causes serious eye irritation."** |
| Precautionary statements | P264, P270, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Autoignition temperature | 1010°F (543°C) |
| Lethal dose or concentration | LD50 (oral, rat): 3,000 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral rat: 3,000 mg/kg |
| NIOSH | WS5600000 |
| PEL (Permissible) | PEL not established |
| REL (Recommended) | 3 mg/kg bw/day |
| Related compounds | |
| Related compounds |
Isocitric acid Citric acid cycle Citrate Tricarboxylic acids Malic acid Succinic acid |

