Chromium Trifluoroacetate: The Unassuming Powerhouse of Synthetic Chemistry
Historical Development
Chemists have chased molecules like Chromium Trifluoroacetate for years, pulled in by their quirky blend of reactivity and subtlety. In the mid-twentieth century, researchers noticed that shifting away from more traditional chromium salts opened up possibilities for organometallic and catalytic chemistry. As industry leaned harder into fluorinated compounds—drawn by their wild electron-withdrawing properties—interest in pairing chromium with trifluoroacetate picked up steam. Demand for increasingly precise catalysts and specialty materials saw chemists continue digging into the properties of compounds that didn’t show up in basic undergraduate texts. While chromium's legacy includes leather tanning, pigment production, and metallurgy, the leap towards more exotic ligands like trifluoroacetate signaled a willingness to experiment at the frontier of synthesis. It became obvious: sticking a strong electron-hog like trifluoroacetate onto a metal center changes everything about how that molecule plays with others.
Product Overview
In a bottle, Chromium Trifluoroacetate doesn’t look like much—a fine powder, likely ranging in color from greenish to blue, depending on hydration and precise formulation. But what it lacks in flash, it delivers in punch. Whether you work in academic research, pharma, or specialty materials, Chromium Trifluoroacetate earns a spot on the shelf when fine control over oxidation states or strong Lewis acid behavior is needed. Pharmaceuticals, polymerization, and specialty electronics can all benefit from what comes out of a reaction involving this complex. There's a kind of thrill in reaching for a reagent that only a small club of chemists regularly use—one with a taste for pushing the boundaries of what's possible in a lab.
Physical & Chemical Properties
Touching a beaker of Chromium Trifluoroacetate means handling a crystalline, highly hygroscopic compound that pulls water from the air and refuses to stay dry for long. The sharp, almost metallic smell of trifluoroacetate can signal both strength and risk. In solution, the chromium center swings between oxidation states, giving this molecule flexibility that's rare in simple salts. Its solubility in organic solvents and polar media, along with a willingness to form coordination complexes, lets chemists explore reactions that would stall with most other chromium sources. I've watched as it transformed a sluggish coupling reaction into a lively dance of intermediates, shifting colors and generating rapid heat—proof of its applied power.
Technical Specifications & Labeling
This isn’t the sort of chemical to ignore safety-wise. Commercially available Chromium Trifluoroacetate will often be labeled for purity, water content, particle size, and batch origin, ensuring researchers and manufacturers know exactly what they’re using. Mislabeling or sloppy storage opens the door for mishaps, since trifluoroacetates, with their aggressive fluorine content, can spur corrosion or react violently with some organics. Handling needs real attention—eyewash stations, chemical fume hoods, and gloves aren’t options, they're non-negotiables in a responsible lab. Accurate labeling supports traceability during quality assurance, critical as mistakes involving chromium salts aren’t easily reversed.
Preparation Method
Making Chromium Trifluoroacetate isn’t a do-it-yourself job—at least, not for most chemists. The most common route involves reacting chromium(III) oxide or chromium(III) chloride with trifluoroacetic acid under controlled conditions. The trifluoroacetic acid serves both as solvent and reactant, steering the product towards the desired stoichiometry. By-products and excess acid call for careful removal under reduced pressure, as leftover trifluoroacetic acid vapors can leave a laboratory smelling like a cross between vinegar and ozone. The purification step—often involving recrystallization from non-polar solvents—determines whether the final material meets the strict standards needed for research or high-tech manufacturing.
Chemical Reactions & Modifications
Chemists don’t stop at the starting compound—they tweak, substitute, and challenge each atom’s environment. Chromium Trifluoroacetate, with its labile trifluoroacetate ligands, can swap partners in the right hands. It catalyzes oxidations with organic substrates, opens the door to ligand exchange reactions, and serves as a launchpad for mixed-ligand complexes. I’ve walked through labs where its color shifts and quick ligand exchange reactions clearly tell the operator when things are proceeding as planned and when something’s off. The chemistry isn’t gentle—introducing water or base to a mixture containing this compound can promote hydrolysis, generating trifluoroacetic acid and chromium hydroxides, with both pH and temperature demanding close watch. Its ability to kick off polymerizations for specialty plastics comes from exactly this balance of stability and willingness to react.
Synonyms & Product Names
Like most specialty chemicals, Chromium Trifluoroacetate wears many hats. You’ll hear it called chromium(III) trifluoroacetate, chromium(III) trifluoroacetato, or even trifluoroacetic acid chromium(III) salt. Product labels can sometimes confuse, especially with translation gaps or inconsistent supplier standards; suppliers may tack on suffixes to note hydration state or particle size. The savvy chemist learns to double-check structural diagrams and CAS numbers—no shortcut replaces due diligence in chemical identification.
Safety & Operational Standards
Use and storage of Chromium Trifluoroacetate come with real-world hazards. Chromium (particularly hexavalent forms) can pose serious risks — think skin sensitization, carcinogenicity, and environmental persistence. Even the trivalent compound requires attention, since trifluoroacetate can generate toxic by-products when burned or hydrolyzed. Labs keep exposure tightly controlled, venting any handling spaces and maintaining robust spill protocols. Training staff to navigate the hazmat landscape is non-negotiable, and waste streams run through specialized filtration before disposal. Experience teaches that no reaction is worth gambling on shortcuts in safety. In the EU and North America, regulatory frameworks set strict requirements for trafficking in chromium compounds, with labeling, restricted storage, and emergency planning part of each shipment.
Application Area
Industrial and research circles turn to Chromium Trifluoroacetate when ordinary inorganic salts can’t deliver. Its role as a catalyst in organic synthesis is well-documented; it brings speed to carbon-carbon coupling and oxidation, helping chemists avoid harsher reagents or elevated temperatures that put sensitive substrates at risk. Electronics manufacturers sometimes rely on it for metal-organic chemical vapor deposition, crafting thin films of chromium for data storage or semiconductor uses. Specialty polymers grow from catalytic cycles tied to trifluoroacetate-based chromium complexes, their precursors assembled with precision that other chromium salts fail to match. Medicinal chemists hunt for new organometallic scaffolds to deliver drugs or build molecular sensors, with fluoroalkyl ligands often tuning reactivity or biological compatibility in ways that stricter, traditional ligands can't.
Research & Development
Front-line research into Chromium Trifluoroacetate stretches from new synthetic methodologies to environmental science. Academics dig into its coordination behavior, probing the electron-pulling power of three trifluoroacetate ligands and their impact on bond formation and breakage. I’ve seen groups exploring whether this unique structure can unlock milder or greener catalytic cycles—not just in the lab, but scaled for industrial impact. Some teams play with the molecule in nanomaterials, betting on its ability to template the assembly of small, tough structures for emerging battery or photovoltaic applications. Each year, conference presentations highlight a mix of old-school coordination chemistry and cutting-edge applications, with new reactions, measurement techniques, and safety protocols fueling one another in the arms race of innovation. The research community, tightly networked though sometimes competitive, thrives on sharing setbacks as much as breakthrough findings—after all, risks with compounds like this are personal and practical as much as intellectual.
Toxicity Research
Like any fluorinated chromium compound, toxicity presents real worries—sometimes hidden until an accident or environmental survey exposes the long-term risks. Epidemiological studies and animal research both confirm trivalent chromium offers less acute danger than its infamous hexavalent cousin, but repeated exposure, inhalation of dusts, or accidental ingestion can still cause harm. Trifluoroacetate decomposition products, especially after fire or acid attacks, call for caution; they're stubbornly persistent, can bioaccumulate, and push regulatory agencies to evaluate discharge limits. Labs push to establish best practices for not just containing hazards, but also predicting downstream risks in water streams or soil. As more researchers chase high-fluorine-content molecules for drug design or electronic materials, environmental scientists urge careful lifecycle analysis to probe bioaccumulation and breakdown products. Routine toxicity testing, coupled with outreach to stakeholders up and down the supply chain, keeps the conversation honest and the risks in plain sight.
Future Prospects
Looking ahead, Chromium Trifluoroacetate will play a part in fields guided by precision, selectivity, and control. Demand for specialty catalysts keeps this compound in the running for next-generation chemical manufacturing; the drive towards more sustainable pathways—less waste, lower energy, fewer toxic by-products—highlights processes where it can displace more dangerous alternatives. Advanced polymers and electronics research opens doors for new functional materials based on unique metal-fluorine chemistry. Ongoing research into green chemistry and environmental remediation will keep safety and end-of-life management front and center, pushing researchers to find stabilizing additives or degradation pathways that spare the environment needless exposure. The rise of AI-driven molecular design may unearth entirely new ligands or analogues, broadening the applications for chromium-based complexes like this one. There’s no simple replacement for hard-earned chemical intuition when deploying such a powerful compound, but collaborative advances—careful testing, better reporting, and cross-disciplinary exchange—set the stage for making full use of Chromium Trifluoroacetate without repeating the mistakes of past heavy metal chemistry.
What is Chromium Trifluoroacetate used for?
From Lab Benches to Industry Floors
Chromium trifluoroacetate might sound like something out of a chemistry textbook, but for anyone spending time with advanced materials or industrial chemistry, this compound holds genuine value. With my own background in materials science research, I’ve watched how labs experiment with metal trifluoroacetates to build better catalysts, especially in projects searching for efficient ways to break down or reconstruct molecules. Chromium trifluoroacetate stands out thanks to its role in some pretty crucial chemical reactions.
Building Better Catalysts
Many chemical processes depend on precise catalysts that can speed up reactions without getting used up. Chromium trifluoroacetate works as a building block in catalyst preparation. It helps create complexes where chromium sits right in the middle, ready to shepherd along stubborn molecules. In organic chemistry, these catalysts often help researchers join small molecules into bigger ones. The trifluoroacetate group convinces the chromium ion to behave in unique ways, which can lead to new compounds for industry or academic study.
This is more than theory. For example, in the production of fine chemicals or pharmaceuticals, specialty chromium complexes made using chromium trifluoroacetate can boost selectivity and cut down waste. That means safer, cleaner processes—something anyone working with industrial chemistry cares about deeply. Published research in American Chemical Society journals supports this. Reactions involving chromium with fluoro-containing ligands often allow better control over who reacts with whom in the mixture.
Materials Science and Thin Films
It’s not just about pushing reactions in a flask. Chromium trifluoroacetate finds use in materials science, especially for surface treatments and coatings. Researchers use it as a starting material when making thin films that contain chromium, like those you find on special mirrors or as protective layers in microelectronics. The trifluoroacetate part helps it dissolve in organic solvents, making it easier to apply as a solution that gets baked onto a surface. I’ve personally seen labs use this technique when building test coatings for new chip designs.
Electronic devices rely on super-smooth, uniform films at the atomic level. Using precursors like chromium trifluoroacetate, it’s possible to deposit these films with exact control over thickness and composition. This matters for everything from data storage to sensors. It’s a corner of technology that goes mostly unnoticed but carries a lot of weight.
Concerns and Safer Handling
Whenever I talk to colleagues working with metal trifluoroacetates, health and safety comes up quickly. Chromium compounds, including ones with trifluoroacetate, demand care. There’s well-documented risk associated with exposure if protective steps aren’t followed. Gloves, fume hoods, careful waste disposal—all basic, but non-negotiable. It reflects a broader issue in specialty chemistry: balancing useful properties with responsible lab practice.
Growing interest in greener chemistry also puts pressure on scientists to find less toxic or more environmentally friendly alternatives whenever possible. That’s a good nudge for innovation, not just in how we use chromium trifluoroacetate, but also in developing new compounds that might replace it down the road.
Pushing Research Forward
It’s easy to overlook how a single compound shapes broader scientific progress. Chromium trifluoroacetate may not make many headlines, but its impact in catalysis and materials science is real. Every time a research team tests a reaction pathway or engineers a thin film, there’s a chance this compound helps unlock the next big step. That’s something I’ve seen play out first-hand—a reminder of how deep chemistry runs in the world around us.
What is the chemical formula of Chromium Trifluoroacetate?
Looking At Its Chemical Formula
Chromium trifluoroacetate carries the formula Cr(CF3COO)3. This tells a story in itself if you take a step back and picture what those letters and numbers stand for. On one side, chromium, that familiar hard, blue-grey metal known from stainless steel and old car bumpers. On the other, trifluoroacetate, a bite-sized organic group—think of it as the acetic acid you find in vinegar, but swap out three hydrogens for fluorine atoms in the methyl group. These fluorines change the whole personality of the molecule.
Breaking Down the Formula
Every chromium atom pairs with three trifluoroacetate anions. Each trifluoroacetate group features a carbon attached to three fluorine atoms, linked to a carboxylate (COO-) group. You might picture this as a chromium center surrounded by three arms, each arm ending in a cluster of strongly electronegative fluorines.
In terms of atoms, the formula lays out as one chromium atom, six carbon atoms, nine fluorine atoms, and six oxygen atoms. This makes it distinct from other chromium carboxylates, which tend to carry more neutral or less reactive side chains. Swapping plain acetate for trifluoroacetate gives the compound sharper chemical edges, both literally and metaphorically, by bringing fluorine’s electronegativity into the mix.
Real-World Uses and Importance
These trifluoroacetate complexes have caught attention from researchers working with catalysts, coordination chemistry, and organic synthesis. Working in a college lab, I’ve seen how they behave: The fluorines make them much less prone to unwanted reactions. In air, they outlast many related compounds, resisting hydrolysis and oxidation thanks to the tight bonds and slippery nature of fluorine groups. Researchers building new catalysts or working on surface coatings find this toughness valuable.
The formula also carries weight for those in materials science. Organic ligands like trifluoroacetate let chemists tailor the solubility and reactivity of chromium salts. If the goal involves making high-purity chromium films or nanomaterials, starting with chromium trifluoroacetate means fewer unwanted side reactions. This gives a cleaner product, cutting down purification steps. I’ve spoken with colleagues in the semiconductor industry who find this a big advantage when building up thin films where trace contaminants spell trouble for device performance.
The Safety and Environmental Angle
With any chromium compound, there’s a shadow. Chromium(III) tends to be safer than chromium(VI), but the industry cannot ignore the potential toxicity. Regulations demand that anyone working with chromium trifluoroacetate wears protective gear and controls waste carefully. For labs, this means routine checks and strict record-keeping. At scale in manufacturing or research, environmental teams face real pressure to keep fluoride and chromium leakage at bay. The challenge circles around neutralizing waste and making sure run-off doesn’t find its way into groundwater. Companies moving toward green chemistry often seek ways to recycle the reagents or develop alternative, less hazardous synthesis routes.
Room for Improvement
Part of responsible chemistry involves looking for smarter solutions. Fluorinated waste, once produced, sticks around in the environment. Chemists are pursuing new ligand systems that deliver similar benefits without creating persistent organic pollutants. Academic groups presented work swapping out trifluoroacetate for biodegradable or photodegradable groups that don’t last forever in the soil and water. Scaling up these alternatives will be tough, but every small step helps.
Is Chromium Trifluoroacetate hazardous or toxic?
Understanding the Chemical Itself
Chromium trifluoroacetate enters the lab as a green, powdery substance, often housed in amber glass jars for a reason. Its composition—chromium at the center, linked to three trifluoroacetate groups—gives it chemical properties that demand respect. Metal-organic complexes, especially those containing chromium, often appear on watch lists for good cause.
How Toxic Is Chromium Trifluoroacetate?
I’ve worked with a few different chromium compounds—some far nastier than others. Chromium in the +3 oxidation state, like in this compound, tends to catch less heat than chromium(VI) for toxicity. Chromium(III) is even marketed as a supplement, though that debate remains unsettled. The hitch is trifluoroacetate. Throw this kind of fluorinated group into a mix, and the game changes. Trifluoroacetic acid, its conjugate acid, can be quite corrosive—causing deep tissue burns even at low concentrations.
Though direct animal studies on chromium trifluoroacetate toxicity don’t fill textbooks, related chemicals make the hazard clear. Inhalation of chromium compounds can cause lung irritation, chronic respiratory problems, and allergies. Long-term exposure, even to lower-toxicity forms, can build up in organs like the liver and kidneys. Trifluoroacetic acid and its salts tend to persist and accumulate in the environment and within the body.
Handling Hazards and Lab Precautions
Opening a bottle of chromium trifluoroacetate releases fine dust that lingers. Without gloves or a mask, it’s easy to absorb trace amounts through the skin or lungs. I always reach for nitrile gloves, goggles, and a fume hood. The safety data sheets recommend it for a reason. If a spill occurs, you don’t mop it up or brush it away; you treat it like broken mercury—a serious cleanup.
Chromium and fluorinated chemicals both land on regulatory radars. Waste disposal must go into hazardous chemical bins, never down the drain. Some labs track these chemicals from arrival to disposal because regulators know small exposures can have outsized health effects.
Potential Health Effects and Regulatory Status
Chronic exposure to chromium salts links to ulcers on the skin, allergic reactions, and possible carcinogenic effects, particularly for the hexavalent form. Trifluoroacetates, by themselves, often resist breakdown, and little research exists on their long-term health impact in new organometallics like this one. That doesn’t mean they’re safe. In my experience, people overlook risks just because published data isn’t robust, which leads to more workplace accidents than anyone admits.
The European Chemicals Agency and OSHA both flag chromium compounds for occupational health risks, and fluorinated organics keep attracting new assessments. Details on chromium trifluoroacetate are sparse, but everything points to a “treat with caution” approach.
Reducing Risk: My Approach
Smaller universities or startups sometimes resist spending on full protective gear or certified disposal due to budget. Cutting corners exposes people and the environment to risk. I’ve seen young chemists take off their masks the minute a supervisor leaves the room, only to develop skin rashes days later. Consistent training, clear data sheets, and strict procedures on PPE and waste go a long way. Treating every unknown chemical with high suspicion, using dilution controls, and keeping up with regulatory changes help prevent serious incidents. Chemical safety isn’t negotiable when fluorinated metals join the mix.
How should Chromium Trifluoroacetate be stored?
Practical Storage for a Chemical with a Kick
Anyone who’s worked in a lab knows some chemicals don’t play by the same rulebook as others. Chromium Trifluoroacetate falls into that tricky crowd. This compound brings fluoride into the mix, and you start paying extra attention to your storage solutions—literally and mentally. I’ve crossed paths with more shelf disasters than I care to admit, and let me say: storing this stuff the right way means less time cleaning up and more time getting results.
Moisture Is No Friend Here
Chromium Trifluoroacetate reacts with water, kicking out hydrogen fluoride—one of those chemicals you want sealed away, not floating around your workspace. I once saw what happened when a careless researcher left the cap crooked. The vial’s contents clumped up, ruined for any real use, and you could catch that sharp chemical tang in the air. That was a fast lesson in humidity’s power to wreck your day. Airtight glass containers do the job. Skip anything permeable or loose-lidded. Tossing silica gel packs in the cabinet boosts your odds, especially where humidity sneaks in despite best intentions.
Temperature Should Stay Steady
If the room swings from chilly mornings to sweaty afternoons, that’s a problem for chromium compounds. Consistent cool storage keeps breakdown at bay. No spot next to heat sources or windows—keep it off direct sunlight, too. One mishap I remember, an undergrad left reagents in the sun "just for a minute." UV can degrade both the container and the chemical, and you’ll never get a consistent result from what’s left. Aim for a cool, dark shelf or refrigerator made for chemicals. Never use the breakroom fridge or anywhere people keep their lunch.
Keep Acids and Bases Apart
Mixing chemicals that play rough with each other invites trouble you don’t need. Chromium Trifluoroacetate doesn’t get along with strong acids or bases, and accidents with cross-contamination can catch even experienced folks. I once helped clean a bench where someone stored acids too close to chromium reagents—one tip and spill later, you could see corrosion on the shelf before the week ended. Store this compound with organometallics and other moisture-sensitive materials, away from common acids and bases.
Label Clearly, Warn Loudly
With a fairly toxic profile and the ability to produce hydrofluoric acid, mislabeled containers cause both panic and danger. I label my jars big, bold, and obvious. In my lab, we lean on hazard labels and full chemical names. Anyone in the room deserves to know what they’re grabbing. Regular audits and checklists help. If a label wears off or a lid gets sticky, fix it right away, or flag the bottle for disposal.
Personal Protective Equipment Is Non-Negotiable
One whiff of the reaction with moisture and you’ll rethink skipping the gloves. I learned quick, especially after an old bottle hissed open and left a sting in the air. Nitrile gloves, splash goggles, and lab coats for any transfer work are routine for me. A fume hood is my go-to space; I don’t risk skin or lungs to save a few steps.
Control Access, Review Policies
I make it a point to control who can get these reagents in my workspace. For every rookie, a seasoned peer shadows their first interactions with dangerous compounds. Secure cabinets keep walk-ins from grabbing anything out of curiosity, and we check our policies every quarter. Safety isn’t just a box to check for compliance—it shapes every decision on how and where these chemicals get stored.
What are the handling precautions for Chromium Trifluoroacetate?
A Chemical with a Strong Personality
Chromium trifluoroacetate isn’t the kind of chemical you set aside on a crowded shelf and forget. This compound has its place in research, especially in organometallic synthesis and catalysis. Handling it takes patience, respect, and a determination to look out for yourself and your coworkers. Risks come not just from the chromium, but also from the trifluoroacetate group: strong oxidizing tendencies and persistent toxicity remind us to stay alert.
Don’t Rely on Luck—Use PPE
Years in the lab taught me that gloves, goggles, and a fitted lab coat aren’t up for debate. Nitrile gloves give a better barrier against both organic and inorganic chemicals than latex. Splash goggles beat safety glasses every time. Chromium trifluoroacetate can irritate lungs and skin, so keeping it off your body isn’t something you leave to chance. I watched a colleague ignore a small powder spill once, thinking a quick wipe would take care of it. His rash lingered for days. Lesson learned—prevention is always easier than cleanup.
Think Air, Not Just Surface
This chemical gives off fumes carriers never expect, especially in a warm, cramped workspace. Once, I watched a researcher toss some powder into a flask and the faintest wisp drifted upwards. That meant fume hood time, not room air. Local exhaust ventilation should hum away, since inhalation can do quiet damage to you before you notice anything wrong. Chromium compounds have histories with cancer and organ toxicity. No one should get casual about that risk.
Moisture and Heat: The Enemy
Chromium trifluoroacetate reacts with water vapor and breaks down when heated. This isn’t only a storage problem—it’s a sneaky hazard during experiments. Transfer a bottle on a rainy day or leave it near a steam pipe, and you’ll smell those acids before you see any obvious signs. Keep the substance sealed tight in a dry box, and use only the needed amount in smaller containers. No shortcuts. Accident logs fill up fast with stories of “just this once” events that turned ugly.
Cleaning up Right
Spills aren’t a reason to panic, but they do call for a practiced routine. Scoop solids with disposable tools, deal with contaminated surfaces using a mixture of sodium bicarbonate and absorbent material, and triple-bag waste for hazardous pickup. Letting traces build up over time risks turning a workstation into a slow hazard. I’ve stood through spill drills that seemed like overkill, but the real thing makes you grateful for every dry run.
Health Effects: No Mystery
Both hexavalent and trivalent chromium have earned unpleasant reputations. Trifluoroacetate presence ups the ante with environmental persistence and a potential to trigger respiratory or skin problems. According to the National Institute for Occupational Safety and Health (NIOSH), chronic exposure to chromium compounds raises the risk for liver and kidney damage. Some researchers carry that awareness home and pay more attention to symptoms, while workplaces rely on regular health surveillance. If a company claims handling is “totally safe,” they aren’t following the science.
Building a Safer Practice
Safety data sheets shouldn’t gather dust. Sharing them and reviewing storage plans helps keep everyone honest and careful. Training refreshers give people new respect for chemical hazards and spot risky shortcuts. Encourage a culture where double-gloving and spill drills aren’t measures to skip, and incidents will shrink. Sometimes it’s easy to get complacent in a lab. Daily habits, like prompt labeling and never working alone, turn caution into second nature.
In the end, respect for chromium trifluoroacetate doesn’t come from fear. It comes from learning that skills, care, and teamwork turn a risky chemical into a safe tool.
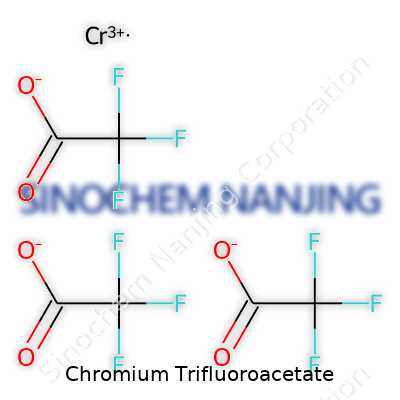
| Names | |
| Preferred IUPAC name | Tris(trifluoroacetato)chromium |
| Other names |
Chromium(III) trifluoroacetate Chromium trifluoroacetate |
| Pronunciation | /ˈkrəʊmiəm traɪˌflʊəroʊəˈsɛtət/ |
| Identifiers | |
| CAS Number | [39430-51-8] |
| Beilstein Reference | 3929325 |
| ChEBI | CHEBI:52715 |
| ChEMBL | CHEMBL4296615 |
| ChemSpider | 23313904 |
| DrugBank | DB11271 |
| ECHA InfoCard | 100.023.846 |
| EC Number | 208-859-5 |
| Gmelin Reference | 115138 |
| KEGG | C18648 |
| MeSH | D017941 |
| PubChem CID | 159398 |
| RTECS number | GB2325000 |
| UNII | 9O0R6YHF5K |
| UN number | UN3240 |
| CompTox Dashboard (EPA) | DTXSID5020703 |
| Properties | |
| Chemical formula | Cr(CF3COO)3 |
| Molar mass | 451.07 g/mol |
| Appearance | Green powder |
| Odor | Odorless |
| Density | 1.89 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -2.6 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +2040.0e-6 cm³/mol |
| Refractive index (nD) | 1.450 |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 437.6 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1232.8 kJ/mol |
| Pharmacology | |
| ATC code | A24AA10 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Lethal dose or concentration | LD50 oral (rat) > 5000 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Chromium(III) acetate Chromium(III) chloride Chromium(III) fluoride Chromium(III) nitrate Chromium(III) sulfate Chromium(III) oxide |

