Chromium Nitrate: A Closer Look at Its Path, Properties, and Role in Modern Science
Historical Development
The journey of chromium nitrate traces back to the 1800s, marking a time of rapid expansion in the field of transition metal chemistry. Chemists exploring the colorful world of chromium compounds found this salt offered a unique spectrum of colors and reactivity. Early synthetic work came on the heels of breakthroughs in isolating chromium as an element, after researchers like Louis Nicolas Vauquelin kicked off the first studies by analyzing minerals from Siberia. The field moved forward with developments in producing purer forms and more refined methods, setting the foundation for today’s more precise manufacturing and analytical tools. In labs and industry, chromium nitrate soon stood out for its versatility among metal nitrates.
Product Overview
Chromium nitrate generally points to the nonahydrate form, appearing as deep purple, almost red-violet crystals or granules. Often made available in technical, laboratory, and reagent grades, this compound comes with a high solubility in water and delivers chromium in a moderate oxidation state. It’s a crucial chemical in dye and pigment production, catalysts preparation, and some specialty synthetic applications. Producers focus on purity and moisture content, as impurities impact reactivity and suitability for sensitive work.
Physical & Chemical Properties
This salt, formula Cr(NO3)3·9H2O, consists of green-violet or deep red crystals. Its melting point sits around 60°C, but heating it any further breaks it down and releases nitrogen oxides. It dissolves easily in water or ethanol, and it gives acidic solutions that reflect chromium’s trivalent state. Unlike some other chromium compounds, this salt doesn’t produce toxic hexavalent chromium during regular handling, though strong oxidizers can sometimes make that leap. Nitrate anion brings both stability and reactivity, letting the compound serve varied chemical needs.
Technical Specifications & Labeling
Suppliers stamp containers with purity—often above 99% on a dry basis for lab use—and underline water content, since the nonahydrate changes weight based on storage. The labeling also calls out the batch or lot number, production date, country of origin, and safety information like hazard pictograms and proper storage instructions. Chemical Abstracts Service (CAS) Number and the molecular mass both support traceability in international supply chains. Shipping and storage standards reference United Nations recommendations due to the oxidizing nitrate group.
Preparation Method
Typical preparation involves reacting chromium(III) oxide or hydroxide with nitric acid under carefully controlled temperatures, avoiding excessive acid or heat—otherwise, the compound may decompose. After the reaction, the solution slowly cools, forming crystal hydrates that workers filter and dry. Recrystallization helps to boost purity, which stands out as important for analytical chemistry or catalyst fabrication. Sometimes, chromium nitrate comes as a byproduct of industrial nitrate treatment cycles.
Chemical Reactions & Modifications
Chromium nitrate works as a flexible starting material for several inorganic syntheses. It acts as a source of trivalent chromium in making catalysts or other coordination complexes. Heat with a reducing agent such as hydrogen or charcoal, and it yields chromium oxide—a key pigment. Add reactants from the world of organic chemistry, and the nitrate can support oxidation or electrophilic addition at moderate temperatures. In solution, it swaps ligands easily, paving the way for custom complexes tailored for specific textile dyeing or ceramic coloration jobs.
Synonyms & Product Names
Industry and science both know chromium nitrate under other names. Chromium(III) nitrate, chromic nitrate, or the systematic chromium trinitrate all refer to the same compound. In catalogs, it sometimes appears as chromium(III) nitrate nonahydrate when specifying the hydrate’s full form. International labeling standards like GHS list it by its full name or UN number for shipping.
Safety & Operational Standards
Handling chromium nitrate calls for proper gloves, eye protection, and good ventilation. The compound’s oxidizing properties mean it must stay apart from flammable or reducing agents. Although trivalent chromium is far less risky than its hexavalent counterpart, dust or accidental ingestion still prompts irritation or—over repeated exposures—potential organ trouble. Wastewater treatment guidelines require neutralization and precipitation before disposal, following both local and national hazardous waste rules. Safety Data Sheets detail first aid and emergency information with clear, actionable directions.
Application Area
This compound finds use in several factories and research labs. In textile dyeing, it acts as a mordant to lock pigments into fibers. Ceramic and glassmakers use its rich hues for glazes and coloring, and metallurgists use its chemical reactivity to generate anti-corrosion coatings. Catalyst developers lean on its role as a chromium source, particularly in polymerization and selective oxidations. Some niche synthetic fields utilize the nitrate to produce specialty chromium complexes or ionic liquids for research.
Research & Development
Scientists continue probing new uses for chromium nitrate, especially in the nanoscience and materials sectors. Nanoparticle synthesis research tests it as a precursor for magnetic, catalytic, or photonic materials. In green chemistry, teams analyze its performance as a catalyst source in selective oxidation with minimal waste. Environmental researchers monitor its mobility and breakdown in water and soil, supporting cleaner production cycles and waste treatment upgrades. As renewable technologies advance, the metal’s redox chemistry offers promise for solar cells or energy storage.
Toxicity Research
Chromium(III) compounds, including this nitrate, show lower toxicity than chromium(VI) types, but occupational health studies don’t ignore them. Respiratory and digestive irritation remains a risk through dust or accidental ingestion in unprotected settings. In plant and aquatic systems, some buildup can affect growth, so environmental chemists keep watch near discharge points. Chronic high-dose exposure connects to organ effects, so ongoing biological monitoring and stricter workplace air standards minimize risks. Research continues into environmentally benign alternatives for large-scale applications.
Future Prospects
Looking ahead, the world of chromium nitrate faces a blend of opportunity and challenge. As demand grows for precision catalysts and safer materials, new production methods promise cleaner output and tighter impurity control. Regulations push for lower emissions and stricter toxicity profiles, spurring companies to design closed-loop handling and zero-waste recycling. In science, expect further study of its coordination chemistry in advanced materials or medical imaging contrast agents. Graduate students and industry chemists likely will find chromium nitrate at the heart of electrochemical, catalytic, and environmental innovation for years to come.
What is Chromium Nitrate used for?
Behind Many Science-Lab Doors
Chromium nitrate doesn’t turn up in the average home—I’ve only seen it in school and industrial settings. Right away, that tells you it’s not something people use in daily cooking or cleaning. Its main role shows up in chemistry classrooms and research labs. Teachers and students use it for experiments that show chemical reactions, changes in color, and the behavior of transition metals. In my high school chemistry class, I’ll never forget seeing a deep purple solution appear from thin air, just because the teacher sprinkled in a bit of chromium nitrate. That color isn’t just eye-catching; it helps chemists spot differences between metals and work out what’s dissolved in a liquid.
The Industry Side: Coatings and Dyes
Factories also carve out a spot for chromium nitrate in their processes. Metal companies use it to create protective coatings through a method called electroplating. What that means: they cover one metal surface with a thin layer of another. This process can help fight off rust or give a smooth, shiny finish. If you’ve ever noticed the gleam on tools or machinery, sometimes chromium nitrates do the trick behind the scenes. By building up a line of defense, manufacturers cut down on corrosion that can lead to breakdowns or wasted resources.
On top of coatings, textile industries turn to chromium nitrate for dyeing and printing fabrics. Dyestuffs mix with chromium nitrate to set bright colors—think about your favorite shirt that hasn’t faded after fifty washes. That vibrant, lasting hue often relies on a tough bond formed with this compound. Painters, glass-makers, and even the creators of ceramic glazes value it for the color shifts it brings. Chromium gives a green tint to glass and ceramics, something I spotted once during a glassblowing workshop.
Learning to Tread Carefully
No matter how useful a chemical might be, there’s always a flip side. Chromium nitrate belongs in the category of substances to handle with care. Tiny particles and dust can cause harm—skin irritation, breathing problems—if care slips. Safety policies from groups such as OSHA stress limits for exposure. They recommend gloves, goggles, and solid ventilation in the workplace. In community college, every chem lab charted clear rules: spills were never ignored, and cleanup supplies had to be within arm’s reach. Stories circulate about “just a little dust” causing trouble, and they’re no exaggeration.
Potential Paths Forward
Many industries keep looking for safer or more environmentally-friendly chemicals. Some manufacturers test new coatings that rely less on metals or produce less waste. In textile and ceramic fields, scientists work on dye bonds and glazes that don’t use chromium at all. These tweaks don’t happen overnight; old ways sometimes die hard. Encouraging better training, stricter storage protocols, and broad chemical recycling programs might help shrink the risks further.
Why Paying Attention Pays Off
Every industrial chemical has a story that stretches well beyond the lab. Chromium nitrate offers safer, long-lasting products, vivid colors, and deep insight into chemistry, but it also reminds us to weigh benefits against risks. In labs, classrooms, and workshops, the best outcomes come from people paying attention and taking responsibility for what’s in their hands.
What is the chemical formula of Chromium Nitrate?
Why Knowing the Chemical Formula Matters
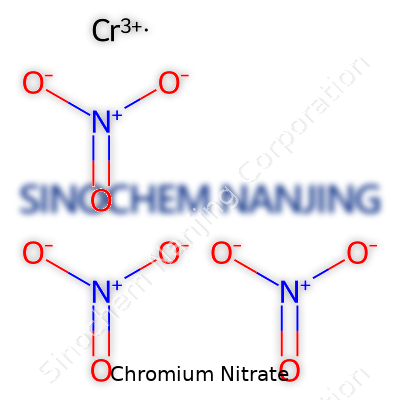
Walking into a science classroom or an industrial facility, you might spot bottles labeled with all kinds of chemical names. One that pops up in both education and industry is chromium nitrate. The formula, Cr(NO3)3, at first looks complicated. It really just shows that each chromium atom bonds with three nitrate groups. This information gives much more than a shortcut for chemistry students. It tells anyone handling the compound about the composition, how it could react, or what to expect if it comes in contact with other chemicals, or even water and air.
Real-World Uses and Hazards Rely on Formula Knowledge
Understanding chromium nitrate’s structure guides how people apply it. In a lab setting, I’ve seen chromium nitrate play a role in preparing chromium salts for use in other chemical reactions. It finds a spot in textile dyeing and printing because of its ability to provide a deep, steady color. People working with chrome plating processes know it as a tool that can help produce bright, hard metal surfaces, which often go unnoticed in car parts and tools.
Its chemical formula matters because chromium exists in several forms, each carrying a different risk. Chromium nitrate, following the formula, contains Cr3+. This trivalent state makes it less poisonous than the infamous Cr6+ found in compounds like chromium(VI) oxide. Misunderstanding or mislabeling could lead to confusion— with health consequences and legal trouble. I’ve seen a colleague stop mid-experiment, double-checking, because he noticed the color was off from what the formula suggested.
Environmental and Health Insights
Chromium nitrate’s solubility, a direct result of its formula and structure, means it dissolves quickly in water. This boosts its usefulness, but also raises questions about disposal and environmental impact. In high concentrations, as the environmental protection agency warns, chromium compounds can harm aquatic life. So, factories using chromium nitrate in their processes have to treat their waste carefully to avoid letting dangerous run-off into rivers.
The occupational safety and health administration in the US lays out precise rules for handling chromium compounds, especially in powder or aerosol form, to shield workers from respiratory risks. Technicians who understand what Cr(NO3)3 means can read labels and safety data sheets and make smart decisions about gloves, ventilation, or eyewash stations. Having spent hours in chemistry prep rooms, I know the formula on a bottle is more than a dry fact; it's key to making sure everyone leaves work safe and healthy.
Paving a Safer Path Forward
Better training and clearer labeling in both schools and industries could narrow the gap between knowing a chemical’s formula and knowing how to handle it— that’s a step toward fewer accidents and healthier people. Companies could run refresher sessions, focusing not only on “what the formula is” but also “what it means in this process or product.” Solutions don’t have to cost a fortune; hands-on demonstrations and on-the-spot quizzes during safety meetings help workers remember the difference between a trivalent and hexavalent chromium salt when it counts.
If everyone in a lab or on a shop floor understands why Cr(NO3)3 matters, from its formula to its real risks and uses, fewer surprises come up. That’s a win for science, for business, and for anyone who wants to keep learning more about what’s in the bottle in front of them.
Is Chromium Nitrate hazardous or toxic?
What We Know About Chromium Nitrate
Chromium nitrate pops up in labs, classrooms, and sometimes in the manufacturing world. Mixing chromium and nitrate ions brings out a bright violet chemical, known for its strong oxidizing powers and solubility in water. Schools often include it in chemistry kits. Some companies rely on it for dyes, catalysts, and certain surface treatments. But curiosity doesn’t always mean safety, and folks often ask: how risky is this stuff, really?
Human Health Risks: Not Just a Lab Problem
The health hazards start with chromium’s form. Here, it comes in the trivalent form—chromium (III)—which acts much less aggressively than its notorious cousin, chromium (VI). Still, that doesn't mean you can just shrug it off. Prolonged or careless exposure has ever-present dangers. For instance, breathing in dust or mist can sting the respiratory tract, causing coughing or shortness of breath. Skin doesn't fare much better. Direct contact can irritate or even cause rashes, especially in people sensitive to heavy metals. If swallowed, nausea and stomach pain might follow.
Occupational exposure sheds more light. Studies have clocked in higher chronic conditions among workers regularly handling water-soluble chromium nitrates, including allergies, eczema, and, in poorly ventilated spots, possible kidney or liver issues after long-term high-level contact. Accidental spillage or improper disposal boosts those risks to whole communities down the line through water or soil contamination.
Environmental Impact: More Than Meets the Eye
Chromium nitrate doesn't just disappear when poured down the drain. It breaks apart and slips easily into soil and groundwater, threatening aquatic life. That violet color isn't just pretty — it signals the presence of heavy metals, which can travel with rain or runoff to reach rivers and lakes. Fish and insects take the first hit, disrupting food webs and threatening drinking water quality.
The U.S. Environmental Protection Agency groups water-soluble chromium compounds as hazardous waste for a reason. The stuff makes its way into landfill leachate or wastewater, and often ends up in places it shouldn't. Regular monitoring and safe storage become critical steps. Ignoring these means dealing with polluted soil or poisoned streams — and cleaning that up costs far more than prevention.
What Works for Safer Handling?
Anyone dealing with this substance ought to suit up with gloves and goggles, practice good housekeeping, and avoid eating or drinking in the work area. Good ventilation turns out to be a real difference-maker. Proper labeling and locked storage shelves keep chemicals out of the wrong hands. Training helps, too—folks who know the risks rarely cut corners, especially if everyone supports a culture of safety.
Disposal brings another layer. It isn’t something to flush away or toss in the trash. Hazardous waste collection sites exist for a reason, and companies must stick to legal protocols. If an accident does happen, spill kits, showers, and clear emergency procedures go a long way in minimizing harm.
What Needs Fixing?
The answer isn’t locking up every bottle or banning chromium nitrate altogether. Schools and industries can enjoy the benefits without cutting corners on safety. Real education and transparency, enforcing rules, and regular checks can make a huge difference. If oversight slacks off, the costs—health, environment, and legal—pile up quick. Replacing hazardous chemicals with safer alternatives stands as a goal worth chasing, but for now, careful management remains the frontline defense.
How should Chromium Nitrate be stored?
Why Storage Matters
Some chemicals cause more headaches than others. Chromium nitrate lands in that category. Working with it for years in research and teaching labs, I’ve learned shortcuts don’t pay off. The bright purple crystals look harmless, but things go sideways fast if ignored on a shelf or left in the wrong spot. Explaining the reasoning behind good storage isn’t just about following rules; it’s about staying safe every time someone walks by that cabinet.
The Real Risks Behind the Name
Chromium nitrate pulls moisture out of the air like a magnet. Leave the lid loose, you’ll come back to find a sticky mess eating through packaging, staining everything around it. Any contact with skin can bring irritation, and inhaling dust or fumes from an accidental spill can mess with your lungs. Toss some heat into the mix during a storage accident, and it’ll decompose into toxic and corrosive gases — definitely not something to ignore.
Best Storage Practices from the Trenches
Put away the notion that ordinary shelves or open jars will do. Genuine safety begins with a solid, airtight container. Thick glass or correctly labeled plastic works well. Keep the jars off floor level. Cabinets designed for storing oxidizers (not just acids) provide a solid layer of insurance. Separation keeps reactions from happening. Never shelve chromium nitrate close to anything flammable — paper, solvents, or even strong reducing agents. Mistakes multiply when chemicals mingle in the wrong way.
Humidity matters more than most folks think. Storage rooms should stay dry, cool, and out of direct sunlight. Humid labs speed up caking, corrosion, and leaks. I learned the hard way during a summer heatwave: the old storage closet sweated so badly that containers bloated and rings of residue formed under lids.
Label Everything, Mind the Records
Clear labeling saves lives and time. Every bottle gets a tough, legible label with the chemical name, concentration, and hazard details. Relying on memory or faded stickers creates confusion, especially for new staff or emergency responders. Good records track usage and remind staff to inspect containers for cracks, buildup, or unusual color shifts.
Regular Checks Keep People Safe
Weekly inspections catch trouble before it grows. I set aside fifteen minutes at the end of each week to run through chemical storage, scout for leaks, and replace worn-out labels. Most bad accidents start with a lid not screwed down tight or someone forgetting to wipe up crystals from the top.
Training: The Missing Piece
Every new lab tech watches storage in action — not just a slideshow, but a walk-through with real containers. Discussing mishaps I’ve seen firsthand drives home the point. Knowing what happens when a bottle seeps in a forgotten drawer keeps everyone alert. This kind of training works better than thick binders left on a shelf.
Disposal and What Comes Next
Nothing lasts forever, and old chemical stockpiles pose risk. Used or expired chromium nitrate belongs in a sealed, labeled waste container, handled by trained professionals. Local regulations spell out disposal protocol, but the basics remain: keep the stuff contained and out of regular trash streams.
Take these steps, and the risk drops fast. Chromium nitrate demands respect. Storing it right turns a dangerous compound into a manageable tool for science and industry.
What are the safety precautions when handling Chromium Nitrate?
Understanding the Risks
Chromium nitrate belongs in the family of chemicals that demand respect. Anyone who's worked around it knows how crucial good habits are in keeping people and property safe. It brings a bright green color to the table, but its risks don't appear on the surface. Problems often start small: a splash to unprotected skin, a sniff of unseen dust, or a moment of carelessness. That's all that’s needed to ruin a day—or much worse, a career. I’ve learned that proper precautions make a bigger difference than most folks realize.
Protecting Your Body
No casual dress code fits here: lab coat, gloves made for chemical resistance, and snug-fitting goggles should become second nature. Forgetting just once opens the door for a burns, rashes, or damage to the eyes. Even people who work with Chromium nitrate every day know they can’t become careless. The National Institute for Occupational Safety and Health puts Chromium VI compounds in the category of occupational carcinogens, so respiratory protection also enters the picture if any chance of dust or vapors exists. I always remind new lab members: it’s impossible to tell how tiny particles or unseen fumes move around. A real respirator—fitted, not one of those blue surgical masks—brings peace of mind during trickier jobs.
Air Quality and Ventilation
The stories of accidental inhalation don’t get shared at safety meetings as often as spills, but they cause real harm. Fume hoods aren't optional. Proper airflow strips harmful particles from the workspace, reducing risks that can build quietly over time. I’ve replaced clogged filters, wiped sticky residues, and seen what happens when a fume hood isn’t working at full strength. Taking time to check airflow before starting pays off every time. Simple airflow devices easily show if a hood draws enough air, helping people avoid slow poisoning from inhaled compounds.
Storage Matters More Than You Think
Too many believe that after closing the bottle, the danger disappears. Storage—away from heat, sunlight, or anything reactive—becomes part of the safety routine. I’ve seen bottles sitting on hot windowsills or jammed next to acids. That’s more than a mess; it invites corrosion, fire, or dangerous chemical reactions. Clearly labeled containers, locked cabinets, and a well-organized chemical storage area don’t just streamline work—they stop accidents born of confusion or laziness.
Spill Response and Waste Disposal
A small spill feels harmless, but even a few grams of Chromium nitrate demand attention. Absorbent pads, neutralizing agents, and waste bins stand ready for a reason. I’ve watched someone try to towel up a spill before, only to realize days later that mishandling left toxic residue. Reporting every spill, even mistakes that might bruise the ego, helps keep everyone out of harm’s way. Waste heads into special bins for hazardous materials—not down the sink, never in the trash. Following local regulations for hazardous waste brings peace of mind and avoids damaging the water supply or local environment.
Knowledge and Habit Save Lives
Nobody starts a career in chemistry planning to get burned or sickened by a bright green salt. Reading the safety data sheet, asking questions, and practicing good habits—those simple steps add up to decades of healthy work. The lessons come from real experience: the scars, the close calls, and the coworkers who slowed down to show me the right way. Chromium nitrate isn’t a villain or a boogeyman—it’s just a chemical. With the right respect, it stays safe.
| Names | |
| Preferred IUPAC name | Chromium(III) nitrate |
| Other names |
Chromium(III) nitrate Chromic nitrate |
| Pronunciation | /ˈkroʊ.mi.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13548-38-4 |
| Beilstein Reference | 14639 |
| ChEBI | CHEBI:63071 |
| ChEMBL | CHEMBL1201117 |
| ChemSpider | 16215778 |
| DrugBank | DB14655 |
| ECHA InfoCard | ECHA InfoCard: 03c3bd365e-b2ae-4fb3-87ce-03cb885447c5 |
| EC Number | 231-907-1 |
| Gmelin Reference | 83722 |
| KEGG | C18516 |
| MeSH | D017681 |
| PubChem CID | 24594 |
| RTECS number | GB5420000 |
| UNII | 97LMA81YKA |
| UN number | 1466 |
| Properties | |
| Chemical formula | Cr(NO3)3 |
| Molar mass | 238.01 g/mol |
| Appearance | Dark purple crystals |
| Odor | Odorless |
| Density | 1.85 g/cm3 |
| Solubility in water | 642 g/L (20 °C) |
| log P | -1.2 |
| Basicity (pKb) | -5.0 |
| Magnetic susceptibility (χ) | +100.0e-6 cm³/mol |
| Refractive index (nD) | 1.205 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 240.6 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | −404.6 kJ/mol |
| Pharmacology | |
| ATC code | A24AB10 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, toxic to aquatic life |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS01,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H334, H335, H350 |
| Precautionary statements | P273, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 325 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 52 mg/kg |
| NIOSH | 0237 |
| PEL (Permissible) | 0.5 mg/m3 |
| REL (Recommended) | REL (Recommended): 0.01 mg(Cr)/m³ (as Cr) |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Chromium(III) oxide Chromium(III) sulfate Chromium(III) chloride |

