Looking Closer at Chloromethyl Chloroformate: Beyond the Chemical Formula
How Chloromethyl Chloroformate Took Shape Over Time
Chemistry textbooks keep making room for compounds that shape lives in ways many people never consider. Chloromethyl chloroformate started to show up in labs and research reports decades ago. As the chemical industry expanded across the twentieth century, the pursuit of more specialized reagents began to drive attention toward molecules like this one. Scientists searching for tailored reaction steps needed building blocks that would unlock new pharmaceutical pathways, advanced polymers, and specialty materials, and chloromethyl chloroformate stepped out from a background of similar derivatives. Research journals from the 1960s already catalogued reactions using it to fine-tune pharmaceuticals, and by the 1980s, its role in peptide chemistry grew increasingly clear. My experience paging through old chemical catalogs shows that its listing moved from obscurity to routine in just a few generations. Looking at how it evolved underscores how demands from medical and industrial innovation steadily push new chemicals into the spotlight.
Understanding What Chloromethyl Chloroformate Really Is
To many in the science world, its structure is almost iconic: a reactive carbonyl flanked by a methyl chloride group on one side and a chloroformate tail on the other. Chloromethyl chloroformate sits as a colorless or slightly yellow liquid, often with a pungent, acrid odor that warns anyone nearby to put on gloves and masks. This compound isn’t something you want to spill or breathe, which highlights just how serious its chemistry can be. With boiling points near 74°C and low vapor pressure at room temperatures, it’s volatile but still manageable in a controlled lab setting. Flammability and compatibility issues come into play, especially if water or alcohols are in the same room. What always sticks with me after handling this substance is the sensation that even small quantities demand respect, both for their reactivity and for potential hazards.
The Rules and Labels that Keep Labs Running Safely
Safety protocols around chloromethyl chloroformate reflect a long history of tough lessons learned. Over the years, labels changed from cryptic abbreviations to bold hazard pictograms, giving even non-chemists a fighting chance at understanding what’s at stake. Regulatory bodies like OSHA and the European Chemicals Agency stress eye and skin protection, point out the corrosive and irritant risks, and call for strong ventilation. Material Safety Data Sheets outline detailed storage instructions, recommending cool, dry conditions and regular inspection since this kind of chemical can break down or leak. Handling it reminds me of training new lab members: once someone reads about the toxic byproducts and sees what exposure risks mean in the real world, care improves dramatically. The moment someone lapses, accidents follow—an unfortunate constant in chemical work.
How Preparation Has Evolved—And Why That Matters
Back in the early days, making chloromethyl chloroformate involved reactions that left behind plenty of problematic residues and took more effort to control. Modern synthesis often starts with phosgene as a key reactant, working alongside chloromethanol or related intermediates under strictly anhydrous conditions. Phosgene itself carries notorious safety concerns, so today’s chemists press for alternative preparation routes or automation wherever possible. This isn’t just about process efficiency—it's a matter of survival. Even now, process improvements come from reducing operator contact and tightening controls around emissions. I’ve seen research labs upgrade fume hoods and invest in new personal protective equipment just to keep up with best practices. In industry, continuous-flow chemistry sometimes replaces batch methods, reducing the total amount of hazardous material in one place at any time. With each adaptation, labs edge closer to safer, more sustainable production.
Why Its Chemical Reactivity Drives Research and Industry
Chemical reactivity lands at the heart of what makes this molecule valuable. The two key functional groups—chloroformate and chloromethyl—open several possibilities for reactions with amines, alcohols, and thiols. This capacity underpins its use in coupling chemistry, especially for forming carbamate and urethane linkages. In the biotech world, it’s used to protect or activate sensitive groups as part of peptide synthesis, enabling the assembly of complex bioactive molecules with carefully controlled steps. Chemists appreciate reagents like this for their selectivity, and over time, methods improved to give higher yields and fewer side products. Research always circles back to how these precise modifications lead to new drugs, polymers, or molecular probes. Talking with chemistry grad students, I hear complaints about its strong reactivity—one small slip, and you ruin a synthesis or set off a dangerous side reaction. But without it, many research advances would crawl to a halt.
The Many Names and Faces: Synonyms Keep Cropping Up
Ask around in different labs, and someone is bound to call this chemical by a different name. Chloromethyl chloroformate, methylcarbonochloridate, and CMCF all refer to the same compound. In journals and older textbooks, names like chloroformic acid chloromethyl ester appear, sowing confusion for newcomers or those combing through the literature. This tangle of nomenclature forces researchers to double-check safety sheets and reaction protocols, especially given how easy it is to confuse similarly named chemicals—some of which act very differently in a flask. I always check both CAS numbers and IUPAC names before ordering or using any specialty reagent, because a small translation mistake can derail months of effort. Consistent naming conventions form the backbone of data-sharing, peer review, and reproducibility in experimental science.
Practical Safety and Habits in Working with Chloromethyl Chloroformate
Lab routines that handle highly reactive compounds call for more than a checklist. Chloromethyl chloroformate demands a well-trained team comfortable with full personal protective wear, strict protocols for chemical transfers, and thorough waste management. Splash incidents need immediate response gear, and storage requires secure, well-labeled containers away from water, strong bases, and heat sources. Toxicity hovers in the background, and local exhaust ventilation becomes essential, as does rapid access to eyewash and drench showers. My own experience with chemical safety drills—painstaking and tedious as they may feel—gave me the muscle memory to act fast during real emergencies. Proper disposal procedures, air monitoring, and periodic refresher training help keep complacency in check. In the end, constant vigilance and a willingness to revise protocols save both lives and resources.
Application: Where This Chemical Proves Its Worth
Chloromethyl chloroformate stands out in specialty organic synthesis. As a reagent, it helps build complex molecules by facilitating the introduction of carbamate groups—these show up in a wide range of drugs and agricultural chemicals. Medicinal chemists lean on it for forming linkages in protecting-group strategies when building peptides and nucleotides. I’ve seen research groups use it to synthesize diagnostic imaging agents or develop new enzyme inhibitors. Beyond healthcare, it finds a niche in materials science by linking polymer subunits or modifying surfaces for better performance. Demand rises with advances in precision medicine because these reactions let drug developers tweak molecular structures with greater specificity. As new generations of pharmaceuticals and biodegradable plastics crop up, the tools that enable these advances—including this compound—take on greater significance.
What’s Happening at the Cutting Edge of Research and Development
Chloromethyl chloroformate keeps showing up in academic conferences and grant proposals centered on novel synthesis methods. Newer routes focus on minimizing hazardous precursors and streamlining purification, which often aligns with green chemistry goals. Collaboration between industry and university labs sometimes brings pilot-scale processes online much faster than a decade ago. Research teams now use robotic systems for micro-scale reactions, tracking which procedures reduce risks without sacrificing yield or purity. After seeing how minor changes to reaction conditions can unlock massive improvements, it’s clear why development work often centers on optimizing every step. Analytical chemists track impurities more closely, and computational models simulate reactions before lab trials start. This new toolkit shrinks both cost and environmental footprint, pushing the field forward in practical, measurable ways.
Toxicity: The Uncomfortable Side of Progress
Any enthusiast raving about new chemistry must confront the darker side—chemical toxicity and environmental hazards. Chloromethyl chloroformate stings the eyes and skin, burns the throat, and harms the lungs if inhaled. Chronic exposure ties to organ damage, and accidental spills can lead to serious injuries or worse. Some decomposition products bring even greater risks, which is why emissions control and spill response matter as much as reaction yield. Regulatory scrutiny intensifies as more data emerges, driven by experimental toxicology and workplace incident reports. Proper engineering controls, improved air handling, and the substitution of less dangerous reagents hold the key to safer workplaces. Developing strategies for safer disposal and containment now draws as much interest as novel reactions. Those who ignore these risks rarely last long in the field—most experienced chemists carry stories of close calls that left lasting lessons.
The Road Ahead for Chloromethyl Chloroformate
Looking toward the future, the fate of chloromethyl chloroformate will ride on efforts to manage its risks while expanding its utility. Companies and universities invest in alternative reagents that deliver the same precision in coupling chemistry but with less baggage from toxicity and handling hazards. Meanwhile, the regulatory landscape shifts as new safety data prompts updated guidelines and stricter controls. Advances in continuous manufacturing, micro-reactor technology, and computational chemistry might enable reactions that sidestep the need for such volatile intermediates entirely. On the research side, every breakthrough in safer or more selective chemistry raises the bar for what investigators and manufacturers expect. For now, chloromethyl chloroformate sticks around as a valued tool in the arsenal of modern organic synthesis, but the search continues for safer, greener substitutes that don’t compromise performance or accessibility. Until then, those who work with it do so with vigilance, always weighing its power against its perils.
What are the main applications of Chloromethyl Chloroformate?
The Building Block in Chemical Synthesis
Chloromethyl chloroformate looks like just another chemical on a list, but most chemists know it stands out on the lab bench. This compound acts as a real workhorse in the synthesis of various agents, and it owes its value largely to the reactive nature of its structure. In pharma labs, chloromethyl chloroformate jumps in where others fail, helping to bridge simple molecules and complex drugs. If you’re talking about making antiretroviral medicines or anti-cancer compounds, more than a few of them rely on this chemical during early-stage reactions.
Scientists usually seek out chloromethyl chloroformate because of its capacity to introduce the so-called “chloromethoxycarbonyl” group onto nitrogen or oxygen atoms. That might sound technical, but in practical terms it lets drug designers build scaffolds for new treatments, link different bioactive units together, and produce compounds that could eventually land in a hospital pharmacy. In 2021, for example, some recent antineoplastic agents depended on reactions made possible by this very molecule.
Peptide and Protein Modification
A chemist working on peptides often hears about chloromethyl chloroformate almost as a rite of passage. It opens the door to protecting the amine group on amino acids. This temporary “blocking” step is essential, because most syntheses fall apart if you try to rush without protection — literally and figuratively. Peptide chemists who skip this step tend to lose yield and spend more hours at their benches.
This practice matters for companies racing to develop therapeutic peptides, hormones, or enzyme inhibitors. Chloromethyl chloroformate shapes the process, letting them build complex molecules without side reactions making a mess. Today, the biotech industry sees growing demand for synthetic peptides, with projections heading over $25 billion by the late 2020s. So, the role of efficient protective groups in this chemistry isn’t just academic — it translates to real jobs, innovation, and hope for patients.
Agrochemical Production Gets a Boost
A lot of the world’s food security rests on the back of plant protection, and once again, chloromethyl chloroformate plays its part. Agrochemical manufacturers lean on its ability to deliver specific modifications, tailoring molecules to knock out crop diseases but spare beneficial plants and insects. This precision couldn’t be achieved using blunter synthetic tools.
Chloromethyl chloroformate enters the scene early in agrochemical development, especially during the crafting of carbamates. Many herbicides, insecticides, and fungicides rely on these core structures for their effectiveness. Just last year, a crop protection startup in Iowa built an entire line of products with this chemistry at its core. Better crop yields, lower environmental footprint, and jobs in rural economies all start with reactions made possible by this fine chemical.
Safety, Waste, and Looking Forward
Handling chloromethyl chloroformate demands real caution. It causes nasty respiratory and skin reactions, so strict safety protocols come into play. My own time in a chemical plant — oversized gloves, triple-sealed hoods, all of it — taught me where shortcuts can backfire. Companies invest in scrubbers, sealed reactors, and employee training not because they want to but because accidents cost lives and stall projects.
Disposal also poses challenges. Waste management teams use neutralizing agents and high-efficiency capture systems to prevent environmental harm. There’s a real push now toward greener substitutes or better closed-system technology, because sustainable transitions don’t happen by accident. The chemical industry can anchor itself on a foundation of best practices, but it thrives when innovation pushes both safety and environmental solutions.
What are the safety precautions when handling Chloromethyl Chloroformate?
Understanding the Challenge
Chloromethyl chloroformate demands attention the moment the bottle comes out of storage. In my time working in a laboratory, I saw how even experienced chemists show real caution around it. This chemical releases toxic fumes—some folks call it “phosgene in a bottle.” There’s no guesswork here. Your skin, eyes, and lungs all become targets when exposure happens. Breathing the vapors can trigger coughing, chest tightness, and headaches. Splash it on your skin and burns show up almost instantly. It’s comforting to know that scientific research, such as accident investigation reports from OSHA and NIOSH, highlight these dangers after laboratory mishaps.
No Substitute for Proper Gear
Working with chemicals like chloromethyl chloroformate brings out protective instincts. Every person I’ve worked with trusts in their PPE—not as an afterthought, but as a central routine. Nitrile gloves and a full lab coat come first. Goggles shield eyes, but I always go one step further and choose a full-face shield during transfers. Labs with chemical fume hoods make a huge difference. The fume hood’s sash stays down; my hands do the work underneath the shield of moving air that grabs escaping vapors. Those hoods rescue people more often than you’d think. Respirator masks go beyond what a simple dust mask offers—research shows certain filters block out phosgene-like gases far more effectively. Laboratory SOPs often spell all this out, but shortcuts put people at risk.
Mindful Storage and Handling
Getting careless with containers changes the day from routine to emergency. I label every bottle right away, with date and concentration. Caps screw on tight, bottles sit away from acids or any reactive chemicals. Heat turns those vapors loose, so everything stays below room temperature. Refrigerators marked for chemical storage—never the ones used for food—become the safest bet. Even a brief lapse can create a chain reaction. I’ve seen a storage mixup trigger an evacuation, just because someone put a bottle in the wrong bin. Reviewing storage guidelines from manufacturers like Sigma-Aldrich or Fisher Scientific highlights how critical these details become.
Emergencies and Spill Response
Spill kits save hours and heartbeats. Materials like vermiculite absorb liquids fast, letting us scoop up spills and dispose of them as hazardous waste. Emergency showers and eyewash stations stand within a few steps for good reason. I’ve jumped for that pull handle before when I saw a colleague splashed. Speed makes a difference: rinsing for at least fifteen minutes gets recommended, but you want that first rinse within seconds. Training sessions, whether run by in-house safety officers or external consultants, shape how confidently people react. Reviewing incident case studies, published in journals or shared internally, shapes best practices and builds group memory around what not to do twice.
Building a Safer Culture
Lab safety with chloromethyl chloroformate links to how seriously people treat training. In one internship, staff told stories about what went wrong in earlier years—it anchored the message. Encouraging coworkers to speak up about unsafe behaviors changes outcomes. Peer checks before starting reactions bring a second set of eyes on complex procedures. Documentation from regulatory bodies points out one fact: shared responsibility matters as much as any single protocol.
Making the Choice to Respect the Risk
Respecting chloromethyl chloroformate means taking every step seriously, from wearing gloves to logging bottle locations. Shortcuts promise efficiency but deliver problems. Attention to detail, continuous education, and open communication guard against disaster. Every chemist builds these habits into muscle memory, not just for personal safety but for colleagues and the environment as well.
What is the proper storage condition for Chloromethyl Chloroformate?
Risks That Demand Respect
Chloromethyl chloroformate isn’t the kind of compound anyone takes lightly. People who use it, whether in a research setting or on the manufacturing floor, know it acts aggressively in the wrong conditions. I remember walking into a lab as a young intern, catching the end of a cleanup after a chemical leak. The silence in the room told its own story. Handling a substance like this calls for more than just technical knowledge; it asks for respect and constant vigilance. Without that, the risks escalate in ways you don’t want to see unfold.
Temperature and Storage Location Shape Safety
Stashing reactive chemicals in any cool, dark shelf is not a solution. Chloromethyl chloroformate needs a dedicated cold storage space. Not just any fridge will do. Laboratory-grade refrigerators, designed for flammables or hazardous chemicals, cut down the risk of accidental sparks or heat. Keep it below 8°C. Warmer conditions tip the odds in favor of decomposition, and nobody wants to meet its toxic fumes or face an explosion. The Laboratory Safety Institute documents accidents caused by simple power failures—room temperature storage can spell trouble overnight.
Containers: More Than a Formailty
Glass bottles with tight, chemical-resistant seals remain the trusted choice. The cap shouldn’t corrode or allow vapors to seep out. Ordinary plastic won’t hold up over time; some types degrade, letting the contents react with moisture or air. I’ve seen colleagues open old bottles where the cap literally fused to the top, thanks to chemical attack. Stick to manufacturer advice for container types, and check seals during every inventory. It saves time and headaches later.
Humidity and Ventilation Help Keep Hazards Contained
Chloromethyl chloroformate’s relationship with water gets ugly—fast. Even humid air can trigger corrosive byproducts and release gas. Shelves in storage rooms should stay dry, and desiccants help ward off moisture. Good ventilation matters just as much. Local exhaust and general airflow mean leaks or accidental releases won’t build up to unsafe concentrations. Labs that skip on airflow invite exposure incidents, which hit hard with chemicals as reactive as this one.
Isolation and Security
Mixing incompatible chemicals on the same shelf tempts fate. Stash chloromethyl chloroformate far from bases, strong acids, amines, and oxidizers. An extra containment tray works wonders in case of drips or spills. You don’t need many substances to set off dangerous reactions—a single overlooked bottle is enough. After one too many cautionary tales, I make a point to separate everything by type and label every space in shared storage units.
Building a Safety Culture
Warnings on the label only go so far. Training sessions, regular audits, and straightforward storage protocols translate to fewer accidents. The American Chemical Society and national occupational safety agencies urge the same—treat high risk chemicals with a kind of healthy paranoia. It’s not exaggeration; the harm from chloromethyl chloroformate mishaps lingers, both physically and mentally. Double-checking storage once and again beats any rush job or shortcut.
Better Habits, Safer Outcomes
Safe storage comes down to forming habits and sticking with them. Think cold, dry, sealed, isolated, and well-ventilated. For anyone curious about the specifics, go straight to the SDS or major chemical safety databases—they’ll back up every recommendation with case studies and years of hard lessons. Understanding the nature of chloromethyl chloroformate, then storing it with purpose, keeps both people and projects on the right track.
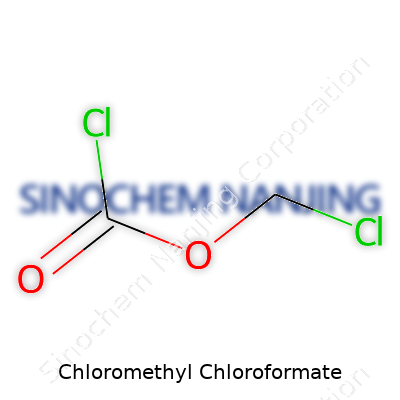
What is the chemical formula and molecular weight of Chloromethyl Chloroformate?
Looking at the Numbers: The Chemistry Behind the Name
Chloromethyl chloroformate shows up whenever someone wants to push the edges of organic synthesis. If you pick up a lab notebook, you’ll see its formula scribbled down as C2H2Cl2O2. Some days, this is all a chemist needs on the board to set off a chain of reactions. For folks who live in the world of molecular diagrams, this compound checks in with a molecular weight of 130.94 g/mol.
More than just numbers, that formula and weight carry a punch. A single extra oxygen, an added chlorine, means its reactivity shifts in ways you don’t always see coming. Decades ago, the first time I set out to work with chloromethyl chloroformate, the lab felt different – hood fan running full blast, glassware cleaned twice, hands wrapped tight in nitrile gloves. Its structure makes it more than a theoretical curiosity. The two chlorines and two oxygens flag danger and possibility all in one whiff. This isn’t a molecule for daydreamers. Mistakes leave a mark, and not just on paper.
Why Chemists Keep Talking About This Compound
Pharmaceutical pros and researchers know chloromethyl chloroformate for its role as a linking agent. In medicinal chemistry, it’s a building block. The molecule’s design – reactive, but not wild, tough, but still handleable with the right approach – fits well in stepwise synthesis. Its use jumped up in peptide chemistry, especially when you need to protect amino groups. Remove an obstacle, allow a new bond to form, then pull away the covering when you’re done. Having this tool makes it easier to piece together large, complex chains like those used for modern drugs or biotech solutions.
Alongside the high points, every bottle sits with warning symbols: toxic, corrosive. Store it wrong, a drop of humidity creeps in the bottle, and suddenly it’s doing more than intended. I knew a chemist who spent weeks tracking down byproducts—just water in the cap, just a hint of light, enough to unleash a mess.
Weighing Safety With Progress
Chemical labs walk a tightrope: discovery on one side, safety on the other. The molecular formula points to risks: two chlorines mean fumes no one wants to inhale, and that carbonyl chloride tandem can turn a mistake into a hospital visit. My own experience—reading MSDS two or three times, rehearsing disposal steps out loud—shows how vital it gets to plan ahead. Schools and labs should drill safe handling skills constantly, push beyond the minimum OSHA poster. My old advisor said, “You get one set of lungs—use your mind, not just your hands.” I’ve seen how goggles, careful air flow, double-tagged waste containers change the outcome for everyone in the lab.
Better Practices and the Path Forward
There’s pressure to challenge old methods. Chemists look for safer alternatives or try to invent more selective reagents that cut down side reactions and waste. Green chemistry initiatives encourage cutting hazardous waste and swapping out toxic building blocks. Even simple steps—shorter storage, smaller batch sizes, tailored ventilation—pile up to make an impact.
Understanding the formula and molecular weight of chloromethyl chloroformate doesn’t stay on the exam sheet. In every lab, every new synthesis scheme, the numbers steer choices on safety, design, and responsibility. Take it seriously, and you get progress. Forget the details, and you’ll wish you hadn’t.
How should Chloromethyl Chloroformate be disposed of after use?
Understanding What You’re Handling
Chloromethyl chloroformate doesn’t grab headlines like everyday household chemicals, but handling it demands respect. Folks working in chemical labs or industrial settings know this compound as a powerful reagent, prized for its reactivity — and that same energy means things can go south if it winds up in the wrong place. With heavy exposure risks, including severe damage to skin, eyes, or lungs, there’s no shortcut when talking disposal.
Why Getting Rid of It Wrong Hurts More Than Just the Environment
A few years back, a college down the road had to evacuate a wing after students poured leftover chemicals down the drain. Sewer pipes, treatment plants, and rivers ended up carrying the brunt, with local wildlife paying the price. With chloromethyl chloroformate, water isn’t its friend. Mixing the two shoots toxic gases like phosgene and hydrochloric acid right into the air. The danger leaps from the flask into the breathing space of anyone nearby.
Workers in chemical storage facilities know spills aren’t just cleanup jobs. A little mistake can fill a room with volatile fumes, and cleanup suits don’t erase the aftereffects on air quality. Hospitals sometimes see the fallout in the form of respiratory injuries. That’s a warning — not only for big companies but also for anyone with a forgotten bottle on a shelf.
Real Solutions for Real Risks
Most places with a responsible chemical management policy have a basic rule — leave toxic disposal to the pros. Licensed hazardous waste vendors have the gear, training, and permits to transport and process chemicals like chloromethyl chloroformate. Sending it to these experts shouldn’t feel optional. Routes involving sealable containers, clear hazard labeling, and airtight transfer help avoid spills.
Chemical destruction often uses specialized incineration. High-temperature furnaces break down molecules under controlled conditions, trapping anything nasty in advanced filtration systems. Not every incinerator fits the bill — smaller labs can partner with regional hazardous waste centers. Tracking manifests and digital logs keep everyone honest about where waste ends up.
Some research centers run chemical neutralization prior to disposal under a fume hood. This approach demands a skilled chemist, precision weighing, and copious safety gear — gloves, eye shields, air monitors. Rushing risks bigger trouble. Even after neutralization, the waste needs evaluation before lab staff pour or pitch it.
Why It’s Worth Doing Right
Incidents with dangerous chemicals rarely stay contained. Airborne accidents reach coworkers, neighbors, or kids at a nearby playground. The fines from environmental agencies only sting more if clean drinking water suffers or wildlife vanishes downstream. Insurance doesn’t always cover bad judgment.
Staff training doesn’t just tick a compliance box. It gives people the sense and confidence to catch mistakes before they scale up. Supervising leftover stock, mapping storage dates, and running disposal drills sharpen that instinct. Emergencies don’t wait for manuals. Knowing that a slip-up can haunt the company and community makes proper disposal less of a chore and more of a duty we all share.
Looking Forward
Newer research in green chemistry pushes for substitutes less likely to turn ordinary mistakes into long-term harm. Until that shift takes hold, the answer stays clear: treat every bit of chloromethyl chloroformate with the caution it earned. Local hazardous waste collection isn’t just a box to check, but a promise to coworkers, neighbors, and nature. Respect for the process protects everyone involved.
| Names | |
| Preferred IUPAC name | Carbonochloridic acid, chloromethyl ester |
| Other names |
Carmofur intermediate Chloromethyl carbonochloridate Chloroformic acid, chloromethyl ester Chloromethoxycarbonyl chloride Methyl chloroxymethyl ether |
| Pronunciation | /ˌklɔːroʊˈmɛθəl ˌklɔːrəˈfɔːrmeɪt/ |
| Identifiers | |
| CAS Number | 22128-62-7 |
| Beilstein Reference | **1362302** |
| ChEBI | CHEBI:132682 |
| ChEMBL | CHEMBL135885 |
| ChemSpider | 10812 |
| DrugBank | DB08334 |
| ECHA InfoCard | 03c84d734e-f3b2-4c13-aadf-b5fe7f664ed1 |
| EC Number | 205-702-4 |
| Gmelin Reference | 82217 |
| KEGG | C01583 |
| MeSH | D002694 |
| PubChem CID | 6367 |
| RTECS number | GF9100000 |
| UNII | 8T49P47P43 |
| UN number | UN2735 |
| CompTox Dashboard (EPA) | DTXSID6060328 |
| Properties | |
| Chemical formula | C2H2Cl2O2 |
| Molar mass | *110.49 g/mol* |
| Appearance | Colorless to pale yellow liquid |
| Odor | Sharp odor |
| Density | 1.48 g/mL at 25 °C |
| Solubility in water | Decomposes |
| log P | 1.80 |
| Vapor pressure | 24 mmHg (20°C) |
| Acidity (pKa) | 4.0 |
| Basicity (pKb) | 12.62 |
| Magnetic susceptibility (χ) | -5.07×10⁻⁹ cm³/mol |
| Refractive index (nD) | 1.4560 |
| Viscosity | 1.310 mPa·s (20 °C) |
| Dipole moment | 2.26 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -205.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -623.6 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V3D |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin. Causes severe burns and eye damage. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H317, H334, H335 |
| Precautionary statements | P210, P260, P261, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-1-W |
| Flash point | 72 °C (Closed cup) |
| Autoignition temperature | 570 °C |
| Explosive limits | Explosive limits: 5.6–14% |
| Lethal dose or concentration | LD50 oral rat 36 mg/kg |
| LD50 (median dose) | LD50 (median dose): 94 mg/kg (rat, oral) |
| NIOSH | SAF77900 |
| PEL (Permissible) | 0.1 ppm (0.5 mg/m³) |
| REL (Recommended) | 0.05 ppm (0.2 mg/m³) |
| IDLH (Immediate danger) | IDLH: 2 ppm |
| Related compounds | |
| Related compounds |
Methyl chloroformate Ethyl chloroformate Chloroform Phosgene |

