Chloroacetic Anhydride: A Commentary on Its Footprint, Importance, and Future
Historical Roots and Development
The story of chloroacetic anhydride stretches back to a period when chemistry relied more on brute force and grit than on neatly controlled lab conditions. Industrial chemists hunted for efficient ways to manipulate acetic acids with chlorine, trying to squeeze new molecules into the growing field of organochlorine chemistry. That long arc of experimentation, trial and error, bred both innovation and mishap. Chloroacetic anhydride started as a niche compound, but the qualities it brought to organic synthesis allowed it to slip quietly into the toolkit of laboratories and chemical manufacturers across continents. Its development shadows the evolution of agricultural chemistry, pharmaceutical research, and even the more shadowy corners of chemical manufacturing. These roots didn’t unfurl in a vacuum; global demand for specialty chemicals surged in the twentieth century, and researchers reached for whatever compounds could help unlock new products, better intermediates, or simply cheaper ways to access difficult transformations.
Product Overview and Chemical Identity
At first glance, chloroacetic anhydride does not draw much attention. Its sharp odor and reactive bite make it memorable only to those who work directly with it. As a close relative of acetic anhydride, but bearing that crucial chlorine atom, it serves as a cornerstone for a range of acylation and chlorination reactions. Chemists commonly refer to it by names like acetyl chloride anhydride or monochloroacetic anhydride. Its formula and structure sum up to a simple, almost modest molecule, but that unassuming profile belies a potent ability to activate other chemicals and introduce the vital chloroacetyl group into more complex structures.
Physical and Chemical Properties
Most labs that handle it recognize its appearance—clear, colorless to slightly yellow liquid, volatile, and quick to react with water. Its fumes catch not just the nose, but also the back of the throat, forcing even the most seasoned chemists to keep the bottle capped tightly and handled with care. Chloroacetic anhydride is highly unstable in moist air, breaking down to release not only chloroacetic acid but also corrosive byproducts. This compound’s tendency to hydrolyze makes it a candidate for single-use, on-demand production rather than long-term storage. Boiling points hover in a zone that challenges simple distillation setups, demanding controlled conditions and careful monitoring.
Technical Specifications and Labeling in Practice
Specific labeling for transportation and storage reads like a litany of hazard warnings, reflecting real incidents over the decades where improper handling led to costly cleanups or severe health effects. Every drum or bottle arrives stamped with clear markings for corrosivity, acute inhalation risk, and environmental hazards. Industrial users pay close attention to purity, watching out for hydrolyzed byproducts and unreacted feedstock. For those on the lab bench, handling protocols involve full gloves, face shield, and a respect for its aggressive reactivity. Mistakes here can mean more than ruined experiments; the cleanup can haunt a lab for days.
Preparation Methods and Chemical Pathways
Producing chloroacetic anhydride often starts with the raw materials: chloroacetic acid and established acylation agents. Classic routes employ dehydrating agents such as phosphorus pentachloride, thionyl chloride, or acetic anhydride itself. Industrial pathways trade off between cost, yield, safety, and waste management—every kilo produced generates streams that must be treated with rigor. In an age obsessed with green chemistry, these methods face growing pressure. Process engineers and synthetic chemists have begun to re-examine legacy methods, looking for catalytic alternatives or pathways that reduce reliance on the most hazardous reagents.
Chemical Reactions and Flexible Modifications
Chloroacetic anhydride delivers a potent one-two punch: it acts both as an acylating agent and as a source of the chloroacetyl group. This property enables transformations on alcohols, amines, phenols, and a host of other functional groups. Organic syntheses involving this compound fuel the production of herbicides, dyes, pharmaceuticals, and specialty polymers. Its use in the manufacture of carboxymethyl cellulose, a thickener found in everything from toothpaste to ice cream, shows how a harsh reagent on the bench can have gentle fingerprints in daily life. Nevertheless, its reactivity sometimes comes at a cost—side products or unwanted over-chlorination that challenge even seasoned chemists to fine-tune reaction conditions.
Synonyms and Alternate Names in Chemical Literature
Walk through the archives or scan through product lists, and you’ll see chloroacetic anhydride appear under several guises—monochloroacetic anhydride, chloroacetyl anhydride, or even CAA. Its synonyms reflect a practice in chemistry of naming by structure, function, or use. Clarity is key, considering the close relationship with other acylating agents whose properties may differ just enough to matter enormously to a given synthesis.
Safety, Operational Protocols, and Human Impact
Few compounds have shown so starkly how the line between industrial application and human risk can blur. Chloroacetic anhydride’s capacity to cause serious burns to skin and mucous membranes demands more than just gloves and goggles. Many a chemist has learned the hard way that vigilance trumps even the best-designed safety gear; a vapor leak can sicken a whole lab. Industry surveys and accident reports highlight the need for real-time monitoring systems and airtight containment, not just for legal compliance but for worker health and long-term facility integrity. Strict EU and US regulations control not just use but also discharge and disposal. Waste streams must pass through rigorous neutralization and filtration before entering the environment.
Application Areas Across Industries
While the public rarely encounters chloroacetic anhydride directly, its reach extends far. Agrochemical producers value its role in making herbicides and insecticides, which feed back into the food supply and shape the economics of farming around the world. Pharmaceutical synthesis draws on its utility for introducing specific chemical functionalities, helping forge the rings and chains that make up antibiotics or painkillers. Textile manufacturing employs its chemical power in dye intermediates, which mean that the clothes on a store rack might trace back to a chloroacetic anhydride batch produced continents away. The complex web of supply chains means that disruptions or regulation changes in any region can ripple outward—raising prices, stalling production, or demanding rapid scrambling for alternatives.
Investment in Research and Evolving Chemical Strategies
Research teams chase innovations that tame some of the brute force associated with chloroacetic anhydride. Modern academic and industrial labs look for catalyst systems that lower reaction temperatures, cut waste generation, or open doors to as-yet-unimagined transformations. I’ve been part of a lab staring down environmental regulations that forced us to justify every gram used and every waste stream created. Patent landscapes grow denser year by year, as new applications, derivative products, and clean-synthesis strategies emerge. Collaboration between universities and industries produces a feedback loop that quickens the pace of improvement, but these advances only reach commercial scale after rigorous safety and environmental impact trials.
Toxicity and Health Impact Research
The toxic profile of chloroacetic anhydride cannot be shrugged off. Animal studies and case reports alert us to acute and chronic dangers—eye and skin irritation quickly give way to more serious consequences with repeated or unprotected exposure. Accidental inhalation brings a punch to the lungs, while skin exposure leads to rapid, deep burns. Long-term effects on workers handling the compound daily have pressed industry leaders to rethink old ways and invest in containment improvements. Regulators look not just at immediate toxicity but also environmental persistence, given concerns about waterway and soil contamination downstream from manufacturing sites. Public health campaigns urge robust training and preparedness among users—from the smallest academic labs to sprawling chemical plants.
Prospects and Directions for the Next Generation
Looking ahead, the future of chloroacetic anhydride will ride on a mix of pressure and possibility. Increasing environmental oversight pushes the field toward greener, less hazardous production methods. Governments and consumers alike grow weary of compounds that place health and ecosystems in the crosshairs. The search for alternatives—whether milder reagents for acylation or downstream substitutions in herbicides and pharmaceuticals—intensifies with every regulatory tightening or high-profile safety incident. Some startups and university groups chase engineered enzymes or designer catalysts that could sideline traditional chemical routes. Still, underlying demand for the molecular features it enables will keep industry returning to the drawing board, experimenting with everything from process intensification to replacement chemistry. Experience teaches that complex chemicals like chloroacetic anhydride do not simply fade from importance; innovation or regulation may shift its position, but for now, its mix of utility and challenge keep it in the chemical conversation.
What is Chloroacetic Anhydride used for?
What It Does in the World of Chemistry
Chloroacetic anhydride doesn’t show up on supermarket shelves, but it’s no stranger to anyone who works with chemical manufacturing or advanced synthesis. Its main job is to help produce chemicals that eventually end up in things we use every day. For folks like me, who spent time in research labs, this compound means progress—only after a few careful safety checks because it reacts fast and isn’t something you want to spill.
Why Chloroacetic Anhydride Matters More Than You Think
I’ve watched colleagues put on extra gloves for just this substance. Because of its powerful reactivity, companies rely on it to make products that spread far beyond labs. Think herbicides, pharmaceuticals, and dyes. Without intermediates like these, crops would struggle more against weeds, and factories would have a harder time creating certain painkillers or cough medicines. With proper handling, chloroacetic anhydride speeds up chemical reactions that might take ages to complete or need more resources.
Shaping Pharmaceuticals and Protecting Crops
One key role for chloroacetic anhydride is production of chloroacetic acid, which leads to medicines and materials that shape today’s healthcare and agriculture. Researchers and manufacturers depend on reactions involving this compound to get high yields with fewer impurities. For example, some antibiotics wouldn’t even exist in their current form without a step built on this anhydride. On the agriculture side, the journey from raw chemical to finished herbicide can get technical, but it often involves a batch of intermediates born from chloroacetic anhydride. This supports global food supplies and lets farmers keep up with demand.
Balancing Progress and Safety
The progress comes with challenges. In the lab, my own mistake was nearly cutting corners on ventilation once—I learned from more experienced chemists that vigilance keeps people healthy. This compound needs solid planning. It’s harsh to skin and lungs. Factories that work with it invest in closed systems and protective barriers to keep staff safe. History has shown that accidents result from skipped steps—not from the chemical itself, but from people trying to move too quickly.
Regulation and Environmental Stewardship
No one wants to see chemicals like this get into soil or waterways. Regulators put firm controls in place: transport records, strict labeling, rigorous waste treatment. I remember seeing colleagues document every milliliter of outgoing waste for this exact reason. And industry groups update safety guidelines often, learning from small mishaps so bigger ones stay rare. This focus on stewardship benefits everyone, even those far away from chemical plants.
Moving Forward with Chloroacetic Anhydride
Science and industry will keep relying on intermediates like this compound while looking for greener ways to produce them. Synthetic chemists continue to experiment with alternative reagents and recycling of byproducts. Each improvement in process safety or cleaner manufacturing can ripple outward, affecting not just labs but communities and industries down the line. Nothing about working with strong chemicals comes easy, but with the right respect and procedures, their benefits reach well beyond their reactive origins.
What are the safety precautions for handling Chloroacetic Anhydride?
Why Chloroacetic Anhydride Deserves Respect
Chloroacetic anhydride isn’t the sort of chemical you sweep aside on a shelf and forget. It bites back with toxic vapors, severe skin burns, and potential organ damage. A splash or a whiff can start trouble fast. I remember my early lab days—how the odor would sting the nose, the way an accidental drop melted a hole clean through plastic. Anyone who spends time near this substance should know what they’re up against.
The Must-Have Gear
I always tell newcomers, the right gear isn’t about following rules—it’s about protecting your skin, your lungs, and sometimes your career. Full nitrile or butyl gloves are non-negotiable. Chloroacetic anhydride chews through cheap latex fast. Face shields or tight-fitting goggles keep burns off the eyes. I have seen too many folks wipe sweat from their face, only to regret it with a chemical rash.
Lab coats mean nothing if they’re unbuttoned or rolled up. Chemical-resistant aprons add a layer that can mean the difference between an annoying accident and a hospital visit. Shoes cover the whole foot. No sandals ever. If the ventilation hoods hum, everyone feels safer. A good fume hood makes all the difference because vapors from chloroacetic anhydride can hit hard and linger.
Smart Practices That Keep People Safe
I learned early: never get comfortable with a chemical’s dangers. Good training sticks with people. You show them how to store chemicals away from anything with water or strong bases. Chloroacetic anhydride reacts fast and fierce if it finds moisture. Sealed containers, properly labeled, go on ventilated shelves. If someone leaves a lid loose, others could end up breathing in corrosive fumes.
It helps to develop a habit of checking equipment before use. Glassware should show no cracks; hoses can’t leak. Spills clean up best with neutralizing agents—not water and not just paper towels. If the exposure happens, rapid action reduces injury. Safety showers and eye wash stations should always be clear of clutter.
What the Facts Tell Us
Government health agencies and chemical manufacturers highlight the same dangers year after year. Reports of skin burns, chemical pneumonitis, and even deaths from vapor exposure come from labs and production plants that cut corners. Statistics show that training workers just once isn’t enough—regular refreshers reduce injuries.
Personal experience matches the research. In facilities with active safety programs, incidents drop. Teams that run emergency drills react faster. Routine checks of ventilation and spill kits ensure nothing gets missed if something does go wrong.
The Path Toward Fewer Accidents
Safer workplaces start with a culture where no one feels embarrassed to double-check a lid or ask about glove thickness. Leaders can make that the norm. Companies should invest in regular maintenance for safety gear and proper training for new and veteran staff alike. Employees who know the real risks and see respect for safety in action speak up more and take fewer risks. Strong policies backed up by steady supervision keep dangerous shortcuts out of the workplace.
Chloroacetic anhydride is unforgiving. With the right habits, gear, and teamwork, people can handle it and go home in one piece at the end of the day.
What is the chemical formula and structure of Chloroacetic Anhydride?
Chemical Formula and Structure
Chloroacetic anhydride comes with the formula C4H4Cl2O3. Its structure deserves a closer look since this compound isn't something most folks see in regular life, but for anyone working in labs or industries that handle organic synthesis, it pops up in some surprising applications. The molecule consists of two chloroacetyl groups bonded through an anhydride linkage. Picture two carbonyl groups (C=O) connected by an oxygen atom, with each carbon also bonded to a chlorine atom and a methyl group. The arrangement mirrors that of acetic anhydride, but with a chlorine substituting one hydrogen on each methyl group. This small change brings out markedly different reactivity.
Why Its Details Matter
I remember my early days in the lab testing some anhydrides. People sometimes overlook the weight of a “simple” formula, but tiny differences, like swapping hydrogen for chlorine, often alter reactivity and hazards by more than a little. Chloroacetic anhydride isn’t just a puzzle piece for organic chemists. It’s reactive enough that a single drop can set an entire flask’s chemistry on a new path. The chlorine introduces extra electrophilicity, which matters in synthesis, because it can make forming new bonds easier or sometimes more dangerous.
Safe handling is never just a checklist. This compound can release corrosive fumes. Even quick skin contact can bring burns, so I’ve always stressed proper gloves and ventilation in training new lab members. The formula spells out more than a recipe—it bears a warning: handle with respect, and double-check your safety gear.
Use in Industry and Science
Manufacturers turn to chloroacetic anhydride most often to make specialty chemicals. Crop protection products, pharmaceuticals, and dyes have all leaned on it at some point. The reactive anhydride group is a powerhouse for introducing chloroacetyl groups into other molecules, which means downstream products might inherit some of its intensity. In my work with fine chemicals, finding a reliable supplier for sensitive intermediates like this makes or breaks a project’s schedule. Even a small impurity in the anhydride can taint an entire batch, costing weeks of work.
The formula also enables researchers to predict and troubleshoot side reactions by mapping out what functional groups sit where. Since both ends of the anhydride can react independently, side reactions crop up if you ignore the structure. This specificity holds real value for people aiming to make their reactions greener and their processes safer.
Challenges and Solutions
Waste disposal creates an ongoing challenge, especially with halogenated compounds like this. Chlorinated byproducts hold real environmental risks, and wastewater must be managed carefully. Many labs now recycle what they can and neutralize what they can’t. Training staff to catch spills before they spread stands as one front-line solution.
Maintaining tight storage protocols also pays off. Chloroacetic anhydride reacts with water in air. Keeping the material tightly sealed in a dry place avoids nasty surprises when you next open the bottle. I've seen entire flasks foam over from just a few stray drops of water during open transfers. Proper staff training, and storing reactive anhydrides in a regulated space away from humid environments, goes a long way. Knowledge of the formula breeds respect and, more importantly, safety.
How should Chloroacetic Anhydride be stored and transported?
Getting Serious About Chemical Safety
Chloroacetic anhydride acts aggressive. A whiff of this stuff can sting the eyes and trigger coughing fits. Anyone who has ever worked near storage tanks or drums of strong acids knows that chemicals with bad attitudes deserve careful handling. Chloroacetic anhydride belongs in this camp, and common sense matters just as much as regulations. No business wants a leak, no worker wants a burn, and nobody wants to end up in the emergency room.
Storing Chloroacetic Anhydride the Right Way
This chemical reacts with water, releasing corrosive fumes and heat. So don’t keep it where humidity can creep in. A dry room with proper ventilation stands up to the challenge best. You keep the temperature cool, ideally under 25°C. Heat makes vapors worse and corrosion more likely. Thieves and unauthorized people don’t get near it, so locked storage is a must. Rusty or corroded containers create trouble down the road, so only high-quality glass or fluorinated plastics hold up. Metal containers corrode, and anyone who’s cleaned up after a drum failure won’t forget the smell.
Shelf-life shrinks if light or air gets involved. Opaque containers keep out UV. Secure caps and seals limit contact with air, which lowers the risk of unwanted reactions. Spill kits stay close because accidents happen, and fast cleanup protects people and property. All containers need clear labeling—mistaking one chemical for another never ends well.
Transportation Calls for Extra Caution
On the road, vibrations and shaking test every drum, tote, or tanker. Secure fastening avoids spills. Drivers need to carry safety information in the cab, not in the trunk. Unlike boxes of printer paper, chemicals require paperwork at every checkpoint—shipping documents, emergency contacts, hazard class codes. Chloroacetic anhydride counts as a hazardous material, so drivers’ training must cover not just the basics, but also what to do when a leak or crash happens.
Regulations dictate the drum and container types. Using thin-walled or reused containers tempts fate. Businesses invest in containers certified for corrosive chemicals. I’ve visited sites where old, rusty drums waited for pickup—every dent and spot raised anxiety about leaks. Investing in quality packaging and checking it before every shipment saves money and headaches.
Protecting People and the Environment
Workers deserve more than gloves and googles. Full-face shields and chemical suits should stand ready for loading, unloading, and any time there’s a chance of splashing or vapor release. Facilities rely on eye-wash stations and emergency showers positioned nearby. Wastewater and rinse solutions stay out of normal sewer lines. Discharge permits, waste manifests, and proper disposal fees beat a visit from the fire marshal or a hazardous spill cleanup crew any day.
Better Training, Fewer Surprises
Training sessions that mix hands-on drills with real-life case studies keep everyone sharp. Talking about accidents from other plants puts the risks in perspective. Everyone remembers the stories—not just the procedures. Making safe storage and transport of chloroacetic anhydride a routine habit beats dealing with lawsuits or injuries. The best teams make it look easy, but behind the scenes, every step matters.
What are the potential health hazards of Chloroacetic Anhydride exposure?
Chloroacetic Anhydride on the Job and in the Air
Chloroacetic anhydride isn’t just another odd-sounding chemical; its hazards feel close to home for anyone who’s spent time around labs or manufacturing plants. This compound, used in synthesizing dyes, herbicides, and pharmaceuticals, packs a punch both on contact and through the air. Most workers I’ve met don’t expect a clear, oily liquid to be so aggressive, yet stories of sneezing fits, burning skin, or worse show how real the risks get.
How This Chemical Hurts the Body
The sting starts with skin. Chloroacetic anhydride eats through outer layers fast—a splash can burn through gloves and leave nasty wounds. Some guys I worked with thought their usual gear was enough; one day’s distraction proved how quickly that stuff chews through latex or weak plastics. Inhaling its fumes brings even more trouble: sharp throat pain, choking, and damage all the way to deep lungs. Eyes react badly—burning, tearing, even the risk of losing vision if it’s not washed out in seconds. No one shakes off those symptoms with deep breaths or quick rinses.
Systemic Reactions and Long-Term Problems
Absorption doesn’t end on the surface. If chloroacetic anhydride gets under the skin or into the lungs, it starts a chain of health problems. I’ve heard doctors warn about kidney and liver distress after workers spent too long in contaminated air, plus central nervous system symptoms like confusion or tremors. Chloroacetic acid, one breakdown product, hits the body hard and fast. Fatalities haven’t just happened in textbooks—industrial accidents, often due to bad ventilation or bad luck, have turned this chemical’s threat into grim reality.
Why It Matters in Everyday Work
Ignoring personal experience here would downplay real stakes. A small mistake—spilling, leaking drum, fumbled transfer—can mean taking emergency showers, or worse. Many workers don’t see immediate effects and assume the warning signs get overblown. Years of policy reviews and safety trainings show that one lapse can end a career, or a life. This chemical rarely gives second chances.
Keeping People Safe—What Actually Works
Making safety personal gives rules real weight. The best plants set up heavy-duty exhaust systems, keep exposure times down, and supply full-face respirators—not just flimsy masks—along with thick, chemical-resistant suits. Training matters, but I trust crews who treat every drum and hose with respect, double-checking seals, and working in pairs. Spill kits close at hand save minutes that matter. Around chloroacetic anhydride, assuming it’ll leak someday isn’t paranoia—it’s sensible survival.
Making Health a Priority, Not an Afterthought
Regulatory agencies like OSHA have good reasons for tight exposure limits. Medical monitoring adds a backup layer, picking up trouble before workers lose their edge. But real change starts when front-line staff and managers treat every shift as a high-risk operation, not routine daily grind. Sharing stories of close calls and lessons learned keeps risks fresh in everyone’s mind, giving a face to what happens if anyone lets their guard down.
A Call for Respect, Not Fear
Chloroacetic anhydride isn’t a villain if people stay informed and cautious. Solutions don’t come from ignoring hazards or blaming small mistakes—they come from treating each person’s safety as the goal, above speed or convenience. In the end, knowledge, swift action, and teamwork give everyone a fighting chance against a chemical that won’t cut you any slack.
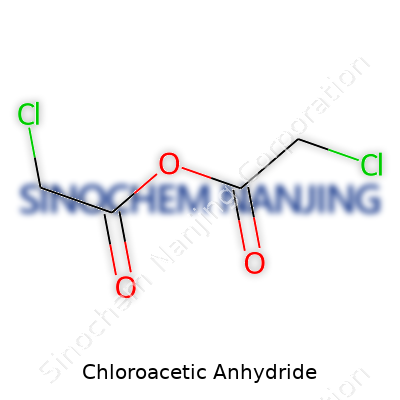
| Names | |
| Preferred IUPAC name | 2-chloroacetic anhydride |
| Other names |
Acetyl chloroacetate Chloroacetic acid anhydride Chloroacetyl acetate |
| Pronunciation | /ˌklɔːroʊəˈsiːtɪk ænˈhaɪdraɪd/ |
| Identifiers | |
| CAS Number | 541-88-8 |
| Beilstein Reference | 1209082 |
| ChEBI | CHEBI:132997 |
| ChEMBL | CHEMBL461172 |
| ChemSpider | 22589 |
| DrugBank | DB11338 |
| ECHA InfoCard | 100.002.937 |
| EC Number | 211-876-7 |
| Gmelin Reference | 8229 |
| KEGG | C06148 |
| MeSH | D002694 |
| PubChem CID | 17613 |
| RTECS number | AF8575000 |
| UNII | KJ7Z8IKW1B |
| UN number | UN1751 |
| CompTox Dashboard (EPA) | DTXSID7050952 |
| Properties | |
| Chemical formula | C4H2Cl2O3 |
| Molar mass | 197.39 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 1.58 g/cm³ |
| Solubility in water | Reacts violently |
| log P | 1.77 |
| Vapor pressure | 0.135 mmHg (25 °C) |
| Acidity (pKa) | 1.0 |
| Basicity (pKb) | -2.30 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.438 |
| Viscosity | 1.47 mPa·s (20 °C) |
| Dipole moment | 2.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -359.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -715.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ11 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H314 + H331 + H373 |
| Precautionary statements | P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 40 °C (104 °F) |
| Autoignition temperature | 185 °C |
| Explosive limits | Upper: 9.2% ; Lower: 2.6% |
| Lethal dose or concentration | LD50 oral rat 730 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 121 mg/kg |
| NIOSH | FJ6475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Chloroacetic Anhydride: 0.05 ppm (0.2 mg/m³) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | 1 ppm |
| Related compounds | |
| Related compounds |
Acetic anhydride Acetyl chloride Chloroacetic acid |

