Cesium Nitrate: A Comprehensive Look
Historical Development
The early history of cesium nitrate traces back to the mid-19th century, a period marked by landmark discoveries in alkali metal chemistry. Robert Bunsen and Gustav Kirchhoff introduced cesium to the world in 1860, isolating it through flame spectroscopy, a method that sparked a series of innovations in chemical separation and purification. The tale of cesium diverged from simple curiosity to practical value soon after, as chemists explored various nitrate salts for use in early optics, pyrotechnics, and analytical methods. Cesium nitrate eventually established its presence as scientists recognized its role in specialty glassmaking, infrared spectroscopy, and as a component in electrochemical devices, standing as a quiet contributor to several breakthroughs in material science and communication technologies.
Product Overview
Cesium nitrate presents itself as a colorless, crystalline powder with a strong oxidizing nature. Chemists and industry specialists find it appealing for its solubility in water and its role as a source of cesium ions, supporting a wide range of laboratory procedures. Strong ionic character places it among the more active alkali metal nitrates, especially in applications requiring rapid and clean thermal decomposition. Its non-hygroscopic nature, unusual among alkali nitrates, minimizes the risk of caking and makes it stable under normal storage conditions.
Physical & Chemical Properties
Examining the essential features, cesium nitrate carries the molecular formula CsNO3 with a molar mass close to 194.91 g/mol. It melts at about 414°C and begins to decompose at temperatures exceeding 500°C, typically releasing nitrogen dioxide and oxygen. Its refractive index, about 1.51 for a saturated solution, gives it a place in precision glassmaking. Dissolving in water at 184g per 100mL at room temperature, it aids various wet chemical processes. On the flip side, it resists dissolving in alcohol, reflecting classic nitrate salt behavior. While stable in its pure form, the compound reacts strongly in the presence of reducing agents or organic material, sometimes causing unwanted fires or explosions.
Technical Specifications & Labeling
Cesium nitrate often ships with purity grades marked at 99% or higher, catering to needs across electronics, optical glass, and pyrotechnics. Batches destined for laboratory or pharmaceutical use face strict scrutiny under guidelines for heavy metal content, moisture levels, and insoluble residue. Labeling flags it as an oxidizer, with relevant hazard symbols and transport codes attached in bold lettering. These designations reflect both the compound’s potential risks and its privileged standing in specialty manufacturing—a duality that keeps both users and regulators alert. Inspections focus on keeping sodium and rubidium cross-contamination below 0.1% for premium-grade stocks.
Preparation Method
Synthesizing cesium nitrate often begins with cesium carbonate or cesium hydroxide as a starting material, neutralized using nitric acid in a controlled reaction. The resulting solution undergoes evaporation or crystallization, yielding a solid that gets washed and purified to strip away residual acid or soluble side products. Large-scale operations may adopt ion-exchange columns, using sophisticated resins to enhance purity and cost efficiency. Science teachers and bench chemists have repeated variations of these protocols for decades, tweaking steps to address scale, available reagents, and end-use purity demands.
Chemical Reactions & Modifications
Redox chemistry sits at the center of cesium nitrate’s appeal. When heated with charcoal, sulfur, or aluminum, the compound quickly surrenders its nitrate ions, producing a spectrum of gases and elemental cesium intermediates. This renovation process allows for the creation of cesium-based explosives, infrared flares, and advanced glass compositions. In laboratories, it helps to drive metathesis reactions, forming salts or separating ions in analytical contexts. Chemists also pursue new modifications by blending it with rare earth elements, pushing the development of photoelectric devices and advanced ceramics forward.
Synonyms & Product Names
You might find cesium nitrate under names ranging from cesium(I) nitrate to caesium nitrate, with regional spelling quirks reflecting the British or American preference. In some databases, it shows CAS number 7789-18-6, giving it a unique tag among chemical seekers. Some vendors label it simply as CsNO3, making it vital to double-check labels to avoid mix-ups with potassium or sodium counterparts, especially in labs that stock all three.
Safety & Operational Standards
Working with cesium nitrate calls for vigilance. As an oxidizer, it turns minor accidents into real emergencies with flammable material or exposed skin. Protective gear, including gloves, goggles, and lab coats, must be worn without fail. Work areas need ventilation hoods, spark-free switches, and strict discipline around incompatible chemicals. Spillage protocols demand immediate containment with non-combustible absorbents and disposal according to local environmental rules. Training covers not just use but storage, emphasizing cool, dry environments and sealed containers away from acids, organic matter, and reducing agents. These standards cut across industry lines, from college labs to defense contractors.
Application Area
The main users of cesium nitrate are glassmakers, who turn to its high solubility and thermal stability to craft specialized filter glass and lens coatings for scientific and medical instruments. Pyrotechnicians value its fierce oxidation power—the compound plays key roles in signal flares, colored fireworks, and infrared decoys used by defense agencies. Electrochemists and battery developers blend it into electrolyte mixes, pursuing improved charge mobility and higher voltage windows. Even X-ray crystallographers lean on its precise lattice patterns in structure analysis, demonstrating that this rare chemical travels far beyond the classroom desk. Beyond industry, researchers exploit its characteristics in studying spectroscopic shifts and high-energy chemical phenomena.
Research & Development
Recent research targets the evolving needs of semiconductors and photonics. Teams are probing cesium nitrate for its ability to enhance ion-exchange processes in advanced glass fibers, supporting faster data transmission and greater resistance to signal loss. Battery researchers work on integrating cesium compounds into next-generation solid electrolytes, chasing better performance in harsh environments. Optical scientists chase the minor tweaks in cesium nitrate’s structure that could improve light filtering in ultraviolet and near-infrared zones. New analytical techniques, including neutron activation and high-res spectroscopy, dig into potential trace effects of cesium nitrate in environmental and medical settings, broadening both utility and understanding.
Toxicity Research
Health studies show that cesium nitrate brings risks if handled carelessly or ingested in significant doses. Acute exposure can irritate mucous membranes, damage the respiratory tract, or trigger skin and eye inflammation. Long-term animal studies point to possible central nervous system impacts, although comparable effects in humans have limited documentation and mostly arise from chronic exposure in poorly regulated facilities. Modern research aims to clarify thresholds for safe air and water exposure, and environmental scientists analyze its mobility in soil and potential to bioaccumulate. Environmental fate studies reinforce strict disposal regulations, citing the compound’s persistence under certain conditions and need for monitored landfilling or neutralization.
Future Prospects
Looking ahead, the world of specialty chemicals sees new calls for ultra-pure and application-tailored cesium nitrate. Clean energy projects and rapidly growing photonics markets bring both challenges and high hopes. Digital infrastructure expansion could amplify demand in glass and optical coatings, and innovative storage materials for energy production might tap deeper reserves of cesium compounds. Ongoing toxicity studies and life-cycle analyses will shape tighter occupational guidelines, balancing innovation with public and environmental health. As more labs diversify into rare alkali salts, cross-border regulations and shared best practices gain importance, urging collaboration and informed stewardship. In all, cesium nitrate stands as a rare but critical catalyst in tomorrow’s most demanding technical fields.
What is Cesium Nitrate used for?
Explosive Color and Specialty Uses
People rarely stop to wonder what makes brilliant reds appear in fireworks. Cesium nitrate, a chemical salt, gets credit for a good part of that vibrancy. Firework makers choose it because it burns with a signature violet or red hue. Beyond color, it works as an oxidizer, which means it helps things burn hot and fast—exactly what a firework needs to launch sky-high and explode on time. Every time the Fourth of July rolls around, or New Year’s lights up the night, there’s a good chance cesium nitrate is lifting the show.
Some pyrotechnicians who spend whole seasons perfecting recipes depend on the purity of chemicals like cesium nitrate. Any contamination could change the color or weaken a display. Clean, consistent results mean better shows and less guesswork. This detail matters—if a firework fails, disappointment hits the crowd and safety can be at stake.
Space and Cutting-Edge Technology
There’s another side to this salt, one tucked away from crowded celebrations. Cesium nitrate plays a quiet but crucial role in scientific instruments and devices that need precision. Scientists who build lasers sometimes use it in specialty glass or in the process of aligning optical instruments. Cesium’s unique properties, including the way it interacts with light, catch the attention of engineers working on atomic clocks and quantum devices. These clocks define today’s understanding of time, and any error in timing can throw off everything from GPS navigation to financial transactions. Trust comes from decades of lab testing—if cesium nitrate didn’t deliver, teams would look elsewhere.
In satellite technology, developers work with cesium-based thrusters for spacecraft. Small, consistent thrust helps keep satellites in the right spot above the Earth. Precision is non-negotiable in space, where repair missions cost millions and basic errors can end a billion-dollar investment. Here again, material quality influences both performance and reliability.
Challenges and Responsible Handling
With benefits come responsibilities. Cesium nitrate’s oxidizing power also brings hazard if handled poorly. Any chemical that speeds up burning can pose risks in storage or transport. I remember working near labs where clear rules on handling oxidizers kept everyone safe. Regulations exist for a reason; people who’ve ignored them in the past have seen warehouses catch fire, and families lose livelihoods. Proper training and respect for procedures mean safer communities and fewer health scares.
Environmental care matters, too. Spills can seep into soil and waterways. Some salts, cesium nitrate among them, dissolve in water and travel far from where they started. Research has tied improper disposal to plant damage and changes in water chemistry. Farms or gardens near disposal sites sometimes show stunted growth or yellowed leaves. Responsible companies invest in containment and cleanup, not just because the law demands it—community trust and long-term business depend on good stewardship.
Looking Ahead: Improving Safety and Access
Innovation should always pair with safety. Chemistry teachers introducing future scientists to cesium compounds must stress both the fascinating possibilities and the need for caution. Manufacturers could support safer storage and packaging. Simple reminders—keep it cool, dry, tightly sealed, away from other reactive chemicals—make a world of difference.
Every material has a story beyond its formula. Cesium nitrate’s tale cuts across street festivals, laboratories, and the furthest reaches of space. Each step in its journey deserves respect, a careful hand, and a thoughtful eye toward the future.
Is Cesium Nitrate hazardous or toxic?
What This Compound Is All About
Cesium nitrate crops up in specialized industries like pyrotechnics, optics, and oil drilling. The chemical mixes easily with water and holds a strong oxidizing nature. Its uses might look interesting, but anyone working with it can’t ignore safety questions. When a chemical gets a spot in fireworks or decoys for military use, it deserves a closer look.
Why Risk Matters in Everyday Handling
People often ask if cesium nitrate deserves the label of hazardous or toxic. My experience in industrial labs and environmental reporting tells me most folks usually focus on direct exposure risks. This compound doesn’t spread radioactive damage the way its cousin cesium-137 does—there’s no radioactive worry in basic cesium nitrate. Still, the substance can bring its own set of troubles if someone ignores the ground rules of safety.
How Cesium Nitrate Acts on Health
Short-term exposure generally leads to irritation. Breathing in dust can leave throats sore or noses stuffed up. Direct contact might sting eyes or skin. Swallowing even a small amount causes burning in the mouth and stomach upset. Symptoms may feel mild at first, but repeated or heavy exposure raises the stakes.
Labs and factories stress that cesium nitrate risks multiply once it meets organic stuff or flammable materials. It can trigger fires or explosions during poor storage or accidental mixing. If a spill happens, cleanup quickly turns into a hazardous task for any untrained worker. I’ve watched cases where overlooked dust clouds inside a storeroom led to costly building damage after one bad spark.
Worker Safety and the Long Game
Safety agencies like OSHA and NIOSH mark cesium nitrate as hazardous, especially because it acts as an oxidizer and brings acute toxicity with high enough doses. Chronic exposure isn’t a daily problem for most people, but lab techs, miners, and fireworks manufacturers see higher odds for risk. Skin rashes and chronic respiratory irritation sometimes show up among staff who cut corners on gloves or respirators.
I remember early days in academic labs, watching newcomers mix cesium nitrate without goggles or ventilation. It often started with just a cough or a burning nose—later followed by red eyes and mild burns before a supervisor laid down stricter safety rules. Poster warnings only sink in after a direct scare.
Solutions—What Gets Risk Under Control
Good ventilation keeps airborne dust low. Workers need gloves and goggles so the compound stays off skin and out of eyes. Spills should be swept up by trained crews—not wiped a rag—because water and cleaning agents can spread the powder around or trigger unwanted reactions. I’ve learned from chemical safety trainers that isolated storage, away from paper, wood, and fuel, matters just as much as personal protective equipment.
Clear labeling, training refreshers, and strict inventory checks prevent compounds like cesium nitrate from ending up in the wrong spot. Facilities also invest in emergency showers and eye wash stations. Disposal can’t be casual; it goes to licensed chemical waste handlers, not down a drain or in regular trash.
In the real world, no shortcut beats routine care. One overlooked scoop in a dusty corner spells more trouble down the line than nearly any other kind of mistake I’ve seen in the field.
What is the chemical formula of Cesium Nitrate?
What Makes Up Cesium Nitrate?
Science class introduced me to the idea that each material around us comes down to its most basic particles: elements arranged in specific ways. Cesium nitrate features a simple and clean formula: CsNO3. It combines one cesium atom, one nitrogen atom, and three oxygen atoms. On the surface, this looks like just a bundle of letters and numbers, but understanding this formula opens the door to real-world impacts—sometimes in fields I never expected to connect with chemistry.
Why the Formula Matters Beyond the Lab
A handful of us have heard about cesium before—its name pops up in fireworks and high-precision clocks. Nitric acid forms the backbone of the nitrate part, lending it explosive and oxidizing characteristics. Combining these gives us cesium nitrate. Recognizing this composition serves more than curiosity; it shapes real decisions in safety, responsible use, and even innovation.
I’ve seen researchers refer to CsNO3 in applications involving pyrotechnics. In fireworks, for instance, cesium salts create a striking blue-violet color that's tough to achieve with other elements. The nitrate’s oxidizing power boosts the burn. Without knowing exactly what’s inside, chemists risk accidents by mixing the wrong blends. One element out of place in a formula can turn a controlled effect into a dangerous mess.
Handling Safety: A Real Concern
Experience in industrial work makes it clear—knowing a material’s formula isn’t just textbook trivia. Storage, transportation, and handling rules hinge on understanding compounds at the atomic level. Cesium nitrate can support rapid combustion due to its nitrate group. In practice, this means that any spill over an organic surface or exposure to flame can start an intense reaction. People working in labs or factories, myself included, trust those formulas for safe handling. Safety data sheets rely on this chemical shorthand, making it possible for workers to identify hazards quickly.
Environmental Decisions Anchored in Formulas
There’s another layer: environmental risk. Nitrates in water supplies trigger alarms because they fuel algae blooms and choke off aquatic life. While cesium nitrate does not have the wide use or environmental footprint of sodium or potassium nitrate, its fate and transport lean on those same nitrogen and oxygen atoms. Regulatory agencies test for specific chemicals—knowing the formula means correct detection and mitigation. Environmental engineers depend on those three oxygens and single nitrogen to model movement and impact. Getting the formula right isn’t a trivial academic pursuit; it impacts decisions that ripple into public health.
Possible Solutions and Personal Responsibility
Everyone involved—engineers, scientists, teachers—faces a challenge. Mistakes in identifying formulas can cause mishaps, spark poor research, or lead to regulations that miss the mark. Education holds the strongest role here. Students need practice in recognizing and using formulas in context, not just memorizing their shapes. Industry can support this by giving clear chemical labeling and access to resources that go deeper than a label on a bottle.
From a hands-on perspective, I’ve found that double-checking formulas before starting work prevents more headaches than any fancy equipment. Mistakenly swapping cesium nitrate for a similar-looking compound changes outcomes, budgets, and reputations. The honest answer is that attention to detail—knowing that CsNO3 contains cesium’s one positive charge balanced by the nitrate's negative charge—strengthens science and safety across the board.
How should Cesium Nitrate be stored safely?
Cesium nitrate grabs attention among chemical workers for its unique use in pyrotechnics, optics, even rocket propellants. I once dealt firsthand with its shipment at a mid-sized lab, and no lab buddy ever forgot: this salt brings real risks. Flammability and reactivity mean one slip-up brings a headline nobody wants. So, safe storage turns critical the moment a shipment arrives.
Understanding the Hazards
Most folks handle cesium nitrate in powder or small crystalline form, but just because a substance looks stable, don’t judge a book by its cover. It reacts aggressively with many organics and reducing agents. Dust in the air poses inhalation risks. Even small spills, if left, can trap moisture and start chemical reactions, sometimes producing toxic gases. That’s not a scare tactic—it’s reality.
Location and Environment
Space matters. Store it away from high-traffic areas and never near those “catch-all” shelves where odds and ends pile up. A locked, designated cabinet separates hazardous material from common supplies. As a younger technician, I saw more than one cabinet open to the hallway—somebody eventually got burned (literally and on their job review). Controlled access means only trained staff handle each jar, and that reduces accidents.
Temperature control also makes a difference. Keep storage cool, dry, and shielded from direct light. Humid rooms or spaces with fluctuating temperatures set the stage for clumping, decomposition, or unplanned reactions. Silica gel packets serve well to reduce moisture inside cabinets, and routine humidity checks catch trends before they cause trouble. I remember a year we lost a whole order after a storm pushed humidity above 70%—no one liked cleanup day after that.
Labeling and Segregation
Clear, honest labeling isn’t just for bureaucratic peace of mind; it keeps staff safe. “CSNO3—Oxidizer—Keep Clear of Organics.” Make it bold and readable. Colored hazard labels signal danger to anyone who walks by, even on a bad day. Mix-ups between containers sometimes happen midway through an inventory check, and only clear labeling prevented emergencies more than once.
Keep cesium nitrate away from flammables, acids, and reducing agents. Storing strong oxidizers next to solvents or acids creates opportunities for runaway reactions. In the lab, we learned not to trust memory—separate shelves, clearly marked boundaries, and inventory logs put everyone on the same page. That kind of segregation works outside the lab, too, in warehouses or university storerooms.
Packaging for Prevention
Sturdy, airtight containers become essential. Glass or compatible plastic works best; thin polyethylene bags tear easily, defeating the whole purpose. Lids should form a tight seal, resistant to moisture and air. Before resealing, inspect containers for chips and leaks. One overlooked chip means more risk than skipping gloves. If secondary containment buckets are available, they add another line of defense, catching spills before they threaten the rest of the cabinet.
Personal Protective Standards
Never underestimate the basics: gloves, goggles, and lab coats. Even for brief container checks, splashes or dust surprise the careless. As much attention should go to the workspace as to the chemical: keep benches uncluttered, spill kits stocked, and respirators close at hand if procedures demand it.
Training and Procedures
Routine practice stops most accidents. Every staff member should run through dry drills: spill response, emergency contact steps, and monitoring for exposure. Annual review sounds tedious until you avoid one more “close call.” Over the years, every team I’ve worked with benefitted from open discussion, clear communication, and checklists visible right on the cabinet door.
Chemicals like cesium nitrate ask us to do the simple things well—consistent attention pays off every day.
Where can I buy Cesium Nitrate?
What to Know Before Shopping for Cesium Nitrate
Cesium nitrate draws interest from folks in science, engineering, and sometimes even pyrotechnics. In my college chemistry lab, handling anything with the word “cesium” attached always kicked up curiosity among students—mostly because of its reactive qualities and its role in specialty applications. Sourcing this compound, though, gets tangled up with federal laws, safety, and supply chain limits.
Sourcing Isn’t Simple
You can’t stroll into a regular hardware store and ask for cesium nitrate. The compound isn’t blacklisted like some controlled substances, but it gets enough regulatory attention that companies steer clear unless they work directly with research labs or specialized manufacturers. Strict rules exist because cesium salts play a part in sensitive electronics applications and sometimes in pyrotechnic products—raising the bar for responsible distribution.
Companies That Sell Cesium Nitrate
Most suppliers that carry cesium nitrate tend to focus on science and research markets. Businesses like Sigma-Aldrich (now part of MilliporeSigma in the US), Alfa Aesar, or Thermo Fisher Scientific come up most often. These companies require a lot of paperwork before they’ll sell to you. Customers get vetted for credentials and end-use declaration. If you’re a college student or hobbyist without institutional backing, a sale rarely goes through. Even legitimate research teams sometimes jump through hoops to keep records clear and protect the company from liability.
Regulatory Roadblocks
Homeland security regulations, as well as those from agencies like OSHA and the DEA, heavily influence how chemical suppliers market and move products. Cesium nitrate sits on lists that trigger extra scrutiny—not outright bans, but extensive tracking and documentation requirements. The US, for example, tracks certain chemicals due to their potential in weapons development or industrial sabotage. Even internationally, European suppliers will ask for identity verification and end-use justification.
Risks and Public Safety Factors
Safety is a double-edged sword with chemicals. Cesium nitrate doesn’t explode on contact with air, but in high concentrations, many nitrates act as oxidizers. Throw a reactive alkali metal ion like cesium into the mix and the risk increases in less-experienced hands. Past accidents push regulators and producers to watch who is buying and how they're using it. From my time working with high school science teachers, I saw plenty of requests for restricted chemicals shut down—safety in an educational environment always came first.
Pursuing Legal and Safe Access
For academics, the best route always runs through official procurement offices, not back-alley internet shops. That means finding a professor or research manager who knows how to file paperwork and justify purchases. Hobbyists without formal ties to a lab or company almost always hit a dead end for good reason. Trying to source cesium nitrate from dubious online sellers can open you up to scams or, worse, criminal charges.
Building Trust and Transparency
Companies with long track records and open documentation set the standard for safety and compliance. They educate their customers, make clear what is required, and help legit users get access. This fosters trust and keeps questionable actors at bay. Sourcing sensitive chemicals—cesium nitrate included—remains an area where transparency wins over secrecy every single time.
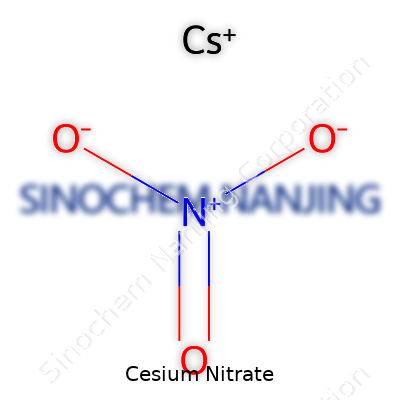
| Names | |
| Preferred IUPAC name | Cesium nitrate |
| Other names |
Nitric acid, cesium salt Caesium nitrate Cesium saltpeter Nitrate de cesium |
| Pronunciation | /ˈsiːziəm ˈnaɪtreɪt/ |
| Identifiers | |
| CAS Number | 7789-18-6 |
| Beilstein Reference | 358998 |
| ChEBI | CHEBI:32515 |
| ChEMBL | CHEMBL3306692 |
| ChemSpider | 6736 |
| DrugBank | DB14655 |
| ECHA InfoCard | ECHA InfoCard ID: **100.028.776** |
| EC Number | 232-146-4 |
| Gmelin Reference | 67636 |
| KEGG | C01738 |
| MeSH | D002584 |
| PubChem CID | 24856 |
| RTECS number | WL4370000 |
| UNII | SY7Q814VCS |
| UN number | UN1451 |
| Properties | |
| Chemical formula | CsNO3 |
| Molar mass | 194.91 g/mol |
| Appearance | White Crystals |
| Odor | Odorless |
| Density | 3.68 g/cm³ |
| Solubility in water | Very soluble |
| log P | “-3.3” |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.71 |
| Magnetic susceptibility (χ) | +35.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.522 |
| Viscosity | 1.14 cP (20°C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 132.67 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -370.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −295.3 kJ/mol |
| Pharmacology | |
| ATC code | V09IX02 |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, may cause irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H272, H319 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P337+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3 0 1 |
| Autoignition temperature | 410 °C |
| Lethal dose or concentration | LD50 oral rat 2,060 mg/kg |
| LD50 (median dose) | Oral rat LD50: 2,380 mg/kg |
| NIOSH | WT4825000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | “REL: 2 mg/m³” |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Potassium nitrate Sodium nitrate Rubidium nitrate Lithium nitrate Ammonium nitrate |

