Cerium Selenite: A Comprehensive Look
Historical Development
Cerium selenite stands as a unique compound within the broader family of rare-earth metal selenites. Cerium itself first came into scientific awareness at the dawn of the nineteenth century, thanks to the hunt for new elements that pushed both industrial and academic chemistry forward. Selenium entered the scene a bit later, celebrated for its peculiar properties and applications in glassmaking and electronics. During the twentieth century, chemists explored a huge range of cerium compounds, primarily fascinated by their reactivity and electronic structure. The first documented synthesis of cerium selenite came out of research focused on optical materials and catalysis. Over the decades, this compound has popped up across many technical fields, boosted by its unusual blend of stability and reactivity.
Product Overview
Cerium selenite gets attention as a pale, powdery solid that easily blends into research and industrial settings. As a product, it arrives in air-tight bottles or sealed drums, labeled for laboratory use and deeper applications in manufacture. Chemists and engineers value it for its intermediate place between pure metal salts and more aggressively reactive selenides, offering opportunities to harness the characteristics of both cerium and selenium in one body. Its production runs span from small research quantities through to kilogram-scale batches for pilot projects in electronics and materials science.
Physical & Chemical Properties
On the physical side, cerium selenite presents as a fine, light grey or off-white powder, often showing a subtle pinkish hue due to its selenium content. Its crystal lattice holds firm at room temperature, resisting breakdown in dry air. It dissolves slowly in strong acids, releasing both cerium and selenium ions in solution. It stays remarkably stable compared with cerium nitrate or selenite, rarely giving off vapors or fumes unless exposed to intense heat. Its melting point sits above 450°C, and, unlike some of its selenite cousins, it resists both oxidation and reduction outside extreme environments. This resilience makes it reliable for rigorous experimental setups and potential commercial applications.
Technical Specifications & Labeling
Product specifications put a strong focus on purity, with tech-grade cerium selenite topping 99% assay by combined metal content. Labs look for sharply defined particle size distributions, often between 1 and 200 microns. Shipping labels mention moisture sensitivity and shelf life, prompting prompt use after opening. Containers bear hazard pictograms flagging oxidizer risk and potential toxicity. Modern labels also show batch number, country of origin, and any known contaminants—trace rare earths, sulfur, or alkali metals typically register below 200 parts per million. Detailed documentation backs up each lot, supporting rigorous traceability and compliance for regulated sectors.
Preparation Method
Making cerium selenite usually begins with cerium oxide and purified selenium dioxide. Cerium oxide reacts with warm, dilute selenous acid—stirred under nitrogen—to yield a suspension that settles into a paste of cerium selenite. Chemists control temperature and pH to curb unwanted side reactions, especially formation of insoluble cerium salts. The wet product gets filtered, washed with cold water, and dried in vacuum ovens to lock in phase purity. Batch scale labs spend as much time on cleaning glassware and verifying sources as they do on the reaction itself, since faint copper or iron traces can shift the product’s color and crystalline form. Once made, product batches undergo X-ray diffraction analysis and wet-chemical titration to confirm that only the desired selenite phase turns up in each run.
Chemical Reactions & Modifications
Cerium selenite responds actively in coordination chemistry experiments—its oxygen atoms readily form bridges to neighboring metal atoms. In catalytic settings, it works as both an oxidizer and a buffer, holding transition states during organic reactions. Sticking the selenite to polymer backbones or metal surfaces generates hybrid materials with tuned properties, a trick seen in both environmental sensors and light-manipulation technologies. In strong reducing atmospheres, cerium selenite drops selenium, changing color and sometimes forming metallic nodules. Adding fluoride or chloride ions can trigger rapid ligand exchange, letting labs tune its solubility profile for advanced separations and recycling schemes.
Synonyms & Product Names
Chemists know cerium selenite by several aliases, including cerous selenite and cerium(III) selenite. Trade catalogs list it under names such as cerium(3+) selenite or simply Ce2(SeO3)3, referencing its formula. Some suppliers offer it in hydrated forms or blend it with minor modifiers, but the substance generally moves in its pure, anhydrous guise. Academic papers sometimes slip into older names from the early rare-earth literature—look out for spellings like "ceriit selenit" or Latin root notations in certain European journals.
Safety & Operational Standards
Working with cerium selenite takes its lead from established rare-earth and selenium compound protocols. Dust inhalation presents a major health concern, so operators wear tight-fitting masks and work in ventilated hoods. Spillage cleanups revolve around damp cloths, never sweeping, which could send airborne particles across a lab space. Contact with skin gets treated with lots of water and soap, and first-aid stations stock up on eye wash bottles, given the risk from even a light dusting of solid. Regulations flag cerium selenite as an oxidizer, with a modest risk of fire if mixed with organics or strong reducing metals. Storage avoids wood and paper products, using metal drums with gasket-sealed lids, lined up away from acids and food areas. Disposal instructions fall under local hazardous waste laws, specifying locked bins and special routes for pickup and incineration.
Application Area
Research labs put cerium selenite to work in studies on solid-state chemistry, catalysis, and advanced spectroscopy. Its use creeps into ceramics, where it tunes the color and transparency of specialty glasses. Glassmakers push the boundaries using small amounts to craft filters that control light transmission across visible and near-infrared bands. Electrochemists pair it with other metal selenites in redox batteries, unstable at room temperature but showing high capacity in lab setups. Its role in photocatalysis grows, since combining cerium’s redox flexibility with selenium’s semiconducting nature unlocks reactions for green energy and pollutant breakdown. Environmental chemists leverage its sensitivity to oxygen for quick probes into trace contaminants, and trial runs in agriculture look at its impact on trace-element balance for certain crops.
Research & Development
R&D projects build on cerium selenite’s unique status at the intersection of rare earth and selenium chemistry. Industry teams examine its role in low-temperature electronic components, hoping its stability translates into improved device reliability. A new wave of research dives into crystal engineering, growing larger, defect-free single crystals for optical testing and specialty lasers. Pharmaceutical teams experiment with it as a carrier for selenium in slow-release dietary supplements—early results show promise, though complex interactions between cerium and the body’s chemistry make this path tricky. Collaboration between university groups and private sector labs continues, crossing boundaries between basic chemistry, physics, and engineering, all eager to uncover hidden benefits in solar cells, magneto-optical sensors, and selective reduction catalysts.
Toxicity Research
The safety profile for cerium selenite remains mixed. Animal tests hint at moderate acute toxicity when doses get high, especially due to selenium content. Chronic exposure can trigger symptoms similar to those seen with other heavy metals: kidney strain, irritation, and impaired enzyme function. The compound’s low solubility limits absorption in most environmental settings, but fine dust can linger on work surfaces and gloves, so thorough training stands as a front-line line of defense. Monitored disposal and thorough personal protective equipment use help avoid accidental ingestion or inhalation. Regulatory agencies review new animal studies and environmental impact reports each year, but gaps persist—especially regarding long-term effects on soil and water microorganisms.
Future Prospects
Cerium selenite holds potential well beyond today’s applications. Materials scientists look to it for roles in next-generation photonic crystals and green hydrogen production. As rare earth markets grow more competitive, recycling efforts for cerium selenite waste look set to sharpen, cutting both costs and environmental footprint. Its unique electronic properties draw interest from quantum technology startups, where even minor improvements in crystal quality could trigger big leaps in sensor accuracy. Efforts to tweak its composition—adding dopants or combining it with carbon nanomaterials—open new paths in catalysis and energy storage. If sustained funding flows into its R&D, cerium selenite may well become a staple in clean energy tech, precision optics, and tailored chemical engineering for decades to come.
What is Cerium Selenite used for?
Digging into Cerium Selenite’s Place in Modern Science
Chemicals sometimes stick to the shadows, and cerium selenite may fly under the radar for most people. I remember learning about rare earth elements in a college lab, noticing how often the cerium group crept into advanced applications—catalysts, magnets, and even glass polishing compounds. Cerium’s chemistry suits industrial needs, and tying this soft, silvery element with selenium builds a new category of compounds serving researchers and niche markets alike.
Scientific Curiosity and Real-World Experiments
One of the strongest reasons cerium selenite stands out happens in the laboratory. Chemists lean on it for its redox behavior. The material often plays a role as either an oxidizing or reducing agent, which means it can accept or donate electrons during chemical reactions. It opens up opportunities for researchers exploring new types of batteries, solar panels, and other electronic devices. The ongoing push for small-scale, long-lasting batteries in medical devices and sensors could use cerium’s redox magic. These features don’t just exist on paper; teams run experiments today, hoping cerium selenite can unlock lower costs in battery production.
Glass and Imaging: More Than Meets the Eye
My old chemistry professor kept a chunk of polished glass on his desk, always ready to show off how some compounds can shift its look and usefulness. Cerium selenite sometimes finds its way into specialty glass, especially where unique optical characteristics matter. Scientists value it for how it filters certain wavelengths and improves image clarity, whether in microscopes or advanced telescopic lenses. In a world now filled with high-performance cameras and scientific instruments, every element that sharpens our vision or makes medical imaging safer builds real value for society.
Healthcare Applications—Small Steps, Big Hopes
I recently spoke with a researcher developing contrast agents for scans and therapies. Cerium selenite, due to its behavior at the cellular level, draws interest for future cancer detection and treatment projects. Its unique properties could help guide where drugs travel after entering the body or provide clearer scan results. No magic cure yet, but early results push teams to look deeper. The race for better, less toxic imaging agents means compounds like cerium selenite won’t stay locked in storage cabinets for long.
Environmental and Safety Concerns
With every promising chemical, someone has to ask tough questions about safety. Cerium selenite combines cerium and selenium, both of which show bioactivity. Handling always needs strict protocols—no researcher wants accidental exposure. Over the years, scientists and regulators have built standards that require tight control and disposal guidelines for these compounds. This safeguards both researchers and the local environment. From what I’ve seen, labs and manufacturers raising the bar on safety training help keep everyone protected, with regular audits and real transparency on risks. There’s no shortcut; you stay safe by respecting the chemical and prioritizing procedures.
Bigger Picture: Where Cerium Selenite Could Go Next
Innovation rarely follows a straight line. Today, cerium selenite carves out space in advanced research and industrial labs, playing supporting roles in energy, optics, and healthcare. Staying up to date with material science and chemistry breakthroughs points to even broader uses on the horizon. By supporting strong collaboration between universities and private industry, more solutions using rare earth compounds take shape. As teams gain hands-on experience, best practices will spread, building both safer and more efficient new technologies for everyone.
Is Cerium Selenite safe to handle?
Understanding Cerium Selenite
Cerium Selenite, a chemical compound made from cerium and selenium, finds use in labs and certain specialized manufacturing. Some chemists might reach for it during research or material processing. Its reputation as a rare-earth compound brings a sense of novelty—maybe even the sense that it’s somewhat exotic. That doesn’t mean it’s safe just because it’s not encountered every day. In fact, handling any selenium compound throws up big red flags for me based on past safety training and stories of mishaps in academic labs.
What the Science Says About Safety
Safety sheets and toxicologists get straight to the point with Cerium Selenite. Selenium compounds can be very toxic, especially if dust or fumes get inhaled, or if the compound finds a way into your mouth. Too much exposure damages organs, with the liver and lungs in the firing line. Irritation of the eyes or skin crops up quickly if you don’t cover up. Having seen researchers regret not taking those warnings seriously, I won’t mince words—you’ll want goggles, gloves, and a well-ventilated space if you need to work with it. Open containers outside a fume hood, and you invite headaches at best, or something much worse.
Past Experiences in the Lab
I remember one slip-up during a teaching session. Someone handled a very small quantity of a selenium compound, not Cerium Selenite but a cousin. Just opening a container generated enough airborne dust to cause a cough that lingered for days. College-aged scientists often see themselves as invincible, but the stories stick. It’s a sharp reminder: even a modest amount of the wrong powder causes real problems if it’s not respected.
Why Proper Handling Is Non-Negotiable
One look through incident reports at big research facilities drives the point home. The worst cases bite when chemists don’t pay attention to safety sheets or when short cuts creep into routines. If Cerium Selenite gets mixed with acids or organic material, things can spiral further—think of it as opening up a can of worms you never wanted. Fire departments and poison control have real-world stories to share. They all say the same: Don’t underestimate compounds you can’t pronounce easily.
Simple Steps for Safe Use
There’s no mystery to protecting yourself. Use goggles and gloves rated for chemical work, not those flimsy over-the-counter types. Keep Cerium Selenite inside a working fume hood. Seal containers tight, and label them so there’s no confusion later on. Wash hands and surfaces as soon as you finish. Never eat or drink in the same room. Disposal isn’t just a trip to the trash—follow hazardous waste guidelines, since selenium can poison ground and water if it ends up outside proper channels.
Alternatives and the Bigger Picture
Some researchers ask if a substitute works instead. In many projects, swapping out Cerium Selenite for something less toxic pays off. Cutting down on toxic inventory lowers accident risks all the way down the supply chain. If you have to use Cerium Selenite, training keeps you sharp. This isn’t just a box to check for risk management—it’s about community safety, the health of researchers, and the trust that lets science move forward safely.
Staying Informed
It pays to revisit data sheets every time new batches arrive. Chemistry isn’t just formulas on a page—it’s about real-world consequences. Cerium Selenite belongs on the list of “do not take lightly” materials. Solid experience and respect for best practices keep you out of trouble. In the end, the most hidden dangers come from familiarity and forgotten caution. Keep risks in the front of your mind, and you’ll stay on the right side of chemistry.
What is the chemical formula of Cerium Selenite?
Cerium Selenite: Breaking Down the Chemistry
Cerium selenite stands as a compound made from cerium, a lanthanide metal, teaming up with selenium and oxygen. It doesn’t show up in the way table salt or baking soda does in everyday life, but its role becomes clearer in specialized fields, from research labs to advanced manufacturing setups. To get right to it, the chemical formula for cerium selenite is Ce(SeO3)2.
Why the Formula Matters
Looking at chemical formulas reminds me a bit of following a good recipe. With the right mix of elements—nothing more, nothing less—you get the unique result you want. That matters a lot in research and industry. Cerium selenite’s formula shows one cerium ion (Ce4+) hooks up with two selenite ions (SeO32–). Getting this stoichiometry right keeps chemists safe and helps avoid waste or even dangerous situations, especially with materials as sensitive as selenium compounds.
What Makes Cerium Selenite Worth Noticing?
Plenty of folks haven’t heard about it in daily conversations, but this chemical finds use as a catalyst, a pigment, and sometimes in certain glass-making applications. Cerium’s position as a rare earth metal brings with it a bunch of interesting traits, including high reactivity and the ability to change oxidation state. Selenium, for its part, draws the attention of environmental scientists because of how it behaves in the natural world—too much can wreck ecosystems, but just enough powers important biological processes.
One aspect I’ve seen trip up beginners is dealing with rare earth elements. Cerium itself can exist as Ce3+ or Ce4+, but selenite only balances with the +4 state. Matching charge and composition matters because the wrong combination leads to the wrong compound—or no reaction at all.
Safety and Environmental Considerations
Selenium, in the form of selenites, isn’t the friendliest chemical to handle casually. Swallowing or even touching enough of it brings health dangers. Disposing of it safely should involve strict attention to chemical waste protocols. While cerium isn’t particularly toxic, the selenium part more than makes up for that in terms of handling demands.
Back in the day, scientists gave little thought to environmental impacts when using rare earth and selenium compounds. Today, the focus shifted. Facilities that use cerium selenite work hard to capture, recycle, and neutralize their wastes. Clear labeling, using the right storage containers, and having an emergency plan make a difference. These steps help avoid workplace accidents and environmental spills.
Paths Forward
Researchers keep searching for safer substitutes or improved recycling methods. Some investigations focus on recovering selenium and cerium from industrial waste streams to cut down raw material use. Keeping an eye on these strategies can boost sustainability. Most of us outside the chemistry world will never directly encounter cerium selenite, but the way labs handle it reflects broader lessons for chemical safety, environmental respect, and scientific accuracy.
How should Cerium Selenite be stored?
Why Storage Matters for Cerium Selenite
Chemistry can feel distant until you’ve had to clean up a spilled chemical that shouldn’t hit the floor. Cerium selenite, a combination of cerium, a rare earth element, and selenium, isn’t something you find at the local supermarket. It shows up in labs and, from experience, it doesn’t get the same attention as household chemicals. That’s a risk. Cerium selenite isn’t as notorious as mercury or cyanide, but mishandled mineral compounds cause real trouble for health and safety. I once watched a researcher ignore a leaky cap on a jar of a different selenite—within days, air moisture turned that powder crusty and basically useless for the experiment.
What Science Tells Us
The key with cerium selenite comes down to moisture, temperature, and chemical compatibility. This compound reacts with water. Exposing it to a humid environment usually triggers slow chemical changes. If that jar sits open long enough in a damp lab, you’ll see evidence: discolored powder, granules caked together, maybe a mineral smell. That’s a sign of lost integrity. Keeping it bone dry isn’t just a preference. It makes or breaks the material’s value in research and manufacturing. A desiccator—a sealed, airtight cabinet with drying agents—keeps the powder dry and stable. Dry cabinets prevent moisture from sneaking in, which preserves purity and reduces risk.
Temperature and Light Control
Cerium selenite won’t melt in your average storage closet, but heat can mess with compounds in unexpected ways. A hot shelf next to a radiator sounds harmless, but prolonged high temperatures might degrade sensitive powders over time, especially with the risk of volatile decomposition. Room temperature works, but away from direct sunlight, which can heat up glass containers and slowly degrade contents. Darkness and ambient indoor temperatures have kept our stocks reliable year after year.
Container Materials and Labeling
Some folks might store chemicals in whatever jar’s handy, but that’s asking for cross-contamination. Glass containers with tightly fitting lids shut out reactions with plastics and make spills easier to spot. Every jar should have a clear, durable label—chemical name, hazard warnings, and date received. People underestimate labeling until they find two unmarked jars and can’t tell them apart. In my lab days, relabeling after accidents always took longer than simply doing it right the first time.
Keeping Cerium Selenite Safe From Other Chemicals
Chemical storage isn’t just about where it sits, but what it sits next to. Cerium selenite reacts with strong acids, and those shouldn’t share a shelf. Mixing up chemical families guarantees unpleasant surprises. Years ago, our storeroom shelves carried acids, bases, and oxidizers separated by category. It took just a single mix-up, an acid bottle left inches from a storage jar, for us to tighten up the whole system. Keeping cerium selenite in a section reserved for rare-earth and selenium compounds stops those accidents in their tracks.
Disposal and Emergency Planning
No storage system works forever—spills and breaks happen. Emergency instructions must hang where anyone can see them, not just buried in a binder. Cerium selenite waste needs specialized disposal procedures to avoid releasing toxic selenium compounds. Keeping spill kits and training staff for chemical cleanups protects everyone in the building. Quick response to a minor spill in our storage area meant no chemical dust spread to nearby offices, saving time, money, and health worries for the whole team.
Why This All Adds Up
Few people notice chemical safety until it fails. A little logic, clear procedures, and remembering stories from the lab all feed into safer, more reliable storage. Cerium selenite, like any specialized material, rewards careful handling. The end goal: keep it pure and safe so it performs as intended in the field—not as a costly cleanup project.
Where can I purchase Cerium Selenite?
Understanding Cerium Selenite and Its Uses
Cerium selenite doesn't appear on many shopping lists. In my own research, most folks searching for it work inside labs, tech companies, or universities. The compound has a place in advanced glassmaking, chemical research, and sometimes niche electronics. It isn’t something you’ll see stocked on regular store shelves or in hardware stores. Handling and sourcing chemicals like this involves real risk, so a responsible approach matters.
Where Scientists and Companies Go Shopping
The easiest way to buy cerium selenite involves heading to specialized chemical suppliers. Companies like Alfa Aesar, Sigma-Aldrich, and Strem Chemicals serve professionals daily. Their catalogs usually list purity, forms, and fair warnings about safety. In the US and Europe, only organizations with proper credentials can order. Individuals don’t usually clear those hurdles, except by enrolling in a university with a legitimate reason to purchase.
In some cases, smaller lab supply businesses like Oakwood Chemical or ChemShuttle have stock too. Their reputation matters. I would never buy a chemical from a seller without a known track record because it puts both research and safety at risk. Check reviews, ask for certificates of analysis, and confirm the supplier meets local regulations. Labs rely heavily on these documents for traceability and consistent results.
Risks Outweigh Convenience
If you poke around online marketplaces, sooner or later you’ll spot sketchy listings—sometimes on eBay, Alibaba, or lesser-known websites. Most professionals I know avoid these sources. Besides the risk of low-quality chemicals or even dangerous substitutions, customs offices might seize shipments or pursue charges. Selenite compounds contain selenium, which brings strict handling and transport restrictions in many countries. The law doesn’t care much whether you handle it for art, since public safety trumps convenience every time.
Buying from a recognized supplier keeps everyone honest. They provide proper labels, guidance for disposal, and help with questions. Health authorities expect this level of care, especially for compounds with both rare-earth metals and selenium. No doubt about it: standards exist because mistakes with chemicals can harm the environment and living things quickly. I learned this firsthand during lab training, where mistakes led to costly delays and tricky cleanups.
The Importance of Training and Compliance
I know a few graduate students who thought they could handle everything themselves. All it took was one spill for everyone to appreciate those boring-sounding safety lectures. Cerium selenite deserves respect. Even in properly equipped labs, protective gear and strong ventilation come standard. Disposal presents another challenge, and many universities partner with hazardous waste companies to deal with leftovers safely.
Ordering with the right paperwork also helps with audits. Labs get visits from inspectors who expect to see records for every shipment. Cutting corners makes future experiments harder, and trust in your research takes a hit if anyone catches errors.
Better Ways to Access Cerium Selenite
People with a genuine need almost always work within universities, startups, or industry labs. Collaborating with these institutions streamlines everything—access, safety plans, technical advice. Sometimes it’s possible to reach out to academic contacts who can help you find a place at the bench. If the goal involves personal curiosity rather than research, consider visiting a local lab instead of risking mail-order chemicals.
Taking shortcuts tempts some folks, but with rare-earth compounds like cerium selenite, the risks rarely pay off. Safe, responsible sourcing supports future discovery. It keeps everyone out of trouble, from the lab technician to the nearby community.
| Names | |
| Preferred IUPAC name | cerium(3+) selenite |
| Other names |
Cerium(III) selenite |
| Pronunciation | /ˈsɪəriəm ˈsɛlɪnaɪt/ |
| Identifiers | |
| CAS Number | 13597-46-1 |
| Beilstein Reference | 80537 |
| ChEBI | CHEBI:131738 |
| ChEMBL | CHEMBL4296772 |
| ChemSpider | 22587291 |
| DrugBank | DB11125 |
| ECHA InfoCard | 100.033.643 |
| EC Number | 234-599-9 |
| Gmelin Reference | Gm. 778 |
| KEGG | C18609 |
| MeSH | D045073 |
| PubChem CID | 166864 |
| RTECS number | VV8780000 |
| UNII | L7Z8G6G10Y |
| UN number | UN2655 |
| CompTox Dashboard (EPA) | DTXSID3046934 |
| Properties | |
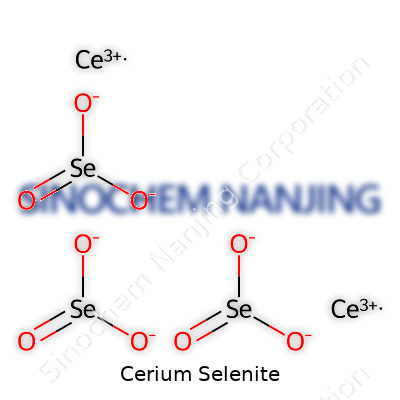
| Chemical formula | Ce2(SeO3)3 |
| Molar mass | 379.09 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 4.947 g/cm³ |
| Solubility in water | Insoluble |
| log P | -4.63 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 10.84 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 2.45 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1347 kJ/mol |
| Pharmacology | |
| ATC code | V07CI |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-0-OX |
| Lethal dose or concentration | LD50 oral rat 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 5,000 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Cerium Selenite: "15 mg/m3 (total dust), 5 mg/m3 (respirable fraction) as nuisance particulate |
| REL (Recommended) | 0.02 mg Se/m³ |
| Related compounds | |
| Related compounds |
Cerium(III) selenide Cerium(IV) oxide Sodium selenite |

