Carbonyl Fluoride: A Closer Look
Historical Development
Carbonyl fluoride didn’t appear overnight. The compound’s discovery shaded into the era where chemists scoured for new building blocks in organic and inorganic chemistry. Chemists investigated fluorinated gases in the early twentieth century, looking for both their industrial potential and behaviors in the laboratory. Carbonyl fluoride owes a lot to the tradition that produced early refrigerants, uranium enrichment agents, and nerve agents, showing both the promise and peril that fluorochemicals brought to chemical industries. Laboratories built the earliest samples as offshoots of sulfuryl and phosgene chemistry, later refining production as a chemical intermediate. Important chemists and chemical companies published findings as the utility for fluorinated compounds grew, especially in the manufacture of plastics, pharmaceuticals, and agrochemicals.
Product Overview
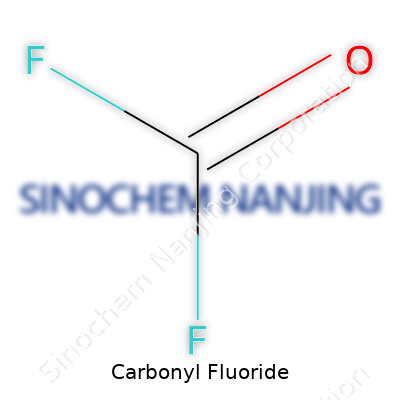
Carbonyl fluoride stands as an acyl fluoride, with the formula COF2. In appearance, it is a colorless, toxic gas, strongly reminiscent of phosgene but marked by its aggressive fluorinating power. Commercial and academic labs keep it for its ability to insert both fluorine and the carbonyl group into other molecules. Producers ship it in pressurized steel cylinders, sometimes with stabilizers depending on shipment duration and region. Companies measure purity using gas chromatography, keeping impurities — like hydrogen fluoride and phosgene — under strict control. Only a handful of manufacturers produce significant volumes, with buyers clustered mostly in the specialty chemical and advanced material sectors.
Physical & Chemical Properties
This small molecule boils at -83°C and freezes below -136°C, highlighting its volatility even at freezer temperatures. A direct sniff can be fatal. The molecule is linear, similar in shape to carbon dioxide but more reactive. Its molecules pack a heavy punch even in minute concentrations, breaking down in sunlight or in the presence of moisture into hydrofluoric acid and carbon dioxide. Carbonyl fluoride dissolves poorly in water, but reacts rapidly on contact — a matter that drives much of the safety-focused research in the field. When handled in pure streams, it corrodes metals like steel and attacks glassy surfaces, so suppliers use corrosion-resistant alloys for storage and transport. In practical applications, its chemical stability in dry environments makes it useful in controlled fluorination and as a predecessor in advanced polymer synthesis.
Technical Specifications & Labeling
Shipping containers bear strict hazard warnings in large fonts. Labels specify that the gas asphyxiates and corrodes tissue, while trace contaminants must not exceed thresholds set by both industrial standards and national regulations. Canisters include batch numbers, point-of-origin codes, and UN transport codes — often UN3304 for toxic gases. Customers receive certificates of analysis detailing composition, emphasis on hydrogen fluoride and phosgene residues. In research and industry, safety data sheets stress instant evacuation and respiratory protection on leaks, and most institutional protocols require two-person handling at all times. Rigid labeling supports tracing, not just for workplace safety but also to ensure chain-of-custody in cases of spills or exposure. Government watchdogs, including the U.S. Department of Transportation and European Chemicals Agency, publish publicly available labeling protocols.
Preparation Method
Labs and factories prepare carbonyl fluoride mainly from the reaction of phosgene (COCl2) with hydrogen fluoride (HF). This process happens in corrosion-resistant reactors. Trickling phosgene gas over anhydrous hydrogen fluoride at elevated temperatures strips the chlorine atoms from phosgene and substitutes them for fluorine, yielding carbonyl fluoride and hydrogen chloride. Since hydrogen fluoride eats through glass and damages skin, the entire reaction setup uses specialized linings, gaskets, and remote control automation. In clean-up steps, chemists scrub by-product gases with alkali or concentrated base solutions to neutralize acids before venting. In smaller amounts, chemists have explored alternate routes, such as thermal decomposition of difluoroformaldehyde or electrochemical methods, but phosgene fluorination has stuck as the commercial favorite due to reliability.
Chemical Reactions & Modifications
In synthetic chemistry, carbonyl fluoride behaves as both an electrophile and a fluorinating agent. When it reacts with alcohols, it forms fluoroformates and liberates hydrogen fluoride. Reaction with amines gives carbamoyl fluorides, which open up paths to pharmaceuticals and chemical protection groups. In the presence of water, it hydrolyzes instantly, making it useful for generating hydrofluoric acid or as a one-pot carbonylating agent. Inhalation, even at very low levels, damages lung tissue due to the rapid hydrolysis product, so chemists work under fume hoods and closed nitrogen systems. Industrial routes tap carbonyl fluoride as an intermediate converting to fluorinated monomers like tetrafluoroethylene (TFE). Fluorochemists appreciate its controlled reactivity, driving research into newer transformations that minimize unwanted by-products in advanced synthesis.
Synonyms & Product Names
Commonly referred to as carbon oxide difluoride or carbonofluoridic acid, its best-known names in commerce are Carbonyl Fluoride and COF2. German literature records it as Kohlenoxiddifluorid, and older documents sometimes list it as Fluorocarbonyloxy. Global chemical indexes assign it CAS number 353-50-4. Companies brand it under catalog names for high-purity labs, but workplace jargon often truncates it simply to COF2. Occasionally, labels display safety codes as a first identifier due to hazard class, reflecting the regulatory framework more than marketing flair.
Safety & Operational Standards
Handling carbonyl fluoride places immense responsibilities on both companies and workers. Errors result in burns, blindness, pulmonary edema, or death. Facilities enforce rigorous leak detection, ventilation, and respiratory protection requirements. Technicians go through regular safety drills; equipment undergoes scheduled maintenance and pressure testing. American Conference of Governmental Industrial Hygienists advises short-term exposure limits in the low parts-per-million range and forbids any unprotected handling. Medical teams on site need training in hydrofluoric acid exposure, with calcium gluconate gel always stocked for field treatment. Incident logs show that violations rarely go unpunished — not by machines, but by the brutal chemistry of the substance itself. Reliable sensors, double-walled pipes and full-face respirators come standard. Where regulations lack specifics, industrial consortia set their own, knowing that even a single oversight holds company reputation and worker safety alike on the hook.
Application Area
One key use pops up in the synthesis of fluoropolymers, like polytetrafluoroethylene (PTFE), which turn up in everything from non-stick pans to aerospace cable insulation. Carbonyl fluoride acts as a way station in converting cheap precursors to high-value monomers by carefully substituting hydrogens or chlorines with fluorine atoms. In specialized organic synthesis, labs use it for carbonylation and fluorination steps otherwise hard to pull off, sidestepping harsher or less selective reagents. Some agricultural chemistries once explored it as an insecticide precursor, but the health hazards shifted development elsewhere. On the cutting edge, researchers test its use for regulated introduction of fluorine into drug molecules or high-energy materials. Its gas-phase behavior, combined with acute reactivity, makes it more prominent as a tool among experts than a mainstay for general chemical production lines.
Research & Development
Modern research circles dig into new, greener preparation methods, hoping to supplant phosgene-based routes and reduce waste. Continuous-flow reactors, membrane separations for gas purification, and in situ generation draw attention as factories chase both safety and cost savings. Chemists want cleaner, more selective reactions to limit hazardous by-products. Academic groups work to tame the reactivity and harness carbonyl fluoride for next-generation fluorinated organics, especially where selective carbonylation and fluorination unlock better pharmaceuticals or materials. Open literature shows that, despite strong hazards, research funding persists since the need for tailored fluorine insertion remains unsolved by other chemicals.
Toxicity Research
Toxicologists have documented the ways carbonyl fluoride attacks the body. At low levels, it causes respiratory tract irritation, headaches, and delayed lung damage. Inhalation leads to quick hydrolysis, and the concurrent formation of hydrofluoric acid explains much of its corrosive effect. Occupational health researchers report that even trace exposure builds up in enclosed environments, sharpening the focus on continuous air monitoring. Skin contact leads to burns, and any accidental inhalation or splash needs immediate medical response. Most lab studies happen in animal models, but field accidents and incident records in industry support the case for exclusion zones and robust training. Regulators cite animal data showing lung damage, inflammation, and lethality at only slightly above recommended occupational limits, a fact that keeps safety gear at the center of any operation.
Future Prospects
The role of carbonyl fluoride in the chemical sector sits in tension between its rapid action and inherent danger. Engineers pursue process intensification and safer plant designs aiming to limit fugitive emissions. Next-gen synthesis seeks in situ, on-demand generation, turning away from stockpiling and bulk transport. New regulations push up the cost for large-scale use, steering routine chemistry toward less hazardous alternatives or toward automated, enclosure-driven processes. Specialists keep their sights on even narrower targets — precision fluorination, advanced drugs, functional materials — accepting trade-offs if risks can be managed. I’ve seen shifts in the academic community move away from legacy reagents with bad safety records, but haven’t yet seen a true replacement for the unique carbonyl and fluorine chemistry offered here. As technology marches forward, the hope centers on making the essential chemistry safer by default, not just by discipline or luck.
What is Carbonyl Fluoride used for?
A Chemical With Hidden Influence
Carbonyl fluoride doesn’t grab headlines, but its impact reaches into parts of life that most folks never realize. This colorless, highly toxic gas shows up mostly in the world of industrial chemistry, and it earns its spot by getting tough jobs done—especially jobs other chemicals can’t handle.
How Industry Puts Carbonyl Fluoride to Work
I first learned about carbonyl fluoride through a friend who worked at a semiconductor plant. He mentioned they used the stuff to etch patterns onto silicon wafers that go into computer chips. In these cleanrooms, workers wear protective gear, since even tiny leaks would cause big trouble. The reason they put up with the danger? Carbonyl fluoride does the job quickly and with precision. In electronics, accuracy means profit, and this compound holds its own against stiff competition.
This chemical also plays a part in making pesticides and synthetic lubricants. Anyone who has worked on a farm or in a garage probably hasn’t thought much about what went into the lubricants keeping machines running or the crop protection products on store shelves. A byproduct or reactant, carbonyl fluoride shows up in these supply chains more than most people would guess. Its unique ability to add specific fluorine atoms to molecules gives it an edge in these processes.
The Price of Power: Health and Safety Worries
Sometimes, the story of a chemical is as much about the problems it brings as the jobs it does. In college, my roommate did a co-op job at a chemical plant. He told me that even seasoned chemists treat carbonyl fluoride with respect, because exposure risks go beyond the usual discomforts. Inhaling this gas damages the lungs and eyes within minutes. As a gas, it escapes easily, so leaks remain a real threat to workers unless they’re armed with top-notch ventilation and sensors.
Over the years, the industry leaned on stronger rules. Good companies invest in training and equipment that cut down on exposure, both for the folks on the line and people living nearby. Still, accidents remind us that vigilance matters every shift.
Climate Impact and Environmental Concerns
As someone who cares about clean air, I keep an eye on chemicals with greenhouse potential. Carbonyl fluoride can break down into hydrofluoric acid and carbon dioxide, both of which raise alarms. The acid can burn skin and corrode metal; the carbon dioxide fuels climate change. Neighboring communities pay the real price if mishaps release these into the atmosphere.
Most countries now set stricter limits on how much makes it out of a plant, but like with many specialized gases, loopholes and enforcement gaps persist. Investment in better capture technology and on-site destruction methods can go a long way toward shrinking pollution risk.
The Path Ahead: Safer Alternatives
Finding safer replacements for carbonyl fluoride remains a challenge. Its chemical skills give it an edge that can’t always be matched. That said, research continues on green chemistry approaches—ways to build up molecules without reaching for the nastiest tools in the box. Industries can push for better processes, more transparency about what goes into our products, and tighter controls on every pound produced and used.
Behind every device, lubricant, and crop protection product sit stories most people never hear. Paying attention to chemicals like carbonyl fluoride means respecting the invisible work and learning to ask how manufacturing can leave a lighter mark on our world.
What are the safety precautions when handling Carbonyl Fluoride?
Understanding What You’re Dealing With
Stepping into a lab, tank farm, or any facility with carbonyl fluoride always gives me a chill. This colorless gas has a sharp, biting odor, and the science backs up the gut feeling—this stuff can mess you up quickly. Working in environments packed with toxic gases, one never forgets that carbonyl fluoride can hit the lungs, eyes, and skin hard and fast. The best tech in the world won’t help if you ignore the basics.
Personal Protection Means More Than Gloves
Goggles, chemical-resistant gloves, and a splash apron might sound standard, but that’s not enough. A full-face respirator or, better yet, a positive-pressure self-contained breathing apparatus, keeps the gas out of your system. Face shields and lab coats help too, but exposed skin or a cracked mask seal turns a routine job into a trip to the hospital. I’ve seen someone react to just a whiff—not pretty. Respiratory protection needs a serious fit check every shift.
Workplace Controls Can’t Get Lax
At sites handling carbonyl fluoride, I expect to see solid ventilation systems humming quietly, backed by gas detectors blinking in every corner. Local exhaust takes priority, with fume hoods tested regularly. I always check manuals to know where the emergency shutoff valves sit. Accidents happen in places with blocked vents or busted fans—maintenance must be on point. Storage rules make sense: secure cylinders in upright racks, keep them out of sunlight, and watch for temperature spikes.
Emergency Protocols Make a Real Difference
The emergency eye wash and shower stations get their weekly test, and the best facilities have a plan that everyone can recite forward and back. I feel uneasy if workers can’t find the nearest wash station blindfolded. If carbonyl fluoride splashes or leaks, you want copious water at arm’s length. Spill kits—absorbent pads, neutralizing agents, heavy-duty disposable gloves—should sit ready at the perimeter. Call the trained hazmat team, don’t improvise yourself as the hero.
Medical and Environmental Hazards
Direct exposure tears up lungs, corrodes skin, and triggers severe eye damage. If you’ve seen fluorine burns or felt your lungs seize from inhaling a reactive gas, you learn to respect those safety sheets. Even low-level leaks matter. Health teams keep baseline and periodic medical exams for anyone near the material. I recommend companies invest in training showing real-world footage—words rarely hit home the way a few seconds of chaos do.
Training Matters More Than Fancy Equipment
All the best engineering controls fade if folks don’t follow the rules. I’ve watched new hires get overconfident, skipping one step from the checklist, and that’s where injuries come from. Leadership sets the tone. Management that visits the shop floor, joins safety drills, and brings in stories from real incidents changes the culture. Clear signage, regular retraining, and strict job briefings should be standard. Written policies mean little if nobody believes they matter. Companies that talk about safety once a year never achieve the zero-incident goal. Hard lessons from the past only mean something if they change what happens today.
Taking Responsibility, Every Day
Treating carbonyl fluoride casually risks lives and reputations. Respect starts with personal discipline but grows with a shared culture. Teams who check on each other spot mistakes and save lives. A healthy safety culture means open talk about failures as well as successes. Real progress begins on the ground, not in the boardroom.
What is the chemical formula and molecular weight of Carbonyl Fluoride?
The Basics Behind the Formula
Carbonyl fluoride carries the chemical formula COF2. It's a straightforward mix, combining one carbon atom, one oxygen atom, and two fluorine atoms. Scientists use it in all sorts of research and manufacturing scenes—often behind the scenes in chemistry labs and specialty industries.
Molecular Weight: A Crucial Number
Molecular weight isn’t just a lab curiosity. People working with dangerous or reactive gases rely on it to plan safe storage, shipment, and every handling step in between. COF2 clocks in at about 66.01 grams per mole. For anyone who works in industrial settings with gases—especially toxic ones—every decimal point can end up making a real-world difference.
Experience in the Lab and Industry
I still remember my first real encounter with specialty chemicals. You see a tiny vial labeled with unfamiliar formulas. Safety procedures get drilled into your head because mistakes aren’t just paperwork headaches—they can be physical emergencies. Carbonyl fluoride stood out to me because of its unusual combination of toxicity and reactivity. Even now, I don’t take lightly any project that mentions fluorinated carbon compounds. I’ve seen teams spend hours just reviewing molecular weights to prevent miscalculations that could trigger leaks or exposure.
Safety and Handling Precautions
Most folks don’t cross paths with carbonyl fluoride. It isn’t something you stumble on at the hardware store. For the select few who do work with it, the risks demand respect. At a molecular weight of 66.01 g/mol, even small amounts pack a punch. The substance gives off a sharp, suffocating odor, and its fumes can chase anyone from a poorly ventilated lab in seconds. Prolonged exposure, even at low doses, attacks the lungs. I’ve watched coworkers go through nearly military-grade drills to contain minor spills or leaks.
Why This Chemical Draws Concern
COF2 doesn’t get the public attention that, say, chlorine gas receives. In chemical circles, it triggers alarm bells. Its volatility means that broken seals, faulty equipment, or rushed calculations can escalate fast. With fluorine in play, the dangers aren’t all acute; long-term issues stack up in lungs and tissues.
Responsible Use and Possible Solutions
Tighter regulations and better monitoring equipment have made workplaces safer. These days, sensors reading molecular weights and concentrations automate part of the vigilance. Still, no machine replaces hard-earned caution. Every technician I’ve known learns to double-check the math and the gear before opening a cylinder. Some teams have pushed for substitution with less hazardous gases—an alternative gaining ground where possible. A few companies have switched to on-demand generation of COF2 at the place of use, limiting how much sits around waiting for an accident.
Staying Ahead of the Risks
Each detail matters. Even the difference of a gram in molecular weight gets factored into pressure calculations and ventilator settings. I’ve watched new safety plans unroll—and the best ones always hinge on making everyone understand these numbers, not just the folks in charge. In chemical safety, awareness of seemingly small details like formula and molecular weight turns academic information into practical protection. It’s real science, real concern, and very real solutions forged day by day.
How should Carbonyl Fluoride be stored and transported?
Understanding the Risks
Carbonyl fluoride comes with a reputation in the chemical world. This colorless gas causes sharp irritation to eyes, skin, and the lungs. After decades around industrial safety protocols, I’ve watched people underestimate toxic, reactive materials. Just because a gas is invisible doesn’t mean it keeps quiet. A single accidental release can put workers at risk for severe burns and breathing problems. Inhaling enough can even be fatal. Remembering the Bhopal tragedy or smaller mishaps closer to home, I’ve learned how a lapse in storage or transit creates consequences the community feels for a long time.
Storage: It’s Not About Chasing Perfection
Let’s get straight to the point—carbonyl fluoride doesn’t play nice with moisture. It reacts with water vapor in the air to form corrosive and toxic compounds like hydrofluoric acid and carbon dioxide. So, storing it demands strict dryness. From experience, specialized carbon steel or nickel cylinders made for pressurized gases provide the best bet against leaks. Seals, joints, and valves must hold up to frequent checks. Even the pressure gauge has to be kept in top working order.
Temperature matters more than most folks realize. Leaving gas cylinders in a sun-drenched yard or a cramped hot storeroom turns them into possible bombs. A well-ventilated, climate-controlled building—think bright lights, dry floors, no moisture—is what industry safety officers insist on. Any sign of corrosion, bulging, or faulty valves should be a red flag. Paying attention to these warning signs beats dealing with a leak in the dead of night or as staff are heading home.
Transport: Trust Only the Prepared
Moving cylinders of carbonyl fluoride isn’t like hauling bags of fertilizer. Department of Transportation (DOT) regulations require high-integrity containers, clear hazard labels, and documentation tracking every handoff. Equipment should match the standards set by regulatory agencies like the U.S. DOT or the International Air Transport Association (IATA) for international moves. After so many years reviewing shipping logs, the honest truth is that the weakest link isn’t always the packaging—it’s the gorilla handling on the loading docks or the driver on edge from too many deadlines.
Spare parts, neutralizing agents, and protective gear must go in the truck with the gas, not left behind. Splitting hairs over a few hundred dollars in safety investment only sets up bigger legal and financial pains down the line. Tightly regulated transit routes and regular emergency drills give everyone a better shot at heading home safe at the end of the shift.
Lessons from Real-World Grit
Across chemical plants and transit hubs, most mishaps link to skipping basic safety checks or ignoring staff concerns. I’ve stood beside weary engineers grumbling about “just one shortcut.” Next thing, someone’s suiting up to patch a leak with shaky hands. Iron-clad protocols and empowered employees often stop problems cold. In my own experience, companies that treat their people like partners, not replaceable cogs, see the fewest accidents.
I’ve seen regulators step in hard after incidents, levying legal action and public shaming. That kind of fallout lasts years. Staff, townsfolk, and investors suffer. Proactive training, solid communication channels, and a budget line that covers the little things—better gaskets, unannounced audits—go a long way in keeping carbonyl fluoride where it belongs.
Moving Toward Safer Futures
Chemical safety isn’t some abstract exercise. Every leak, every accidental exposure cuts into real lives, company reputations, and often, local economies. To move the dial, industry leaders, regulators, and frontline staff need to stick together. A mix of robust gear, practical training, and a bit of plain stubbornness about never cutting corners makes the difference. I’ve watched it in action. It works.
What are the health hazards and first aid measures for Carbonyl Fluoride exposure?
Why Carbonyl Fluoride Matters in Workplace Safety
Carbonyl fluoride shows up in labs and industries that need strong chemical agents. This compound gives engineers and researchers a handy tool, but the risks aren’t just theoretical. Getting exposed even once can cause lasting trouble, and not just for folks in white coats. I’ve worked near chemical storage before, and there’s never a day when safety doesn’t cross your mind. It only takes a small leak to cause a real panic.
Knowing the Health Hazards Up Close
Breathing in carbonyl fluoride brings burning in the nose, throat, and lungs. It doesn’t care about gloves or goggles if it finds an open path. I once watched a coworker double over, coughing after something leaked in the lab. His eyes watered up, and his face turned red fast. Even low amounts can set off headaches, dizziness, or a heavy feeling in the chest. Higher shots of this gas cause choking and severe shortness of breath. Lungs might not recover if the exposure keeps up, and people have landed in the hospital with pulmonary edema – the lungs fill with fluid, and oxygen simply can’t get in.
Touching carbonyl fluoride causes burns and blisters on the skin. The gas can sneak through clothing if nobody suited up properly. Eye contact brings searing pain and blurred vision that lasts for hours. Any chemical strong enough to etch glass has no place inside your body.
Delayed symptoms trick people into going back to work too soon. Some folks feel better after a small exposure, only to wake up a few hours later with pain or breathing trouble. That long tail lulls teams into a false sense of security.
Effective First Aid on the Spot
If carbonyl fluoride gets released, escape from the fumes beats anything else. Running for fresh air sometimes feels basic, but it remains the best move. Anyone feeling short of breath or weak shouldn’t try to tough it out. Get them outside, and let medical staff handle it from there. Oxygen helps a lot in severe cases, so keep an oxygen tank nearby if your workplace handles this chemical.
People exposed on their skin should strip off any contaminated clothing. It sounds embarrassing, but wasting time means the burn gets worse. Wash the affected area with lots of water, using up every emergency shower and eye-wash station in reach. Rubbing or scrubbing just drives the chemical deeper. Wrap up in a blanket for warmth, as shock tends to come fast after chemical injuries.
Eyes exposed to carbonyl fluoride demand at least 15 minutes of steady rinsing. Aim for the stream to hit from the inside of the eye out, never the other way around. Keep eyelids open, even if it feels impossible. Don’t let someone head home or try to sleep it off—eye doctors need to take over immediately.
Anyone who breathed the gas should see a doctor, even if they feel fine at first. The lungs may need X-rays and follow-up care. Letting things slide can mean suddenly getting hit by swelling or scarring that didn’t show up right away.
Actions and Real Solutions
Teams often wait for a scare to review their safety drills, but that makes no sense with a gas this nasty. Make safety gear—real face shields, chemical suits, and working exhaust fans—a daily habit. Don’t let worn-out equipment sit in the corner. Quick training on emergency showers or evacuation needs to stay fresh in every worker’s mind, not just the new hires.
Regular air quality testing doesn’t just tick a box for regulators. I’ve seen surprise leaks caught before anyone felt symptoms, thanks to alerts from detectors. Every saved life or averted injury proves that investing in monitoring and safety routines pays off.
| Names | |
| Preferred IUPAC name | carbonyl difluoride |
| Other names |
Carbonyl difluoride Carbon oxyfluoride Carbonic difluoride |
| Pronunciation | /ˈkɑː.bə.nɪl ˈflʊə.raɪd/ |
| Identifiers | |
| CAS Number | 353-50-4 |
| 3D model (JSmol) | _C=O;F\C(F)=O |
| Beilstein Reference | 1209225 |
| ChEBI | CHEBI:29438 |
| ChEMBL | CHEMBL133165 |
| ChemSpider | 42581 |
| DrugBank | DB02398 |
| ECHA InfoCard | EC 207-310-9 |
| EC Number | 206-845-1 |
| Gmelin Reference | 770 |
| KEGG | C14108 |
| MeSH | D002246 |
| PubChem CID | 7916 |
| RTECS number | FG9625000 |
| UNII | 86038Y22D6 |
| UN number | UN3300 |
| Properties | |
| Chemical formula | COF2 |
| Molar mass | 66.01 g/mol |
| Appearance | Colorless gas |
| Odor | Pungent |
| Density | 2.54 kg/m³ |
| Solubility in water | Reacts |
| log P | -0.38 |
| Vapor pressure | 5470 mmHg (20°C) |
| Acidity (pKa) | -1.1 |
| Basicity (pKb) | pKb = 7.89 |
| Magnetic susceptibility (χ) | -30.4×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.254 |
| Viscosity | 0.034 cP (20°C) |
| Dipole moment | 1.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -604.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -677.7 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V3CA01 |
| Hazards | |
| Main hazards | Toxic if inhaled, causes severe skin burns and eye damage, may cause respiratory irritation, reacts violently with water. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H330, H314 |
| Precautionary statements | P260, P264, P271, P304+P340, P310, P320, P403 |
| NFPA 704 (fire diamond) | 3-0-2-Tox |
| Flash point | -57°C (-70°F) |
| Autoignition temperature | 530 °C |
| Explosive limits | Explosive limits: 9.9–21% |
| Lethal dose or concentration | LD50 oral rat 44 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,000 mg/m³ (inhalation, rat) |
| NIOSH | PC1400000 |
| PEL (Permissible) | 2 ppm |
| REL (Recommended) | 0.0002 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Phosgene Carbonyl sulfide Carbon disulfide Thionyl fluoride |

