Octachlorocamphene: History, Properties, and Future in Perspective
Tracing the Roots: The Emergence of Octachlorocamphene
Octachlorocamphene didn’t pop out of nowhere. Its origins track back to a time when chemical research moved fast, and our understanding of pest control changed overnight. In the mid-1900s, production of organochlorine compounds took off, promising cheaper, more powerful attack against insects in fields, forests, and homes. Chlorination of natural camphene generated a new crop of highly chlorinated products, including octachlorocamphene. Researchers and industry leaders, eager for breakthroughs, incorporated it into pesticide blends—following the hard lessons of DDT and chlordane, hoping to strike balance between impact and public safety. Countries facing serious crop losses and disease transmission started using it where traditional control methods failed, so any new chemical that protected harvests or households grabbed attention. I sometimes hear people wonder why such chemicals had a heyday—after all, the environmental costs eventually forced a rethink. Yet, in those years, balancing food security and public health often outweighed emerging environmental risks.
What We’re Looking At: Understanding Octachlorocamphene
Chemically, octachlorocamphene packs in chlorine, making it a member of the broader chlorinated camphene family. It stems from camphene’s reactive structure—throwing a host of chlorine atoms onto the skeleton and creating a molecule with serious staying power. As a white, crystalline powder with a faint chemical odor, octachlorocamphene does not dissolve easily in water. Instead, it lingers in soil, clings to organic matter, and doesn’t break down in ordinary weather or water. In real-world use, that makes disposal and residue problems a headache. Historical records suggest it was sometimes hard to tell octachlorocamphene from its close relatives—chlorinated camphenes often go by trade names, and purity varied from batch to batch. Analysts spent years wrestling with chromatographic separation and characterization, learning how to fingerprint the exact makeup of each sample.
The Technical Landscape: Preparation, Reactions, and Labeling Insights
Making octachlorocamphene draws on a simple recipe: camphene meets chlorine gas with the right catalyst, producing a crowded molecule bristling with chlorine. Technicians found that process conditions heavily influenced the exact profile of the final product. Ratios, temperature, and reaction time shaped everything—one misstep and you’d get mixtures with unexpected impurities or degraded quality, leading to inconsistent performance. Chemists noticed that octachlorocamphene could swap halogens in certain reactions, allowing for chemical tailoring. Modifications gave birth to derivatives with subtle shifts in behavior, but also kicked off debates over environmental persistence. From a labeling standpoint, early containers displayed hazard warnings later supplanted by regulatory diktats mandating poison symbols and precise chemical content. Newer standards reflect decades of learning about storage and accidental release risks, influenced by industry accidents and soil contamination episodes that left communities demanding stricter oversight.
Names in the Field: Synonyms and Aliases
Few chemicals collect aliases like this one. Octachlorocamphene travels under a wild batch of synonyms. Old trade names, like Toxaphene, conjure up images of the old dusters and sprayers in southern U.S. fields. Sometimes labels read “chlorinated camphene,” though this can create confusion since it covers a range of products with variable chlorine load. I’ve even run into older publications that jumble it among camphene chlorinated pesticides in general, so tracking the literature gets tricky. Such naming confusions signal the way pesticide regulation and marketing danced out of step with real-world chemistry for years, leaving agricultural workers and local governments to sort out the aftermath long after the industry had shifted priorities.
Staying Safe: Handling, Standards, and Human Hazards
Any commercial handler quickly realizes the importance of strict safety routines. Handling octachlorocamphene calls for gloves, eye protection, and a well-ventilated workspace because dust and particles can aggravate skin and eyes, while inhalation raises bigger concerns. There’s a reason governments called for locked chemical storage and mandatory signage as evidence mounted about long-term persistence and ecosystem effects. I’ve seen communities downwind from old mixing plants actually carry that caution into daily life years after facilities shut down. Air and water monitoring regulations got stronger, driven by research linking residues to animal deaths and, in some places, human illness. From my experience listening to rural extension agents, trust in official assurances still runs low, and for good reason: it takes only a few mishaps for whole neighborhoods to reevaluate their relationship with chemical interventions. Over decades, international agencies stepped in to issue stricter guidelines—including outright bans—citing evidence of bioaccumulation in food chains and open questions about endocrine disruption and carcinogenicity.
Where It Got Used: Agriculture, Vector Control, and Beyond
Farmers desperate for less crop damage and fewer livestock losses leaned heavily on octachlorocamphene. Cotton, corn, and a swath of fruit and vegetable fields routinely saw applications, especially across the southern United States and parts of the former Soviet Union. Mosquito abatement programs mixed it into their fight against malaria and yellow fever, banking on its knockdown effect and long-lasting action. Fisheries and aquaculture shied away out of concern for waterborne accumulation, but less-regulated settings saw mixing right up to waters’ edge. Forestry managers sometimes used it to stave off beetle outbreaks. As the risks came into focus—the chemical’s heavy hand tipping the balance of aquatic and soil life—regulatory clampdown erased most legal markets in North America and Europe, although environmental justice debates still follow those old application zones.
Inside the Lab: Research, Toxicity, and the Chase for Safer Alternatives
For decades, toxicologists chased down case after case of environmental and human exposure. Studies showed that octachlorocamphene moved up the food chain, clustering in animal fat where it persisted for years. Laboratory mice and aquatic test organisms developed a slew of chronic effects: liver tumors, reproductive issues, and immune suppression. In communities near use sites, researchers reported higher rates of certain cancers and neurological complaints, though proof of causality stayed elusive in many cases. Regulators responded, sometimes lagging behind the latest findings, restricting application rates and mandating clean-up. I worked with college students who combed through waterway sediment cores—every layer a silent record of decades-old use—documenting contamination that outlasted the initial chemical’s economic value by generations. Seeing those data up close, it’s tough to overstate the challenge: the very persistence that made this chemical attractive to farmers made it a multi-decade headache for the next generation of scientists and policymakers.
What Tomorrow Brings: Prospects and Responsible Innovation
Modern crop science stands at a crossroad. No large-scale future remains for octachlorocamphene itself as environmental and health risks outweigh the benefits when safer, targeted pest management tools exist. Yet the legacy of its chemistry still shapes how new products get evaluated. Regulatory agencies watch for environmental build-up and long half-lives now, asking for rigorous pre-market testing and transparency. Research shifted toward biodegradable molecules, biological controls, and integrated pest management that values ecosystem health as much as pest suppression. Communities living with the aftermath of older chemical regimes push for cleanup funds, better site monitoring, and education about lingering risks—which signals a new kind of E-E-A-T in science: experts not just in labs, but in affected neighborhoods, working to restore trust and capacity. As a writer reflecting on these cycles, I see lessons not just for chemists, but for any profession trying to balance innovation and responsibility in a complicated world.
What is Octachlorocamphene used for?
Digging into Its Main Purpose
Octachlorocamphene, often grouped with other chemicals called organochlorine compounds, has served a pretty specific niche in the world of pest control. Older generations who worked on farms or around public health pest mitigation probably remember its role as an active ingredient in some insecticides. These chemical tools found their prime back in the mid-20th century, tackling everything from crop pests to malaria-carrying mosquitoes. Many houses and storage sites, especially in tropical zones, saw widespread spraying across ceilings and dark corners.
Coming from a chemistry background, the use of chemicals like this always feels like living in a double-edged sword scenario. Science delivered a solution to pest outbreaks, especially where food security hung by a thread. At the same time, the comfort of quick victories brought on bigger issues—land, water, and people absorbed the cost.
Health and Environmental Red Flags
Growing up with tales about the “miracle chemicals” that cleared out insects, I’ve seen how those stories shifted as research rolled in. Extended exposure to these organochlorines has raised alarms for decades. They tend to linger for years, both in the ground and in living tissue, and the body’s not great at breaking them down. There’s clear scientific consensus about risks—neurotoxicity, hormone disruption, even cancer connections surface in long-term studies.
Wildlife never asked for a chemical bath, but they got one anyway. The thing is, octachlorocamphene doesn’t stay put. It drifts in wind and water, and then it accumulates up the food chain. Bird eggshell thinning, fish kills, and massive drops in beneficial insects tell a pretty clear story. Neighbors downstream never consented but ended up with residues just the same.
Cracking Down and Moving Forward
Governments picked up on the risks after enough warning signs. International treaties like the Stockholm Convention pushed for bans and tight regulation of persistent organic pollutants, including octachlorocamphene. Trade declined, with most countries switching to other pest control options. Newer products on supermarket shelves rely on compounds that break down faster and target bugs more specifically.
Plenty of people grew distrustful. The shift didn’t happen overnight. Some regions still have stashes of octachlorocamphene, either forgotten or hoarded as a “last resort.” Old habits take time to replace, but the science makes the need obvious: there’s no free lunch when dumping chemicals into shared environments.
Better Options Take Root
Safer ways to handle pests have sprouted up as farmers try to avoid repeating past mistakes. Mixing up crop planting, reintroducing natural predators like ladybugs, or relying on biological controls brings a lighter touch. For those who grew up in farming families, those methods don’t sound exotic—they sound like old wisdom that’s made its way back into the light. Chemical use should be the tool of last resort, not the first knee-jerk reaction.
Targeted education, tight tracking of chemical sales, and transparent reporting help keep harmful compounds on the sidelines. The more people push for cleaner produce and safer ecosystems, the harder it gets for heavy-duty chemicals to sneak back into use.
Looking at octachlorocamphene’s history, it’s clear the real value comes from learning, not forgetting. Putting health and environmental impact first lets everyone stay safer while keeping our crops and communities thriving.
Is Octachlorocamphene toxic or hazardous?
Understanding Octachlorocamphene
Octachlorocamphene belongs to that family of industrial chemicals that crop up in places people don’t always expect. It’s had its moments as a pesticide, a role that often signals a need for caution. Anyone who’s worked around older warehouses, farms, or even some municipal projects knows these compounds don’t just fade away quietly.
Health Risks Tied to Chlorinated Compounds
From years spent volunteering with environmental groups, patterns become obvious: chlorinated chemicals tend to raise red flags for health. Octachlorocamphene fits the bill. Studies have shown chlorinated camphors carry risks like skin and eye irritation — not just for workers. Once these chemicals move through soil and water, the exposure risk can jump from factories to neighborhoods.
Scientists point to animal research where even low doses bring about problems, mainly with livers and kidneys. There are reports of breathing dusts or vapors causing headaches or dizziness. Nobody wants those after a shift. The thing about chlorinated pesticides is their tendency to stick around. Breakdowns are slow, which brings a chance for bioaccumulation up the food chain, including fish and crops.
Environmental Hazards
Having seen how runoff behaves after a heavy summer rain, it gets clearer how such chemicals travel. They move from field edges into nearby streams and even affect distant groundwater. Over time, persisting contaminants like octachlorocamphene can disrupt aquatic species, leading to loss in fish stocks and subtle shifts in local biodiversity. The United Nations Environment Programme flagged many chlorinated pesticides as persistent organic pollutants. International bans haven’t always stopped their use everywhere. Old stocks remain in circulation, and disposal sometimes lands these compounds in poor communities.
Regulatory and Workplace Realities
The Occupational Safety and Health Administration recommends strict handling rules for anyone near such chemicals. Still, in real-life shops, gloves and masks sometimes get skipped under pressure to meet deadlines. Small manufacturers, especially outside big cities, often lag in upgrading gear or training. Community activism helps some, but regulation only works with real enforcement.
If a chemical can travel and linger, the risk grows past workers to children, pets, and neighborhoods. Medical science keeps finding more evidence connecting long-lived pesticides and chronic health problems. The EPA updates their guidance all the time, but the science often lags behind what daily exposure looks like.
Seeking Safer Practices
Companies must improve labeling and tracking, so nobody’s left guessing what’s in the drum. Governments ought to provide more accessible hazardous waste pickup—visited a town in the south last year that had community collections, which cut dumped barrels by a huge margin. For farmers and industrial users, moving to newer, less persistent alternatives sounds simple, but change costs money. Better public incentives and education campaigns go a long way for convincing smaller operations.
Teachers, nurses, and local leaders need to know what dangers lurk in forgotten sheds or empty lots. About five years ago, a schoolyard cleanup revealed dozens of overlooked pesticide containers. That experience proved the value in routine checks and community involvement.
Keeping Eyes Open
Staying aware matters most. Don’t let slick technical labels or faded packaging mislead you. Octachlorocamphene carries risks that trail behind its production. Real progress comes from pairing tough policy with the everyday vigilance of families, workers, and local groups. If you see old chemicals collecting dust, ask about proper disposal. Sometimes the difference between harm and safety is a call to a local waste service or a heads-up at a town meeting.
What is the chemical formula of Octachlorocamphene?
Understanding Octachlorocamphene
Octachlorocamphene doesn’t pop up in conversation, but in the field of industrial chemistry, it’s more than just a mouthful. Its chemical formula, C10H10Cl8, speaks volumes to people who work with pesticides, environmental regulations, and chemical manufacturing. If you’ve spent any time near citrus groves or old agricultural reports, you might have run across its other name: toxaphene. Decades ago, farmers turned to compounds like these to fight pests and save crops. The idea seemed pretty straightforward—protect the food, support the farm, feed the world.
Breaking Down the Formula: C10H10Cl8
This formula shows you get ten carbon atoms, ten hydrogens, and eight chlorine atoms fused together in this molecule. The heavy load of chlorine gives octachlorocamphene its pest-killing power, but it also makes the compound persistent and tricky once released into the air or water. Years ago, I watched community meetings about groundwater safety—chemicals like this always draw questions about health and legacy pollution. Even if you’ve never seen a lab up close, hearing concerns from families about water safety brings home how formulas on a page turn into real issues.
Health Impacts and Environmental Concerns
Research links chlorinated pesticides to health risks, especially with long-term exposure. The United States Environmental Protection Agency and international groups flagged toxaphene compounds for their ability to hang around in the environment. They move through soil and water, and they eventually enter the food chain. Eat enough fish from contaminated water, and toxic effects add up. Stories from fishing communities on the Great Lakes painted a vivid picture: the chemical formula affects lives for generations after the spraying stops.
Balancing Benefits and Risks
Chemicals like octachlorocamphene made farming more reliable for a time, but there’s a bill to pay. The formula brings upfront benefits for yield and crop value. Over years, though, people noticed that boosted production came with pollution that doesn’t break down easily. Toxaphene and similar chlorinated organics get labeled as persistent organic pollutants, traveling around the globe. I remember reading about Arctic wildlife testing positive for compounds made thousands of miles away—chemistry’s reach often surprises people.
Searching for Smarter Solutions
No single law or restriction deals with this for good. It takes smarter chemistry, better monitoring, and support for farmers to transition to safer options. Companies developing alternatives look at formulas that degrade naturally and don’t build up in humans or animals. Universities and government labs now focus less on brute chemical force and more on integrated pest management—mixing biology, chemistry, and technology to minimize harm.
Moving Forward
Octachlorocamphene’s formula isn’t just academic detail. It reflects a moment in history and the lessons learned about weighing immediate needs against lasting impacts. For the students and scientists tackling tomorrow’s pest control, understanding how formulas like C10H10Cl8 shaped our environment can steer new efforts toward something cleaner and safer.
How should Octachlorocamphene be stored and handled?
What Octachlorocamphene Demands from Us
Most people out there haven’t heard of octachlorocamphene until it lands on a Material Safety Data Sheet or shows up in a niche lab project. Its heavy chlorine load gives it staying power. That stickiness means trouble if folks don’t treat it with respect. Growing up around great science teachers, I saw how cavalier handling of solvents and chemicals left marks on benches and, sometimes, lungs. No matter the pedigree or intent, octachlorocamphene shares the rules that all persistent organochlorines expect: airtight containers, chemically resistant gloves, and genuine accountability.
Storage: Out of Sight, Never Out of Mind
Octachlorocamphene likes cool, dry spaces. I’ve seen labs turn a back closet into a dumping ground, but smart teams use ventilated chemical storage cabinets with clear hazard labeling. Any place storing organochlorines benefits from solid spill trays and secondary containment. This isn’t about fear-mongering. Chlorinated compounds, by reputation and by accident, have played into contamination scares before, from abandoned drums in warehouses to leaky shelves that nobody checked for months. Keeping lids tight and locking doors doesn’t just meet some line in a binder—it keeps everybody else in that building out of the hospital.
Personal Protection Isn’t Optional
Sloppy handling of octachlorocamphene has consequences. Run-of-the-mill lab gloves sometimes fail, especially with solvents or extended contact. Butyl gloves or thick nitrile gloves offer better protection. Goggles, not just glasses, and long sleeves block contact if a spill splashes. During my time shadowing industrial hygienists, cheaping out on face protection never paid off. Skin rashes and odd smells that lingered after the shift served as the early warning signs everyone wished they’d noticed sooner.
Good Ventilation Stops Problems Before They Start
Odorless, invisible fumes don’t let you know something’s wrong until you cough or your head aches. Fume hoods—scrubbed and with new filters—make a lab safer. Airflow isn’t cosmetic; it’s essential. Anyone decanting octachlorocamphene needs those fans on and sashes down. In old university labs, people would try to get away with a cracked window. Proper ducted hoods beat improvisation every time. Beyond that, spill kits belong nearby, with absorbent pads and chemical neutralizers that match octachlorocamphene’s profile. Plain old paper towels only make things worse.
Training Solves a Lot of Dumb Mistakes
The best rule I picked up early in my lab time: don't handle what you don’t fully understand. Interactive safety drills—fire, eye-wash, chemical splash—always leave more of an impression than dry lectures. Workers should recognize the SDS icons on containers, and know exactly what to do if exposure occurs. Emergency showers and eyewash stations need regular checks and easy access. No one appreciates these stations until a split-second choice saves skin or vision.
Waste: What Leaves the Room Matters
Octachlorocamphene won’t just disappear down the drain without consequences. Disposal through licensed hazardous waste handlers keeps it out of water supplies and landfill. A lot of small labs treat waste as afterthoughts, but trace contamination adds up—just one leak can cause headaches for years. Records for all waste transfers matter, not just for regulations, but as proof that the chain of custody doesn’t break.
What Helps: Practical Solutions That Stick
Safe storage starts with clear signage and the right containers, and keeps going with routine inventory checks. Good ventilation trumps shortcuts. Check gear twice before each use, hold daily toolbox talks, and never fudge labels for “just this once.” Investing time in habits, not just equipment, delivers real protection. In a world full of shortcuts, doing things right still keeps people healthy, compliance officers happy, and labs running smoothly.
What are the safety precautions when working with Octachlorocamphene?
Understanding What You’re Handling
Octachlorocamphene turns up in some corners of the chemical world where pest control or industrial chemistry calls for a heavy-duty answer. It looks like a harmless white powder at first glance, but the hazards run deeper than its appearance suggests. Inhalation, skin contact, or swallowing even a small amount can stir up real trouble. Headaches, nausea, and irritation happen fast. Chronic exposure means long-term health effects. That’s not just based on the safety data sheets—longtime workers in the field have seen colleagues develop respiratory or liver problems after careless stints around this stuff.
Personal Protective Equipment: Non-Negotiable
Nobody should skim on PPE. Standard lab coats and nitrile gloves are the starting line. Splash goggles seal out dust and fumes, and a proper respirator does a better job than any dust mask. Experience working around volatile compounds tells me: once you let your guard down, accidents repeat themselves. The EPA keeps strict lists for a reason. Over time, companies that require regular fit-tests and routine replacement of gloves and masks see fewer incidents. Good employers invest in these because the cost of treating a chemical injury is steeper than the bill for safety gear.
Good Ventilation Matters
Open-air workstations pull triple duty—protecting your lungs, avoiding flammable vapor buildup, and cutting down on cross-contamination. I’ve learned that a simple exhaust fan won’t cut it. Fume hoods with active airflow, regularly inspected filters, and airflow monitors help. Colleagues at plants with dedicated HVAC controls talk about the difference one system upgrade makes: fewer lingering smells, clear air, and reduced sick days. Lab managers who push for modern ventilation upgrades protect their teams and their bottom line.
Safe Storage Prevents Accidents
Shelving lined with chemical-resistant trays. Clear labels with hazard icons. No stashing containers next to food or drink—no exceptions. Many incidents trace back to lazy storage. One misplaced jar mixed with organic material has set off fires. I once saw a minor spill inside a locked cabinet spark a cleanup day for an entire wing. Store substances like this in sealed, dedicated cupboards away from heat. Locking the area adds peace of mind in busy labs where non-authorized hands sometimes wander in.
Spill Response: Be Ready Every Day
Even with sharp awareness and layers of protection, spills happen. Training makes a difference: teams who drill their response steps don’t freeze when a jar tips. Rapid access to absorbent materials, goggles, and extra gloves cuts risk. Strict rules—such as evacuating non-responders—keep confusion outside the danger zone. I’ve watched well-drilled teams contain and decontaminate with confidence because the training wasn’t a checkbox but a real part of the job.
Health Monitoring and Reporting
Regular screening for chemical exposure forms never go to waste. Clinics catch elevated liver enzymes or respiratory symptoms early, saving people from irreversible damage. Open reporting channels encourage workers to spot weak points: blowing fuses in ventilators, worn-out gloves, unmarked containers. A solid safety culture builds from this kind of honest feedback, not from punitive crackdowns. Leaders who listen to feedback and act quickly set up everyone for long careers instead of long recoveries.
Looking Ahead
Staying safe doesn’t end with written guidelines. The workers who treat every shift as a personal investment—checking their own gear, looking out for each other, pushing for safer conditions—end up writing the best stories of long-term health in high-risk jobs. No shortcuts. That simple truth keeps families, colleagues, and companies whole.
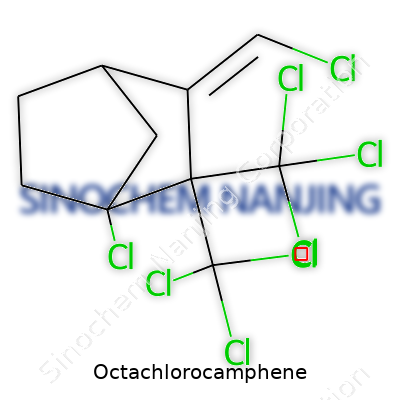
| Names | |
| Preferred IUPAC name | 1,2,3,4,5,6,7,7-Octachlorobicyclo[2.2.1]heptane |
| Other names |
Toxaphene Camphechlor |
| Pronunciation | /ˌɒk.təˌklɔːr.əˈkæm.fiːn/ |
| Identifiers | |
| CAS Number | 17066-46-5 |
| Beilstein Reference | 1755206 |
| ChEBI | CHEBI:34828 |
| ChEMBL | CHEMBL2106611 |
| ChemSpider | 21803997 |
| DrugBank | DB13630 |
| ECHA InfoCard | 200-066-5 |
| EC Number | 206-098-3 |
| Gmelin Reference | 6768 |
| KEGG | C19275 |
| MeSH | D010020 |
| PubChem CID | 96239 |
| RTECS number | GL6180000 |
| UNII | U11W6386P4 |
| UN number | UN2761 |
| Properties | |
| Chemical formula | C10Cl8 |
| Molar mass | 402.77 g/mol |
| Appearance | White crystalline solid |
| Odor | Characteristic odor |
| Density | 1.70 g/cm³ |
| Solubility in water | Insoluble |
| log P | 5.9 |
| Vapor pressure | 1.5 × 10⁻⁷ mmHg (25°C) |
| Magnetic susceptibility (χ) | -0.000105 |
| Refractive index (nD) | 1.617 |
| Viscosity | Viscous liquid |
| Dipole moment | 1.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 763.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -27.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3595 kJ/mol |
| Pharmacology | |
| ATC code | Pesticides |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause respiratory irritation, harmful to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P264, P270, P271, P273, P301+P312, P304+P340, P308+P311, P330, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-0 |
| Flash point | Flash point: >100°C |
| Autoignition temperature | 539°C |
| Lethal dose or concentration | Lethal dose or concentration: **LD50 oral rat 1500 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 3200 mg/kg (rat, oral) |
| NIOSH | SY5950000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
Hexachlorocyclopentadiene Chlornaphthalene Endosulfan Mirex |

