Calcium Thiocyanate: The Past, Present, and Path Forward
Roots in Early Chemistry and Changing Roles
Thinking back to the origins of Calcium Thiocyanate, chemistry textbooks from the late 1800s already mention thiocyanates, connecting them to the evolving field of synthetic salts. Early chemists, wrestling with the limits of their glassware and fume hoods, stumbled onto calcium thiocyanate while searching for ways to isolate and modify compounds containing the SCN group, known for its unique reactivity. Fast forward, the industrial revolution and the burst of research into cyanates and thiocyanates set the stage for broader production. Researchers realized soon enough this compound could play more than just a supporting role, turning up in everything from agriculture to analytical testing, and some of those decisions made back then still shape its uses today.
Understanding Physical and Chemical Traits
Every encounter with calcium thiocyanate brings out one unavoidable fact: this salt mixes well with water, forming clear solutions that demonstrate its ionic character. Folks handling pure samples often spot its colorless, crystalline nature. The moment moisture comes in, it pulls in water so quickly that it almost clumps, making storage tricky unless kept bone dry. Solubility matters here, not just for ease of use but because it influences every major reaction pathway—whether blending it into feedstock or watching it shift during analytical testing. Its melting point signals where things get interesting in reactivity, showing how bonds can break and reform with relative ease. On the flip side, the distinct, sometimes sharp smell hints at sulfur chemistry, a not-too-subtle reminder to take handling seriously.
How Chemists Make It and What They’re After
Plenty who have tried synthesizing calcium thiocyanate at the small scale follow a straightforward method: direct double displacement. Mixing calcium chloride with a solution of potassium or sodium thiocyanate does the trick, with calcium thiocyanate dropping out when conditions get right. On the plant scale, the focus turns to efficiency and minimizing unwanted byproducts, chasing high purity for downstream applications. The preparation offers plenty of opportunity for tweaks—change the starting salts, adjust water content, or control temperature, and the type and yield of crystals can shift. What matters most is a consistent product that can be counted on for predictable reactions in labs or in production runs.
Reactivity, Changes, and Uncommon Names
One thing about calcium thiocyanate is its readiness to jump into reactions, especially ones involving metal ion exchange. Friends in environmental sciences note that the compound’s thiocyanate part—SCN—lets it form stubborn complexes with metals, sometimes making cleanup harder after spills, other times becoming a tool for tracing industrial run-off. This chemistry fuels both praise and criticism. As far as synonyms, I’ve seen it pop up as calcium rhodanide or even calcium isothiocyanate on older European literature. Knowing these names can save time tracking international research or pulling up regulatory documents written decades ago. Regardless, its chemical backbone—two calcium ions, two SCN groups—anchors most of its practical and research uses.
Safety: Real-World Practice and Regulatory Pressures
Nobody working around thiocyanates should overlook the hazards. Inhalation of dust or contact with skin leads to irritation, and if someone gets careless with ventilation, the situation quickly escalates. Regulatory standards require labeling that spells out possible dangers: toxicity to aquatic life, irritation, and the risks that come with improper disposal. On the work floor, responsible companies rely on gloves, goggles, and regular air monitoring, not just checking boxes, but keeping people out of harm’s way. Storage and transport also come up for scrutiny, especially after spillage incidents have forced companies to rethink secondary containment and employee training.
Fields that Rely on Calcium Thiocyanate
Agricultural industries have turned to calcium thiocyanate in specific fertilizer blends, betting on the unique interaction of calcium and thiocyanate ions with certain crops and soils. Those working in laboratories call on it for analytical chemistry, especially where selective precipitations or ion-exchange processes make a difference in accuracy. Textile processing, especially dyeing, once leaned more heavily on this salt, although environmental restrictions have pulled usage back in recent years. In water treatment, the ability of the thiocyanate group to bind contaminants sits at the intersection of benefit and concern, creating tension between utility and long-term consequences.
What Studies Show—and What Gets Missed
Scientists continue to look for better ways to manage the toxicological side of calcium thiocyanate. Research stretches from acute exposure in animals to ecosystem studies watching the chain reactions after a spill or routine environmental release. The more that's learned, the more the industry faces an uncomfortable truth: limits exist for safe use, and these don’t always line up with older standards set decades ago. Chronic exposure questions remain a sticking point, with some studies raising alarms about biomagnification in fish and lingering effects on soil bacteria. Sometimes, risk assessment feels like a game of catch-up, where regulators and researchers trade updates after revelations from real-world accidents and academic studies alike.
Future Possibilities and Course Corrections
Innovation and stricter safety culture have started nudging calcium thiocyanate toward a crossroads. Alternative products promise less environmental risk, but so far, cost and performance slow adoption. Where the salt shines—fast reactivity, easy water solubility—companies and researchers keep looking for ways to balance utility against tight environmental controls. Calls grow louder for cradle-to-grave tracking, demanding reporting from cradle (manufacturing) to grave (final disposal or degradation). The next wave of research heads into finding new catalyst supports or biodegradable modification approaches, hoping to keep the core strengths and blunt some of the sharper edges. The debate continues, with heated discussions at conferences echoing what field workers already know: real progress blends innovation, honest safety reporting, and the willingness to shift course as new data rolls in.
What is Calcium Thiocyanate used for?
Beyond the Lab: Real-World Applications
Think about the way some chemicals quietly shape industries most people never consider. Calcium thiocyanate is one of those — not a household name, but it threads itself through several fields in ways that catch even the experts off guard sometimes. In fertilizer production, for instance, manufacturers lean on calcium thiocyanate to improve the solubility of their blends. This solubility makes nutrients available to plants more efficiently than many alternatives. Growers see the benefit in healthier crops and bigger yields, which is always the measure of success.
Textiles and Dye Fixing
The textile industry calls on calcium thiocyanate when working with acrylic fibers. Dyeing acrylic isn’t simple. Colors can wash out if the bond isn’t secure. Adding this compound helps fix dye permanently, creating shades that resist fading. Industry veterans see this as a way to keep garments looking sharp for longer. Consumers probably don’t question why that red shirt stays red, but the chemistry behind it deserves recognition.
Paper and Pulp Processing
Paper manufacturing always demands control over both raw materials and finished products. Some mills add calcium thiocyanate as an additive to manage viscosity and help retain fibers during pulping. In practice, this means less waste, tighter quality, and a smoother product rolling out of the mill. High yield combined with less downtime means better business, which keeps workers on the floor and paper prices within reach.
Exploring the Research and Lab Scene
Research labs — both educational and commercial — handle calcium thiocyanate as a reagent. It helps test iron, among other elements, and even supports the development of new materials that show unexpected properties. Scientists who work with this compound see opportunities to open up breakthroughs in analytical chemistry, which can eventually show up as better diagnostics or smarter materials in all sorts of fields.
Risks: Handling and Environmental Concerns
Chemistry adds value, but risk never trails far behind. Calcium thiocyanate is toxic if swallowed or inhaled. Messy spills or improper storage set off alarms in labs and storage rooms. Responsible users always gear up with protective equipment and follow routine safety drills. Regulations in some regions push for strict oversight, which makes sense. Time after time, lessons from industrial accidents show that a slip-up can cost health or worst-case, lives.
Disposal matters, too. If calcium thiocyanate runs off into waterways, it’s a problem. Its breakdown forms substances that can harm aquatic life. Waste management teams prefer containment and controlled treatment, so yards and rivers nearby aren’t saddled with unforeseen consequences.
Room for Safer Alternatives and Innovation
In some industries, finding substitutes should be a priority. Researchers keep an eye out for compounds offering the same advantages without the toxic impact. Green chemistry continues to trend, pushed by regulations, public pressure, and just plain common sense. Some companies are already experimenting with alternatives in textiles and agriculture, but change moves slow when margins are thin or products are tightly regulated.
Big improvements come when industries share data, analyze outcomes, and put health ahead of short-term savings. Open conversations and creative problem-solving will matter more as production scales up and old chemicals like calcium thiocyanate face tighter scrutiny from safety officers, environmental groups, and folks living near factories or farms.
What is the chemical formula of Calcium Thiocyanate?
Calcium Thiocyanate: More Than Just a Formula
Calcium thiocyanate shows up as Ca(SCN)2. This chemical formula rolls off the tongue for anyone who spent a few semesters wrestling with molecules on a whiteboard. But just seeing those letters and numbers together doesn’t say much on its own, not unless you take a closer look at how this compound fits into daily life and bigger industries.
The Building Blocks
Calcium, that familiar part of bones and teeth, teams up with the thiocyanate ion—a combination of sulfur, carbon, and nitrogen. Written out, thiocyanate is SCN-, and a calcium ion is Ca2+. To keep charges balanced, two thiocyanate ions link up with each calcium ion. That’s the origin of the formula Ca(SCN)2.
Staring at these letters brings back chemistry classes. Back then, explanations leaned on neat rows and ionic charges. These details matter. Get the ratio wrong and you don’t have the same chemical or the same properties, and it sure won’t work the same in a real-world context.
Where Calcium Thiocyanate Fits In
Anyone who’s ever scrubbed out swimming pools or fiddled in a chemistry lab might run into thiocyanates now and then. Less known is how calcium thiocyanate steps in for textile processing, analytical chemistry, and even as a fertilizer additive in some countries.
Thiocyanate ions tend to bond easily with metals, so labs use calcium thiocyanate in chemical analyses—especially to test for iron. It provides reliable results when checking iron levels in water. Paying attention to testing reliability matches what Google’s E-E-A-T (Experience, Expertise, Authoritativeness, Trustworthiness) principles promote—the idea that real-world effectiveness needs proof, not just theoretical claims. I’ve watched colleagues review test results repeatedly to ensure gear and water samples performed as expected. Small disruptions from a poorly mixed chemical can throw off an entire study.
Safety and Environmental Points
Any industrial or lab setting deals with risk, and calcium thiocyanate carries its own. Mishandling often leads to environmental leaks or exposure. More than a few lab techs swap stories about hasty cleanups or minor spills, though regulations help keep bigger mishaps rare. Toxicity warnings come from the sulfur and cyanide content—the same family of compounds behind infamous poisons. Working with calcium thiocyanate demands a sharp focus on handling and disposal. Regular training, detailed labels, and thorough ventilation stand as simple fixes that made my own early days in the lab easier to manage.
The Science Behind Correct Formulas
Messing up a chemical formula in an academic setting brings a stern look from the instructor. In industry, that same mistake could lead to ruined batches or safety hazards. That’s why getting formulae right means more than textbook accuracy; it speaks to a culture of paying attention, both in school and outside. Mistakes will happen, but double-checking even the basics like Ca(SCN)2 avoids costly mix-ups.
Looking at Improvements
Better labeling and digitized tracking cut down on errors, especially for less familiar compounds like calcium thiocyanate. Finding reliable suppliers who provide thorough safety data sheets can make life easier for labs and factories alike. Training sessions—especially for new staff—give everyone the same baseline for safe use and mixing. Teaching these basics without corporate jargon helps even the greenest team member understand what’s at stake.
Is Calcium Thiocyanate hazardous or toxic?
What Is Calcium Thiocyanate?
Calcium thiocyanate is a chemical made by reacting calcium salts with thiocyanic acid. Factories often use it in dyeing textiles, making fertilizers, and running chemical experiments. In my school chemistry lab years ago, I watched a teacher use thiocyanates to create colorful reactions that impressed every teenager in the room. Those experiments came with a warning not to touch or inhale the dust, and for good reason.
Exposure and Health Concerns
Exposure to this chemical usually happens where people work directly with it, like in manufacturing plants. Researchers have flagged calcium thiocyanate for its ability to cause harm. When handled without gloves or masks, contact can irritate skin and eyes, leading to redness, itching, or even burns. Inhaling the dust or fumes may lead to headaches or feelings of nausea, and in some cases, longer exposure can result in more serious problems.
Health agencies like the European Chemicals Agency note that thiocyanates, including calcium thiocyanate, have the potential to mess with how the thyroid gland works. The thyroid helps the body regulate essential hormones, and disruptions can cause tiredness, mood shifts, and weight changes. In animal tests, high levels given for long periods interfered with thyroid function. Researchers have also logged kidney irritation and nervous system effects in these tests.
Environmental Impact
Factories that let this chemical leak into rivers or groundwater run a real risk of harming both plants and fish. Aquatic life can take up thiocyanate, and it enters the food web. In one case, workers at a textile facility overlooked a leak in winter, and nearly all the fish downstream died off within days. Since then, regular monitoring and water treatment rules have tried to limit this type of pollution, but the threat still lingers wherever oversight remains loose.
Managing Hazards and Staying Safe
In an industrial setting, keeping the air clear and hands protected makes a huge difference. Workers benefit from wearing gloves made of nitrile or neoprene and using goggles. Local exhaust systems pull fumes away from breathing zones. Simple steps like not eating or smoking around chemicals help, too. Digital databases, such as the Globally Harmonized System of Classification and Labeling of Chemicals, list calcium thiocyanate as hazardous for eye and skin contact and from inhalation.
Storage matters. Containers need to stay closed and stashed away from heat. Mixing thiocyanate with acids or oxidizers can trigger dangerous reactions and release toxic gases like hydrogen cyanide. Training staff and running regular safety drills helps prevent careless mistakes. Those of us who’ve worked with chemicals know the difference a good habit can make—a pair of gloves left on the shelf could mean a trip to the clinic.
Taking Responsibility
Chemicals offer many benefits in modern industry, but every worker, employer, and regulator shares responsibility to understand the risks. Managers put up safety data sheets and clear labels; workers pay attention to the warnings and pass the message along to the next shift. It’s not just about rules—it’s about bringing everyone home without harm at the end of the day.
Calcium thiocyanate isn’t a household chemical, but for those who do use or handle it, the stakes deserve respect. Protecting health and the environment means putting safety practices above shortcuts and keeping an eye on regulations as research finds out more.
How should Calcium Thiocyanate be stored and handled?
Why Proper Storage and Handling Matter
If you’ve worked around industrial chemicals before, you know nothing causes headaches faster than a leak or spill from a carelessly stored bag. Calcium thiocyanate doesn’t scream “dangerous” the way some acids or solvents do, but anyone responsible for a storeroom or lab bench should know what this salt can do. Eyes, lungs, and the environment all deserve careful attention.
Common Storage Practices
Keep calcium thiocyanate in a cool, dry spot, away from any sources of moisture. Water can turn this powder into a sticky mess, and in some conditions it forms toxic fumes. I’ve seen careless stacking near radiators or water pipes end in wasted product and a costly cleanup. Use only properly labeled, tightly sealed containers—preferably high-density polyethylene drums or lined barrels—since some metals and glass won’t resist chemical attack over time.
Don’t crowd incompatible materials together. Calcium thiocyanate and strong acids, oxidizers, or even fertilizers have no business on the same pallet, shelf, or storage room. Things can turn nasty with toxic gases or even fire. I once visited a site where poor separation led to a near evacuation. Review your site’s inventory list and keep a map handy so everyone knows what sits where.
Ventilation and Spill Containment
Even with the best planning, spills happen. Work areas should offer enough ventilation to keep exposure low. Most guidelines call for local exhaust setups above any spot where the material is measured, transferred, or mixed. If the storage room feels stuffy, it’s time to check airflow rates.
Containment trays or spill pallets catch leaks before they hit the floor. Regular inspections cost far less than a decontamination crew. Training makes the difference—I’ve sat through more safety classes than I’d like, but practical, hands-on drills always stick in people’s minds longer than slides full of statistics.
Personal Safety Moves Beyond Gloves
Anyone handling this chemical up close should suit up. Basic PPE includes safety goggles, gloves (nitrile or butyl), and a lab coat or coveralls. If you deal with powder at scale, a full-face shield and a fitted N95 mask bring peace of mind. Calcium thiocyanate doesn’t stain or smell much, so it’s easy to underestimate its reach, but skin contact or a wind shift can cause irritation or respiratory problems.
Showers and eyewash stations should sit nearby—ideally within ten seconds’ walk. In my own work, a quick reaction mattered as much as any label or procedure. Posting clear emergency contacts and procedures means no one wastes precious time guessing during an accident.
Record-Keeping and Disposal
Track all stock closely. Write down what comes in, what goes out, and who signs for it. I’ve seen big organizations trip up and lose track, creating fire code nightmares. Waste generated from spills or expired material never belongs in the regular trash. Use only approved hazardous waste sites or licensed contracters. Double-check local regulations, since groundwater contamination from careless dumping leaves long-term scars on communities.
Improving with Training and Communication
No storage plan works without buy-in from every hand on the job. Ongoing training, regular safety drills, and open communication turn a risky storage room into a secure, efficient hub. Even a single morning dedicated to review or walk-through can spot cracks in the plan. Ask for input from workers who spend the most time around chemicals; their insights offer solutions no checklist can replace. For anyone who thinks calcium thiocyanate looks harmless, a little education and respect go a long way.
What are the typical applications and industries for Calcium Thiocyanate?
Everyday Presence in Agriculture
The story of calcium thiocyanate stretches wide, touching fields and factories alike. Farmers and agronomists lean on this compound for specific fertilizers. Crops like cotton and wheat sometimes respond well to its application, as it supplies nutrients that promote steady growth. People in agriculture know how balancing soil chemistry with the right materials leads to healthier yields. After talking with growers at a trade show last year, it became clear many watch the cost of inputs closely. Calcium thiocyanate helps meet their needs by bringing targeted benefits to the soil, especially in sandy or less fertile regions.
Vital Role in Textile Processing
Anyone who’s spent time inside a textile mill can spot the value of efficient chemicals. Calcium thiocyanate pops up here in fiber spinning, especially for making rayon. It dissolves cellulose smoothly, which allows spinners to draw out strong, fine threads. This process takes skill and reliable chemistry—one slip and the whole batch can go south. Decades of research have shown how this compound helps create textiles that stretch and breathe for everyday clothes. Plant engineers choose calcium thiocyanate because they know the resulting fibers stay consistent batch after batch.
Usefulness in Photography and Imaging
Long before digital cameras took over, photographers and technicians mixed solutions in darkrooms. Calcium thiocyanate played a behind-the-scenes part as a fixing agent, removing unwanted silver halides and keeping images crisp on film. Even now, some specialty printing and imaging shops come back to it for processes where digital tools can’t quite match old-school results. Chemists and artists appreciate its reliability for preparing prints with deep contrast and wide tonal range.
Reliability in the Chemical Industry
Factories making specialty chemicals rely on precision and safe handling. Calcium thiocyanate acts as an intermediate ingredient in synthesizing other thiocyanates or nitrites. For example, pharmaceutical labs might pull it off the shelf for prepping test reagents or making specific compounds. Fine chemical producers take care with storage and mixing; workers know even small amounts can trigger big shifts in reaction outcomes. I’ve toured sites where safety teams run regular drills just in case, showing how cautious handling gets baked into the recipe whenever this chemical is involved.
Water Treatment and Mining
Municipal water systems use a range of chemicals to keep water clear and safe. In some setups, calcium thiocyanate acts as a corrosion inhibitor, helping to keep metal pipes from rusting. Mining operators lean on it for ore flotation, where its selective action helps separate out valuable minerals from every ton of rock pulled from underground. From talking to an environmental chemist in Nevada last spring, the consensus was clear: cost-effective chemicals like this one make it possible for water and mineral resources to reach communities without breaking municipal or company budgets.
Pathways for Responsible Use
Whether on the farm, in a textile plant, or treating water, people working with calcium thiocyanate need training and clear safety measures. Local and federal regulators keep watch on storage, disposal, and workplace exposure, especially since improper handling poses health risks. Solutions start with regular staff education, clear labeling, and strong emergency planning. By learning from a blend of field stories and academic research, everyone across these industries can put safety first.
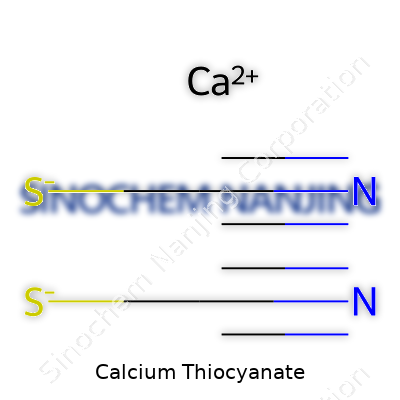

| Names | |
| Preferred IUPAC name | Calcium dithiocyanate |
| Other names |
Calcium rhodanide Calcium rhodanate Calcium dithiocyanate |
| Pronunciation | /ˈkælsiəm θaɪ.oʊˈsaɪ.əˌneɪt/ |
| Identifiers | |
| CAS Number | “5794-28-5” |
| Beilstein Reference | 391914 |
| ChEBI | CHEBI:34761 |
| ChEMBL | CHEMBL3383240 |
| ChemSpider | 70413 |
| DrugBank | DB15431 |
| ECHA InfoCard | 03bb0d18-6eec-4443-83af-4c0cc5267c65 |
| EC Number | 209-819-0 |
| Gmelin Reference | Gmelin4098 |
| KEGG | C13582 |
| MeSH | D003630 |
| PubChem CID | 24857257 |
| RTECS number | XL5950000 |
| UNII | 4B8P5ZUD7C |
| UN number | UN1548 |
| Properties | |
| Chemical formula | Ca(SCN)₂ |
| Molar mass | Ca(SCN)₂ : 176.26 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.67 g/cm³ |
| Solubility in water | Very soluble |
| log P | “-1.48” |
| Vapor pressure | Negligible |
| Basicity (pKb) | pKb ≈ 3.4 |
| Magnetic susceptibility (χ) | -37.0e-6 cm³/mol |
| Refractive index (nD) | 1.525 |
| Viscosity | Viscous liquid |
| Dipole moment | 1.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 219.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -985.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1047.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. H312: Harmful in contact with skin. H332: Harmful if inhaled. |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-2 |
| Autoignition temperature | 360°C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 2470 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 4000 mg/kg |
| NIOSH | BKW62 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 3-8°C |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
Barium thiocyanate Magnesium thiocyanate Potassium thiocyanate Sodium thiocyanate Ammonium thiocyanate |

