Calcium Selenite: Insight into a Chemical Compound Shaping Industries
Historical Development
Interest in calcium selenite began to grow during the broader exploration of selenium compounds in the nineteenth and twentieth centuries. Early chemists were drawn by selenium's curious behavior, flipping between helpful trace nutrient and toxicant, depending on context. Calcium selenite emerged as researchers looked for forms of selenium that would balance stability and utility. Over decades, industries pulled knowledge from universities and government studies—often slow, patient work translating selenium chemistry into real-world forms. Traditional glassmaking and metallurgy sometimes toyed with selenite salts, but a shift toward precise nutritional and technological uses brought calcium selenite to the table for more than just academic curiosity.
Product Overview
In the commercial world, calcium selenite turns up in feed additives, some specialized supplements, and acts as a laboratory reagent. Companies market it to animal nutrition and feed industries because selenium is tough to balance: animals need only a trace, yet deficiency or excess cuts into herd health and farm profits. Most often, calcium selenite fits into micronutrient enrichment, allowing nutritionists to tweak intake reliably. Some regions have relied on calcium selenite to fight selenium-poor soils, fortifying diets through livestock and even human supplements, though always within narrow tolerances.
Physical & Chemical Properties
We’re talking about a white, crystalline powder with the formula CaSeO3. While not flashy, these off-white grains hide unique reactivity and solubility quirks. In cold water, calcium selenite holds together, but warming speeds up dissolution. Unlike some selenium salts, calcium selenite gives off very little odor and won’t stain hands on contact, but don’t let looks fool you—selenium’s story has always demanded respect for subtlety. Under a microscope, the monoclinic crystal system pops out, separating it from other selenites and selenates. Calcium selenite carries a molecular weight of around 183 grams per mole and keeps stable in dry storage, away from acids and moisture which prompt breakdown.
Technical Specifications & Labeling
Bags or drums of calcium selenite, labeled for technical or feed-grade use, usually spell out selenium purity as a top-line metric. Most suppliers specify not just selenite percentage, but limits on lead, arsenic, and heavy metals—because buyers want to avoid bunk or contaminated stock. Labels should signal batch number, best-before date, and source, since traceability reduces safety and compliance headaches. In countries with tight selenium rules, manufacturers tend to register their products and offer third-party assay results. Because nutrition and safety standards vary by region, a supplier with European reach may carry different documentation—and slightly tweaked formulas—than a US or Chinese producer. Labels rarely tell the product’s full story, but savvy buyers hunt for details on solubility and a consistent white color—small clues for quality and adherence to requirements.
Preparation Method
Calcium selenite does not spring from the earth, but comes from a handful of chemical syntheses. Most preparative routes flow through reaction of sodium selenite and calcium salts. Technicians typically dissolve sodium selenite in water, then add a source of calcium like calcium chloride or calcium nitrate. The resulting mixture brings about precipitation of calcium selenite, which then gets isolated by filtration, washed, and dried. Much care goes into this process: sodium and calcium impurities tend to shadow selenium, and even the water matters. Some suppliers recycle selenium sources from industrial waste streams, provided purity standards promise safe final products. Chemists keep acid or base at bay, because pH swings can push selenium into unwanted oxidation states or dissolve the selenite right back into solution. Each step in this preparation trickles down to the stability, safety, and efficacy of calcium selenite.
Chemical Reactions & Modifications
Calcium selenite sits at the crossroads between stability and reactivity. Exposed to strong acids, it decomposes to form selenous acid, a compound known for its biting toxicity and volatility. Mix it with powerful oxidizers, and the selenium center bumps up to selenate—sometimes desirable, other times not, depending on use. Reductive conditions can drop selenium down to its elemental state, forming a powdery red or black precipitate. In nutrient formulations, people sometimes blend calcium selenite with chelating agents or other salts to tweak solubility or absorption in feed, especially for ruminants and poultry. Every chemical twist or tweak holds risk and promise—one misstep, and the selenium moves outside safe ranges, or loses biological activity.
Synonyms & Product Names
Calcium selenite rarely goes by a flashy street name, but don’t get tripped up by technical jargon. You’ll see “calcium selenious oxide” or just “calcium salt of selenious acid” in literature. Feed formulations prefer “selenium (as calcium selenite)” on their ingredient lists. Other terms—like EINECS 232-256-6, or in older research “selenious calcium”—pop up in registries or regulatory filings. All roads lead back to CaSeO3. If a supplier pitches anything labeled “calcium selenium oxide,” run a double-check: confusion sometimes swirls between selenite and selenate, and product quality or regulatory status depends on that little change.
Safety & Operational Standards
Working with calcium selenite takes more than gloves and goggles. While rarely an immediate irritant, the real risk sneaks up through accumulation. Selenium ranks among nutrients with a narrow gap between required and toxic doses for livestock and humans. The World Health Organization, European Food Safety Authority, and local regulatory agencies all set sharp maximums for selenium exposure—typically micrograms per kilogram body weight—driven by selenium’s effect on the heart, kidneys, and nervous system. Facilities handling calcium selenite must prevent dust spread and accidental ingestion. Spills demand scrupulous cleanup, not just to protect workers, but to avoid trace contamination in animal feed stores. On farms, feed-mixing rooms sometimes build in extra ventilation, separate weighing rooms, and strict logbooks for selenium products, all to keep accidental overdoses at bay. Medical personnel also watch for symptoms like hair loss, hoof disorders in livestock, or garlic breath–clear red flags for selenium intoxication. In the lab or factory, safety data sheets direct responses to exposure or fire, since heated calcium selenite can give off toxic fumes. Following these operational standards keeps the compound a help rather than a hazard.
Application Area
Calcium selenite circles closest to animal nutrition. Most commercial use targets the livestock sector, allowing precise selenium supplementation for cattle, sheep, swine, and poultry. In parts of the world with selenium-poor soil, such as the Great Plains in the United States or certain Chinese provinces, calcium selenite steps in where forages and grains fall short. Veterinary professionals and feed formulators argue over optimum dosing, since even small variations nudge animals from health to trouble. In lab research, scientists sometimes use calcium selenite along with radioisotope labeling to trace selenium movement in plants, animals, or even microbes. A few dietary supplement lines for humans have chosen calcium selenite for short-term therapeutic use, although less frequently than organic selenium forms due to bioavailability debates and tight toxicity margins. Outside nutrition, only rare technical uses emerge, such as in certain photographic processes or specialized glass coloring.
Research & Development
Research teams keep probing the metabolic pathways and comparative bioavailability of different selenium sources, including calcium selenite. Animal nutritionists track how quickly and efficiently livestock draw selenium from selenite versus selenomethionine or selenate. Genetic and environmental studies dig into why certain breeds or species respond differently to the same supplemental regimen. Analytical chemists work to design new test kits and field assays for faster, cheaper detection of selenite in feed and tissue, lowering the barriers to safe use in rural settings. Meanwhile, regulatory agencies and producers try to balance food-chain safety with farm productivity, pushing for ever tighter production controls and transparent reporting. From the researcher's bench to the feed mill, every step links toward broader selenium stewardship.
Toxicity Research
Toxicology studies of calcium selenite reveal both value and risk. Acute exposure in animals or humans leads to symptoms ranging from gastrointestinal upset and hair loss to, at higher doses, systemic organ failure. Chronic ingestion, even in small surpluses, prompts selenosis, marked by hoof deformation, tooth decay, and neurological signs in livestock. Regulatory benchmarks draw on decades of trial and error, including tragic lessons from natural seleniferous regions and accidental manufacturing mishaps. In public health, toxicity episodes in livestock often force mass culling or quarantines. Researchers continue to fine-tune safe ranges for each livestock species: cattle, for instance, tolerate higher daily selenium levels than sheep, due to differences in metabolism. Toxicity research presses forward, looking into chronic low-dose effects and possible links to cancer, diabetes, or autoimmune disease, especially in humans. These findings ripple through regulatory circles, pushing the industry to improve product purity and labeling.
Future Prospects
Looking forward, calcium selenite faces both challenge and promise. As food systems globalize, precision micronutrient management grows, and climate shifts threaten soil selenium status in new regions, reliable and affordable selenium sources attract more questions than they solve. Sustainable sourcing, tighter process controls, improved product traceability, and new analytical techniques get top priority, driven by food safety and consumer trust. Innovation pushes for blends or formulations that maximize uptake, minimize waste, and eliminate toxicity risks. As the world learns more about the subtleties of selenium biology, the spotlight falls on both new and classic compounds like calcium selenite—too useful to ignore, too risky to leave on autopilot. Farmers, regulators, and researchers each carry part of the story, shaping how this simple white powder supports health, productivity, and scientific progress in the years to come.
What is Calcium Selenite used for?
Understanding Calcium Selenite
Calcium selenite doesn’t exactly pop up in everyday conversation, but its role stretches across industries and our food supply. As a chemical compound, it combines two elements the body can’t really function without—calcium and selenium. Calcium is the building block for strong bones, healthy teeth, nervous system activity, and muscle contraction. Selenium supports immune health, fertility, and shields against oxidative stress. Together, calcium selenite offers a way to supplement diets and boost certain industrial processes.
Feeding Livestock and the People They Feed
A huge percentage of calcium selenite ends up mixed into feed for livestock. Cattle, pigs, and poultry don’t pick out minerals on their own. If they graze in regions with selenium-poor soil, they risk a range of health problems—weak growth, reproductive struggles, poor immune response. Farmers notice these issues and trust selenium supplements to fill the gap. This shows up indirectly on dinner tables; humans depend on animal-based food for nutrients, and selenium deficiency doesn’t do anyone any favors. Poor animal health translates to weaker, less nourishing meat, milk, and eggs. The World Health Organization highlights selenium as an essential trace mineral for people too. Not every supplement suits every species, but calcium selenite stands out because it’s stable, supplies both calcium and selenium, and gets absorbed fairly well by animals.
Industrial and Agricultural Uses
Beyond the farm, calcium selenite helps in chemical manufacturing. Glassmakers employ it to add color to glass, neutralize unwanted tints, and sometimes even to work with specialty ceramics. In agriculture, some use it to fortify soil in areas where selenium deficiencies harm crops or pasture. Plants seem to grab onto selenium from the soil and pass it up the food chain, so the right balance at ground level really matters. Scientists study how much selenium crops need, how much farmers should add, and which forms of selenium offer the best bang for the buck.
Risks, Oversight, and Real-World Challenges
Most stories around trace minerals like selenium remind me it’s easy to get too much of a good thing. Add a pinch to animal feed, everyone wins. Dumping too much turns selenium from helpful to harmful—think kidney failure, neurological problems, even sudden livestock death. Humans aren’t immune either; supplements can cause selenium poisoning if taken the wrong way. I’ve seen insurance reports from farmers detailing entire flocks gone sick from accidental overdoses. The Food and Drug Administration regulates how much selenium makes it into feed and supplements. Safety checks help, but errors still slip through where oversight is thin or misinformation spreads. Educating feed mill operators, veterinarians, and farmers stands out as a practical step. Knowing how much livestock needs—and sticking to it—keeps problems at bay.
Better Practices, Safer Outcomes
Science shapes how people use calcium selenite. Research badges doses that work in different diets and species. Government bodies set legal limits to keep animals and people safe. Regular lab testing ensures feed contains what labels promise. From my experience visiting farms, improvements come down to training and communication. Misreading a label or mismeasuring mineral powder sounds small but turns up in too many cases of accidental poisoning. Automatic feeders with digital controls help reduce human error. Some feed suppliers run workshops for farmers on supplement safety.
Calcium selenite might seem like another shelf chemical, but its trajectory cuts through the food system, glassworks, and agricultural science. With the right education, careful measuring, and continued research, its benefits can outweigh its hazards.
Is Calcium Selenite safe for human consumption?
What is Calcium Selenite?
Calcium selenite is a chemical compound that brings selenium and calcium together. Selenium helps support the immune system and works as an antioxidant. Calcium, as most people know, keeps bones strong. Combining the two seems like an efficient way to deliver both minerals at once. Food manufacturers and supplement brands often try to jump on these compounds to save space in pill bottles and claim extra benefits.
Looking at the Facts
The Food and Nutrition Board recommends a daily selenium intake of about 55 micrograms for most adults. Go over 400 micrograms and you’re at risk for selenium toxicity, which shows up as hair loss, nail brittleness, fatigue and in extreme cases, nerve issues. The form of selenium matters. Most supplements use selenomethionine or sodium selenite because these have been studied for years. Calcium selenite doesn’t have this same track record in supplement use.
Some studies on selenium compounds show that different types affect absorption rates and toxicity. The European Food Safety Authority lists sodium selenite and selenomethionine as acceptable selenium sources for food and supplements. Calcium selenite skips the list. Scientific reviews rarely mention it, which raises questions. Oversight agencies treat “unproven” compounds carefully, especially when toxicity could come into play.
What Experience Teaches
Folks in the nutrition or pharmacy world learn to treat new compounds with caution. Just because two minerals are good individually doesn’t mean their combo is safe or even helpful to the body. Selenium is essential, but it’s powerful stuff. Years back, supplement trends hyped up selenium-based blends. I saw a couple cases where people tried high-dose selenium blends thinking it would supercharge their health. The results were rough: headaches, skin rashes, stomach upset. Going overboard, especially with unfamiliar forms, often does more harm than expected.
One challenge is tracing the source of raw ingredients. In the United States, companies must follow dietary supplement rules, but enforcement doesn't always catch everything. Unknown compounds can slip into the market. That shakes consumer trust and leaves people guessing about what’s actually in the bottle and what it could do to them.
Safety Concerns and Regulation
Right now, major health authorities don’t give the green light for calcium selenite as a food additive or supplement ingredient. Lack of approval doesn’t always mean outright danger, but it does mean more research is needed. Some chemicals can cause long-term effects even at low doses, and with selenium, the line between helpful and harmful gets thin fast.
Smart Steps for Consumers
People who want to get selenium can lean on food sources: Brazil nuts, tuna, eggs, and sunflower seeds. These foods have a safety track record, and it’s easier for the body to regulate natural forms. If you’re considering selenium supplements, look for ones that use selenomethionine or sodium selenite, since there’s solid evidence for their safety if the dose stays reasonable.
Unfamiliar forms, like calcium selenite, belong in the science lab a while longer before hitting grocery shelves or supplement aisles. Trust in health means only using substances that have survived tough scrutiny. Life’s too short to take big risks for small gains, especially where nutrition is concerned.
What is the recommended dosage of Calcium Selenite?
What Calcium Selenite Does
Calcium Selenite stands out as a mineral supplement often used to boost selenium intake. Selenium means more to the body than just a trace element—it plays a part in making antioxidant enzymes, shielding cells, and supporting the immune system. Over the years, labs and supplement brands have put different forms on the market, but dosing always brings debate, confusion, and a real risk of mistakes.
The Science Behind the Recommendations
Experience in nutrition shows that more isn’t always better, especially with minerals like selenium. The Recommended Dietary Allowance (RDA) for selenium in adults rests at around 55 micrograms (mcg) per day, regardless of the form. Calcium Selenite usually enters feed and supplements in microgram doses, never grams or even milligrams, because selenium’s benefits quickly turn to harm at higher levels.
Studies show that selenium toxicity can happen if you take above 400 mcg daily. Early signs like hair loss, stomach upset, nerve trouble, and fatigue start appearing once you cross that threshold. Rural communities using well water high in selenium sometimes run into these exact problems, showing the unintended risks of natural sources. Case studies after certain supplement recalls make it clear—sticking to recommended values matters.
Risks and Who Should Decide Dose
Personal experience helping people with minerals points to a commonsense approach: Dosing isn’t one-size-fits-all. Children, older adults, pregnant people, or those with certain health issues (dialysis, thyroid disorders) don’t process selenium the same. If you’re thinking about adding Calcium Selenite, get a doctor involved. A health professional probably will do a blood test to check your selenium status before saying yes or no. In some rare medical conditions, doctors may advise higher doses, but these situations come with close lab monitoring.
Facts from Food and Supplements
In many countries, local diets already supply enough selenium through foods. Brazil nuts, seafood, meats, and grains usually deliver what people need. Adding Calcium Selenite just because you read an article or saw a product on the shelf doesn’t make sense for most. Anyone thinking about supplementing should dig into the numbers. For reference, one Brazil nut covers your entire daily requirement (and then some).
Manufacturers of supplements—with guidance from EFSA, FDA, and other agencies—usually cap individual Calcium Selenite doses in products at 55 mcg to 100 mcg per serving, matching safe daily intakes. Labels sometimes show confusing “high-potency” claims, but that doesn’t translate to better health. Supplements should never deliver more than 200 mcg of selenium (total from all sources) without medical signoff.
What Works: Sensible Supplementation
Selenium isn’t like vitamin C, where excess flushes out easily. The body stores what you take, so daily overdose and mistakes add up. If you decide to use Calcium Selenite, check the label, track your total intake from all foods and pills, and talk honestly to your doctor. If you live in a place with naturally low selenium in soil (parts of Europe or China), doctors sometimes run population screenings and recommend fortified foods before supplements.
Smart choices and real data—plus a nod to medical advice—help you dodge problems while getting the benefit. Nobody needs to take risks with a mineral that works best in tiny doses.
Are there any side effects associated with Calcium Selenite?
A Quick Glance at Calcium Selenite
Calcium selenite falls into the group of selenium compounds, showing up sometimes as a dietary supplement or feed additive. Most folks hardly notice it unless flipping through the ingredient list of their multivitamin or livestock feed. Selenium itself helps our bodies in a bunch of ways — boosting immune defense, working against cell damage, and helping with reproduction.
Not All Selenium Is the Same
You’ll see selenium pop up in several forms: sodium selenite, selenium yeast, and calcium selenite. Each type gets processed a little differently in the body. What stands out about calcium selenite is that it’s more often found in animal feed. The body grabs the selenium from these supplements and tries to make use of every bit, but getting too much can be risky.
Possible Side Effects — Signs You Are Getting Too Much
Every nutrient walks a line between helpful and harmful. Selenium is no different. The problem kicks in with overexposure. Even in regulated feed, mistakes happen, and a little too much can build up. Stack on a multivitamin or other source, and it doesn’t take much before problems show up.
Signs of too much selenium, called selenosis, read like a list nobody wants: stomach pain, nausea, fatigue, and sometimes brittle hair or nails. In severe cases, folks may face nerve problems or a metallic taste every time they eat. These symptoms match up with what researchers found in both people and animals, especially in areas where natural selenium levels run high.
Is Calcium Selenite Dangerous at Normal Doses?
Stick to the right guidelines, and problems rarely come up. In the United States, the Food and Drug Administration limits selenium additions to food and animal supplements. Studies comparing different forms show that calcium selenite does its job, but tends to hold more toxicity risks than selenium yeast, which comes from natural sources and is a bit softer on the body.
I remember seeing livestock operations getting hit with accidental overdoses — not often, but sometimes enough to land on the local news. Most outbreaks come down to mixing errors or formulas meant for different animals. It’s no mystery; too much of a good thing can turn bad fast.
Simple Steps for Safety
Nobody wins from selenium poisoning. Sticking within safe levels matters, and this is where professional oversight steps in. Dietitians and animal nutritionists check nutritional plans, and feed mills have to show test results as proof. It all circles back to solid science and common sense. Supplements, even for people, should always match what real research and regulation suggest.
Most people eating a balanced diet or using normal supplements will never run into issues with calcium selenite. If you ever plan to add more selenium to your routine, checking with a real expert helps keep you on solid ground. A few minutes of smart planning spares you a world of headache later on.
Better Alternatives and Future Direction
Researchers keep looking at other forms of supplemental selenium, hoping to find safer, more absorbable compounds, especially for use in high-risk places. For most people and animals, sticking with food sources — nuts, whole grains, and seafood — stays the safest bet. Real food brings along other nutrients, avoiding worries about tipping the scale with a single mineral.
Calcium selenite has its place, just so long as doses match need and careful eyes keep watch. This push toward supervision matches what modern health care and animal husbandry both aim for: more safety, fewer surprises, and a steady focus on real evidence.
Where can I buy Calcium Selenite?
Understanding the Demand for Calcium Selenite
Many folks hear about calcium selenite when looking to improve their soil mix or explore nutritional supplements for farming. Some also stumble onto discussions about its use in animal feed or even laboratory research. The conversation often drifts toward buying options, but too often, safety and sourcing get glossed over in favor of a quick transaction. It’s worth slowing down and taking a closer look before plugging in credit card details on the first site that pops up in a search.
Checking Legitimacy Counts More Than Ever
Walking into a shop for vitamins at the mall doesn’t translate to specialty chemicals. Calcium selenite sits within a tricky area, given its toxicity and regulatory controls. Selenium, the element in selenite compounds, acts as a trace nutrient—too much causes serious health problems. This point isn’t just a legal formality—it’s life-or-death for livestock and, if misused, for humans too. Supply stores usually keep this product under lock and key for good reason.
My own run-in with specialty chemicals involved trying to source fertilizer-grade micro-nutrients for a small garden project. One agricultural co-op wouldn’t even quote me until I proved my intended use and credentials. Their caution wasn’t about red tape—it was learned experience from others ignoring warnings and ending up with contaminated stock or legal headaches. Reputable vendors—especially in agriculture or laboratory supply—follow strict sourcing rules, ask for proof of business or professional use, and insist on clear documentation. If a site sells calcium selenite without any of those guardrails, skepticism saves more than money.
Regulations and Health Risks
The U.S. Food and Drug Administration (FDA) restricts the sale of selenium supplements to certain concentrations, mostly due to the risks from overexposure. The Occupational Safety and Health Administration (OSHA) also lists strict handling protocols for selenium compounds. My research for this piece led me to peer-reviewed warnings about calcium selenite toxicity and bioaccumulation in ecosystems. Countries differ in their stance—Europe, for example, often limits these compounds even more aggressively than the U.S. Ignoring these guidelines can put buyers on the wrong side of the law, not to mention the risks to health and the environment.
Finding Reliable Suppliers
Actual suppliers for calcium selenite tend to fall into a handful of categories. Major chemical distributors (think Sigma-Aldrich, Alfa Aesar, Fisher Scientific) will vet buyers. These vendors supply laboratories and industry. Sometimes, agricultural supply chains stock trace mineral additives for livestock feed, but a farm tax ID and business credentials usually form part of the process. Don’t expect to find it in the supplement aisle at the health food shop. Take time to look for a safety data sheet (SDS); real vendors post them alongside product details.
Alternative Solutions and Considerations
For home gardeners interested in soil health, a conversation with an agricultural extension agent or a credentialed agronomist can open doors to safer, approved options. Often, other selenium sources serve the same need—safer, legal, better supported by science. In one instance, a family friend learned that their real problem had less to do with selenium and more with poor soil drainage, which simple composting fixed at a tenth of the cost. If a supplier offers calcium selenite to the public with little to no oversight, skepticism trumps convenience every time.
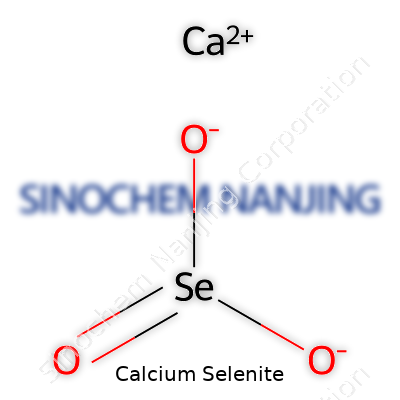
| Names | |
| Preferred IUPAC name | Calcium selenite |
| Other names |
Calcium Selenite Calciumselenit Calcium selenium oxide Selenous acid, calcium salt Calcium selenious oxide |
| Pronunciation | /ˈkæl.si.əm ˈsɛl.ɪˌnaɪt/ |
| Identifiers | |
| CAS Number | 10102-18-8 |
| Beilstein Reference | 120696 |
| ChEBI | CHEBI:86462 |
| ChEMBL | CHEMBL1232993 |
| ChemSpider | 22644058 |
| DrugBank | DB15856 |
| ECHA InfoCard | 03c6e835-ea4a-478d-862b-cef8f1616d5a |
| EC Number | 233-250-6 |
| Gmelin Reference | 53140 |
| KEGG | C18710 |
| MeSH | D017661 |
| PubChem CID | 24963 |
| RTECS number | VS8575000 |
| UNII | 867K6SW9HF |
| UN number | UN3264 |
| Properties | |
| Chemical formula | CaSeO3 |
| Molar mass | 199.07 g/mol |
| Appearance | White powder or crystalline solid |
| Odor | Odorless |
| Density | 2.91 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | -4.17 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 11.2 |
| Basicity (pKb) | 6.7 |
| Magnetic susceptibility (χ) | −38.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.63 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | “87.8 J·mol⁻¹·K⁻¹” |
| Std enthalpy of formation (ΔfH⦵298) | -1207.7 kJ/mol |
| Pharmacology | |
| ATC code | A12AX |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. Suspected of causing genetic defects. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P330, P391, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Lethal dose or concentration | LDLo oral rat 7 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 7 mg/kg |
| NIOSH | WI6700000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Calcium Selenite: 0.2 mg/m³ (as Selenium, OSHA TWA) |
| REL (Recommended) | 0.02 mg/kg |
| IDLH (Immediate danger) | IDHL: 1 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium selenite Selenium dioxide Calcium sulfate Calcium selenate |

