Calcium Permanganate: A Deep Dive Into Its Past, Present, and Promise
Historical Development
Calcium permanganate doesn’t show up in the usual stories of chemistry’s greatest hits. Still, this compound holds roots back to the nineteenth century, in those energetic days when curiosity fueled new salt discoveries. Potassium permanganate became a staple in water treatment and disinfection, and calcium’s variant followed as researchers searched for better oxidants with unique properties. Even if textbooks mention potassium or sodium more often, some research hubs and forward-thinking labs kept returning to calcium permanganate, looking for different ways to tackle tough environmental and industrial problems. Shifts in regulatory requirements and advances in purification technologies kept its relevance bubbling under the surface. The compound’s background reflects how chemistry keeps evolving, responding to both scientific imagination and society’s needs.
Product Overview
Calcium permanganate isn’t what most folks keep under the kitchen sink, and its vibrant purple color signals the strong oxidizing punch it packs. This salt comes as a free-flowing powder, and you usually catch a whiff of its metallic, slightly earthy scent if you get close. Most people never see it in daily life, but those who work with oxidizers, water purification, or specialty chemical reactions know its deep value. It’s the kind of product you encounter in advanced labs, water remediation projects, or specific manufacturing setups where operators value its stability and oxidizing strength. People working with it read labels carefully, track batch numbers, and treat spills with just as much respect as they give its better-known cousins. Its story is more than niche curiosity; it points to chemistry’s direct impact on clean water, clean air, and modern industrial processes.
Physical and Chemical Properties
If you’ve ever handled calcium permanganate, you won’t forget the rich violet hue. Its crystalline form looks dramatic, standing out on any shelf. Unlike potassium permanganate, the calcium salt brings less solubility to aqueous solutions, and that aspect shapes both how it can be applied and what results you can squeeze from it in treatment systems. Calcium’s heavier backbone keeps it a bit more settled, less likely to zip into solution than sodium or potassium forms. The substance reacts strongly with reducers and fuels. It holds up well in dry storage, but introduce moisture and things get lively. Left exposed, it tends to clump as it reacts with atmospheric humidity, a trait operators watch closely. The smell—stark and memorable—carries a faint reminder of iron or even rust, hinting at the power packed into its lattice.
Technical Specifications and Labeling
Most bottles and drums of calcium permanganate arrive stamped with the usual hazards: keep away from heat, avoid contact with organic matter, and protect contents from moisture. Labels warn about strong oxidation and incompatibility with acids and flammable substances. Proper documentation tracks purity, usually listed at 97% or above, with some lots reaching technical or even analytical grade. Moisture content gets special attention, and suppliers expect users to store material in tightly sealed containers. From experience, anyone who’s worked with strong oxidizers checks for manufacturing dates and batch traceability—problems can arise if old stock lingers on high shelves. That detail marks the difference between a safe chemical and a shelf hazard just waiting for a slip-up.
Preparation Method
Manufacturing calcium permanganate runs through a familiar cycle for anyone in chemical production. Industrial producers start by reacting potassium permanganate with calcium chloride or calcium nitrate in aqueous solution under chilled conditions. Double displacement swaps the ions, and after filtration and careful washing, producers collect crystals with patience, sticking to careful drying schedules to preserve the material’s pristine state. The method promises decent yields if controls on temperature, concentration, and mixing speed get dialed in. Everything from the reactor’s material—glass or high-grade plastic—to precise washing techniques influences the end product. Strict adherence to these procedures is non-negotiable, since shortcuts mean impurities, moisture, or mixed hydrates that both spoil product quality and increase the risk during application.
Chemical Reactions and Modifications
Calcium permanganate exists for one main reason: it transfers oxygen. It breaks down in water, releasing oxygen and forming insoluble manganese dioxide, a reaction used to oxidize organic and inorganic contaminants in water. You can push the material to react with a long list of reducers—think hydrogen peroxide, iron salts, and even certain persistent pollutants. This makes it handy in environmental cleanup jobs. Modifications can involve using blended oxidants, adding stabilizers to delay oxygen release, or changing particle size for specific flow-through filters. Some scientists push the envelope by coupling calcium permanganate with catalysts or binding it to substrates for slow-release applications. Every tweak brings a trade-off between strength, safety, handling, and reaction rate.
Synonyms and Product Names
Ask for calcium permanganate in the lab and somebody might call it “permanganic acid, calcium salt” or just plain “calcium manganate(VII).” On chemical inventory systems, it shows up under CAS number 10118-76-8. Commercial invoices sometimes shorten the name to “Ca(MnO4)2.” Other synonyms pop up here and there, including “permanganic acid calcium salt hydrate” or “dicalcium permanganate,” depending on who manufactured it or in what context it’s sold. Regardless of the label, those in the know recognize the purple powder and approach with the same caution granted to any strong oxidizer.
Safety and Operational Standards
Nobody familiar with chemical safety takes calcium permanganate lightly. Direct contact with skin means irritation, and the compound stains clothing, floors, and even skin in a matter of seconds. Goggles, gloves, and splash aprons stand as standard gear in any room where it’s handled. When spills occur, users mop up spills with inert material, keeping organics far away since the salt can ignite combustible material. Storage in cool, dry areas, away from acids and reducing agents, becomes a matter of day-to-day protocol. Emergency shower stations sit close at hand. Every operator understands the risk of prolonged exposure—manganese absorption brings neurological symptoms—and any heated arguments about shortcuts fade in the face of these health realities. The culture around strong oxidizers like this shapes entire safety modules for new technicians.
Application Area
Calcium permanganate sits in the toolbox for tough remediation jobs, where strong but steady oxidizing agents are needed. Water treatment professionals rely on it for breaking down pollutants in groundwater and tackling persistent organic pollutants. Its ability to oxidize and precipitate out manganese dioxide gives it a unique role in sites with heavy metal or organic contamination. Some research labs test its mettle in controlled chemical syntheses, especially where a milder or less soluble oxidant would outperform its more reactive cousins. The food industry, the pulp and paper industry, and select segments of chemical manufacturing incorporate it sparingly, recognizing both its power and its hazards. On rare occasions, it features in analytical methods, but safety procedures tend to limit widespread adoption where gentler compounds will suffice.
Research and Development
Much of the recent research around calcium permanganate focuses on tackling environmental pollution, especially sites with groundwater contaminated by chlorinated solvents. Research groups across the globe design injection strategies to deliver permanganate deep into soils and fractured bedrock, trying to maximize contact time without risking secondary contamination or toxic byproducts. Advanced studies push for safer formulations, sometimes micro-encapsulating the oxidant for longer shelf life or more targeted release. Publications share data on new ways to couple calcium permanganate with iron or activated carbon, hoping to trigger synergistic breakdown of industrial contaminants. In these real-world tests, researchers fine-tune methodologies, hoping to outsmart both technical and regulatory obstacles. Every new paper helps keep the compound relevant, especially as governments demand stricter pollution control and new contaminants challenge current cleanup technologies.
Toxicity Research
Toxicology reports make it clear: manganese compounds taken up via inhalation or ingestion cause health problems. Exposures over time, even at low levels, create headaches, tremors, and neurologic effects ranging from simple clumsiness to symptoms that mimic Parkinson’s disease. Calcium permanganate brings all the same dangers as its potassium cousin, plus the challenge of keeping dust under control during handling and mixing. Company safety data sheets repeat the warnings, grounded in animal studies and long-term occupational exposure records. Short-term eye and skin irritation gets most of the attention in safety training, but serious inhalation or ingestion means immediate medical attention. Regulators upped requirements for air monitoring, medical surveillance, and personal protective equipment, tightening compliance for any workplace where the stuff gets used in bulk.
Future Prospects
There’s no sign that strong oxidizers will fade from industrial, municipal, or environmental playbooks. Calcium permanganate’s unique mix of reactivity and manageable solubility keeps it in the running as new cleanup challenges surface. Nanotechnology, advanced encapsulation, and even drone-assisted environmental delivery promise new ways to deploy the compound safely while keeping costs in check. As researchers develop more selective oxidation techniques and as regulations reshape contamination standards, companies and government labs keep an eye on old salts like calcium permanganate. Its future hinges on the balance between fixing environmental messes and managing health risks—a reminder that advances in chemistry solve today’s toughest headaches, but only with the right respect for the power lurking inside each purple grain.
What is Calcium Permanganate used for?
What It Brings to the Table
Anyone with a toe dipped in water treatment, chemical manufacturing, or environmental cleanup knows how vital the right oxidizer can be. Calcium permanganate, often showing up as a purple granular powder, lands on shelves for one clear reason—it packs a strong oxidative punch. Its presence often hints at a tough job needing efficient, powerful chemistry.
Why Water Treatment Trusts It
In water purification, oxidizing agents do the heavy lifting. Calcium permanganate steps up in places where regular chlorine struggles. Algae blooms, sulfur smells, iron and manganese that stain fixtures—these problems cling to water sources all over the world. Municipal water treatment plants use calcium permanganate as a front-line defender. It breaks down foul-smelling and taste-forming compounds, making water cleaner and safer. This matters for people who expect fresh-tasting, healthy tap water every time they turn on the faucet.
Years spent around Florida’s lakes showed me how smelly, iron-stained water crushes trust in local water supplies. Only strong oxidizers like potassium and calcium permanganate proved stubborn enough to fix those problems quickly. Talking to utility workers, I heard time and again how calcium permanganate made a real difference during emergencies—when warm weather spiked algae and left water sources reeking.
Industrial Cleaning and Disinfection
Factories and wastewater facilities face their own set of headaches—biofilm on pipes, lingering industrial waste, and relentless bacteria. Regular bleaches often don’t cut it. Calcium permanganate, with its higher stability and slow, sustained release, brings a different sort of muscle to the job. Operators favor it for cleaning tough-to-reach systems, scrubbing down contaminated surfaces, or even treating certain industrial spills. Strong oxidization knocks out organics and pathogens, keeping workspaces safer and processes running smoother.
Environmental Remediation
Environmental cleanup isn’t a glamorous field, but it plays a crucial role in community health. Soil and groundwater contaminated by pesticides, wood preservatives, or petroleum products call for specialized tools. Calcium permanganate ranks high for treating chlorinated solvents—common in old industrial lots and leaking storage tanks. It reacts with toxic compounds, breaking them down into much less harmful substances. Field techs like it because it handles tough jobs where other treatments stall. Good outcomes help neighborhoods reclaim land once written off as hazards.
Cleanup work across the Midwest cemented my respect for what calcium permanganate achieves. Homeowners near dump sites wanted their land back, and only a handful of chemistries, this one included, could handle the depth and volume of contamination. Every successful remediation job meant fewer risks for children playing outside and cleaner water for families.
Challenges and Safer Usage
While its benefits stand out, using calcium permanganate safely requires respect for its oxidizing strength. Mishandling sparks fires or hazardous reactions. Folks who work with it need training and proper gear—no room for shortcuts. Sourcing and transportation require strict care due to regulatory limits around strong oxidizers. Manufacturers who take shortcuts risk incidents that hurt everyone.
Better safety training, improved packaging, and smart storage help minimize the risks. Regulatory agencies, such as the EPA, routinely audit sites handling it. That oversight keeps standards high and people safer.
Looking Ahead
As water sources get squeezed, industry and municipalities turn more often to robust tools like calcium permanganate. Having spent years watching its impact across everything from rural water systems to big-city wastewater plants, I get why so many professionals trust it to get the job done right. It fills a niche that cleaner, greener chemistry alone can’t reach—yet. Looking toward the future, safer handling and responsible application remain top priorities to balance progress with protection for both workers and the environment.
Is Calcium Permanganate safe to handle?
Let’s Talk About Handling Chemistry
Handling chemicals in any lab or industrial setting brings a blend of fascination and risk. Calcium permanganate shows up in places people don't always expect, like water treatment and disinfecting processes. Some folks might think of it as just another purple compound, but there’s more going on. Getting hands-on with this chemical means stepping into some real hazards, and it’s important to understand those risks upfront before opening any bottles.
Understanding the Hazards
I’ve watched over the years as students and newcomers underestimate the hazards of oxidizing compounds, especially those with permanganate. Calcium permanganate hits hard on that list—it reacts strongly with organic material, sometimes leading to spontaneous ignition or explosions if people get careless. Add a little moisture, and you ramp up possibilities even more. This compound stains skin and surfaces, causing not only headaches for cleaning but also delivering a pretty effective irritant punch. If someone breathes in the dust, potassium permanganate’s cousin calcium permanganate can irritate the lungs and airway, triggering coughing or worse, especially in untreated, confined spaces.
The Science Is Clear
Scientific reviews back up these concerns. Research published in the American Chemical Society journals points out the stability issues and safety incidents tied to permanganates as a group. Oxidizers in the workplace account for regular reports of accidental burns and fires. When calcium permanganate gets handled without respect, these statistics start to look a lot less abstract. In my experience, the professionals who’ve worked safely around this compound all treat it with real caution, keeping safety goggles, gloves, and lab coats as a daily uniform, never just for show.
Why Anyone Should Care
People might wonder why they should worry—after all, it’s found in some products sold for home use. That’s the tricky part. Commercial and industrial concentrations differ from hobbyist amounts, but accidents happen fast. Just last year, a case in a Midwest town traced a small fire in a school laboratory to a forgotten open jar of calcium permanganate left next to sugar. Small lapses or taking shortcuts build up toward big problems when strong chemicals are involved.
Practical Solutions for Safer Handling
One overlooked habit can make all the difference: storing calcium permanganate away from anything flammable, never skimping on ventilation, and keeping work areas scrupulously clean. Spills need cleaning with water right away, and not just wiped up. Hands need washing, even after gloves come off. Local fire codes usually require oxidizers like this to sit in separate storage cabinets. Schools and hobby labs benefit from checking chemical inventories every month. If a bottle’s old or the crystals look unusual, best practice means disposing of it through an approved chemical disposal service, not just in the trash.
Turning Knowledge Into Trust
Staying safe with calcium permanganate isn’t about paranoia—it's about habits and respect for the science. The real experts in chemical safety trust their eyes, their noses, and their routines, earned through both training and experience. Relying on clear facts, solid procedures, and a healthy dose of skepticism about “shortcut” solutions keeps accidents at bay, whether in big facilities or in high school chem labs. Teaching these habits early and repeating them often will always beat relying on luck.
What are the storage requirements for Calcium Permanganate?
Treating Calcium Permanganate with Respect
I’ve spent enough time around labs and supply rooms to know that chemical storage isn’t just a checklist. A handful of mistakes, or one bad label, and suddenly you’re dealing with an accident that shouldn’t have happened. Calcium permanganate, a purple-black powder, reacts quickly with all sorts of materials. That’s one reason storage gets so much attention from experienced chemists, safety experts, and anyone in the habit of handling oxidizers.
The Dangers Sitting on the Shelf
Calcium permanganate does not tolerate moisture or heat. Left in a humid space, it starts to clump or break down, sometimes releasing oxygen that triggers fires. Moisture is not the only problem. This chemical reacts dangerously with organic materials. Sawdust, cloth, paper, and even simple sugars are all on the list of things to keep away. Stories are out there about mop heads catching fire or clouds of purple smoke billowing into storage areas. Calcium permanganate is not forgiving of sloppy habits.
Temperature and Lighting
Heat speeds up chemical reactions—everyone learns that early in chemistry class. Storage for calcium permanganate belongs in a cool, temperature-controlled room. Anything above normal room temperature adds risk. Sunlight isn’t ideal either. Direct light causes slow decomposition of this compound. That turns into a storage headache in warehouse windows or on open shelving. Secure cabinets help, especially those that keep out light and allow the air to circulate.
Containers Matter
I’ve opened containers that ruptured because someone used the wrong lid or picked a can with a loose seal. Calcium permanganate needs strong, airtight containers—plastic or glass, not anything made from metals like iron or aluminum. Iron reacts and throws off sparks and smoke if things go wrong. Safety data sheets recommend keeping containers well-labeled, and—based on experience—double-checking the caps or plugs saves hassle later.
Segregation Sounds Extreme, But It’s Smart
Stacking different chemicals to save space sounds clever, but in practice it leads to accidents. You’ll find that experienced lab managers keep oxidizers like calcium permanganate separate from combustibles, acids, or reducing agents. In small-scale settings, a dedicated cabinet with clear signage works well. Large warehouses rely on separate bays or even rooms built away from the main storage. It’s not paranoia—there are enough fire reports out there to justify it.
Safety Isn’t Just a Rule—It’s a Habit
No matter how strict the guidelines, lapses happen. I’ve caught myself rushing and nearly setting incompatible bottles too close. Regular inspections help, but habits make the biggest difference. Posting handling instructions, training staff, and marking areas with hazard tapes all create reminders. Emergency gear should sit nearby, not locked away across the building. Spill kits, goggles, and gloves—these protect against surprises.
Solutions That Work
Some storage concerns get solved by switching to smaller packaging. Less volume means fewer problems if a leak or reaction starts. Electronic monitoring handles humidity and temperature in larger storage spaces. Local fire authorities often review plans, and third-party audits catch issues missed by those too close to the daily routine. Build in redundancy—labels in two languages, locks with checklists, inventory controls that verify every bottle’s location. Relying on just one level of safety rarely holds up long-term, especially with chemicals like calcium permanganate.
In the end, storing an oxidizer well just takes consistency. Not just following protocols, but living by them, every time. That is where safety moves from theory to daily practice.
How should Calcium Permanganate be disposed of?
Looking Beyond the Label
Most folks spot the word “permanganate” and think about water treatments or pool maintenance. In real life, calcium permanganate packs a much bigger punch. The powder’s intense purple color means oxidizing strength. If it ends up in the wrong place, that reactive character stirs up plenty of trouble.
The Hazards Nobody Wants in Their Backyard
Working around campus labs and small manufacturers, I have seen firsthand how someone’s forgotten shelf stash can lead to headaches later. Calcium permanganate is no friend to the local ecosystem. Pantry disposal or pouring it down the drain brings on big problems for wastewater facilities. Life in streams or ponds takes a hit—the compound doesn’t just disappear, and aquatic life suffers quick.
Getting careless with strong oxidizers has another downside: fire risk. Once this stuff mixes with flammable materials—cardboard, rags, cleaning agents—fires start easily and escalate even faster. Nobody wants the “boom” of chemicals mishandled, let alone toxic fumes.
Learning from Real Mistakes
Many years ago, a well-meaning neighbor tried to clean out his garage. He tossed several jars—one of them with leftover calcium permanganate—into the city garbage. The regular waste truck mixed it in with grass clippings. The result: a small fire, then evacuation for the whole block. Firefighters sorted it out, but the story spread fast. Education matters way more than a quick sweep-and-toss cleanup.
Reliable Disposal: The Smarter, Safer Approach
So much of responsible disposal comes back to following the rules. Government agencies call the shots here for a reason. The EPA and OSHA both label permanganate compounds as hazardous. Hazardous waste drop-off days, local municipal collection, or licensed waste contractors all know what’s at stake. That specialized know-how takes on the job from there.
Leaving it for trained folks limits chemical exposure for everybody else. They neutralize oxidizers with care, often using reducing agents to turn the purple menace harmless. Sodium thiosulfate or hydrogen peroxide sometimes serve as antidotes, bringing the compound down to manganese dioxide (a far less reactive solid). At every step, the waste remains contained, labeled, and accounted for—preventing runoff, accidents, or unintended reactions.
Staying Proactive: A Community Effort
Clear labeling of every chemical container makes a difference, but it takes more than that. Safety training helped our local lab prevent unnecessary mishaps. New employees got the rundown on proper containment and emergency steps. That hands-on learning shaped smarter habits and stopped trouble before it started.
It isn’t always about big industry, either. Schools, hobbyists, and maintenance staff need guidance just as much. Open communication with environmental services, using clear instructions, and promoting collection sites gives everyone a fighting chance to do the right thing.
Protecting Land and Water for the Next Generation
Looking back, skipping disposal steps never pays off. Community safety depends on responsible handling. Trusting trained professionals with calcium permanganate keeps the environment cleaner, homes safer, and keeps those “garage fire” stories out of the headlines. Simple steps—reading the label, asking the right folks, and making the safe call—carry real weight for the health of our neighborhoods.
What is the chemical formula of Calcium Permanganate?
Digging Into Calcium Permanganate
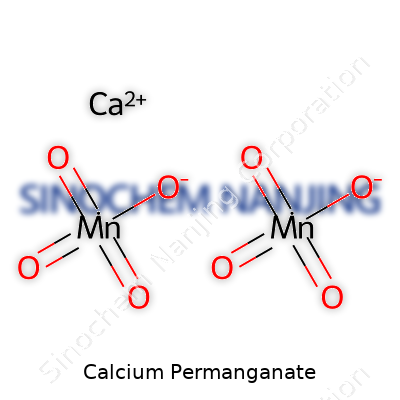
Calcium permanganate turns up in textbooks and labs, but most don’t stop to think why its chemical formula—Ca(MnO4)2—matters to everyday life. This deep purple compound pairs one calcium ion with two permanganate ions, making the whole thing both reactive and a little bit rare to stumble across outside a chemical supply store. Folks working in water treatment, organic synthesis, or even pool cleaning actually link up with this formula more often than it seems.
Why the Formula Isn't Just for Trivia Night
Some years ago, a neighbor asked about pool chemicals, suspecting something was up with the dark stains around his tiles. He didn’t realize permanganates can act as oxidizers—breaking down organic gunk and keeping things clean. Calcium permanganate offers a more stable alternative to its cousin, potassium permanganate, especially when the water system has hard-water issues or calcium shortages. You can’t swap one for the other based on looks; that formula matters. Ca(MnO4)2 delivers two powerful permanganate groups for every calcium ion, letting it oxidize stubborn substances and disinfect in one swoop.
Real-World Use Depends on the Formula
Permanganates have the power to neutralize tastes, odors, and certain harmful microbes in a way chlorine can’t always manage. That’s why cities and industries lean on the correct formula—mixing up calcium with, say, sodium or potassium versions risks weak outcomes or even dangerous results.
Chemical formula precision ties directly to environmental safety. Calcium permanganate decomposes into insoluble manganese dioxide that settles out—handy for removing iron, manganese, and hydrogen sulfide from well water. During my school lab days, getting sloppy with the formula sometimes ruined results, and in real-world cleanup, that error can linger long after the purple fades from view.
Chemical Safety and Consumer Awareness
People hear about pool shocks or water purification tablets, but not many get the difference between calcium and other permanganates. It comes down to the formula: Ca(MnO4)2 gives two permanganate units, pushing stronger oxidizing power than some cheaper alternatives. This makes it more effective, but also means handling calls for respect. Eyes, skin, lungs—it all matters. The right formula tells trained hands how much to use and when to back off.
Solutions to Chemical Confusion
Chemistry classes and supply companies could do a better job breaking down why these details matter, starting early and making sure safety data sheets and labels are clear. Using the wrong chemical in a pool, a water well, or an experiment leads to headaches and real harm. Walking people through the role of formulas, not just the “what” but the “why,” would make everyday life safer.
Out in the field or the factory, someone always asks, “Can I just use what I’ve got?” Reading up and sticking to formulas like Ca(MnO4)2 means getting results that work and keeping health risks out of the picture. Confidence with formulas isn’t just book smarts—it’s how anyone from a maintenance crew to a science student stays one step ahead of problems.
| Names | |
| Preferred IUPAC name | Calcium manganate(VII) |
| Other names |
Permanganic acid, calcium salt Calcium manganate(VII) Calcium permanganate (Ca(MnO4)2) |
| Pronunciation | /ˈkæl.si.əm pərˈmæŋ.ɡə.neɪt/ |
| Identifiers | |
| CAS Number | 10118-76-6 |
| Beilstein Reference | 3561349 |
| ChEBI | CHEBI:66352 |
| ChEMBL | CHEMBL1201649 |
| ChemSpider | 20802 |
| DrugBank | DB15568 |
| ECHA InfoCard | 02-211-995-725-33 |
| EC Number | 232-187-0 |
| Gmelin Reference | Gmelin Reference: 83322 |
| KEGG | C18767 |
| MeSH | D017822 |
| PubChem CID | 24857 |
| RTECS number | OO9625000 |
| UNII | 983L9V6T1A |
| UN number | UN1458 |
| Properties | |
| Chemical formula | Ca(MnO4)2 |
| Molar mass | 238.07 g/mol |
| Appearance | Purple crystalline solid |
| Odor | Odorless |
| Density | 2.703 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.64 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 12.5 |
| Magnetic susceptibility (χ) | +2250.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 174.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –812.1 kJ/mol |
| Pharmacology | |
| ATC code | A12AX |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage, may intensify fire. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318, H332, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 Oral rat 325 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 220 mg/kg |
| NIOSH | Not Listed |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium permanganate Sodium permanganate Barium permanganate Ammonium permanganate |

