Calcium Perchlorate: A Closer Look at Its Role, History, and Future
Historical Development
Calcium perchlorate doesn't get much attention outside of specialist circles, but its origins and uses cut across a lot of familiar ground in science and industry. Chemists figured out the basics of perchlorate salts in the nineteenth century, finding them among other oxidizers that could spark reactions and drive chemical changes. It caught more attention in the twentieth century as industries and laboratories looked for stronger and more reliable sources of oxygen. Calcium perchlorate joined the lineup because it offered a steady, crystalline form with strong oxidizing power. The military and rocket researchers, always hungry for better propellants and emergency oxygen, started studying it in more depth. Academic labs saw value in perchlorates as clean-burning sources for demonstrations, making the salt a minor but notable player in experimental chemistry.
Product Overview
Ask most people what perchlorate does, and you’ll get blank stares, but for those in the know, calcium perchlorate stands out for its sheer oxidizing strength. It looks like a white crystalline powder, dissolves in water with ease, and supplies pure oxygen at a pace that draws the interest of engineers and chemists. Producers standardize forms by particle size, moisture content, and purity, with the highest grades used in research settings and lower grades in bulk chemical processes. Calcium perchlorate doesn't have the PR buzz of lithium and sodium cousins tied to batteries or fireworks, but its value sits in its safer handling and stable storage properties compared to other perchlorates.
Physical & Chemical Properties
With a molecular formula of Ca(ClO4)2, calcium perchlorate lines up as a heavy hitter in the world of oxidizers. It comes in solid form—usually a hygroscopic, colorless to white crystalline material that draws water from air and forms hydrates easily. Aqueous solutions turn slightly acidic, and the crystals break down slowly if left in open air. Its melting point sits lower than expected for a salt, melting into a viscous liquid long before decomposition hits. In water, it dissolves completely, releasing perchlorate ions ready to drive oxidation in reactions ranging from rocket propulsion to analytical chemistry. What sets it apart is the strong oxidizing nature, which means storage needs tight control, especially with organic materials and reducing agents nearby.
Technical Specifications & Labeling
In professional settings, labels mark calcium perchlorate as a strong oxidizer, and every bag or jar comes stamped with purity percentages, hydrate content, and batch data. In laboratory supply houses, the highest purity grades top 98 percent with minimal chloride or sulfate contamination. Labels flag hazards, especially fire and health risks, often with the classic oxidizer symbol. Moisture content ranks high as a concern because the salt loves water, and keeping it dry isn't just about convenience—it’s about safe performance and reliable outcomes in the lab. The technical specs handle the tough job of keeping shipping, storage, and handling safe and predictable.
Preparation Method
Chemists make calcium perchlorate by reacting calcium salts such as calcium carbonate with perchloric acid, driving off carbon dioxide and water under tightly watched conditions. Industrial-scale setups use closed systems and careful temperature control, since perchloric acid brings serious risks of explosion if things go wrong. The process turns out a highly soluble product, filtered and dried to prevent hydrate formation and caking. Purity gets a boost by recrystallizing from water and gently drying under controlled humidity to lock in the right chemical balance. Clean production keeps byproducts—especially chloride and sulfate—at bay, a detail crucial for high-consequence uses.
Chemical Reactions & Modifications
Calcium perchlorate doesn’t just sit on a shelf; it brings out the heavy artillery when mixed in chemical reactions. Inorganic chemists use it as a robust oxidizer, seeing its effect in everything from qualitative tests to synthesis of specialty materials. It breaks down to release oxygen, especially under heat or catalytic influence, and reacts strongly with organic compounds, producing intense exothermic effects. In making other perchlorates, it often serves as a precursor, reacting with sodium or potassium salts to swap ions and turn out new compounds. Adding calcium ions also shapes double salts, hydrates, or mixed oxides. Environmental chemists watch closely when perchlorate enters natural water cycles, knowing these reactions can contaminate drinking water or affect microbial communities.
Synonyms & Product Names
As with many industrial chemicals, calcium perchlorate shows up under a wide batch of names. You’ll hear chemists refer to it as "calcium(II) perchlorate," or by its old-school name, "perchloric acid, calcium salt." In some academic circles, a catalog number stands in for a name, but the substance itself rarely strays from the Ca(ClO4)2 formula. Labels tend to show its hydrate status—monohydrate, tetrahydrate, hexahydrate—each with their own minor tweak in properties and storage needs. These alternate names mostly stretch across technical documentation, ensuring nobody swaps the material for a less reactive cousin.
Safety & Operational Standards
Workers treating calcium perchlorate with casual indifference run into trouble fast. Standard training always covers its role as a top-shelf oxidizer. Any organic residue—grease, dust, even a forgotten scrap of filter paper—can touch off a flash fire or explosion if paired with calcium perchlorate. Storage rules push for sealed, moisture-proof containers kept far from combustibles. Proper ventilation is non-negotiable, since heat or acid contact ramps up decomposition, throwing off toxic chlorine oxides and oxygen. Handlers wear gloves, goggles, and lab coats, and all operations take place far from sources of friction or impact. Disposal rules treat calcium perchlorate as both an environmental contaminant and a fire hazard, banning it from drains or landfills without specialized treatment. Every year, authorities adjust the rules as new research uncovers more about its risks, especially around water systems and food chains.
Application Area
Most people won’t spot calcium perchlorate at the store, but its touch shows up in a surprising number of fields. Fireworks designers tap its oxidation strength for flash components, propellants, and signal flares needing a clean, hot burn. In the lab, researchers lean on it as a primary standard for titration and as an oxygen source for chemical analysis. The environmental sector watches perchlorates closely in water treatment, both as pollutants and as agents for advanced oxidation processes. Space agencies—think Mars rovers and life-support research—study perchlorates as possible life-support elements, since similar salts show up in Martian regolith and could unlock future air and water solutions. Specialty glassmakers and ceramics experts borrow perchlorates for unique coloration and high-temperature processing, stretching its role well beyond a simple lab reagent.
Research & Development
Interest in calcium perchlorate keeps cycling up, especially as environmental and safety stakes rise. Clean synthesis paths that cut extraneous byproducts, reduce environmental leakage, and produce hydrate-free salts drive most lab investigations. Environmental engineers probe its breakdown and interactions, aiming to keep perchlorate out of soil and water. Rocket science circles search for stable, high-energy oxidizers that boost thrust but limit pollution—which means new blends based on calcium perchlorate show up every few years. Water chemists dig into the balance between perchlorate removal and selective recovery, always looking for filters, ion-exchange systems, or microbes that chew up perchlorate for good. Medical research pokes at tiny dose effects, wondering if long-term exposure changes thyroid function or growth patterns in living things. Each advance builds on a century’s worth of technical know-how, but the pace of fresh research never seems to slow down.
Toxicity Research
Toxicologists have watched perchlorate families closely, since the key risk circles back to impacts on thyroid health. Perchlorates interfere with iodide uptake in the thyroid gland, which matters most for infants, pregnant women, and folks with preexisting thyroid disorder. Chronic low-level exposure through contaminated water or dust raises alarms in farming and municipal water systems. Researchers sample air, dust, and water around industrial sites, tracking levels and how they move through ecosystems. Animal studies flesh out the biochemical pathways, giving risk assessors a grounded sense of what real-world exposure can mean. Regulators draw on this work to set legal limits, and ongoing work refines the models as exposure data trickle in from far-flung places.
Future Prospects
Calcium perchlorate looks set for a bumpy but fascinating road ahead. Tighter environmental regulations will force factories and labs to rethink use, storage, and disposal. Startups and university labs tinker with ways to recover, recycle, or neutralize perchlorate with minimal waste. In aerospace, the salt holds promise for next-generation fuels and oxygen supplies, and Mars missions might one day use locally-sourced perchlorates for life support or farming. Advances in water treatment could flip the script, turning an environmental risk into a valuable raw material. Safer packaging, smarter detection, and targeted breakdown strategies promise to lower risks for workers and the wider public. New analytical tools and genetic engineering also raise hopes for cleaning up existing contamination, hinting at greener ways to move forward. All these shifts reflect broader changes in how we weigh chemical benefits against social and environmental costs, making calcium perchlorate a touchstone for twenty-first century science and policy.
What is Calcium Perchlorate used for?
Real-World Applications of a Powerful Chemical
Calcium perchlorate doesn’t turn up in everyday conversation, yet its impact stretches across industries and decades. This chemical brings a punch: it's got oxidizing power, reliability, and a name that pops up in labs, factories, and even space missions. Most folks probably catch a whiff of its story only through news about water purification or space gadgets, but there’s more beneath the surface.
Boosting Rocket Science and Engineering
Growing up, every kid who liked space knew about rocket fuel, but few realized how vital solid oxidizers like calcium perchlorate are for propulsion. You stack it with fuel, and you get a reaction that launches payloads into the sky. The stuff keeps engines burning hot and clean, cutting down on leftover soot and turbocharging liftoffs. NASA and other agencies rely on perchlorates to get probes off the ground, and they do it because of reliability and stability. This isn’t just a science story—success in these launches opens up discoveries about space, medicine, and even climate. Every time a mission lands photos from Mars, some chemical like calcium perchlorate stands in the background.
Pure Water and Industrial Cleanups
I used to volunteer with a local water-testing group, and knowing what’s in river water always mattered to me. Calcium perchlorate steps into water treatment, helping scrub out impurities that would spell trouble for fish, crops, and people. It breaks down pollutants, kills off bacteria, and brings cleaner water to towns and farms. Industry goes through tons of water, and cycling it through massive tanks laced with chemicals is a daily routine where this compound often plays a role. When spill after spill pushes toxins into the soil, perchlorates can help break things down or keep contamination in check.
Life Support and Space Survival
Lately, interest in moon and Mars missions is roaring back. Calcium perchlorate has a starring role there, not just in rockets, but in keeping astronauts breathing. It can release oxygen, turning into a tool for life support on long missions. NASA’s experiments exposed this chemical to Mars conditions. It doesn’t just clean; it can pull water from thin air—a big deal if you’re hundreds of millions of kilometers from home. In 2015, researchers picked up signs of perchlorate in Martian soil. That triggered talk of using it for future habitats: pulling water vapor out, releasing oxygen, and making settlements possible with local resources. The dreams of off-world survival get a small nudge from this unglamorous compound.
Risks, Challenges, and Smarter Use
All this power comes with problems. Calcium perchlorate is classified as a hazardous material. Mishandling it can lead to accidents—there’s real risk in fires, and health regulations treat it seriously. Prolonged exposure links to thyroid problems. In some parts of the United States, perchlorate runoff from factories and military sites left water tainted for years, and the fight over cleanup costs still stirs up locals and lawmakers.
Safer storage, stricter limits, and better cleanup technology lower these risks. I’ve seen community groups press city officials for stricter compliance and regular testing. More public funding goes to monitoring perchlorate levels in water near factories, and researchers are always searching for substitutes or better disposal methods. The goal: keep the benefits, cut the harm, and make sure local families don't pay the price for global progress.
Looking Forward
Calcium perchlorate’s story runs through the veins of industry, medicine, and exploration. It’s not going away soon, and better education on its safe use will help keep accidents in check. Transparent reporting and open science encourage trust, and staying informed—no matter your job—matters more than ever.
Is Calcium Perchlorate hazardous or dangerous?
Unpacking the Facts
Calcium perchlorate doesn’t often make headlines, but you’ll find it quietly at work in laboratories, sometimes water treatment, and specialty industries. It shows up thanks to its strong oxidative kick, which makes it useful in rocket propellants, pyrotechnics, and analytical chemistry. Few people outside these fields ever see the stuff, but if someone dropped a drum of it in your neighborhood, you’d want to know more.
What Goes Wrong?
The simple truth: it’s not a friendly substance. Its main danger comes from its willingness to react, especially with organic material or anything that can burn. Here’s what sticks out most from my own science background—one careless move can mean a violent reaction. Mix it with the wrong cleaning rag, a common solvent, or let it contact moisture when conditions are right, you get fire or explosion hazards most folks can't handle without training or proper equipment.
The Health and Environmental Angle
People worry about getting exposed, sometimes thinking a trace is fine. Studies show perchlorates—including calcium perchlorate—aren’t harmless at low doses over time. They disrupt how the thyroid uses iodine, putting children and pregnant women at risk of developmental deficits. Iodine is no luxury for the thyroid, so if perchlorate competes for absorption, basic hormone balance falters. The U.S. Environmental Protection Agency and several public health sources have pointed to real risks, even at levels once thought safe, especially through contaminated water.
Spill and Storage Hazards
Years ago, I witnessed a poorly stored oxidizer—different compound, similar risks—cause a minor fire at a research facility because someone ignored a leaking bag. Calcium perchlorate isn’t forgiving. A wet floor, stray fragments of organic debris, a lack of ventilation, and you’re set up for either a slow chemical problem in the air or a sudden emergency. You can’t just toss containers in any closet and walk off. Strict labeling, special containers, temperature control, and trained staff make all the difference.
Solutions Grounded in Reality
Keeping the hazards low means strong training and clear rules. Chemical manufacturers bear the biggest responsibility—they need to sell only to vetted, trained buyers. In labs, checklists should get respected, not just signed off because “it’s always worked before.” Water systems treating for perchlorates must keep a close watch, using activated carbon or special resins to pull perchlorate ions from anything destined for taps and wells. Regular soil and groundwater tests help spot problems before they get too big to solve. If communities learn about older industrial sites or surplus dumping in their area, they’ll push for cleanup or monitoring—sometimes they have to fight for that, but it’s safer in the long run.
The Takeaway
Calcium perchlorate poses genuine risks. Calls for respect, not just fear. People who work with it need proper gear, strict discipline, and honest respect for its power. The rest of us count on science-backed regulations and public oversight, not hand-waving reassurances. Keeping this chemical in line isn’t high drama most days—but it takes one slip to prove “safe enough” thinking dead wrong.
How should Calcium Perchlorate be stored?
Understanding the Risks
Calcium perchlorate sets off red flags for most people in lab coats. This salt loves grabbing moisture from the air and brings serious oxidizing power into the room. A forgotten container easily clumps up, forcing all sorts of headaches the next time you reach for the bottle. If any fuel or organic dust picks up a few stray grains, heat doesn’t just mean a little smoke. Often it means flames, maybe worse.
What Everyday Labs Teach Us
Back in my university days, we had a few incidents with perchlorates, though not quite as lively as the stories circulating online. I remember coming into the stockroom to find a container sweating from the humidity, its cap rusted, the label half falling off. Little things like steam boilers and dehumidifiers made a difference, and folks started paying closer attention to that corner of the shelf. Not everyone wants to shell out for inert gas cabinets, but that doesn’t mean you can leave dangerous chemicals to chance.
Dry, Cool, and Controlled
Humidity invites clumping—and that means the chemical won’t dissolve or react the way the paperwork promises. A sealed container, real tight, does the job, especially with a proper plastic liner or glass that shrugs off corrosion. Many labs stash these in desiccators, sometimes alongside a moisture-indicator pak for a little peace of mind. Tossing it near a steamy sink or beneath air-conditioning vents pretty much guarantees an accident in slow motion.
Direct sunlight turns many chemicals nasty, and calcium perchlorate isn’t an exception. Keeping it off the benchtop and away from windows helps. People sometimes overlook HVAC vents blowing dust or heat directly into storage areas. Even a few degrees makes a difference over months. If the chemical room’s thermometer is creeping toward the thirties Celsius, or the air smells like mold, it’s time for either a new location or an environmental control fix.
Labeling and Separation
Mislabeled bottles cause panic. That thrill of surprise in an emergency isn’t something you forget, especially when you crack open a jar expecting salt and instead get an oxidizer. I’ve seen folks forget to check dates and clear out expired stock. Every inventory shuffle means inspecting for cracked seals, powder at the bottom, or weird stains on the shelf. That sort of discipline keeps everyone safe far more than meet-and-greet safety talks ever will.
Never store perchlorates near flammable liquids, paper, or any organic compounds. Some labs color-code shelves or mark them with big, bold stickers. The old timers run oxidizer storage cabinets set apart from everything else, mounted with signs you can read from a hallway. These habits aren’t just for regulations—they’re built on sweat and real-world close calls.
Emergency Planning
Taking time to set up sand buckets, fire blankets, or chemical spill kits near the storage area helps most during a surprise flare-up. Even once in a career, that gear matters more than a thousand bureaucratic checklists. Training drills aren’t just rituals—sooner or later, everyone drops something, and practicing a quick, calm response sticks more than any memo.
Think Fresh, Check Often
Storing calcium perchlorate safely doesn’t come from one rulebook or a single memo. Watch for leaks, adapt during heatwaves or cold snaps, and check up on your oldest containers. I’ve learned that a good chemical inventory—honest, up-to-date—saves lives, wasted money, and a whole lot of paperwork, too. The best storage solutions grow from equal parts respect and experience.
What is the chemical formula of Calcium Perchlorate?
Calcium Perchlorate: More Than Just a Formula
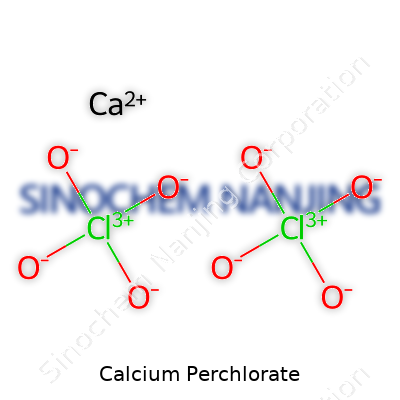
Calcium perchlorate carries the chemical formula Ca(ClO4)2. You see those numbers and letters together, and it might just look like a line out of a college textbook. But every piece of that formula means something about how the substance behaves and where it shows up in daily life. The calcium ion, Ca2+, pairs with two perchlorate ions, each of which looks like ClO4-. Together, you get a white, crystalline salt that dissolves quickly in water and brings a strong oxidizing kick—something I learned firsthand in chemistry class, after an accidental small spill sizzled when mixed with the wrong material.
Why Knowing This Formula Is Worth Your Time
At first glance, it might seem trivial. But knowing the formula makes a direct difference in how people handle, store, and use this compound. Calcium perchlorate’s oxidizing ability belongs in rocket propellants, fireworks, and some disinfectants. Anyone working in those spaces needs a strong grasp on its behavior, and that starts with its formula. Misunderstandings can lead to serious safety mistakes. In 2015, a warehouse fire in China involved several oxidizers and caught workers off guard. Not knowing the chemical makeup of what’s being handled can turn a regular workday into a disaster.
Risks Tied to Calcium Perchlorate
My own work in industrial safety has shown just how easy it is for substances like calcium perchlorate to cause trouble. Its ability to release oxygen when heated makes it powerful—but also dangerous in the wrong hands. The Environmental Protection Agency points to perchlorate as a contaminant of concern. If it gets into water supplies, it can disrupt thyroid function, which is especially risky for infants and pregnant women. The formula guides researchers and water treatment professionals in tracking and removing perchlorate contamination from drinking water. Without accurate chemical identification, efforts can miss the mark entirely.
Safe Handling and Regulation
Handling a substance like calcium perchlorate isn’t just about gloves and goggles. It’s about understanding what reactions can occur, what storage methods are safe, and what legal standards exist. The Occupational Safety and Health Administration (OSHA) enforces strict guidelines for oxidizers in the workplace. Knowing the specific formula means professionals can look up the exact requirements, from storage temperature to compatible materials. In my own experience, clear chemical labeling, regular training, and up-to-date information make a difference on the ground, keeping both teams and facilities safe. The more we understand the underlying chemistry, the better decisions we make in real situations.
Real Solutions for Real Problems
Ensuring safety with calcium perchlorate starts with education. Trainers need to emphasize not just the formula, but what it means in terms of chemical reactions and hazards. At the policy level, clearer regulations can help keep this and similar oxidizers out of vulnerable environments. For communities facing contamination, water treatment facilities have found success with ion exchange systems and bioreactors—approaches that depend on precisely understanding the chemistry behind the contaminant.
Final Take
The formula Ca(ClO4)2 isn’t just memorization material. It’s a crucial tool for safety, public health, and industry. By focusing on what this formula truly represents, from risk to innovation, communities build resilience against chemical hazards and find better ways to protect workers, families, and the environment.
Is Calcium Perchlorate soluble in water?
Everyday Curiosity and Practical Science
Watching my high school chemistry class, I remember adding all kinds of salts to water. Some just vanished. Others clumped up, refusing to budge. Students expect salt to blend with water like sugar in tea, but not every material follows that rule. Out comes calcium perchlorate, a chemical some folks haven’t heard much about.
This salt, made from calcium and perchlorate ions, doesn’t shy from water. Pour it in and you won't spot it for long—water grabs those ions fast, separating them with ease. The reason takes us to the core of chemistry: electric charges attract, and water loves latching onto ions floating around. Each perchlorate ion dances away from the brand new calcium ion as soon as contact happens.
Solubility Isn’t Just for the Lab
It’s not just curiosity or science class fun. Calcium perchlorate’s reputation for dissolving sparks many industries. Firefighters, for example, use perchlorates to fuel chemical oxygen generators. That’s life-saving work, counting on the stuff’s ability to dissolve smoothly, react on cue, and then disappear into solution.
Astronauts have another way to look at this compound. In planning for Mars missions, engineers and scientists examine Mars’s regolith and perchlorate salts buried in the soil. The catch? With calcium perchlorate’s solubility, Mars explorers could pull water right out of the ground using salt—an ancient trick brought to space age frontiers.
Is It Really That Soluble?
Looking up the actual numbers, calcium perchlorate dissolves extremely well. At room temperature, it dissolves at over 160 grams per 100 milliliters of water. Compare that to common table salt, which caps out at about 36 grams per 100 milliliters. Staggering difference. With that kind of ability, chemists often use calcium perchlorate any time they need a strong source of calcium ions where clogs or slow dissolving would mess things up.
Risks and Responsibility
Strong solubility also brings a shovel of risk. Perchlorate ions mix into ground and drinking water more easily than less soluble salts. That’s where public health steps in. Perchlorate can block iodine from entering the thyroid, which changes hormone levels. Young children and pregnant women run higher risks. Regulatory agencies set limits for perchlorate in water, but once it’s out there, cleaning it up takes work—reverse osmosis, ion exchange, advanced biodegradation. Every step needs oversight and investment.
Thinking Toward Solutions
My own experience in a water quality lab reminds me that the most powerful tools against perchlorate pollution lie in monitoring, quick response, and being honest about where perchlorates go after use. Many researchers are searching for new bacteria that munch on perchlorate, turning it into harmless chloride. These bioremediation tricks have promise, but cost slows adoption. It’s worth pushing for more funding, more testing, and better rules on where and how industries handle these salts.
Every dissolved grain of calcium perchlorate tells a story—science, industry, and the bits of daily life that count on smart chemistry balancing practical use with health. For anyone touching this salt, knowing about its enthusiastic dissolving habit isn’t just an answer on an exam but an invitation to look deeper at how chemistry touches water, life, and responsibility.
| Names | |
| Preferred IUPAC name | Calcium perchlorate |
| Other names |
Perchloric acid calcium salt Calcium diperchlorate Calcium perchlorate hydrate Calcium(II) perchlorate |
| Pronunciation | /ˈkæl.si.əm pərˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 13477-36-6 |
| Beilstein Reference | 1201345 |
| ChEBI | CHEBI:63354 |
| ChEMBL | CHEMBL3341502 |
| ChemSpider | 153164 |
| DrugBank | DB15625 |
| ECHA InfoCard | 03b0d832-550a-4687-935f-86447b08142d |
| EC Number | 231-842-9 |
| Gmelin Reference | 82213 |
| KEGG | C18635 |
| MeSH | D018122 |
| PubChem CID | 24827 |
| RTECS number | FH8575000 |
| UNII | W0H8SR04BR |
| UN number | UN1457 |
| Properties | |
| Chemical formula | Ca(ClO4)2 |
| Molar mass | 238.98 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.71 g/cm³ |
| Solubility in water | Very soluble |
| log P | -2.0 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 8.38 |
| Magnetic susceptibility (χ) | +36.0e-6 cm³/mol |
| Refractive index (nD) | 1.44 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 239.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -795.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS03,GHS05 |
| Signal word | Danger |
| Hazard statements | H271, H272, H302, H319, H335 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-1-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 1173 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Calcium Perchlorate: "382 mg/kg (oral, rat) |
| NIOSH | SC9825000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | Store at 2°C to 8°C |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Perchloric acid Magnesium perchlorate Potassium perchlorate Sodium perchlorate |

