Calcium Nitrite: A Closer Look
Historical Development
Manufacturers have relied on calcium nitrite for nearly a century, tracing back to the earliest struggles against concrete corrosion and rebar decay. Concrete engineers in the mid-1900s, bracing for urban expansion, discovered calcium nitrite’s value as a corrosion inhibitor at a time when cities desperately sought ways to extend the life of bridges and roadways. Major flooding and corrosive urban air were eating away at steel structures beneath the surface. Instead of letting that damage run unchecked, engineers at Japanese and American firms came upon calcium nitrite and put it to work preserving concrete investments. Tracking its adoption, research from the sixties shows countries all over the world grabbing onto the technology, trying to keep community infrastructure standing longer with every new mix design.
Product Overview
Calcium nitrite, often showing up as a white or light-yellow crystalline solid, is best known in construction circles as an admixture for concrete. Folks also use it in select chemical processes, but its value in infrastructure maintenance overshadows those roles. Anyone in the industry can spot the simple product: typically sold at high purity levels, often blended for easier dosing at batching plants, but the heart of the technology remains the nitrite anion guarding steel from rust. Manufacturers keep the focus sharp—pure substance, clear labeling, ready to pour straight into big concrete batches—from municipal projects to high-rise decks.
Physical & Chemical Properties
The substance comes out as flakes or granular bits, dissolving rapidly in water, turning up as a colorless solution. Calcium nitrite melts at about 390°C, stays stable under standard storage if kept dry, and isn’t volatile. Unlike several other nitrites, it carries relatively low hygroscopicity, lending itself well to packaging and long-term storage. Chemically, calcium nitrite holds Ca(NO2)2·xH2O as its main composition, splitting into calcium and nitrite ions in water. Teams in the lab confirm that the nitrite part does most of the heavy lifting, halting iron oxidation reactions that start rust. Counting on this interaction allows engineers to handle rebar corrosion aggressively, especially in high-chloride environments where saltwater invades the infrastructure.
Technical Specifications & Labeling
Industrial products usually list purity above 90%, with moisture content labeled carefully: many suppliers keep it below 1% to prevent accidental clumping. Typical labels mention compatibility with cementitious systems and warn about incompatibilities with ammonium compounds. Storage suggestions tend to stress watertight conditions, clear batch labeling, and warnings regarding interaction with acids or oxidizers. Regulations in the EU and US demand safety pictograms and emergency procedures right on each container, giving job sites the critical details on handling spills or exposures. Field operators handle bags and drums in line with chemical product standards used throughout the concrete industry.
Preparation Method
Production hinges on a straightforward chemical reaction—mixing calcium hydroxide (commonly known as slaked lime) with nitrous acid or a sodium nitrite solution creates calcium nitrite, shifting impurities into an insoluble phase so they can be filtered out. Most factories adopt a wet process, using continuous stirred tanks with temperature and pH tightly controlled. As calcium nitrite forms in solution, engineers use vacuum evaporation or spray drying to turn it into solid form. Research by chemical engineers has fine-tuned the process over decades, cutting waste, raising yield, and keeping contaminant levels extremely low, with waste streams often recycled for secondary products.
Chemical Reactions & Modifications
The real power of calcium nitrite as a corrosion inhibitor grows from its chemical behavior. Adding it to fresh concrete, the nitrite ions react with steel to form an iron oxide layer that blocks further rust, even if sodium chloride or other aggressive chemicals are around. This passive film shields the steel, making corrosion slow to a crawl—multiple university studies show a marked reduction in rusting rates over time. Chemists sometimes tweak calcium nitrite with other admixtures or blend it with compounds like sodium nitrite, adjusting reaction rates or enhancing compatibility with different types of cement. Modifications focus on balancing initial setting times and long-term corrosion resistance.
Synonyms & Product Names
Buyers may notice other trade names on drums, such as Nitrocal, Nitcal, or even just the more generic “concrete hardening additive.” Chemists refer to it in lab notes as dicalcium dinitrite or calcium nitrate(II), but commercial buyers mostly care about the reliability of the source. Regional suppliers operate under different branding, though the safety and quality standards converge due to international codes and specifications, especially on transportation documents moving through customs.
Safety & Operational Standards
On construction sites, safety officers develop precise guidelines to limit dust inhalation, eye contact, or accidental ingestion, even though calcium nitrite rates as a relatively low-toxicity chemical compared with other nitrites. Workers use nitrile gloves, dust masks, and sometimes eye protection, especially in poorly ventilated areas. Training emphasizes storing the product away from acids, flammable materials, or organic goods like flooring solvents. In case of spills, teams sweep up the material while dodging direct contact. Waste disposal meets national and EU hazardous chemical rules, sending waste to licensed plants equipped for high-nitrite loads.
Application Area
Construction draws the biggest share. Engineers blend calcium nitrite into bridge decks, tunnels, parking structures, and anywhere that deicing salts might sneak through the concrete cover. Rehabilitation projects on older highways trust it to restore steel integrity without tearing up and replacing whole structures. Some water treatment facilities add it to stave off microbial growth, and limited niche applications show up in specialist chemical manufacturing. Yet its legacy remains with roads and infrastructure—helping governments and contractors save billions by keeping reinforced concrete stable in the harshest climates.
Research & Development
Laboratories in Europe and East Asia lead the push to strengthen calcium nitrite’s role in next-generation concrete mixes. Studies published over the past few decades highlight progress in understanding mechanisms of steel passivation, breakthroughs in admixture compatibility, and efforts to combine calcium nitrite with new hybrid corrosion inhibitors. The industry presses for admixtures that work faster and last longer in increasingly aggressive marine and urban settings. Several active patents point to proprietary blends using nano-additives or surface treatments, built to further stretch the safe life of rebar. University labs team up with cement producers, running full climate-testing cycles to predict which formulas best resist salt, CO2, and freeze-thaw damage.
Toxicity Research
Toxicology panels rate calcium nitrite as far less hazardous than more reactive nitrites, though issues arise when large quantities enter water systems or if workers fail to respect safe handling rules. Animal models point to mild acute effects from ingestion but raise flags about nitrate-nitrite conversion risks in groundwater, especially in heavy industrial or agricultural districts. Health departments track environmental releases, set groundwater benchmarks, and require heavy site monitoring. Teams responding to accidental exposure watch for signs of methemoglobinemia, especially in children or livestock drinking contaminated water. Most large-scale users invest in spill containment, air monitoring, and worker health checks, limiting danger in practice.
Future Prospects
The demand for longer-lasting infrastructure gives calcium nitrite strong momentum. Governments everywhere face mounting costs from rusted-out bridges and water mains. Innovation centers zero in on refining admixture efficacy, building synergy with recycled cements, and blending with low-carbon building materials. Policy trends in green construction seed funds into R&D for inhibitors with a lighter footprint and greater performance under climate extremes. Some startups push for digital dosing solutions, tying batch plant operations to sensor feedback on-site. With every structural collapse, public and political appetite for durable concrete grows, propelling calcium nitrite deep into the plans for next-generation cities and replacement of aging highways worldwide.
What is calcium nitrite used for?
A Closer Look at Construction
Calcium nitrite often makes its way into the world of construction, especially in making concrete last longer. Folks who have worked on job sites in rough winters know that low temperatures and salty roads can ruin slabs, pillars, and bridges faster than most people imagine. Road salt and cold weather cause rebar inside concrete to rust, and the rust expands, cracking the concrete. Calcium nitrite steps in here. Mixed right into the wet concrete, it protects the steel inside from rust when conditions get tough.
Spending years walking construction sites has taught me the real value of good concrete additives. Long stretches of highway in northern climates show the difference between mixes with and without corrosion protection. The roads with calcium nitrite in the concrete hold up better, keeping the costly repairs away for several extra years. Industry research backs up these observations; the National Ready Mixed Concrete Association reports that anti-corrosion additives can double or triple the service life of reinforced concrete.
Going Beyond Concrete
Factories making fertilizers also use calcium nitrite. It works as a source of nitrogen, which helps crops grow strong, especially in soils that struggle to keep nutrients. Farmers facing poor harvests often see noticeable improvement when this kind of fertilizer enters rotation. Nitrogen helps plants build proteins and energy, leading to healthier crops and bigger yields.
The chemical isn’t just limited to plant growth. Water treatment plants keep tanks and pipes in better shape by adding calcium nitrite. The chemical helps stop bacteria from generating bad odors and prevents the pipes from turning rusty. Over the years, countless cities have switched to this type of protection after facing angry residents complaining about smelly water or brown stains in bathtubs. By focusing on simple solutions like these, water authorities build trust with their communities and deliver safer water.
Safety and Handling
Nothing about calcium nitrite is magic. It brings real benefits, but it comes with its own set of risks. Both construction workers and farmers need proper training before handling any bulk chemicals, and local rules set clear storage guidelines. Safety data from the Occupational Safety and Health Administration highlight the need for gloves and goggles to avoid skin and eye irritation, and it’s important to keep dust from spreading in confined spaces.
Room for Better Practices
Looking at the big picture, calcium nitrite can keep highways open, crops growing, and water clean. Yet, like every chemical in building and agriculture, smarter use matters. Construction firms benefit from regular team training, not just at hiring time, but as part of yearly refreshers. Farms and treatment plants do better when they document how much additive goes into the process, tracking weather, soil, and crop response over the seasons. The science behind these materials continues to grow, and we’re all better off when folks using them share what works—and what doesn’t—with their neighbors in the field.
Is calcium nitrite safe to handle?
Everyday Encounters With Industrial Chemicals
Calcium nitrite doesn’t show up in household kitchens, but you’ll find it at construction sites and in specialty labs. It helps protect reinforced concrete from corrosion, especially where salt and moisture threaten steel inside buildings and bridges. Most cement workers and chemical handlers will cross paths with it at some stage. Folks sometimes ask how safe it is, and there’s no shame in looking for solid information before using it at work.
How Calcium Nitrite Behaves
The white or light yellow granules look about as threatening as table salt. They dissolve easily in water and have no obvious smell. This makes the mistake of underestimating their hazards pretty common. Inhalation, accidental swallowing, or even skin contact bring real risks. Breathing in the dust can irritate the lungs and nose. Letting it stay on your skin too long sometimes leads to redness, itching, or even burns. If it gets in your eyes, pain and watering are likely, and severe exposure sometimes means longer-term eye issues without a good rinse.
What Happens Inside The Body
Eating or inhaling enough calcium nitrite is dangerous. The body turns nitrites into compounds that block red blood cells from carrying oxygen. Nurses and doctors call this methemoglobinemia. Early symptoms are blue lips or fingers, headaches, rapid heart rate, and shortness of breath. At higher doses, confusion, fatigue, or even collapse can come on quickly. Healthcare professionals use specific antidotes, but being careful in the first place beats a trip to the ER.
Worksite Realities and Safety Steps
Jobs handling dry chemicals always call for gloves and safety goggles, but a simple mask can prevent lung irritation. Hand washing starts feeling routine after every shift. Even experienced workers sometimes skip the step—and they usually regret it later. Training goes a long way. Teams that know chemical hazards stay safer than those that learn the hard way. Safety Data Sheets (SDS) lay out what to do in an accident, and anyone handling calcium nitrite ought to have read one at least once.
Environmental Impact
Spilled calcium nitrite poses problems outside of personal safety. Once it washes into groundwater or drains, it can break down into nitrites and nitrates. Those nitrates end up feeding algae, which choke rivers and lakes and rob fish and bugs of oxygen. Cleanup after a spill goes beyond just hosing things down. Sites should trap and dispose of runoff, rather than push problems further away. Proper storage—inside clearly labeled containers away from acids—makes leaks and accidents less likely.
Better Handling Practices Benefit Everyone
Good gear, training, and a bit of old-fashioned vigilance prevent most harm. Supervisors who reward safety, listen to team feedback, and walk the floor set the best example. Open talk about incidents, near misses, and better ways to work help everyone keep out of trouble. Those new to chemistry should never assume a chemical’s harmless just because it’s common. Your skin and your lungs will thank you for a careful approach—no shortcuts, no guessing. The price of ignoring safety is always far higher than a pair of gloves and a bucket of soapy water.
How does calcium nitrite prevent corrosion?
Corrosion: The Silent Enemy
Ask anyone who’s worked with concrete rebar or buried steel piping—they’ll tell you that rust never sleeps. Catching corrosion early—or better yet, keeping it from starting—matters a lot. Crumbling parking garages, cracked bridges, and failing utility pipes often have a common root: rusted steel inside what’s supposed to be a solid structure. I’ve seen repairs cost many times more than the original job. Nobody likes dealing with hidden bills, so stopping corrosion before trouble begins always struck me as common sense.
Where Calcium Nitrite Steps In
Lots of folks use coatings, sealers, or stainless steel to fight corrosion, but price and practicality rule those out for big jobs. Calcium nitrite doesn’t work as a paint or wrapper; it gets mixed straight into concrete. Once it’s in the mix, it doesn’t wash away or peel off. Most folks only notice its effect after decades, when reinforced concrete lasts longer than expected.
What stands out about calcium nitrite is its hands-on, chemical way of protecting rebar. Try pouring a slab without it in a place that gets a lot of salt—coastal towns or snowy climates using road salt—and you’ll see rusty streaks and spalling sooner or later. I’ve walked through older parking garages, and the worst damage always traces back to salt-laden water seeping through cracks and working its way to the steel inside.
The Chemistry in Simple Terms
Moisture and oxygen working together eat away at steel—that’s the nutshell version of corrosion. Add road salt, and things go bad even faster. Steel in concrete would usually last decades because the concrete stays quite alkaline at first, creating an invisible shield from rust. Over time, cracks form and open the door for salt and water to reach the rebar. That’s where the trouble brews.
Calcium nitrite interrupts this slow-motion disaster. It reacts with iron at the steel’s surface and helps form a much tougher, more stable “oxide” layer. This layer acts like a lock on the door, stopping the salty water and oxygen from chewing up the steel underneath. In real-world terms, concrete lasts longer—often decades longer—if you add the right amount of calcium nitrite early on.
Real Impact on Infrastructure
Folks running cities and highways watch budgets, but they also look at failures that can’t happen—collapsed overpasses, burst water mains, or corroded supports under a school. So using calcium nitrite might cost more up front, but it often means fewer headaches later. Reports from highway departments and civil engineers back this up. The Portland Cement Association and the Federal Highway Administration both note significantly less corrosion in treated concrete.
Some experts warn that just dumping in inhibitors isn’t enough. Good construction habits—no shortcuts, careful finishing, plus quality aggregates—make the biggest difference. Still, experience says that calcium nitrite buys time and peace of mind, especially where salt or moisture build up fast.
Better Solutions on the Horizon?
People keep searching for easier or cheaper fixes—coatings that last longer, smarter concrete mixes, or even new materials that avoid steel altogether. In my own work, nothing has matched the track record of calcium nitrite for old-fashioned durability. Until something better comes along, most folks in construction view it as one of the simplest, most reliable bets for protecting concrete that needs to last.
What is the recommended dosage of calcium nitrite in concrete?
Why Concrete Needs a Boost
Concrete does some heavy lifting around us, but harsh conditions can tear it up fast. Chlorides from de-icing salts or seawater sneak in, and before long, steel inside the slabs starts rusting. That’s bad news for bridges, parking garages, and buildings near coastlines. Engineers started reaching for calcium nitrite because it slows down corrosion, helping steel reinforcement last longer. Think of it as a shot of vitamins for concrete—helpful, but only at the right dose.
Dosage That Delivers Results
The amount of calcium nitrite used in concrete isn’t just a figure pulled out of a hat. Research carried out by the American Concrete Institute, along with real-world experience on construction sites, points to a practical range. Most of the time, people on the job add between 1% to 2% calcium nitrite by weight of cement. This comes out to roughly 15–30 liters per cubic meter, making it possible to get a real increase in corrosion protection without changing how the concrete pours or cures. Contractors say that going below 1% won’t give enough protection, while loading in more than about 3% starts to mess with the set time and can trigger cracking or shrinkage.
Dosage Depends on the Situation
No two projects look quite the same. Highway bridges over salted roads need more protection than a basic warehouse floor. Cold regions require extra attention since freezing temperatures extend set times. Chloride exposure matters most—locations right by the ocean or busy city roads usually call for the higher end of the dosage range. Scientists and engineers run lab tests to measure chloride content, aiming to keep it under certain levels that research shows won’t cause trouble down the line. By knowing the local threat and reading up on the cement chemistry, they make smarter calls about how much calcium nitrite it takes to protect the steel inside.
Mixing for Strength and Longevity
Getting the amount right means better performance, but adding calcium nitrite changes how the concrete behaves before it hardens. It speeds up hydration, which is great in chilly weather and helps finishers get the job done even in cold snaps. I remember a winter job on a hospital deck where dosing calcium nitrite let us finish days ahead of schedule. Yet, adding too much leads to rapid stiffening, trapping air and shrinking too quickly, which can leave the final slab weaker than intended. Consistency in mixing during batch preparation counts for a lot, as uneven distribution leads to dead spots and patchy corrosion protection.
Smarter Building with Chemistry
While calcium nitrite gives concrete steel a fighting chance against rust, it’s not the only answer. Pairing it with low water-to-cement ratios, high-quality aggregates, and thorough curing leads to better results. Teams who check their materials, watch the chloride content, and follow up with field testing after construction spot early problems and keep structures standing strong. Construction codes in North America, Europe, and Asia now recommend calcium nitrite for certain chloride-exposed structures, but every code reminds builders to tailor dosages for each site instead of picking a one-size-fits-all number.
Looking Ahead: Safer Structures Start with Informed Choices
Smart use of calcium nitrite in concrete hinges on good site assessment, clear local guidelines, and quality control from the mixing plant to the finishers on the slab. Workers with hands-on experience share tips and watch for issues, bringing book knowledge and practical insight together. For lasting roads, bridges, and buildings, the right dose starts with clear information and an eye on real-world results.
Does calcium nitrite react with other chemicals in mixtures?
The Real Deal Behind Calcium Nitrite in Chemical Mixtures
Most folks outside the concrete or construction business might not think much about calcium nitrite. This chemical finds a place in work gloves, specialty fertilizers, and concrete additives. It keeps rebar from rusting in bridges, tunnels, and parking slabs, letting those structures last a lot longer. The catch is, in complex mixes or new product formulas, calcium nitrite doesn’t just sit quietly— it reacts, sometimes helping the process, other times making headaches for everyone involved.
Reactivity in Cement and Concrete
In concrete, calcium nitrite acts as a corrosion inhibitor. It mixes with chloride ions and protects steel reinforcement. But things get wild if you add materials like aluminum powder, sulfates, or certain plasticizers. Several research papers, including a 2021 study in the Cement and Concrete Research journal, show that sulphate ions can react with calcium nitrite and turn it into calcium sulfate and nitrous acid. This might upset set times and durability. I’ve seen pours where unexpected expansion or prolonged set times traced back to surprise chemical reactions. The science backs it up: calcium nitrite and reactive aluminates spark extra heat and, if dose rates get sloppy, microcracking.
Interactions in Agricultural and Industrial Settings
In the ag world, calcium nitrite sometimes steps in as a nitrogen source. It shows up in specialty fertilizers mixed with phosphorus or potassium salts. But there’s a big no-no here. Mixing with ammonium phosphates can sometimes lead to the formation of nitrosamines, which are tightly regulated because of health risks. EPA flagged these reactions as early as the 1980s. Better to ask twice and mix once before tossing chemicals in a tank together.
Water Treatment: Safety and Surprises
People use calcium nitrite in corrosion control for municipal water pipes. The fun begins when you mix it with strong acids or oxidizers. That’s where you risk releasing toxic nitrogen oxides. If handled carelessly, inhaling those gases can land someone in the ER. Water authorities have learned the hard way that calcium nitrite, though stable on its own, needs a careful hand in treatment plants already dosed with variable pH or chlorine levels.
Solutions: More Than Labels and Warnings
It’s tempting to tell folks just to read labels or lean on data sheets, but real safety grows from a culture of curiosity and caution. Manufacturers should run compatibility checks on new product formulations, especially if calcium nitrite is part of the mix. Regular lab testing, clearly marked storage bins, and plain-language warnings make a real difference. In the field, team training, peer review, and old-fashioned troubleshooting save time, money, and sometimes lives. No one wants to pour out a hundred cubic yards of concrete, only to watch it crack up because a chemical mix went sideways. Paying attention to calcium nitrite doesn’t just protect rebar; it protects budgets, health, and sometimes reputations. Mixing chemicals isn’t guesswork—it’s applied chemistry, shaped by experience and respect for what can go right, and what can go wrong.
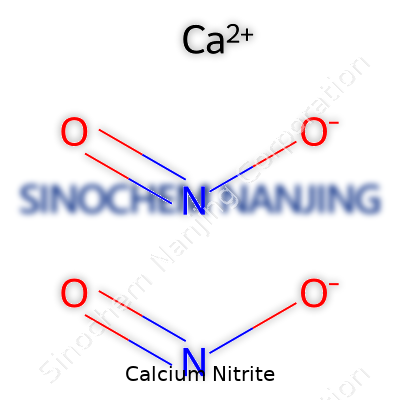
| Names | |
| Preferred IUPAC name | Calcium dinitrite |
| Other names |
Nitrocalcite Calcium dinitrate Ca(NO2)2 Calcium nitrous oxide |
| Pronunciation | /ˈkæl.si.əm ˈnaɪ.traɪt/ |
| Identifiers | |
| CAS Number | 13780-06-8 |
| Beilstein Reference | 63571 |
| ChEBI | CHEBI:32599 |
| ChEMBL | CHEMBL1201591 |
| ChemSpider | 61714 |
| DrugBank | DB11360 |
| ECHA InfoCard | ECHA InfoCard: 03-2119474445-40-0000 |
| EC Number | '233-332-1' |
| Gmelin Reference | Gmelin Reference: 14951 |
| KEGG | C01764 |
| MeSH | D015325 |
| PubChem CID | 24963 |
| RTECS number | NK9300000 |
| UNII | 6ZB63G2Z8H |
| UN number | UN1454 |
| Properties | |
| Chemical formula | Ca(NO2)2 |
| Molar mass | 132.09 g/mol |
| Appearance | White to yellowish crystalline solid |
| Odor | Odorless |
| Density | 2.5 g/cm³ |
| Solubility in water | 550 g/L (20 °C) |
| log P | -3.57 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 10.62 |
| Magnetic susceptibility (χ) | −31.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.45 |
| Viscosity | 1.45 mPa·s (at 20 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 133.0 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | −430.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -420.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P330, P304+P340, P312, P305+P351+P338, P337+P313, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2 |
| Lethal dose or concentration | LD50 Oral Rat 240 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 175 mg/kg |
| NIOSH | SCG |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 200 ppm |
| Related compounds | |
| Related compounds |
Sodium nitrite Potassium nitrite Calcium nitrate Magnesium nitrite |

