Calcium Nitrate: From Origins to Modern Applications
Historical Development
Centuries ago, people noticed that certain saltpeter soils gave better crop yields. Digging deeper, early chemists recognized minerals rich in nitrogen had something to do with healthy fields. This curiosity led to discoveries in the 19th century. At that time, nitrogen fixation became a hot topic as scientists worked to crack the secrets behind synthetic fertilizers. Christian Friedrich Schönbein, famed for his work on gun cotton, also played a role in advancing nitrogen-based compounds. By the start of the 20th century, industry giants such as Norsk Hydro in Norway put calcium nitrate into the world’s hands, helping farmers push past the limits of traditional manure and compost. The appearance of the Odda process in 1927, which extracted nutrients from rock phosphate using nitric acid, cemented the role of calcium nitrate as a by-product, turning a byproduct of Norway’s hydropower industry into a global staple.
Product Overview
Calcium nitrate stands out in the line-up of agricultural and industrial chemicals thanks to its ability to offer both calcium and nitrogen in one package. In my own trips through fields and greenhouses, I’ve seen it packaged as white pellets or prilled granules, marked by their ease of spreading and quick water solubility. In hydroponic setups, growers use it as a key nutrient, mixing it with other salts to keep their tomatoes and lettuces green and upright. Huge factories keep up with demand by turning out millions of tons yearly, serving not just farms, but also concrete plants, wastewater treatment sites, and even refrigeration systems. Each bag follows technical standards that help users track the source, content, and handling rules.
Physical & Chemical Properties
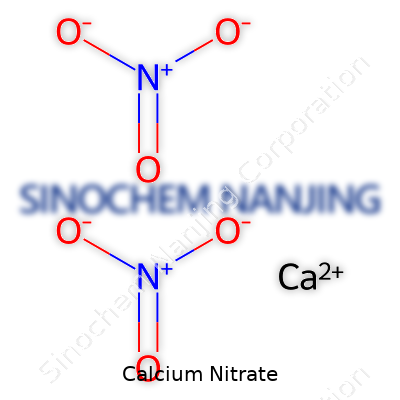
Holding calcium nitrate in hand, you’d notice a colorless or white crystal, dry to the touch, thanks to its hygroscopic nature. This means it draws moisture from the air, clumping quickly unless sealed. The chemical formula, Ca(NO3)2, reveals each molecule carries two nitrate ions alongside one calcium atom. It dissolves almost instantly in water, spiking both calcium and nitrate concentrations with little fuss. It resists burning but will energize flames if mixed with combustible materials due to its oxidizing properties. Its melting point sits at about 561°C, but you’ll rarely see it near that heat unless you’ve trashed your containment protocols. The density falls near 2.36 g/cm3 for the anhydrous form, and it takes on extra water to form a tetrahydrate structure in humid storage.
Technical Specifications & Labeling
Walking into any agricultural supply store, a bag of calcium nitrate carries a wealth of information on its label. Most products in the market advertise a nitrogen content near 15.5% and a calcium oxides content close to 26%, speaking directly to growers looking to target nutrient deficiencies. These figures come backed up by batch analysis, with labels listing both minimum guaranteed levels and acceptable impurity thresholds—think sodium, magnesium, and insoluble residue. International standards such as ISO 8157 guide these specs, and labeling laws demand the mention of water solubility and shelf life due to the product's sensitivity to moisture. Shipping labels often show hazard pictograms for oxidizers and warnings about storage away from fuels, acids, and organics.
Preparation Method
Modern calcium nitrate production leans heavily on the reaction of limestone, nitric acid, and ammonia. In the Odda process, mixing phosphate rock with nitric acid sets off a reaction that produces phosphoric acid, leaving calcium nitrate behind in solution. Some facilities tweak this workflow, feeding calcium carbonate and nitric acid directly without the phosphate step. Large vats churn and bubble as the acid strips calcium from stone, then filtration and evaporation steps concentrate the product. Technicians then granulate or prill the solution, cooling it in towers or drum systems to form market-ready beads or flakes. Thorough washing and drying ensure purity and reduce the risk of caking during storage.
Chemical Reactions & Modifications
In the lab, calcium nitrate behaves predictably but opens possibilities for tailored reactions. Mix it with sodium carbonate, and you gain insoluble calcium carbonate and sodium nitrate—handy for both educators and industry. In fertilizer blending plants, it teams up with ammonium nitrate, urea, or potassium salts to make compound fertilizers. In construction, it reacts in fresh concrete, limiting sulfate attack and reduces the risk of corrosion on steel rebar. Several researchers manipulate its chemistry to craft calcium ammonium nitrate, a favorite in European fields, by adding ammonium nitrate to the manufacturing stream.
Synonyms & Product Names
Over the years, calcium nitrate picked up several names. Some circles call it lime nitrate or Norwegian saltpeter, a nod to its early Scandinavian roots. In the fertilizer trade, it’s known as CN, Nitrocal, or even CalNit, depending on the marketing spin and the country of origin. Look at MSDS sheets, and you’ll spot the CAS number 10124-37-5, helping buyers avoid mix-ups with similar-sounding compounds in vast chemical inventories. Industrial buyers sometimes order it as double salt variants, such as calcium ammonium nitrate, depending on regulatory and agronomic needs.
Safety & Operational Standards
While not explosive on its own, calcium nitrate deserves respect because of its oxidizing punch. Incidents on fertilizer docks and in storerooms usually stem from careless mixing with fuels, organics, or acids. Good safety training calls for keeping it dry, stored in airtight bins under roofs, and away from any potential sources of ignition or incompatible chemicals. Respirators and gloves show up in safety manuals because the dust can irritate lungs and skin. Emergency plans require nearby eyewash stations, and workers should always wash up at shift’s end. Regulating agencies such as OSHA and the EU’s REACH provide guidance, setting limits for employee exposure and storage quantities in warehouses.
Application Area
Farming claims most of the calcium nitrate shipped each year. In my days consulting for greenhouse growers, I’ve seen it mixed with main irrigation water feeds to fix calcium deficiencies, which often show up as blossom end rot in tomatoes or tip burn in lettuces. Outdoor growers use it in fertigation and side-dress programs, rescuing crops from stunted growth or leaf yellowing. In civil engineering, concrete producers add calcium nitrate to new mixes to cut curing times in cold weather and slow rebar corrosion. Wastewater treatment plants rely on it to reduce hydrogen sulfide emissions, which cause the rotten egg smell lurking in sewers. Some refrigeration systems use it for brine solutions because its high solubility chills water more efficiently. Fireworks producers even use it to boost the oxidizer content in their colored flames.
Research & Development
Universities, public agencies, and agrochemical firms pour funds into calcium nitrate research, especially as global food supply pressure mounts. Greenhouse crop studies dig into optimum dosing to avoid tip burn while maximizing yield and shelf life. Environmental scientists examine runoff, looking for nitrate leaching into watersheds and strategizing ways to reduce it through controlled-release formulations. There’s work underway to design biodegradable coatings that slow down dissolution and lower the need for frequent reapplication. Some research teams tune calcium nitrate use to limit heavy metal uptake in urban farming plots, a critical issue in reclaimed land projects. Teams studying concrete chemistry say calcium nitrate can lengthen the lifespan of bridges and tunnels, and prototype mixes offer longer resistance to freeze-thaw cycles.
Toxicity Research
While calcium nitrate is generally low in toxicity, long-term studies track environmental and health impacts, especially for workers and communities near industrial sites. Too much exposure to the dust can irritate skin, eyes, and mucous membranes. Chronic overexposure poses risks for individuals with existing respiratory issues. Researchers monitor drinking water in farming regions for nitrate levels, due to the link between nitrate-rich water and methemoglobinemia, particularly in infants (so-called blue baby syndrome). Regulatory agencies enforce strict guidelines for fertilizer handling and groundwater monitoring, with limits designed to keep concentrations below thresholds that can spark public health scares. Livestock studies try to pin down safe upper limits, as some foraging animals can’t handle high nitrates and show signs of digestive upset or even toxicosis.
Future Prospects
Looking forward, calcium nitrate seems likely to ride new waves of agricultural technology and sustainability efforts. Precision fertigation systems integrate real-time soil and plant data, calling on calcium nitrate to match crop needs hour by hour, which cuts both waste and environmental risk. On the product innovation side, companies explore new prilling technologies and water-resistant coatings, allowing for single applications that last through the growing season. Regulatory changes push factories to invest in even cleaner manufacturing, with energy efficiency and waste recycling as central concerns. Researchers continue to probe for ways to keep nitrate out of rivers, testing bioreactors and cover crop tactics. Across all sectors, pressure continues for products that do their job without polluting food, soil, or water. The story of calcium nitrate runs parallel with the world’s quest for healthy, productive, and resilient food and infrastructure systems.
What is Calcium Nitrate used for?
A Closer Look At Everyday Uses
Many people know calcium nitrate as a fertilizer on farms, but its story doesn't stop there. I’ve seen plenty of folks wonder why this compound keeps popping up in so many places, from the food you eat to the water coming out of your tap. Its uses stretch into several corners of life, and not only for big industrialists or scientists in white coats. Whether you're tending tomatoes or keeping your city’s water clean, chances are you’ve quietly benefited from it.
The Farming World’s Go-To Choice
In agriculture, plants need calcium to hold their cell walls together and nitrogen to keep growing strong. Sometimes, soil just runs low on both. Calcium nitrate steps in to tackle this gap. I remember walking through vegetable greenhouses and seeing workers mixing it into the water for their crops. Tomatoes, lettuce, and peppers especially depend on the extra calcium to avoid problems like blossom-end rot, where the bottom of fruit turns black and mushy. Crops get both nutrients in one swoop without messing up the soil’s salt balance—important for repeated harvests. The demand for it grows wherever growers want fast results with fewer headaches.
Helping City Water Stay Safe
Few folks realize this, but calcium nitrate plays a part in the fight against those rotten-egg odors in municipal water. Sewer pipes can build up hydrogen sulfide gas, making neighborhoods smell bad and damaging metal infrastructure. By adding calcium nitrate into the wastewater lines, city engineers get bacteria to use up nitrate instead of making smelly compounds, so the air is cleaner and pipes need fewer repairs. These changes don’t just help downtown; they improve the quality of life in every community.
Role In Concrete And Construction
Spend any time at a construction site in cold weather, and you’ll see why workers pay attention to what goes in their concrete mix. Calcium nitrate helps concrete cure faster and harder when it’s chilly and humid. It keeps the steel inside from rusting by fighting off chloride attacks. That means bridges and tunnels end up safer and last longer. Skipping this step has caused some big headaches with cracks and corrosion, so adding it just makes sense.
Food Additive And Beyond
You won’t see it mentioned every day, but food-makers sometimes include calcium nitrate as a firming agent. Certain cheeses and canned vegetables keep their texture because of it. Rules keep it at safe levels, and proper handling matters—the food industry tracks this closely because nobody wants unnecessary chemicals on dinner plates. Health agencies worldwide keep reviewing studies to ensure it remains safe, and so far, a track record shows limited risk when used properly.
What Lies Ahead
As cities get bigger and crops face new pressures, the need for smart soil and water management tools grows. Calcium nitrate continues to pull its weight across fields, treatment plants, and building projects. Rather than chase a miracle, making sure everyone—from farmers to city planners—knows how to use it wisely goes a long way. Places that encourage responsible application see better harvests, safer infrastructure, and fewer problems with pollution or waste. If science finds even safer or more effective substitutes in the future, that's great. Until then, the stuff sticks around for a reason.
Is Calcium Nitrate safe for plants?
Understanding Calcium Nitrate in the Garden
People often reach for calcium nitrate to feed their plants or fix deficiencies. It has a mix of calcium and nitrogen, both of which plants crave. Tomatoes, peppers, and leafy greens all seem to perk up when they get a dose of this fertilizer. My own vegetable patch produces firmer, healthier tomatoes if I keep the soil stocked with calcium and nitrogen, and this product delivers both.
The Role of Calcium and Nitrate
Calcium isn’t just for strong human bones. Plants use it for cell wall formation. Gardeners might notice blossom end rot on tomatoes—a condition linked to calcium scarcity. Calcium nitrate stops this in its tracks because it brings immediate, plant-available calcium. The nitrate part comes into play as the main form of nitrogen plants use for robust green growth. Whether in sandy or loamy soil, both nutrients help most crops thrive.
Is It Safe?
Most commercial greenhouse operations rely on calcium nitrate for years, showing that it can be safe when applied with some care. Overuse can build up salts in the soil or runoff into waterways. That’s the real concern. I’ve seen gardeners thinking more is better, but a heavy hand can lead to leaf burn or stunted roots. So, the safety of calcium nitrate hinges on responsible use. Gardeners benefit from reading labels, following dosing instructions, and testing soil before adding anything extra.
Supporting Evidence and Risks
University tests back up its benefits. Research from trusted institutions like the University of California shows that calcium nitrate treats deficiencies quickly, with only rare side effects. It breaks down rapidly, and plants pull up the calcium almost as soon as it dissolves in the soil water.
Still, certain risks stand out. Large spills or careless handling can leach into groundwater. Runoff loaded with fertilizer poses a threat to ponds or streams nearby, leading to excess algae or changes in water quality. This isn’t just a backyard problem. Larger farms face regulations on fertilizer runoff for good reason.
How to Use Calcium Nitrate Responsibly
Home gardeners who test soil get a clearer picture of what their crops actually need. Low calcium or nitrogen calls for supplementation. Some crops, such as potatoes and tomatoes, tend to have higher calcium needs and show clear improvement once corrected.
It’s smart to use small doses, spread evenly over the growing season. Water the fertilizer in thoroughly. Never layer it dry near roots or pile it up in one spot. I try to time my applications for early morning, so the plants can take up the nutrients steadily. Clean up spilled pellets or dust on walkways, and store the bag in a dry place away from pets or children.
The Bigger Picture of Healthy Soil
Long-term soil health means more than just dumping additives. Compost, crop rotation, and cover crops chip away at long-term deficiencies without relying only on any one chemical. Calcium nitrate works well as a targeted fix, not a crutch for poor soil management. By treating soil amendments as part of a tool kit, gardeners give themselves the best chance for abundant, healthy produce every year.
Practical Solutions for Everyday Growers
A smart approach to calcium nitrate involves measuring, monitoring, and correcting deficiencies as they happen. Regular soil tests, proper dosage, and keeping an eye on runoff offer peace of mind. Safe for plants? Yes, absolutely, with thoughtful use and attention to the bigger ecosystem in the garden.
How do you apply Calcium Nitrate fertilizer?
Understanding Why Calcium Nitrate Matters
Gardeners and farmers often deal with blossom end rot in tomatoes, tip burn in lettuce, or general yellowing of leaves. A fair bit of that headache traces back to a calcium deficiency. I’ve spent years troubleshooting stunted growth in both backyard gardens and larger fields. Calcium nitrate delivers a double punch: essential calcium for cell wall strength and soluble nitrogen for leaf growth. That combo gives plants more resilience and bounce.
Applying Calcium Nitrate: Don’t Overthink It
If you’re handling vegetable patches, scatter about one tablespoon of calcium nitrate near the base of each plant every two to four weeks during the growing season. Lightly scratch the granules into the topsoil and water thoroughly. Roots pick up nutrients best when soil moisture stays consistent, not soggy. I’ve tried both early and late afternoon feedings, but morning seems to work out—plants are ready for a drink after a cool night.
For larger crops like potatoes or corn, side-dress in shallow trenches four to six inches away from the main stems. That keeps the fertilizer close enough for roots to grab, without scorching them. In bigger operations, it mixes easily in standard spreaders. Watch for dry spells—if there’s not enough rain, use a hose or irrigation to dissolve the granules into the ground.
Getting Calcium Uptake Right
Just dumping fertilizer rarely fixes serious nutrient gaps. A quick soil test tells you what’s missing and helps avoid waste. Balancing soil pH hovers around 6.5 for most crops. I learned the hard way on my first attempt: too much acidity locks up calcium, too much alkalinity and the nitrate may leach away. Reputable labs or extension services can run a straightforward analysis.
Calcium nitrate dissolves fast. That makes it one of the best choices for “foliar feeding” when you’re dealing with clear symptoms like curled leaves or fruit spots. Mix a tablespoon per gallon of water and mist leaves deeply, aiming for early mornings. Keep an eye out for leaf burn—too strong a solution under hot sun can do more harm than good.
Common Pitfalls and Solutions
Overapplication leads to nitrate pollution, so more isn’t always better. Moderate, regular feeding gets results. In sandy soils or heavy rain regions, it pays to give smaller, more frequent doses rather than one heavy shot. For clay-heavy plots, add sharp sand or compost to loosen up compacted areas. Healthier roots get better access to every dose.
Plants like tomatoes, peppers, and cucumbers show fast improvement with calcium nitrate, especially during fruit setting. Flowers drop less and fruit quality jumps. In my plots, the difference pops out within a couple of weeks. Farmers report steadier yields, fewer disease problems, and stronger crops—outcomes supported by research at universities like Michigan State and Cornell.
Do’s for Responsible Fertilizing
Wear gloves and avoid breathing dust, just for good measure. Store bags in a dry spot—humidity clumps up the product, making it harder to spread. Keep unused fertilizer out of reach of children and pets.
Building soil long-term calls for making compost and using crop rotation. Synthetic inputs like calcium nitrate give plants a strategic boost right away, but nothing matches what healthy, living soil can do over time.
Can Calcium Nitrate be mixed with other fertilizers?
Farmers Face a Chemistry Lesson in the Field
Anybody who has ever fed a tomato patch knows that fertilizer isn’t one-size-fits-all. Calcium nitrate solves two big problems, delivering both calcium and ammonia-free nitrogen. Young leaves stay green, and blossom-end rot rarely appears when plants have enough of these. Still, I’ve seen more than one grower run into trouble after combining fertilizers the wrong way.
Why Mix Fertilizers in the First Place?
Blending fertilizers can save time, and it may seem easier to hit nutrient targets with a single mix in the tank. Some folks also want to tailor their soil or irrigation water by tweaking blends to match specific crop needs. Calcium nitrate looks like a friendly ingredient in those blends. But the chemical game doesn’t stop at the bag.
The Problem with Phosphates and Sulfates
Mixing calcium nitrate directly with fertilizers containing phosphates or sulfates can create real headaches. Calcium ties up with phosphorus or sulfate, forming solid deposits right in your tank or irrigation lines. These clogs slow water flow and lead to wasted cash on fertilizer that plants never get to use.
Research from extension services in California and the Midwest highlights how easy it is to form gypsum (calcium sulfate) or calcium phosphate, both largely insoluble. These look like milk in your solution but settle as sand in lines and drippers. You end up with patchy feeding and extra maintenance.
Practical Mixing Advice from Experience
I learned quickly to keep calcium nitrate on its own, at least until it hits the soil. Most commercial growing operations use multiple tanks—one for calcium nitrate, another for the rest. That’s not just for show; it’s a lesson many picked up the hard way. If all you have is a single mix, experts and smart gardeners stagger irrigation or divide applications over a few days.
For home gardens, apply calcium nitrate separately from phosphate-based and sulfate fertilizers. Give it a few days between applications or water in well before layering something else on top. Most seasoned growers agree that keeping things simple keeps plants healthier and systems running without extra cleaning.
Supporting the Science
A study from the University of Florida demonstrated that separate application delivers better nutrient availability. Tomato plants fed via split applications showed stronger growth than those on a catch-all blend. This has shown up in my own small trial rows, where separate doses led to more even fruit set and less tip burn on leafy crops.
In controlled-environment agriculture, hydroponic growers almost always keep calcium nitrate and phosphate apart, thanks to years of clogged drippers and lost time. They learn from each other and trade tricks—like flushing lines between feeds or swapping out injection points—because these headaches slow down real production.
Solutions for Less Waste and More Growth
Everyone wants more production with less labor. Better education, clearer fertilizer labels, and more user-friendly reference guides for mixing can help. For commercial setups, investing in a simple two-tank injection system pays off within a season just by preventing costly line cleanup. For backyard gardeners, a little patience and careful scheduling are vital.
Calcium nitrate belongs in every grower's toolkit, but it doesn’t play nicely with every fertilizer. The best results come from respecting chemistry, spreading out applications, and not taking shortcuts. Plants—and wallets—end up healthier for it.
Where can I buy Calcium Nitrate?
Why People Care About Calcium Nitrate
Calcium nitrate shows up in all sorts of places. Many gardeners use it to fight blossom-end rot on tomatoes. Big farms depend on it as a calcium boost for soils that need a little something extra. Some folks working in construction add it to concrete to speed up curing times. So, buying the right bag or pail quickly turns into a quest for many types of people, from home growers to pros.
Common Places to Buy Calcium Nitrate
Big-box garden centers sometimes stock smaller bags in the garden fertilizer aisle, right near potassium nitrate or ammonium sulfate. Sometimes, smaller independent nurseries and hydroponic shops on the edge of town have it. Staff there may have useful tips about brands or the difference between prilled and liquid forms. Farm retail stores almost always carry it, because fields often need more calcium than nature offers.
Online shopping sometimes proves the most reliable way. Popular online retailers, garden supply websites, and even major auction or marketplace apps list many options. Some offer bulk deals, well-suited for small farms or hydroponic growers. It’s smart to read reviews carefully and make sure you’re buying from a known, professional seller. Illegitimate vendors sometimes try to repackage or mix products, risking quality and safety.
Regulations and Safety Around Sales
Some countries set limits on who can buy calcium nitrate, often because a person could misuse it to make dangerous materials. In the U.S., smaller bags remain freely available. Farmers and commercial users may face paperwork for larger volumes, especially in areas with tighter environmental controls.
Responsible sellers will ask buyers about intended use, or request details about their business or garden projects. I’ve had conversations with store managers who wouldn’t sell bulk nitrate unless I proved it was for tomatoes. They care about where fertilizers end up.
Handling and Storage
Calcium nitrate works well for its intended purpose, but it’s not something to leave open around kids or pets. I always keep containers on a high shelf in the garage, out of the sun, in a spot that stays dry. Moisture clumps it up, and that can mess with application rates or cause spills. Anyone thinking of using it in a food garden needs to measure and mix carefully, following label instructions. Local extension services often offer free booklets with safe handling tips.
Alternatives and Solutions
People sometimes grab calcium nitrate hoping it’ll fix every plant issue, but there are alternatives. Gypsum provides calcium without adding nitrate. For folks using it in DIY or hobby chemistry, talking with a science teacher or local chemistry club can uncover safer substitutes. If you find it tough to source, check with cooperative extensions, local farm bureaus, or even university greenhouses. Sharing orders with neighbors sometimes reduces shipping costs and quantity headaches.
Real-World Considerations
Few things beat seeing healthy tomato plants thrive thanks to the right nutrition. I’ve learned not every mailbox delivery is the same, and not all sellers treat safety or labeling honestly. Staying curious, asking questions, and reaching out to trusted experts goes further than just clicking “buy now.” In the end, finding and using calcium nitrate calls for a balance of practicality, safety sense, and a little local knowledge.
| Names | |
| Preferred IUPAC name | Calcium dinitrate |
| Other names |
Norwegian saltpeter Norgessalpeter Lime nitrate Nitrocalcite |
| Pronunciation | /ˈkæl.si.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10124-37-5 |
| Beilstein Reference | 320297 |
| ChEBI | CHEBI:32599 |
| ChEMBL | CHEMBL1201732 |
| ChemSpider | 14118 |
| DrugBank | DB09467 |
| ECHA InfoCard | ECHA InfoCard: 030-003-00-2 |
| EC Number | 233-332-1 |
| Gmelin Reference | 18781 |
| KEGG | C01242 |
| MeSH | D002121 |
| PubChem CID | 10182492 |
| RTECS number | CAS6434-01-9 |
| UNII | NRN9J0Y0BP |
| UN number | UN1454 |
| Properties | |
| Chemical formula | Ca(NO3)2 |
| Molar mass | 164.10 g/mol |
| Appearance | White to grayish, hygroscopic, granular solid |
| Odor | Odorless |
| Density | Density: 2.36 g/cm³ |
| Solubility in water | 1,211 g/L (20 °C) |
| log P | -2.29 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | −31.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.505 |
| Viscosity | Low viscosity |
| Dipole moment | 0 |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −940.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –941.2 kJ/mol |
| Pharmacology | |
| ATC code | V07BB |
| Hazards | |
| Main hazards | Oxidizer, causes skin and eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS03,GHS07 |
| Signal word | Warning |
| Hazard statements | H272: May intensify fire; oxidizer. H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Autoignition temperature | 500 °C (932 °F; 773 K) |
| Lethal dose or concentration | LD50 (oral, rat): 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Calcium Nitrate: **300 mg/kg (oral, rat)** |
| NIOSH | RN=10124-37-5 |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 200-250 |
| Related compounds | |
| Related compounds |
Ammonium nitrate Magnesium nitrate Potassium nitrate Sodium nitrate Calcium ammonium nitrate |

