Calcium Iodate: Old Roots, New Purpose
Historical Development
The story of calcium iodate stretches back over a century. Long before supermarkets lined their shelves with iodized salt, chemists poked around with different ways to fortify diets and prevent widespread diseases like goiter. Historically, iodine deficiency haunted inland populations, propelling demand for reliable sources. Calcium iodate, born out of these medical needs, emerged as a stable, less volatile alternative to potassium iodate. Mining operations targeting caliche deposits in Chile—and later, industrial syntheses—secured raw material for health campaigns around the world. In places where logistics and shelf life mattered, this compound’s resilience compared to, say, plain elemental iodine, tipped the balance in its favor.
Product Overview
Calcium iodate shows up as a white crystalline solid. It resists moisture and stands up well to light. These aren't trivial traits in countries battling unpredictable climates and minimal storage infrastructure. The formula, Ca(IO3)2, packs two iodate ions per unit and stashes a decent amount of elemental iodine. Industry players usually source it in technical and food grades, giving animal feed producers and food fortification programs options depending on budget and legal limits. Thanks to its relatively high decomposition temperature and modest solubility in water, calcium iodate survives the rigors of food production, reaching dinner tables with more iodine intact than less robust alternatives.
Physical & Chemical Properties
The fine, odorless crystals of calcium iodate dissolve slowly in water. Compared to iodides, iodates hold onto their iodine, giving them better staying power in processed food and animal supplements. It chars at high heat, releasing iodine and leaving behind calcium oxide. That’s a handy property if technicians need to recover iodine or perform tests for purity. Unlike potassium iodate, calcium iodate brings in calcium ions, which helps sidestep common allergic reactions linked to potassium—important for sensitive populations. The compound doesn’t catch fire or react explosively under typical storage, but it sits among strong oxidizers, so storing it away from reducing agents and organics matters. Anyone handling it catches a faint whiff of its astringency: a subtle, persistent sign of its chemical backbone.
Technical Specifications & Labeling
Manufacturers must tread a fine line with labeling, especially under food and pharmaceutical regulations. Calcium iodate sold for human consumption or animal feed falls under purity requirements, typically above 99% iodate by mass and low limits for nitrites, chlorides, and metals. Labels need batch numbers, production dates, and compliance statements, referencing national and international norms. It’s critical not to treat iodate products as generic white powders. Regulatory agencies, such as the FDA or EFSA, lay down rules about allowable daily intakes for iodine, and product labels need to state the contribution per serving or per kilogram of feed. In my experience, any error here brings costly recalls and nerve-wracking audits.
Preparation Method
Producers rely primarily on oxidizing calcium iodide or treating caliche ore with nitric acid for large-scale calcium iodate production. Chemists once used labor-intensive methods involving hypochlorous acid and iodine, but tighter safety standards and an eye on efficiency nudged the industry toward streamlined oxidations. Electrochemical processes offer better yields and a smaller environmental footprint, with much less waste. In labs, small batches appear by dissolving calcium hydroxide in water and adding iodine solution with controlled oxidation. In factories, though, keeping a tight rein on batch purity and byproduct disposal looms large; too much chlorate, for example, causes trouble at later stages and can slip through into finished products.
Chemical Reactions & Modifications
Anyone working with calcium iodate soon encounters its stubbornness and utility as an oxidizer. Mixed into dough conditioners, it reacts with sulfhydryl groups, helping strengthen gluten bonds and improve the volume of bread. In feed mills, the oxidative properties break down antinutritional factors that would otherwise sap an animal’s growth rate. Scientists sometimes tweak calcium iodate by exchanging its anions or generating mixed salts to tune its solubility or absorption. Some experimental work investigates blending calcium iodate with microencapsulation agents, aiming to delay its breakdown until later digestion stages, a trick employed mainly in feed supplementation.
Synonyms & Product Names
Calcium iodate pops up under a long list of alternative names. You’ll see “calcium diiodate,” “lime iodate,” or catalog designations like “E917” (the European food additive code). In farm and veterinary circles, it’s often lumped together with other iodine sources, risking confusion when farmers talk to suppliers. On chemical supply sites, “calcium iodate monohydrate” frequently appears, distinguishing it from the rarer anhydrous variant. These variants aren’t always equivalent; getting the wrong grade for a production run can raise costs, wreck batch consistency, or even trigger regulatory violations if the iodine content veers out of spec.
Safety & Operational Standards
Handling calcium iodate takes respect and discipline, especially in bulk. As an oxidizer, it demands storage apart from organic matter and reducing agents. Over the years, I’ve seen more than one minor incident traced back to careless storage near solvents or foodstuffs prone to spontaneous combustion. Facility ventilation makes a difference; microcrystalline dust irritates the eyes and throat, so most plants run extractor hoods and supply personal masks. Ingestion risks concern regulators most. While iodine deficiency damages health, excessive intake from accidental spills or bad dosing routines causes toxicity—thyroid dysfunction, skin eruptions, and, in severe cases, organ stress. Training matters as much as equipment: clear procedures for spills, accountability for dosing systems, and regular audits form the backbone of good operations. Any lapse underlines how quickly a supplement can become a contaminant.
Application Area
The most widespread use of calcium iodate sits in fortification. Salt producers use it to stabilize iodine added to table salt, hoping to close the gap in dietary intake, particularly for communities far from the sea. Animal feed companies blend technical-grade product into premixes, providing a consistent iodine baseline to livestock. Dairy operations see the benefit, with improved milk yields and healthier herds. In the bakery aisle, calcium iodate slips into flour treatment agents, improving dough structure. Water treatment plants sometimes use it to sanitize systems and nudge up iodine content in small supply lines, particularly in places where waterborne diseases and iodine deficiency overlap. Laboratories count on it as a primary standard for iodate ion in analytical chemistry, thanks to its stability and purity. Across these uses, the key is consistency: batch-to-batch variations squeak through quickly by way of quality control problems or nutritional discrepancies.
Research & Development
Researchers target calcium iodate both for what it does now and what it could do better. New work looks at slow-release and encapsulated forms, especially for animal nutrition in hot, humid climates where typical iodate mops up moisture from the air. Some teams study its breakdown in the GI tracts of different species, searching for sweet spots that maximize uptake and minimize waste. The compound attracts interest in environmental circles, too. Given its strong oxidizing chops, scientists flirt with it as a low-toxicity alternative in water purification and specialty oxidation processes. The quest for more environmentally friendly production methods, cutting down acid waste and greenhouse gases, anchors recent projects in green chemistry. On the food side, advances in analytical methods, like mass spectrometry, enable tighter quality control, which lets new players produce to high standards without the overhead of enormous QC departments. Regulatory researchers, meanwhile, revisit safe intake thresholds as more data rolls in from population-wide supplementation programs.
Toxicity Research
The double-edged sword of iodine means safety data sits front and center in any discussion. Calcium iodate’s toxicity profile depends mainly on how much is ingested and how often, rather than the compound itself. Overexposure brings acute symptoms tied to iodine overload—metallic taste, stomach pain, and in rare circumstances, kidney or thyroid troubles. In animals, chronic high doses may shrink reproductive rates or skew growth, underscoring the importance of correct mixing and application. Researchers focus on metabolization rates and iodine bioavailability from different carriers, stacking up calcium iodate against potassium and sodium iodates. Human studies underline its narrow therapeutic window: not enough brings deficiency, too much creates new health headaches. The World Health Organization tracks regional intake to guide maximum allowable levels. Evidence keeps mounting, meaning best practices don’t stand still; every few years, the numbers shift as new data matters more than old habits.
Future Prospects
Looking ahead, calcium iodate holds potential beyond its current stable of uses. Health authorities move to tighten fortification strategies in developing regions, where climate and logistics hamper distribution of more sensitive compounds. As plant-based diets trend upward and dairy consumption patterns shift, new types of processed food need dependable iodine sources, and calcium iodate’s stability makes it a prime candidate. Sustainable production methods, including solvent-free syntheses and renewable feedstocks, gather steam—companies figuring out how to outrun regulatory changes will find themselves ahead. Some researchers tinker with hybrid iodine release systems, pairing calcium iodate with organic carriers or nanoparticles to even out absorption or target delivery within the digestive tract. Advances in food testing mean stricter controls and better consumer safety, likely pushing demand for higher purity and tighter specifications. All this points to a future where calcium iodate serves not just as a stopgap for deficiency but as an adaptable ingredient across sectors driven by real-world needs and evolving science.
What is calcium iodate used for?
What Keeps Animals Healthier
Farmers have long watched for signs of iodine deficiency in their livestock—rough coats, swollen necks, tired animals. Calcium iodate helps fix these problems before they start. It doesn’t leave much to chance: this mineral compound delivers a reliable source of iodine to animal feed, plugging dietary gaps that can cause stunted growth or reproductive trouble in cattle, swine, and poultry. The livestock industry depends on healthy animals, so feed additives matter. Calcium iodate isn’t the only iodine supplement out there, but it brings high stability and resists losing potency in storage, which is why many feed producers stick with it.
The story plays out on small family farms and massive commercial operations. A farmer who mixes her own chicken feed knows that lack of iodine will hit egg production and the birds’ overall condition. Bigger farms buy prepared feeds, trusting those mixes deliver enough trace elements—including iodine—to keep thousands of animals in good shape. Calcium iodate does its share in this ecosystem.
Protecting What We Eat
Bakers and food processors keep a close eye on quality. The food industry uses calcium iodate as a dough conditioner to improve bread texture. This matters in places where bread forms part of the daily diet and regulations require a certain level of iodine in food. Public health policies in countries like the U.S., Australia, and parts of Africa push for iodine enrichment to fight nutritional deficiencies, and they often rely on calcium iodate for its ability to stay effective through long transport and storage.
I remember growing up, noticing those little labels on bread packs that mention “iodized dough conditioner.” Most folk skim right past it, not realizing this small mineral addition plays a huge part in fighting thyroid issues. The United Nations and World Health Organization both keep sounding the alarm about iodine deficiency. Roughly two billion people remain at risk worldwide. So a steady supply of calcium iodate can make a big difference, especially in lower-income countries where alternative sources like fresh seafood aren’t always easy to find.
Supporting Clean Drinking Water
Water quality also leans on calcium iodate in a less obvious way. Certain water purification systems use iodine compounds as part of filtration or disinfection processes. Emergency water treatment tablets may include calcium iodate to kill dangerous microbes and reduce the risk of waterborne illness, especially after floods or natural disasters. I’ve watched disaster response teams work in hurricane zones, where clean water becomes gold. In those moments, these small compounds do heavy lifting behind the scenes.
Solutions and Looking Ahead
Addressing deficiencies relies on cooperation—farmers, feed producers, regulators, and scientists working together. Simplifying supply chains keeps calcium iodate flowing into the right hands. Smarter packaging and clear training can help farmers in remote regions give their animals proper nutrition without waste. Governments should update regulations to reflect the latest nutritional science and keep up with local needs. Tracking animal health, monitoring bread quality, and checking rural water systems for iodine content will all help communities stay healthier over generations.
Is calcium iodate safe for animal feed?
Understanding the Role of Calcium Iodate
Walk into any modern livestock farm, and the shelves of nutrition supplements will tell their own tale. Farmers work hard to ensure their herds are healthy, growth rates are up, and milk or egg output matches expectations. Iodine is key for the thyroid gland, and a shortfall means dull coats, poor production, and even reproductive trouble. Calcium iodate, a white, relatively stable crystalline substance, is widely used to provide the required iodine. The key question: does this ingredient offer enough safety for daily feeding?
What the Science Says
Animal health experts put calcium iodate through plenty of testing. Research shows that, in recommended doses, animals digest it easily, absorb the needed iodine, and excrete the rest. The European Food Safety Authority reviewed animal studies and didn’t find chronic negative effects at suggested levels in diets. Feeding trials with poultry, pigs, and cattle showed that growth and development kept pace with animals not taking supplements, which tells us it works as expected and does not pile on health risks.
Feed manufacturers and regulators use a strict margin of safety approach for minerals. Too little iodine triggers deficiency, but too much can poison the thyroid and kick off metabolic mess. Safety means sticking to the established guidelines. In the EU and North America, both the maximum inclusion rate and the way calcium iodate is added are checked carefully through routine oversight. Tight supply chains and regular feed testing pick up potential mistakes before feed reaches farms.
Residues and Food Safety
Iodine, like other trace elements fed to animals, will turn up in milk, meat, and eggs. Studies find that at approved doses, the extra iodine does not cause a spike in residues that would threaten people’s health. Milk and table eggs, even from supplemented animals, stick to safe levels for human consumption. National food safety agencies have set public limits that keep both animal and human health protected.
Possible Risks and Room for Error
Problems start when the balance tips. Accidental overdosing—caused by feed mixing errors—brings trouble for livestock health. Thyroid disorders and drops in productivity and fertility can hit the farm hard. Some breeds and young stock are more sensitive, so experienced farm nutritionists keep a close eye on not just the amount but the source of iodine in the diet. Diversified sources—table salt, kelp meal, and other iodine salts—make it easy to slip past the ‘safe’ limit if calculations aren’t double-checked.
Practical Solutions and Farm-Level Care
No supplement replaces boots on the ground. Farm staff keep nutrient records, and feed mills train workers to measure out minerals accurately. National authorities suggest regular analysis of feed to cross-check for trace mineral levels. Vets run thyroid tests when health slips or reproductive trouble strikes. Feed suppliers also offer nutrient audits, making sure labels match reality.
Animal nutrition is a science, but it’s also built on trust between farmers, feed suppliers, and regulators. Keeping calcium iodate safe isn’t a ‘set and forget’; it’s about ongoing checks, strong communication, and a focus on animal and human health from producer to plate.
What is the chemical formula of calcium iodate?
Understanding Calcium Iodate
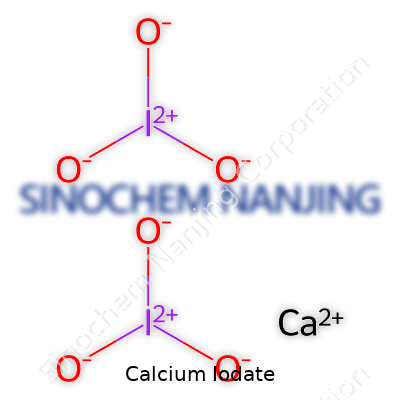
Calcium iodate comes with the chemical formula Ca(IO3)2. You see this compound pop up in food fortification, animal nutrition, and disinfection. Most folks might barely give it a second look, but in the world of nutrition and public health, these few symbols carry serious weight. That’s where my own years in food science actually taught me how big an impact these seemingly minor compounds have in our everyday lives.
Why the Formula Matters
Having clear knowledge about a formula like Ca(IO3)2 can make all the difference between effective supplementation and wasting resources. Take iodine deficiency as an example. Almost two billion people worldwide struggle with this issue, leading to problems like goiter and thyroid disorders. The World Health Organization still keeps a close eye on micronutrients like iodine. They use calcium iodate in table salt, making it a practical source for this important element.
The Chemistry at Work
The structure tells us a lot about function. This formula reveals there’s a calcium ion paired with two iodate ions. I remember how simple tweaks in molecular structure would shift how a nutrient is absorbed or used in the body. The calcium here does more than just "hold" the iodate together. It makes the compound stable under different conditions, protecting the iodine content even if the environment gets a bit rough. In salt or animal feeds where temperature and humidity swing wildly, stability becomes a real-life concern, not just an academic one.
Real-World Applications
In the poultry industry, for example, companies add Ca(IO3)2 to feeds. This helps prevent iodine deficiency in flocks, which can seriously disrupt growth and egg production. On my own small farm growing up, a neighbor ran into a terrible drop in egg yield. A veterinary nutritionist pointed straight at the lack of iodine, which we traced back to a missing supplement in the feed. Swapping in calcium iodate set things right within weeks, and for a small operation, that made the difference between business survival and failure.
The Value of Precision
Wrong information, especially in chemicals and nutrients, leads to wasted money at best and destroyed livelihoods at worst. I’ve seen young science students confuse similar-sounding formulas, leading to lab mistakes. For manufacturers, accuracy in formula means consistency in product, safety for consumers, and predictable nutrition profiles for livestock. It isn’t enough to just eyeball the mixture; you have to hit the right molar ratios, batch after batch. Big brands knew this long before the rest of us, and their long-term reputation depends on quiet details like chemical formulas and purity.
Solutions: Making Access Reliable
Quality assurance in calcium iodate production keeps both people and animals safer. Regulators and public health groups can check up on suppliers, run purity checks, and require transparency in labeling. Farmers and food makers need easy access to lab-verified compounds, not just bulk chemicals from unreliable sources. Training and education help as well. After all, a clear understanding of Ca(IO3)2 goes much further than a dusty textbook entry; it can help someone keep their farm, protect a community’s health, and ensure every breakfast table gets what it needs for a healthy start.
How is calcium iodate different from potassium iodate?
The Story Behind Two Common Iodine Sources
Kids growing up with goiter or developmental issues often lacked iodine—a simple element missing from salt shakers or processed food. After realizing the link between iodine and health, countries began adding it to salt. Salt needs a partner to make this work, and that's where compounds like calcium iodate and potassium iodate step in. I remember learning in high school how vital a sprinkle of iodine in our diet could be. Still today, the delivery vehicle can make a big difference for how well people stay healthy.
How These Compounds Work
Both calcium iodate and potassium iodate carry iodine, but they don’t bring the exact same properties to the table. Calcium iodate shows up as a white, odorless solid. Potassium iodate comes as a little white powder too, but the “potassium” part packs a punch when you want a salt that’s quick to dissolve in water. This makes a big difference in huge salt-processing facilities, where time and consistency mean everything.
Health Impacts and Safety
Both compounds help kick out iodine deficiency, which keeps thyroid glands working right. Most folks encounter potassium iodate in table salt—an effort that's pretty mainstream in places fighting iodine deficiency disorders. But using the right compound isn’t just about solving one problem. For example, potassium iodate can act as a treatment against radiation exposure by blocking radioactive iodine from building up in the body. It’s stocked in emergency kits near nuclear power plants for a reason. Calcium iodate, by contrast, turns up more often in animal feed for poultry and cattle, keeping livestock healthy and productive.
For safety, regulatory agencies like the US Food and Drug Administration and the World Health Organization have both given potassium iodate the green light for fortifying edible salt within recommended levels. Still, like many chemicals, too much can cause harm. That's why quality control matters in production, along with regular monitoring by authorities.
Supply, Shelf Life, and Stability
In big humid warehouses or far-flung towns, anything absorbing moisture becomes a headache. Potassium iodate handles humidity much better than earlier choices like potassium iodide, which can break down and lose potency over time. Calcium iodate also brings solid stability, standing up to heat and light. I’ve seen both compounds used in countries with tough weather conditions, and the decision often comes down to cost, regulations, and shelf life more than chemistry alone.
Issues with Application
In my work with rural health projects, the reality on the ground often creates limits science can't always solve. If you’re in a community with limited infrastructure, distributing the more stable or locally available iodine compound gets priority. Oceanside towns sometimes struggle less with deficiency, but inland and mountainous regions run higher risks. Potassium iodate can get into coarse kitchen salt more easily, so it fits best where folks use loose salt rather than compressed blocks. Calcium iodate can work better in livestock salt licks because it's less prone to caking in big blocks.
What Could Fix Gaps in Access
People working on public health need to keep listening to those using iodized products, not just the policymakers or chemists. Better education helps: local teachers, farmers, and doctors spreading word on why eating enough iodine matters can solve half the battle. Policymakers can help by supporting affordable local supply chains. Collaborations with community organizations make sure fortified salt gets where it’s needed, so nobody pays with their health for what’s missing from their plate.
How should calcium iodate be stored?
Why Proper Storage Matters
Few things can ruin a batch of calcium iodate faster than exposure to the wrong environment. Working with chemicals for years, I’ve seen the mess that results from careless storage. Moisture cakes up the contents, air triggers breakdown, and sunlight leads to discoloration. The risks go beyond product loss—improper storage can affect the quality of animal feed, supplements, or anything else that uses this compound. Poor calcium iodate can mean low iodine in the end product, and that spells health risks for both livestock and people.
Keep It Dry and Cool
Calcium iodate asks for a dry, climate-controlled room. Humidity turns this powder into a hardened mess, almost like concrete in a bag. Storing it near open windows or in sheds with leaking roofs always ends in regret. I’ve watched coworkers scramble, throwing out bags after a rainy spell because they thought “just a little moisture” wouldn’t hurt. It’s best to use sealed containers—think tightly closing bins or drums—and stash them away from plumbing or any area likely to get damp. Cool rooms make all the difference. High heat speeds up chemical breakdown, so keep it away from engines, direct sunlight, or hot machinery. A simple shelf in the shade, in a stable building, wins every time.
Steer Clear of Chemicals and Strong Odors
Many people stack chemicals together for convenience, but calcium iodate picks up strong smells and can react with certain substances. Never park it near acids, bases, or anything labeled as a strong oxidizer. Many years in back rooms have taught me that even keeping cleaning products too close leads to problems. I once opened up a bag that smelled sharply of chlorine thanks to an adjacent bleach spill—the whole stock got binned as a precaution. Assign a dedicated spot for your calcium iodate, ideally with clear shelf labels and some distance from other supplies.
What Happens If You Ignore These Steps?
Ignoring these basics usually ends in waste. Wet calcium iodate clumps and gets moldy. Exposing it to high temperatures means the iodine content can drop out, which destroys the purpose. Any batch that absorbs weird smells or chemicals risks cross-contaminating animal feed or even nutritional supplements. In some cases, improper storage violates regulatory rules and leads to fines—or costly recalls. I’ve watched more than one small business suffer because an easily avoidable storage slip cost thousands in lost stock.
Simple Storage Tips from Experience
- Transfer the powder to airtight, labeled containers right after opening the original bag.
- Keep storage rooms locked and at a steady, cool temperature.
- Never store near cleaning products, pesticides, or corrosive materials.
- Put up clear storage instructions for all staff—fewer mistakes happen when everyone knows the rules.
- Discard any batch showing clumping or discoloration. Better safe than sorry.
Tools and Monitoring
A good thermometer and a simple humidity gauge on the shelf solve most problems before they start. I’ve relied on these inexpensive tools for years to make sure nothing goes unnoticed. Fresh silica gel packets can help with moisture control in smaller storage spaces. If possible, set up routine checks every few weeks—walk the shelves, shake containers, and inspect for leaks or damage. Fewer surprises come up this way.
Conclusion Isn’t Required, but the Lesson’s Clear
How you store calcium iodate shapes its safety and usefulness. The small efforts spent on the right storage technique pay back over and over again, keeping things safe for both users and anyone relying on the end product. Simple habits make the real difference—ask anyone who’s done it wrong, even once.

| Names | |
| Preferred IUPAC name | Calcium diiodate |
| Other names |
Calcium diiodate Lime iodate Iodic acid, calcium salt Calcium iodide oxide |
| Pronunciation | /ˈkæl.si.əm aɪˈoʊ.deɪt/ |
| Identifiers | |
| CAS Number | 7789-80-2 |
| Beilstein Reference | 357962 |
| ChEBI | CHEBI:31343 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 13664002 |
| DrugBank | DB11136 |
| ECHA InfoCard | 100.013.679 |
| EC Number | 231-193-2 |
| Gmelin Reference | Gmelin Reference: 14516 |
| KEGG | C14441 |
| MeSH | D003359 |
| PubChem CID | 25152 |
| RTECS number | FX9450000 |
| UNII | 92W7H0K349 |
| UN number | UN1479 |
| Properties | |
| Chemical formula | Ca(IO3)2 |
| Molar mass | 390.98 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 4.28 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.1 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.45 |
| Magnetic susceptibility (χ) | -72.0e-6 cm³/mol |
| Refractive index (nD) | 2.5 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -795.6 kJ/mol |
| Pharmacology | |
| ATC code | A12CC01 |
| Hazards | |
| Main hazards | Oxidizing solid, harmful if swallowed, causes serious eye irritation |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | Keep away from heat. Keep away from sources of ignition. Take precautionary measures against static discharges. Avoid contact with skin and eyes. Do not breathe dust. |
| NFPA 704 (fire diamond) | 2-0-1-OX |
| Lethal dose or concentration | LD50 oral rat 820 mg/kg |
| LD50 (median dose) | 743 mg/kg (Rat, oral) |
| NIOSH | WA1925000 |
| PEL (Permissible) | PEL: 10 mg/m3 |
| REL (Recommended) | 10 mg/kg |
| Related compounds | |
| Related compounds |
Calcium chlorate Calcium bromate Potassium iodate Sodium iodate |

