Calcium Gluconate: A Deep Dive into Its Development, Use, and Future
Historical Development
Calcium gluconate didn’t just pop up one day in a chemist’s beaker. The journey started back in the 19th century when scientists realized that some forms of calcium worked better than others for treating certain conditions. Jahr’s synthesis of gluconic acid paved the way, catching the eyes of physicians intrigued by calcium salts that didn’t knock patients off their feet with side effects. Hospitals eventually swapped out harsher calcium salts in favor of gluconate, mostly for its grip on safety and compatibility with the body. Surgeons and doctors began calling for calcium gluconate during emergencies, laying a foundation for its clinical uses seen today in both wards and operating rooms. The compound’s long road has built up real trust across generations of practitioners.
Product Overview
Calcium gluconate has worked its way into powders, tablets, injections, and topical gels. Nurses reach for it where sudden drops in calcium demand quick action, especially in settings where IV access changes the game for hypocalcemia or cardiac emergencies. The oral version lands on pharmacy shelves to boost daily calcium intake for bone health and sometimes acts as an antidote for magnesium sulfate toxicity. As a topical gel, it soothes skin after hydrofluoric acid burns during unfortunate chemistry class accidents. Few compounds offer this flexibility: one ingredient spanning medicine cabinets, trauma carts, and industrial accident kits.
Physical & Chemical Properties
Pure calcium gluconate appears as a white, tasteless powder. Unlike other calcium salts, it dissolves better in water, making it a top choice for both oral solutions and injectable preparations. Beyond color and taste, it stands out for stability at room temperature and tolerates a pretty good shelf life under the right conditions. Each molecule brings nine water molecules along for the ride (in its common hydrate form), which matters for dose calculations in pharmacy. The pH hovers between 6 and 8, making it suitable for most physiological applications without causing local irritation, a common complaint with other calcium products.
Technical Specifications & Labeling
Pharmaceutical products require rigorous labeling, not just for safety but due to legal requirements across borders. Vials and bottles get labeled with clear dosing, storage advice, concentration (often as 10% w/v for injection), and batch tracking. The National Formulary and pharmacopeias set identity benchmarks for purity and absence of heavy metals. Laboratories and manufacturers bear the weight of frequent audits, ensuring products don’t carry harmful residues or drift away from standardized calcium content. Hospitals and pharmacies need this assurance, knowing exactly what goes into patient care.
Preparation Method
To make calcium gluconate, chemical engineers bring together gluconic acid and calcium carbonate or calcium hydroxide. The process relies on controlling temperature, pH, and purification steps to coax the salt from solution before spray drying or crystallization. Hydration level (monohydrate or decahydrate) changes the handling and dosing, so technicians watch these variables closely. In the pharmaceutical sector, full traceability covers everything from raw material quality to packaging, keeping every batch consistent. Scale-up from lab bench to factory line has tightened over decades, knocking down risks of contamination.
Chemical Reactions & Modifications
Calcium gluconate reacts as a typical salt in solution, dissociating into calcium ions and gluconate anions. In therapeutics, the available ionized calcium can bind with fluoride or oxalate ions—turning a potentially deadly exposure into a manageable wound or ingestion case. Modifications include using calcium gluconate in buffered mixes for parenteral nutrition, and, less commonly, in gels for topical rescue after chemical burns. Chemical engineers have studied new forms, always exploring less water-soluble alternatives where slower absorption could benefit some treatments.
Synonyms & Product Names
You’ll see calcium gluconate turn up in medical literature under names such as Gluconic acid calcium salt or Calcium D-gluconate. Branded versions show up in hospitals—some call them “Cal-Glu” injections or simply “calcium rescue” when lives hang in the balance. In chemistry circles, the name sticks close to its roots, though international markets sometimes assign local trade names.
Safety & Operational Standards
Healthcare relies on strict standards for production and storage. Manufacturers follow cGMP (current Good Manufacturing Practice) rules, not just to tick boxes but to protect patients from microbial contamination or dosing problems. Nurses check IV compatibility on the spot since mixing calcium gluconate with certain drugs can trigger precipitation. Safety protocols include wearing gloves with gels—especially after hydrofluoric acid burns—since accidental transfer onto unprotected skin brings risks. Regular training and clear labeling keep the accidental misuse rate low in high-stress hospital settings.
Application Area
Most people meet calcium gluconate through hospitals: it lands in crash carts for treating acute hypocalcemia, in vials stashed among antidotes for chemical exposures, or in tablet form for those with chronic low calcium. Dentists sometimes use it for local mouth rinses when oral lesions disrupt mineralization. In industry, companies apply the gel after certain chemical burns, bridging medicine, laboratory safety, and emergency response. Nutritionists occasionally recommend it for patients with trouble absorbing other calcium salts, especially in cases with kidney or digestive issues.
Research & Development
The story’s not finished. Researchers have expanded its use beyond hypocalcemia, running trials in cardiac arrest protocols, exploring lower-dosage regimens for chronic disease management, and testing new intravenous compounds for better bioavailability. Nano-formulations make the news for targeted drug delivery, showing promising application in both medicine and industry. Some teams dig into environmental impacts, probing whether industrial-scale production can clean up processes or reclaim side streams as fertilizer or feed additives. Interdisciplinary research pulls in chemists, doctors, pharmacists, and engineers with a common goal of refining both the compound and the way it reaches those in need.
Toxicity Research
Toxicologists keep a close watch on calcium gluconate’s effects. Most adverse events stem from giving it too fast into the bloodstream; overzealous IV pushes can bring on arrhythmias. Chronic overuse slips calcium into tissues where it doesn’t belong, raising concerns about kidney stones or vascular calcification. Studies dig deep into safety in special populations—pregnancy, liver compromise, and childhood disorders. A major focus stays on interactions, ensuring calcium doesn’t disrupt other therapies. Regular research into toxicity profiles helps adapt dosing recommendations to new findings and patient populations.
Future Prospects
The future points toward smarter, safer, and greener production methods. New research hopes to tweak the solubility and absorption profiles, opening the door for patient groups facing unique challenges. Automated hospital dispensaries and digital labeling may soon ensure traceability far beyond today’s systems. Some teams look outside human health, testing calcium gluconate in veterinary settings and even agricultural uses, betting on the broad utility of a well-understood compound. Keeping a close eye on safety while adapting old chemistry to new purposes will shape how the next generation of healthcare workers sees this time-tested calcium salt.
What is Calcium Gluconate used for?
Calcium Gluconate: More Than Just a Supplement
Calcium seems pretty basic. Most folks think of it as the thing that makes bones strong. Calcium gluconate, though, steps in when ordinary calcium can't do the whole job. Hospitals lean on it, ambulances carry it, and sometimes even folks at home have it in their medicine cabinet. Why? Because it fills some gaps that ordinary supplements just can’t reach.
Medical Emergencies: A Critical Player
Picture the aftermath of a chemical spill or an accident with industrial cleaners. Hydrofluoric acid burns don’t just hurt — they can kill. The acid pulls calcium right out of the body’s tissues, and that’s where calcium gluconate comes in. Topical gels and intravenous forms help prevent tissue destruction and keep a person’s heart from seizing up.
In my hospital days, the need for fast calcium replacement came up during code blues. A patient’s heart rhythm can flip dangerously when potassium goes too high, a problem called hyperkalemia. In those moments, there’s not much room for error. Calcium gluconate helps stabilize the heart, buying time for the more permanent fixes to work.
Low Calcium Conditions: Restoring the Balance
People with kidney problems, post-surgery complications, or certain endocrine disorders can run dangerously low on calcium. Tingling fingers, muscle cramps, even seizures can follow. Tablets and IV calcium gluconate aren’t the most glamorous drugs, but for people with severe hypocalcemia, they’re sometimes life-saving.
It’s not just about bones and teeth. Calcium keeps muscles and nerves working right. A shortage causes confusion or muscle spasms. If someone is getting blood transfusions one after another, citrate in the blood products depletes calcium, and that’s another reason hospital pharmacies stock vials of this everyday mineral in its gluconate form.
Safety in Everyday Products
Calcium gluconate also finds its way into some foods and over-the-counter products. Bread, cereals, and things like sports drinks use it for calcium enrichment. Sometimes cosmetics use it because calcium keeps skin healthy and can help with some rashes or irritations.
It’s easy to take these things for granted. Seeing the sheer number of ways calcium contributes to daily health made me rethink how miraculous some staples are. My own family includes someone with chronic kidney disease, and discussions about calcium get real when bloodwork comes back a bit off.
Risks and How to Use It Right
Too much calcium causes trouble. If doctors push calcium gluconate too quickly through an IV, patients feel their skin flush or even suffer irregular heartbeats. The right dose matters, and careful monitoring makes a big difference. Not everyone with low calcium symptoms needs gluconate in a drip — sometimes, oral supplements or a tweak to diet work just fine.
Because the kidneys handle most calcium clearing, those with kidney damage face a tighter balancing act. Endocrinologists and nephrologists have plenty of experience making these calls, often working with dietitians to make sure food and medicine work together.
Looking Ahead
Public hospitals, clinics, and even medics on the sidelines keep calcium gluconate close for a reason. As we see more people with chronic disease and more industrial exposures, the need for quick and safe calcium correction probably won’t go away. Better training for front-line staff on how and when to use it would make emergency care that much safer. And for people with long-term medical problems, regular check-ins about calcium balance should stay at the top of the list.
What are the side effects of Calcium Gluconate?
Spotlight on Calcium Gluconate Use
Calcium gluconate comes up a lot in hospitals and clinics. Doctors often reach for it to treat low calcium levels or for certain emergencies, such as controlling heart rhythm problems or countering high blood potassium. Most folks see calcium as a friendly mineral—after all, every glass of milk commercial promotes bone health with a big smile. That said, receiving calcium through a tablet, IV, or injection is not quite the same story as sipping it in a morning latte. The risks can grow if someone’s kidneys underperform, or if treatment gets used in high doses.
Short-Term Side Effects: What Patients Usually Notice
After an injection or IV calcium gluconate, many patients feel a flush of warmth that sweeps the body. Sometimes the skin tingles or a metallic taste springs up in the mouth. This reaction can feel strange but often passes in minutes.
Gut troubles can hit soon after taking calcium gluconate by mouth. An upset stomach, bloating, or brief diarrhea show up for some folks. These symptoms rarely last long, but they make it hard to forget that this isn’t just a regular mineral supplement from the health food shelf.
Serious and Rare Effects: Closer to the ER Than the Kitchen Cabinet
For all its routine uses, calcium gluconate can bring real complications. Rapid IV injection can spark an abnormal heart rhythm. This danger grows for older patients or anybody with a shaky heart. Slowing the infusion can keep the rhythm in check. Doctors often monitor heartbeats closely for this reason.
A vein can get irritated after an injection, leading to swelling, redness, or even tissue injury if the medicine leaks outside the vein. Staff members watch the injection site for signs of trouble. If calcium gluconate gets under the skin by mistake, it poses a risk of lasting tissue damage. Hospitals train their teams well to avoid this, but nobody wants to learn the hard way.
Excess calcium—hypercalcemia—can creep up quietly. Once it arrives, it brings constipation, muscle weakness, confusion, and in severe cases, kidney stones or rhythm problems. Long-term use without careful blood checks causes most of these troubles. People with kidney disease need extra care since their bodies struggle to get rid of extra calcium.
Learning from Experience: Staying Safe with Calcium Gluconate
Clear discussion between doctors and patients helps keep surprises in check. Everyone deserves to know what they’re getting and why. I’ve seen cautious teams adjust doses, check bloodwork, and talk openly about symptoms to watch for. These habits catch problems early.
Drug interactions matter. Taking calcium with certain blood pressure medicines or antibiotics can block absorption or change how the drugs work. Patients ought to bring an updated list of everything they take—prescriptions, over-the-counter tablets, even herbal teas.
Hospitals rely on strong checklists and protocols that flag hidden risks. This pays off, especially in the chaos of an emergency department where fast decisions rule the day. I once watched a pharmacist step in after spotting a risky calcium dose for someone with silent kidney trouble. That sharp eye made all the difference.
Looking Ahead: Smarter Choices, Healthier Outcomes
No one wants to shy away from a helpful medicine out of fear, but it isn’t smart to ignore the risks either. By focusing on open communication, regular blood monitoring, and well-trained staff, health teams can keep calcium gluconate in the safe zone. In every setting—wards, clinics, ambulances—safety stands on teamwork and sharing the right information at the right time.
How is Calcium Gluconate administered?
Why Doctors Reach for Calcium Gluconate
Calcium doesn’t get as much attention as some other minerals, but in certain sticky situations, doctors turn to calcium gluconate and not just for a cramp or two. When potassium levels shoot up or a patient gets exposed to hydrofluoric acid, this mineral salt steps up as the frontline fix. In my experience working with health professionals, they treat calcium gluconate with respect—using it only after weighing up the risks and the reason for giving it in the first place.
The Different Ways to Give It
Calcium gluconate isn’t your run-of-the-mill supplement. It’s given in two main ways: intravenously or by mouth. Hospitals favor the IV route. Nurses prepare a syringe or infusion, double-check the vein, and push it slowly. Too fast and the patient complains of a hot flush or, in rare cases, something more serious like a heart rhythm change. In less urgent scenarios, or if the person just needs a bump in levels, tablets or liquid by mouth work well. But those pills don’t act as quickly as a shot to the vein.
What Doctors Watch For
No one likes surprises in a hospital, least of all a nurse giving medicine through a vein. Before starting a dose, many hospitals require a quick check on heart function, especially for older patients. Some days, you see people hooked up to a heart monitor even for a single dose. The reason for this isn’t just caution—calcium can tighten blood vessels or mess up the heart’s rhythm if it builds up too quickly. That’s why nurses and doctors stay close during the administration, watching for sweating, tingling, or any sign of discomfort.
Challenges With the Oral Route
Oral calcium gluconate may sound simple. Just pop a pill, chase it with some water, and get back to your day. The problem is that it doesn’t absorb as well as doctors would like. People with gut issues or who take certain meds—like diuretics—might notice it hardly budges their calcium numbers. That means for real emergencies, like a dangerously low calcium level, swallowing a tablet won’t cut it. Hospitals lean on the IV form for this very reason.
Risks and What Can Go Wrong
Medical teams tread carefully with calcium gluconate. Give too much too fast and veins can burn or, worse, surrounding tissues can take a hit if the needle slips—something called extravasation. Patients with kidney trouble face extra risk because their bodies can’t flush out the calcium like they should. Reports in the literature show this can lead to deposits in soft tissue or contribute to more serious kidney problems.
Staying Safe and Getting Results
Doctors lean on experience, but they don’t go it alone. They rely on lab tests and check calcium blood levels before and after giving medication. In my conversations with pharmacists, many say they double-check dosing especially for kids and older adults, since their bodies don’t handle extra minerals as well. Teams work together to spot side effects early and adjust the plan if necessary.
Improving How Healthcare Handles Calcium Gluconate
Hospitals could keep reducing risks with even tighter protocols around administration. Staff training, regular reviews of injection techniques, and rapid-response kits for accidental leaks near the injection site all make a difference. Better access to pharmacists on busy hospital wards helps, too. More awareness about interactions with other medicines or conditions like kidney disease should become a normal part of patient checks.
Is Calcium Gluconate safe during pregnancy?
Why Pregnant Women Talk About Calcium Gluconate
People hear about calcium supplements during pregnancy for good reason. The body demands more calcium to help the baby’s bones and teeth build up. Shortfalls can affect both mother and child. Not everyone handles dairy or absorbs calcium well, so some turn to pills and powders. Calcium gluconate often shows up on labels and in doctors’ discussions.
What Research Tells Us
Modern prenatal care draws on hundreds of studies covering vitamins and minerals. Calcium salts, including calcium gluconate, have been researched for decades. The American College of Obstetricians and Gynecologists recommends calcium for pregnant women, especially when daily diet falls short. According to the CDC and the National Institutes of Health, calcium gluconate offers a reliable way for people with absorption issues or intolerance to lactose to keep calcium levels healthy.
Current studies and guidelines suggest most women attend prenatal checkups every month in early pregnancy. Doctors frequently check calcium and vitamin D levels, because those two work as a team in bone health. Taking calcium gluconate, in regular oral doses, has not shown risks in people without kidney problems. Injectable forms exist, usually for emergencies like severe low calcium (hypocalcemia) in the hospital and not as a daily supplement.
Personal Experience as a Parent and Patient
I remember my own nerves picking up a bottle of vitamins and seeing a list of ingredients I barely recognized. Talking to healthcare providers always helped. Once, after nausea threw a wrench into eating habits, my OB-GYN discussed calcium gluconate as a backup to food sources. She explained that taking too much calcium won’t give a baby super bones, but taking too little may cause the body to “borrow” calcium from the mother’s bones. It’s a simple matter of protection.
My sister’s doctor suggested calcium gluconate after she developed lactose intolerance. Blood tests tracked her calcium and vitamin D. She stayed within recommended doses and took it with meals. Her pregnancy and delivery stayed on a steady course.
What to Look for in Calcium Supplements
Not every calcium pill is equal. Besides gluconate, stores carry carbonate, citrate, and lactate. Calcium gluconate contains less elemental calcium than, say, calcium carbonate. So people often must take more pills for the same effect. Swallowing several pills per day can be tough for some, especially with pregnancy nausea. Looking for trusted brands with certifications (such as USP Verified) helps sidestep unnecessary additives or dose inconsistencies. Avoid supplements with heavy metals or unknown herbs tossed in.
It makes sense to review label ingredients with a medical provider. They know patient history, what medicines interact, and how much calcium is sensible in light of a personal diet. For women taking prenatal vitamins with calcium already included, doubling up can lead to stomach cramping or constipation.
Risks and Real-World Solutions
Healthy kidneys filter out extra calcium, but high doses over many months—beyond what any reputable source suggests—may build up and cause stones or harm. Anyone with a kidney disorder should not pick a supplement without approval.
Food first still stands as the easiest and safest. Dairy, tofu, dark leafy greens, beans, and almonds all bring nutrients with them that pills can’t match. Supplements, including calcium gluconate, help to fill the gap. Talking it over with a doctor makes sense not because every new supplement is dangerous, but because pregnancy makes the body’s needs unique. Medically guided choices help mothers stay strong and babies grow as they should.
What should I do if I miss a dose of Calcium Gluconate?
Missing a Dose: Don’t Panic, But Don’t Ignore It
Nearly everyone slips up with their medicine at some point. Life gets busy, routines get tossed around, and next thing you know, you realize you’ve missed that scheduled dose of calcium gluconate. And then comes the uncertainty—can you just take it later, or skip it and wait for the next one?
Understanding Calcium Gluconate’s Role
Calcium keeps your bones healthy, your heart beating in rhythm, and your nerves firing just right. For anyone who’s had low calcium—whether it’s because of kidney issues, chronic illness, or medications—you know skipping a dose isn’t ideal. Low calcium can show up as muscle cramps, tingling in fingers, or sometimes more serious heart problems. A missed dose might not be cause for alarm if it happens once, but repeating the pattern brings bigger risks.
What I’ve Learned About Catching Up Safely
If you remember soon after missing your scheduled dose, just take it. Plain and simple. Many doctors—mine included—say if it’s only been a few hours, go ahead and take it with your next meal or water. But if you’re almost due for the next dose, don’t double up. It isn’t worth the risk. Taking too much calcium at once can set off nausea, stomach pain, or even cause your levels to spike, which isn’t harmless either.
Once, I forgot my morning dose and, in a rush, thought about taking two at lunch to “catch up.” My pharmacist gave it to me straight—doubling up does more harm than good, particularly with minerals like calcium that the body doesn’t handle quickly. That stuck with me. Remember, there’s a reason the schedule is spaced out across the day: to keep blood levels steady and avoid overloading the system.
The Importance of Dosage Consistency
Your doctor prescribes calcium gluconate carefully. Too little, and deficiency sticks around. Too much, and new problems crop up—kidney stones being one, or irregular heartbeats. That balance makes it more important to take doses as close to schedule as possible. If you miss a dose once, be mindful, but don’t stress too much. If this keeps happening, have a conversation with your doctor or pharmacist about reminders or an easier schedule.
Tips That Help Me Stay on Track
Phone alarms work wonders. Some folks use a pill organizer. Writing doses on a calendar helps turn it into a habit. Tying medicine to another daily routine, like brushing your teeth, also makes it less likely to forget. If you take other medications, combining them—if your doctor allows—could cut down on missed doses.
Ask Before Changing Anything
Don’t guess. Medical guidance shapes your safety here. If you’re ever unsure what to do after missing or doubling up on your medicine, that’s a good time to speak up. Pharmacists and nurses field these questions every day and can clarify things quickly. For certain folks—older adults, people with kidney problems, or kids—even small changes in calcium intake can matter a lot more, so leaning on professional advice matters.
Nobody’s perfect at sticking with medical routines. Still, understanding what’s at stake, and having a strategy for those occasional slip-ups, means you can keep health worries in check and avoid bigger problems down the road.
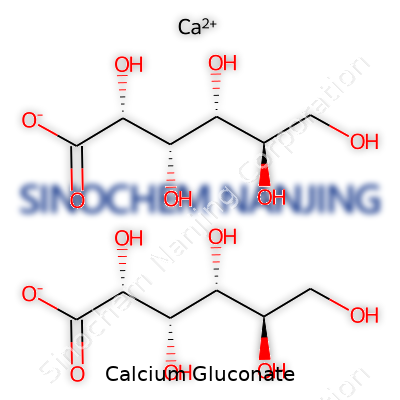
| Names | |
| Preferred IUPAC name | calcium (2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate |
| Other names |
D-gluconic acid calcium salt Calcionate Calcium gluconicum Gluconic acid calcium salt Calcium gluconas |
| Pronunciation | /ˈkæl.si.əm ˈɡluː.kə.neɪt/ |
| Identifiers | |
| CAS Number | 299-28-5 |
| Beilstein Reference | 821158 |
| ChEBI | CHEBI:31344 |
| ChEMBL | CHEMBL1201172 |
| ChemSpider | 6816 |
| DrugBank | DB01373 |
| ECHA InfoCard | 07e6d480-6c31-4a5e-af7d-2e3009c4c07a |
| EC Number | E 578 |
| Gmelin Reference | 1876 |
| KEGG | C00681 |
| MeSH | D002121 |
| PubChem CID | 5284359 |
| RTECS number | RTECS:LZ5850000 |
| UNII | M87A77N3LR |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C12H22CaO14 |
| Molar mass | 430.373 g/mol |
| Appearance | White crystalline or granular powder |
| Odor | Odorless |
| Density | 0.8 g/cm3 |
| Solubility in water | 3.5 g/100 mL (20 °C) |
| log P | -3.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.3 |
| Basicity (pKb) | 12.64 |
| Magnetic susceptibility (χ) | -1.1 × 10⁻⁶ |
| Refractive index (nD) | 1.427 |
| Viscosity | Syrupy liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 417.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2175.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3220 kJ/mol |
| Pharmacology | |
| ATC code | A12AA03 |
| Hazards | |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS05 |
| Signal word | No Signal Word |
| Hazard statements | May cause respiratory irritation. |
| Precautionary statements | Keep container tightly closed. Store in a cool, dry place. Avoid contact with eyes, skin, and clothing. Wash thoroughly after handling. Use with adequate ventilation. |
| NFPA 704 (fire diamond) | 1-0-0 |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 (oral, rat): 7,420 mg/kg |
| LD50 (median dose) | 7,590 mg/kg (rat, oral) |
| NIOSH | WL3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Calcium Gluconate: Not established |
| REL (Recommended) | 10–30 mL of a 10% solution (intravenous injection) |
| Related compounds | |
| Related compounds |
Gluconic acid Gluconolactone Calcium carbonate Calcium chloride Calcium lactate |

