Calcium Dithionite: A Closer Look at a Niche Chemical
The Historical Journey of Calcium Dithionite
Calcium dithionite doesn't turn up often in everyday conversation, but its story goes back. The dithionite salts, especially sodium dithionite, cropped up in the dye industry in the late 1800s. Calcium dithionite arrived a bit later, nudged along by the rise of industrial chemistry in Europe. At first, researchers stuck close to established uses for dithionites: reducing agents in textiles, pulp and paper, and mineral processing. Most people in the field talk more about sodium than calcium, but anyone who has tried to work with calcium dithionite can tell you the differences aren’t just academic. Sodium versions brought punchy reactivity, but calcium dithionite made a name for itself where a lower solubility and less sodium content suited the process better.
Product Overview
You don’t find calcium dithionite sitting on hardware store shelves. Its users know why they pick it over other reducing agents. In dyeing and bleaching, especially with specialized fabrics or certain pulp types, handling sodium can lead to issues involving migration or salt buildup. Calcium dithionite, on the other hand, brings controlled reactivity. It gets the job done for color stripping and specialized reducing needs without dumping as many unwanted ions into vats and machines. That’s a big deal in closed-loop industries, where every input has to count toward a predictable output.
Digging into Physical and Chemical Properties
Calcium dithionite, usually delivered as a white to slightly off-white powder, shares a family resemblance to its more common sodium cousin, yet it’s less soluble in water. Those who’ve worked in a lab see it as a fine, sometimes clumpy powder, giving off a slight sulfur odor. This compound doesn’t hang around for long exposed to air or moisture—dithionites tend to break down, so storage conditions matter. Chemically, it offers a two-electron reducing punch, crumbling azo bonds and breaking chromophores in dyes. During all this, calcium doesn’t interfere with many processing reactions, so its chemical profile stays pretty clean for specialty reductions.
Technical Specifications and Real-World Labeling
Producers tend to shy away from exact numbers in open literature since every application pulls specs in a slightly different direction. One common thread: purity. Impurities, especially iron or heavy metals, mess with the reduction power and end up staining products downstream. Particle size also ranks high—too fine, and you’re dealing with dust; too coarse, and it won’t dissolve fast enough where it matters. Labels may touch on maximum moisture, trace metal content, and shelf life, but trust between supplier and buyer counts as much as a sticker.
Preparation Method: From Raw Materials to Usable Product
Most people won’t whip up calcium dithionite in their garage. It often starts with sulfur dioxide bubbling through a suspension of calcium hydroxide and a reducing agent, like zinc or sodium borohydride. Commercial producers focus on minimizing waste sulfur and watching reaction conditions, because stray air oxidizes the product before it can even hit a package. Since dithionites hate moisture, drying techniques matter too. After synthesis, the powder heads straight into sealed containers with desiccants—every step built with that looming threat of decomposition.
Chemical Reactions and Tweaks
Calcium dithionite shines where direct reduction is needed. It’ll tear down vat dyes, kick off brightening in the pulp and paper mill, and knock Cu(II) ions to Cu(I) without much fuss. In real settings, the chemistry balances between speed and control. Additives tweak reaction rates; sometimes manufacturers blend it with sodium salts to get just the right dissolving profile. Research circles have poked at modifying the structure to get more shelf-stable versions, but air and water still remain its sworn enemies. Chemically speaking, the main appeal stays rooted in predictable reduction, cleaner byproducts, and a calcium cation that doesn’t disrupt downstream chemistry.
Synonyms and Alternate Product Names
In catalogs, you’ll see calcium dithionite go by “calcium hydrosulfite” or sometimes “dithionous acid, calcium salt.” Don’t get sidetracked—this isn’t the same as calcium sulfite or calcium thiosulfate, both of which show up in related applications but bring totally different properties. Sometimes suppliers list it under abbreviated trade names, but anyone in a technical field tends to stick with the IUPAC or CAS names for clarity.
Safety and Operations: Staying on the Safe Side
I’ve learned that you can’t cut corners with strong reducing agents. Calcium dithionite isn’t explosive, but it can decompose if it gets wet, producing sulfur dioxide and potentially flammable byproducts. Handling it means gloves, goggles, and a well-ventilated work area. Safety data sheets highlight danger from dust inhalation and accidental contact with acids, which jumpstarts decompositions in a hurry. Storage stays just as important as use—sealed containers, cool and dry spaces, and strict inventory management keep things stable. Training operators to spot early signs of degradation—clumping, yellowing, odd smells—makes the difference between an uneventful shift and a near-miss emergency.
Where Calcium Dithionite Fits: Applications That Matter
It stands out in the world of reducing agents for textiles and specialty pulp treatments. In the dye house, calcium dithionite strips color from fabrics, prepares fibers for re-dyeing, and pulls down excess chromophores without leaving sodium residue. The pulp and paper crew likes it for controlled brightness improvements, especially where water hardness makes sodium-based products troublesome. Ore flotation sometimes turns to calcium dithionite for selective reductions, picking out metals that would otherwise ride out the process untouched. Its lower solubility turns out to be a feature, not a bug, in processes that demand slow, steady reactivity.
Research and Development: New Ideas in Old Chemistry
Lately, researchers keep pushing for alternatives to high-residue chemicals. Calcium dithionite enters this conversation as industries search for ways to clean up downstream water and reduce environmental burdens. Some teams have tested blends with other reducing salts to stretch product life or tweak dissolving curves. Others get curious about microencapsulation, imagining a world where sensitive compounds stand up better to humidity and air. The literature’s still thin compared to sodium-based products, partly because the market remains smaller. But as cost and environmental pressures climb, interest grows in anything that can bring specialized reduction without complicating waste streams. Every push for green chemistry ends up re-examining legacy chemicals.
Toxicity Research: Understanding the Human and Environmental Burden
Safety researchers haven’t flagged calcium dithionite as especially toxic, but it still produces sulfur dioxide and other irritation-causing gases if mishandled. There’s evidence that runoff from industrial sites ties back to aquatic toxicity, usually due to the breakdown products rather than the original salt. Workers exposed to dust over long periods face the usual risks—skin and eye irritation, respiratory troubles. Regulatory bodies focus on limits for total dithionite use in air and water, pushing for tighter controls on venting and runoff. Researchers keep calling for further studies to track chronic vs. acute exposures, especially in industries that mix calcium dithionite with more reactive partners.
The Road Ahead: Future Prospects for a Specialized Salt
Calcium dithionite sits at the intersection of old-school industrial chemistry and the push for cleaner, more targeted processes. The current uptick in sustainable practices has industries looking twice at every tool in the shed, and legacy chemicals are no exception. Upgrades in storage, handling tech, and microencapsulation stand to extend product life and cut down on waste. If research teams crack the stability challenge, this compound may win a wider audience outside its traditional circles. Its value lies less in replacing all sodium-based reducing agents and more in carving out roles where its specific strengths shine—especially in closed-cycle or low-residue operations. As environmental and cost pressures keep building, I expect interest won’t fade, and new approaches to handling, packaging, and deploying calcium dithionite could take this quiet specialty chemical into unexpected places.
What is Calcium Dithionite used for?
What Exactly Is Calcium Dithionite?
Calcium dithionite sounds like something out of a science lab, but it's got some practical uses rooted in industries that touch daily life. If you've ever noticed how white your clothes can get, or enjoy printing with sharp, crisp images on paper, there's a good chance this chemical has played a part. Chemists mix it up as a pale powder that’s good for reducing, which in simple terms, means it can help strip away color or oxygen from other substances.
Pulp and Paper: Where White Stays Bright
Many people don’t realize how much goes into making paper white and bright. Most office paper starts off brown or yellowish because of the natural materials in wood pulp. Factories turn to chemicals like calcium dithionite to strip out that stubborn color. This process, called bleaching, needs a reliable reducing agent. Calcium dithionite breaks apart chromophores — tiny fragments in pulp that give it color. Without intervention, we’d probably be writing on dingy sheets instead of those clean pages you see every day.
Textile Industry: More Than Color Removal
Dyeing fabric isn’t simply about splashing color on and calling it a day. Mills often need to wash out old dye, prepare materials for new shades, or rescue batches when color goes astray. Calcium dithionite acts fast when it comes into contact with dyes, especially vat dyes and sulfur dyes. It can strip away color to fix mistakes or help with "white-outs” on patterns. Textile workers appreciate its quick-dissolving nature and how it doesn’t leave behind residues that might mess with fibers or future dye jobs.
Role in Environmental Clean-Up
Factory wastewater can come out looking pretty dark after processing textiles and paper. Those ugly shades aren’t just unsightly; sometimes, they also contain pollutants. Some plants use calcium dithionite to break down colorants and other contaminants before letting water leave their facility. Cutting down on toxic material going into streams and rivers makes a difference for anyone who cares about clean water. The process isn’t perfect, but it stands as one of a handful of tools used to tackle industry waste right at the source.
Risks and Responsible Usage
Every chemical has a downside, and calcium dithionite is no different. The compound isn’t especially toxic to people in small doses, but it reacts with water and acids to produce sulfur dioxide. That gas stings the eyes and throats, and factory workers need solid training to stay safe. Facilities make sure the areas are ventilated, and protective gear stays within easy reach. Thinking about long-term exposure, the dust can bother lungs with extended contact. Industry has improved safety training after incidents over the years. Most problems come down to poor storage or not following protocols, which reminds us that knowledge and preparation still go a long way in keeping chemicals safe.
Looking At Sustainable Options
Society wants cleaner methods that don’t add new problems while solving old ones. Researchers keep hunting for bleaching agents that are gentler on the planet. Some promising new products escape the sulfur stink and add fewer other hazards to the supply chain. It takes more investment, but customers and regulations keep pushing companies to make the shift. Until those become mainstream, calcium dithionite stays a mainstay, mainly for its speed and affordable cost.
Is Calcium Dithionite safe to handle?
What the Chemical Brings to the Table
Calcium dithionite pops up in pulp bleaching, textile dyeing, and water treatment. It does a good job as a reducing agent. People working with it usually understand its punch. I spent a summer in a textile plant where the powder showed up on the supply shelf. Most workers had old stories about clouding the air with a messy spill. That chemical experience sticks because safe handling is a constant part of the job.
Looking at Human Health
Breathing in calcium dithionite dust isn’t pleasant. Eyes burn. Throats tingle. Even minor contact with skin may cause rashes, especially if left unwashed. I remember one forklift operator who brushed powder off his arm and went the rest of the day itching. Swallowing calcium dithionite sets off stomach pain, nausea, and sometimes more dangerous reactions. OSHA and NIOSH give guidelines around dust control and skin exposure because of real-world injury reports.
In practice, the risk feels higher where controls are relaxed. Once, a crew switched suppliers and nobody wanted to read the new safety sheet. A bag ripped, and two workers coughed for half an hour before heading home early. Simple actions, like wearing goggles and dust masks, often made the biggest difference in daily safety.
No Magic Bullet for Environmental Hazards
Calcium dithionite decomposes fast in water, which helps lower environmental persistence. It still poses a threat to aquatic life if large quantities spill. I watched wastewater techs sample downstream after an accidental discharge—the fear shows when local fish populations are at risk. Facility manuals often focus on secondary containment. Training staff to recognize early warnings saves ecosystems and headaches down the line.
Sometimes, plant operators cut corners on ventilation or washing stations. This choice turns small accidents into bigger emergencies. Ramping up basic controls, like fume extraction and regular sweep-downs, created real improvement where I worked. I saw fewer accidents the year management invested in training and modern gear.
Safe Habits Matter More Than Just Labels
Labels show calcium dithionite as an irritant and a combustible dust danger. I have yet to meet anyone in industry who skips glove use on purpose, unless they want a supervisor’s lecture. Basic safety—washing hands, closing bags tightly, and sweeping up spills—settles most problems before they start. Fire risk isn’t a joke. The powder can ignite if it piles up or hits a spark. Regular vacuuming and routine safety drills keep risk down.
Training stays as the single best shield against careless mistakes. I remember a veteran supervisor running mock drills every month—teaching new hires how to dodge the most common accidents. These habits transfer to home, too. Anyone handling strong chemicals in their garage or garden could use the same respect for chemical storage, labeling, and cleanup.
Building a Culture of Respect for Chemicals
Facts and experience add up to one truth with calcium dithionite: smart habits build a safer workplace. Companies score higher in safety records when teams learn about the real risks and don’t pretend a powder is harmless just because it’s common. Communities trust manufacturers who take training, transparency, and worker health seriously. Using sharp instincts and open communication about close calls strengthens safety more than any set of posters. Respect for chemicals, not shortcuts, drives every good outcome.
How should Calcium Dithionite be stored?
Why Care About Storage?
Calcium dithionite isn’t just another dusty chemical sitting on a forgotten shelf. I’ve seen more than a few labs treat it like a simple powder, then scramble once they realize it doesn’t behave quietly. As someone who’s worked in facilities where safety is a daily practice, I’ve learned that proper storage makes all the difference. Mishandling calcium dithionite sometimes looks like simple carelessness—then an odd sulfur smell starts drifting through the storeroom. It’s more than annoying; there’s real risk here.
What Makes Calcium Dithionite Tricky?
A big reason to pay attention is instability. Calcium dithionite breaks down if it even gets a hint of moisture. It also reacts quickly if nearby chemicals send off an acid vapor or the air gets warm and humid. One time, someone left a container loosely capped for just an afternoon, and the next morning there was a hard, yellowish crust—the powder had basically ruined itself.
This isn’t only about protecting product quality. Decomposition lets off sulfur dioxide and the area fills with an irritating, choking gas. It’s tough to work safely or efficiently in those conditions. People’s health—eyes, lungs, skin—matters much more than a few wasted supplies.
Storage Tips that Work
From years of experience, I suggest keeping calcium dithionite in airtight, corrosion-resistant containers—plastic ones seal better and stand up to the powder’s stubborn streak. Strong containers stop air and stray moisture, but document any cracks or dents quickly. Rooms set aside for chemical storage do better with dedicated shelving for just sensitive agents—cross-contamination from random bottles spells trouble later.
I know not every storeroom is built like a pharmaceutical vault, but you can always pick the driest, coolest area available. The best range is below 25°C. Humidity sneaks up fast; those small silica gel packets or bigger desiccant canisters inside the cabinets handle the worst of the excess moisture. Don’t crowd the storage space. Proper spacing among containers lets air circulate and makes leaks easier to spot.
Labs or factories that rely on calcium dithionite for large batch work benefit from small, separate containers over one huge drum. Opening a freshly sealed drum pulls humid air inside every time, speeding up breakdown. Smaller batches mean less waste and safer handling if a spill ends up happening.
Don’t Ignore Warning Signs
Some folks wait until they smell rotten eggs or see the powder change color. It’s smarter to set regular checks—look for chemical odor shifts, caked powder, or pressure build-up in containers. Safety training should include information about how this compound behaves when it starts to fail, not just how it looks in a textbook.
Small labs can hold “spot checks” once a week, involving everyone who has access to chemical storage. Bigger organizations often move toward electronic logs to track storage conditions—records help trace problems and prevent repeats. Emergency ventilation systems and personal protective equipment come first if a spill or container failure is noticed.
Supporting Safer Workspaces
Simple steps—spaced containers, sealed lids, dry rooms, and regular checks—bring a sense of order and stress less about accidents. This builds trust not only in a product, but in the people who work side-by-side with it. Seeing co-workers confidently recognize a change in the storage area or an off-smell brings peace of mind. Getting comfortable with the care of calcium dithionite means problems turn up less, and everybody goes home safe.
What are the physical and chemical properties of Calcium Dithionite?
The Basics of Calcium Dithionite
Calcium dithionite draws attention for its role in textile and paper industries, but fewer people realize its presence in many chemical supply rooms. People who work with bulk chemicals notice the difference a well-understood compound can make. This isn’t just a powder in a drum—it interacts with air, moisture, and other materials in ways that demand respect and a good sense of chemistry.
Physical Nature: More Than Meets the Eye
Calcium dithionite appears as a white or pale yellow solid. It doesn’t attract much attention in a beaker. Pick it up and you’ll notice it flows like any fine crystalline powder, but it crumbles if pressed. It doesn’t have a strong odor, making it a bit of a sleeper compared to pungent industrial chemicals. If you leave it exposed to the air, trouble starts fast. The substance reacts in humid air, and pretty soon, you’ll see clumping or a change in color. That means its stability outside sealed containers drops. I’ve lost material to careless storage—those airtight drums aren’t for show.
Solubility stands out. Calcium dithionite dissolves in water, but not quickly. The solution can end up cloudy. Temperature matters—a little heat speeds things up, but push it too far and you risk breaking it down, which destroys the dithionite you want to use. Without stable, cold storage, people lose valuable product to slow decay, often without realizing it’s happening. That’s a lesson straight from the bench—care with storage pays back every time.
Chemical Behavior: Reduction Powerhouse
The real draw comes from calcium dithionite’s strength as a reducing agent. Anyone in dyeing or bleaching knows the value of a reduction step, and calcium dithionite packs a punch. The key: it donates electrons and breaks apart contaminants or dyes, making it a solid option where stronger, more hazardous reducers would be overkill. Throw it into an aqueous matrix and watch the color fade—there’s no need for fancy catalysts or extreme temperatures.
Exposure to oxygen takes that strength away, fast. Calcium dithionite’s instability with air means timing and method matter. If mixed too early, the oxidation kicks in before it hits the stains or fibers. As someone who’s watched expensive batches lose power in open tanks, I know every second counts once it’s mixed with water. Handling the material under nitrogen, or at least keeping it sealed until the last possible moment, can keep its reducing action focused where you want it.
Safety and Sustainability Questions
Nobody loves handling chemicals with tricky safety profiles. Calcium dithionite releases sulfur dioxide and other sulfur compounds when it breaks down, especially if heated or mixed carelessly. You’ll want real ventilation and a plan for spills. I’ve seen the results of poor air flow—tingling eyes and irritated throats. Proper PPE and systems can save everyone’s afternoon and maybe their long-term health.
Waste management brings another layer of concern. Disposal needs a thoughtful approach, as broken-down dithionite can add to the load of sulfur compounds in wastewater. Reusing or neutralizing leftover material pays off for both the budget and the planet. More research into containment and breakdown products can help industries shrink their environmental impact, and it’s worth investing in better testing and secondary treatments. These changes support safer workplaces and healthier downstream ecosystems.
What precautions should be taken when using Calcium Dithionite?
Understanding What You’re Handling
Anyone working in dyeing, water treatment, or some types of paper production will probably cross paths with calcium dithionite sooner or later. This white or pale yellow powder does its job as a reducing agent, but what often gets overlooked is how a person’s safety can hinge on a few simple habits. From the first time I stepped onto a dyeing factory floor, I learned pretty fast that respecting chemicals like this saves you a world of trouble.
Personal Protection Comes First
Normal work clothing offers little defense. Calcium dithionite’s dust annoys skin and eyes and, mixed with moisture, even gets reactive. So, I always reach for gloves, long sleeves, and a pair of solid safety goggles. For heavy work or lots of movement, a dust mask or simple respirator makes breathing easier and keeps headaches at bay from any stray particles. Plenty of folks ignore this, and a single shift with irritated skin will show why that’s shortsighted.
Venting and Air Flow Matter
Any room where you work with this stuff needs solid air flow. Poor ventilation can let sulfur dioxide and other noxious fumes build up. These gases aren’t just stinky—they sting your throat and lungs, and long exposure opens the door to more serious health risks. In every shop I’ve been to, a running extraction fan is non-negotiable. Cracked windows or open doors help, but fans above the workbench make the real difference.
Handling and Storage
Moisture ruins calcium dithionite, turning it unstable. It can break down, releasing sulfur dioxide. I always recommend keeping the powder locked away in sealed containers, away from water sources and out of direct sunlight. Even humidity eats away at the shelf life. If you don’t open bags or drums with care, tiny puffs can spill out, so always open packages slowly and over a tray. Warehouse staff or anyone in supply chains needs regular reminders about this. If you’ve ever watched a colleague fumble a tear in a bag, you’ll know how fast the dust spreads.
Never Mix With the Wrong Materials
I once saw a co-worker dump leftover calcium dithionite down a drain that hadn’t been cleared. The reaction with acids or even rusty pipes led to unexpected fumes and an evacuation. Direct mixing with acids speeds up sulfur dioxide production, and mixing with hot water, oxidizers, or peroxides isn’t just a bad idea—it’s dangerous. Instruct every user on safe disposal. Collect waste according to local hazardous material rules. Never guess.
Preparedness for Spills and Accidents
No matter how careful you are, spills happen. The right tools—a broom, dustpan, plenty of dry absorbent material—not only speed up clean-up but limit how much dust gets airborne. Dispose of swept-up dithionite in sealed bags. Water just spreads the chemical around and makes fumes, so stick to dry methods and call your supervisor or local environmental expert for big spills. An eyewash station and emergency shower, well-marked and open, should always be easy to reach.
Training and Communication
New staff don’t cook up safety rules on their own. Set aside time for specific, hands-on training with calcium dithionite whenever someone joins the team or if the process changes. Post instructions where everyone can see them. Don’t let anyone skip over the details. Knowledge, backed by clear practice, stops accidents before they start.
Looking to the Future: Safer Substitutes?
Some textile and industrial researchers are pushing for alternatives that cut down reactive hazards and environmental impact. Until those become more affordable or widespread, a careful relationship with calcium dithionite and respect for safety routines stand between a normal shift and a trip to the hospital.
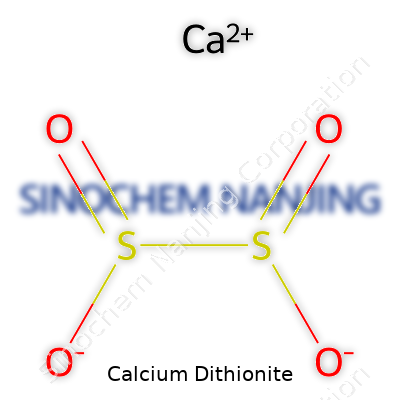
| Names | |
| Preferred IUPAC name | Calcium dihydroxy(vdisulfito)dihydrate |
| Other names |
Calcium hyposulfite Calcium hydrosulfite |
| Pronunciation | /ˈkælsiəm daɪˈθaɪənaɪt/ |
| Identifiers | |
| CAS Number | 16544-54-8 |
| Beilstein Reference | 1206722 |
| ChEBI | CHEBI:91221 |
| ChEMBL | CHEMBL4580962 |
| ChemSpider | 9271425 |
| DrugBank | DB14497 |
| ECHA InfoCard | 100.028.851 |
| EC Number | 231-873-8 |
| Gmelin Reference | 82858 |
| KEGG | C18614 |
| MeSH | Dithionites |
| PubChem CID | 24857 |
| RTECS number | NJ8575000 |
| UNII | R09Q8V1G6A |
| UN number | UN1931 |
| Properties | |
| Chemical formula | CaS2O4 |
| Molar mass | 208.24 g/mol |
| Appearance | White or light yellow crystalline powder |
| Odor | Slight sulfur dioxide odor |
| Density | 2.45 g/cm³ |
| Solubility in water | Soluble |
| log P | -3.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | Approx. 7.0 |
| Basicity (pKb) | 11.0 |
| Magnetic susceptibility (χ) | -53.0e-6 cm³/mol |
| Refractive index (nD) | 1.64 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 179.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -947.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1360 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | Hazardous if swallowed, causes skin and eye irritation, may release toxic gases on contact with acids, decomposes to produce sulfur oxides. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H319, H373 |
| Precautionary statements | P210, P220, P221, P261, P280, P305+P351+P338, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-0-1 |
| Lethal dose or concentration | LD₅₀ oral (rat): 2,500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 2,500 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Calcium Dithionite: Not established |
| REL (Recommended) | REL (Recommended)": "Not established |
| IDLH (Immediate danger) | Unknown. |
| Related compounds | |
| Related compounds |
Sodium dithionite Potassium dithionite Lithium dithionite Calcium sulfite Calcium thiosulfate |

