Calcium Cyanide: Looking at a Double-Edged Chemical
The Story Behind Calcium Cyanide
Calcium cyanide landed in the chemical world more than a hundred years ago, making its entrance with the push from the gold and silver mining industries that were desperately searching for quicker ways to extract metals from ore. Starting in the early 1900s, plant chemists replaced older, slow-leaching techniques with something far less forgiving—using calcium cyanide, which promised both potent results and some notorious risks. Over the years, chemists tinkered with production processes, learning that strong nitrogens and alkali earth metals could open a new chapter in mass gold extraction. Those old mining towns and faded patents remind us that the quest for efficiency has always carried a price.
What’s Inside This Compound?
At first glance, calcium cyanide looks unimpressive: a chalky gray or white solid, often showing up as granules or powder. It barely smells, yet when water or acid gets involved, the reaction brings out a bitter almond odor—a warning sign from the hydrogen cyanide gas released. On the technical side, this compound answers to Ca(CN)2, holding two cyanide ions for every atom of calcium. It dissolves in water, making it handy for solution-based applications. Many in the business call it by other names—black cyanide, nitrocal, even merely “CN calcium”—yet the core dangers and benefits do not change, no matter how it’s labeled on the drum.
How Calcium Cyanide Stands Out
There’s a reason calcium cyanide has kept its spot in mining for all these decades: most commercial blends deliver about 50-60% pure Ca(CN)2 alongside traces of sodium and iron, sometimes leftover from lower-cost manufacturing. Consistency can swing from batch to batch, mainly based on the production route. The technical sheets all stress a key rule—moisture control. Exposure to air soaks up water and triggers the slow but steady release of hydrogen cyanide. The stability issue remains a safety headache, directly connecting the physical traits of the powder to the real risk of toxic off-gassing.
Preparation in the Real World
Calcium cyanide’s usual prep method starts with calcium carbide. That gray solid reacts with nitrogen and carbon sources, like coke or even crude coal, inside a high-temperature furnace. I’ve watched engineers feed raw materials through rotary kilns, battling dust and keeping the temperature clockwork-steady, since a swing above or below the mark reduces yield and produces more byproducts that complicate disposal. The mix sometimes contains residue from upstream processes, which must get filtered out to make the final product usable. Adding lime or sodium cyanide tweaks the recipe, depending on cost, purity requirements, and local safety standards. There’s rarely a dull moment with calcium cyanide plants—tiny leaks or spills draw real-time alarms because of the poisonous gases at play.
Reactive by Nature
Chemists learn early that calcium cyanide doesn’t play well with acids or moisture. When water or a weak acid enters the picture, hydrogen cyanide gets released fast—a classic case of a useful substance that doubles as a hazard. If the powder sits in damp air, trace hydrolysis generates slow but steady cyanide vapor, fueling both industrial use and toxic risks. The ability to form quick complexes with metal ions makes this compound so valuable in gold and silver recovery. It can be modified through careful roasting or by swapping some components with sodium or potassium salts, changing solubility and performance. That versatility may drive innovation, but it creates a minefield of safety headaches for facilities that still depend on older infrastructure.
What’s in a Name?
Calcium cyanide hopped between names as chemistry textbooks grew thicker. Some old miners called it black cyanide because of color from impurities, while technical operators preferred more precise terms. Among chemists, Ca(CN)2 needs no further description. In the lab, folks may cross paths with it under labels like nitrocal, cyanlime, or calcium dicyanide, but all those point back to one family tree of strong cyanide sources partnered with an earth metal—no fancy branding ever tampers down the risks found inside each sack.
Working with Danger—Safety and Operations
Hard truths hit home in any plant or lab that opens containers labeled calcium cyanide. There’s a sharp line between safe handling and disaster. Breathing in the airborne hydrogen cyanide that escapes from careless pouring or wet floors can stop a heart or wreck a nervous system in minutes. Gloves and full suits don’t just fill policy—they save lives when the powder begins to clump on humid days. Emergency kits holding amyl nitrite or sodium thiosulfate have saved many technicians caught off-guard. In my experience, new hires who breeze through safety videos find themselves humbled in drills. Some countries call for full training, from spill containment to lockout-tagout for mixing machines, because one slip, and an entire community feels the consequences. Storage always follows dry, airtight rules, far from food, acids, and untrained hands.
Why Industry Still Leans on Calcium Cyanide
Mining stands as the dominant user, where calcium cyanide’s talent for locking onto precious metals keeps it in business despite rising regulations. Silver and gold leaching work better with this compound, mainly in remote or smaller-scale mines that need local solutions for ore that resists other cyanide blends. In pest control, it gets released in small doses to deal with burrowing animal infestations, though changing rules and worries over non-target species cut this use back over the years. Smaller quantities sometimes end up in specialty cleaning agents or lab reagents, but most buyers stick to the gold circuit. Many current projects aim to reduce spills from tailings ponds and to replace open-air bunds with closed systems, so any slip won’t poison downstream rivers.
Research—Toxicity, Hope, and Tough Questions
Researchers have worked hard since the last century, when tragic accidents drew a direct connection between cyanide leaks and sharp upticks in animal and human poisonings. Toxicity research remains a priority, especially as more studies explore chronic, low-level exposure risks for miners and chemical workers. The cyanide ion, which hides quietly until sparked by moisture or acid, acts as a metabolic disruptor, blocking enzymes vital for energy production in cells. A loaded lungful can kill a person in less than an hour. Studies from the 1970s through today confirm that long-term exposure, even at low levels, can hit memory, mood, and cardiovascular health. Advances in detection—such as electronic nose systems and faster water assays—offer early warnings for small leaks that could turn deadly. The medical community faces the continuing challenge of quick diagnosis, since early symptoms mimic benign conditions like headaches or faintness.
Chasing a Safer Future
Moving toward a world with less cyanide has never been simple. Gold and silver remain crucial for electronics, currency, and medical devices, so many sectors cannot ditch the compound overnight. Funded projects in places like South Africa, China, and Canada search for greener leaching agents—thiosulfate, glycine, even bacteria-based bioleaching. Some miners already switched to closed-loop cyanide management, lowering emissions and capturing more waste. I’ve seen a growing appetite for education and outreach, especially among younger engineers who know firsthand what a careless spill means for their communities. Unless the world votes to abandon precious metals, calcium cyanide will remain part of industry. What matters now is not just profit or productivity, but the guarantee that every worker leaves the shift healthy and every tailings pond holds its poison, not only on paper but in real life.
What is calcium cyanide used for?
How Calcium Cyanide Ends Up at the Worksite
Most folks probably never see a drum of calcium cyanide up close, but this chemical still plays a part in industries that put food on the table for many. Calcium cyanide helps miners pull gold and silver from ore. In my own family, we’ve got folks who spent time in mines, so hearing stories about chemical extraction wasn’t rare. They told stories of big vats swirling with solutions that help separate out precious metals. Calcium cyanide often supplies the cyanide ions that latch onto bits of gold or silver. Without this reaction, those metals would stay stuck in rocky dirt, impossible to pull out and sell.
The Power Plants and Pest Control Angle
Some industrial sites also bring in calcium cyanide for gas production. Factories might use it to create hydrogen cyanide or cyanogen gas, which both end up as building blocks for plastics, dyes, and other essential products. These chemical routes keep manufacturing humming in countries where buying ready-made raw materials can get expensive or slow. It’s a behind-the-scenes role, but when pipelines run dry or shipments stall, production managers notice the squeeze.
Agriculture has a more controversial relationship with this chemical. Decades ago, people turned to calcium cyanide for pest control, especially in fumigating soil and old structures. It proved lethal to pests, but it posed just as big a threat to those handling it. I remember my grandfather talking about the warnings printed in bold on chemical drums—these warnings aren’t for show. The risk explains why safer alternatives now tend to get the nod.
The Real Concerns About Handling and the Environment
Calcium cyanide is nothing to mess around with. If someone breathes in cyanide dust or gets it on their skin, the body can’t just shrug it off. Cyanide exposure can overwhelm organs fast, and medical teams need speed and training to respond. Even in my own town, emergency responders drill for chemical spills, despite the low odds. This training grows out of real respect for how a single misstep can turn tragic. Households don’t handle this chemical, but the risks still reach the wider community through transport, waste, and runoff.
Water sources sit downstream of most mining and industrial projects. Once, a relative working in a cleanup crew checked wells after a spill; lives changed when local water needed hauling in by truck for months. Environmental damage from cyanide compounds can linger, especially if companies downplay their responsibility or cut corners. The world saw this after mining mishaps in several countries, where rivers remained tainted, and fishermen lost their livelihood.
Pushing for Smarter Solutions
Strict rules, better worker education, and transparent reporting keep public trust from cracking. Regular audits, emergency response systems, and modern engineering controls help keep cyanide from moving where it shouldn’t. In some regions, tech improvements shrink the need for calcium cyanide at all. Thiosulfate and other less-toxic approaches can swap in, letting companies dig up metals without as much risk.
People I know working in modern mines say the real focus now rests on protecting both workers and surroundings. Firms that build trust don’t just talk the talk: they invite locals to see safeguards, and they welcome questions. These changes didn’t happen overnight. They came from workers, whistleblowers, and affected communities refusing to stay silent after things went wrong.
Decisions about using harsh chemicals often come down to whether safer, effective choices exist. Calcium cyanide once filled a need, but the world’s moving toward options that don’t sacrifice long-term health for short-term gain.
Is calcium cyanide toxic or dangerous?
Understanding Calcium Cyanide
Calcium cyanide shows up in textbooks as a white powder or granule that carries a sharp, bitter almond smell. It slips into the conversation most often for its role in gold mining and certain pesticide applications. Its reputation doesn’t come from rarity but from what it can do in the wrong hands or under careless conditions.
What Makes It Dangerous
Being around calcium cyanide can turn life-threatening in seconds because this compound releases hydrogen cyanide gas. The gas gets inside by breathing, swallowing, or even light skin contact. Science says hydrogen cyanide breaks down the body's ability to use oxygen. People exposed to enough stop breathing. Even brief exposure in poorly ventilated rooms leads to confusion, rapid breathing, loss of consciousness, and possibly death if help doesn’t arrive quickly.
Real-World Incidents and Health Concerns
Reports from mining sites in Asia and Africa point out workers who handled contaminated water, dust, or improper storage of chemicals suffered from dizziness, skin irritations, and headaches. A few tragic cases involved children playing near open dumps, who showed up at clinics with severe poisoning. Just last year, emergency teams in West Africa responded when calcium cyanide spilled in a community well, forcing entire villages to evacuate the area for cleanup.
Why Should People Care?
Dismissing a toxic compound like calcium cyanide as just another industrial chemical ignores the human factor. Anyone living or working near gold mining sites, agriculture fields, or chemical plants stands at risk if companies skimp on safety. The U.S. Environmental Protection Agency lists cyanide compounds as hazardous, and strict guidelines call for sealed containers, full-body protective gear, and immediate response plans.
Having worked in a laboratory during college, safety talks drilled into me the need for respect and care when handling reactive compounds—especially those tied to hydrogen cyanide. Gloves, face masks, fume hoods, and never letting a drop touch exposed skin were not suggestions, but rule number one. Accidents from overconfidence or fatigue threatened everyone nearby, not just the person making the mistake. Time and again, stories from more seasoned chemists highlighted how swift rescue and strict attention to protective gear saved lives.
Pushing for Better Safety
Communities and workers want assurance that storage and disposal happen far from water sources and food crops. Regulatory agencies play a direct role by running surprise inspections and punishing companies cutting corners. More investment in on-site monitoring gives early warnings before leaks turn into disasters. Companies that use calcium cyanide should offer frequent training, stock up on emergency equipment, and build clear lines of communication so any mishap gets tackled before it spreads.
The facts show calcium cyanide brings risk, and the people near it pay the price if something goes wrong. Practical safety habits, good tools, and respect for the potential danger prevent news stories from turning tragic. The numbers back this up—incidents drop sharply when communities, governments, and businesses stop treating safety as paperwork and face it head-on. Knowledge, preparation, and accountability set the real difference between routine work and a headline nobody wants to read.
How is calcium cyanide stored and handled safely?
Why Calcium Cyanide Demands Respect
Calcium cyanide often comes up in industrial settings, especially in mining and metallurgy. The stuff isn’t all that forgiving. Even a small amount will put workers and the environment at risk if managed carelessly. Contact or inhalation often leads to poisoning, and, as someone who’s spent time around chemical warehouses, I can say even a hint of moisture can turn a safe container into a hazard.
How People Store It Without Trouble
Good storage starts with location. In my experience, leaving calcium cyanide anywhere close to acids or oxidizers breeds disaster. Even airborne acids in the same area will trigger chemical reactions nobody wants to deal with. Factories with the best records keep it in a standalone, locked facility, far from other chemicals, water sources, and food.
Controlling humidity saves lives. Many containers face corrosion or leaks over time, and even a drop of water inside will set off toxic hydrogen cyanide gas. To dodge this, people use containers made of steel drums lined with tough plastic, always sealed tight. Pallet racks keep containers off the floor, just in case any water creeps inside during cleaning, storms, or spills.
Training Makes the Difference
Not all accidents happen because of faulty containers. Sometimes someone new misreads a label or skips personal protection. The smartest managers send every worker through rigorous training before trust enters the equation. Usually this means direct, scenario-based drills: “Here’s how to spot leakage. Here’s how quickly you must respond.” Repetition pays off.
Any reliable training covers protective gear. At minimum, everyone puts on chemical-resistant gloves, goggles, and a well-fitted respirator. Long sleeves, close-toed shoes, and thick aprons turn up in every trusted guide. Nobody cuts corners because even small mistakes can trigger an emergency.
Plans for Spills and Emergencies
Chemical mishaps hit hardest where nobody plans. The best teams post spill kits every few meters—complete with neutralizers like sodium thiosulfate. Big red signs show emergency eyewash stations and showers. I’ve witnessed drills where seconds saved made a clear difference, especially for dangerous substances like calcium cyanide.
In the chaos of an incident, communication stays critical. Supervisors keep radios charged, and all staff members memorize emergency protocols. Nearby hospitals and first responders receive updates that a site stores hazardous cyanide, so they know what to expect before an ambulance even rolls.
Waste Doesn’t Just Disappear
How calcium cyanide gets disposed of matters just as much as how it’s stored. Nobody dumps it down a drain or throws it in the regular trash. Licensed hazardous waste contractors take over, using locked trucks and special permits. Local and federal laws cover every step, from neutralization to tracking paperwork, for full accountability.
Reliable Supply Chains and Labeling
Every package shipped or received gets checked and logged. Labels show health hazards, emergency contacts, and handling rules, readable even after months in storage. Takeaway from my years seeing these deliveries—better labeling means fewer surprises, and extra caution on the loading dock keeps everybody walking home at the end of a shift.
Improvement Never Stops
Smart companies audit procedures after any close call, seeking feedback from workers. Nobody relies on last year’s training or outdated storage rules. Open conversations and lessons learned cut down risks for everyone. That’s what real safety looks like in the world of calcium cyanide.
What industries commonly use calcium cyanide?
Mining and Metal Extraction
You see a gold ring on someone’s finger, or silverware passed down through the family. Most people don’t connect those beloved objects to gritty mining practices. Gold and silver, for example, have to be pulled out from ore, usually with the help of chemicals. Calcium cyanide is one of the main chemicals miners use for this task. Its power lies in how quickly it helps dissolve gold and silver, making it possible to separate tiny flecks from piles of rock. This isn’t the only tool in the mining toolbox, but it’s definitely one of the most potent.
Calcium cyanide can handle large volumes, and its solid form makes it easier to ship compared to other cyanides, which tend to be liquids or powders that spill easily. It’s no surprise mines in remote areas often use it, even if it means taking on some risk. Miners have to train every worker to respect what they’re dealing with. A single spill, if not handled quickly, can threaten workers and the surrounding environment. A few spills in places like Ghana and China have already ruined local streams for years. This is one reason the mining world invests heavily in better containment and stricter site management now.
Chemical Manufacturing
Factories that make chemicals usually run around the clock. While walking through a chemical plant during a college field trip, I noticed stacks of barrels labeled with warnings I hadn’t seen anywhere else. Some were filled with cyanide compounds for industrial use, including calcium cyanide. Their real draw is how easily they trigger reactions needed to create plastics, dyes, and pharmaceutical products.
A lot of the reactions involving calcium cyanide end up producing hydrogen cyanide gas, which gets captured and used as a building block for other industrial chemicals. Hydrogen cyanide itself is the starting point for products like nylon, acrylates, and specialty adhesives. It’s not glamorous—but without chemicals like calcium cyanide, big sections of the manufacturing sector would struggle to keep up with demand for these everyday materials.
Industrial Cleaning
Not every industry relies heavily on calcium cyanide, but some cleaning processes use it to strip metals of impurities or to prepare surfaces for electroplating. Automotive and electronics producers sometimes reach for it, especially if they need a quick, aggressive solution to clear away stubborn residues or oxides.
While it gets the job done, the tradeoff rests in safety. Calcium cyanide can turn toxic quickly, and strong rules about protective gear and fume hoods exist for a reason. A good friend who worked in plant maintenance described the safety briefings as some of the most intense he’s ever sat through—one careless mistake, and there’s no second chance.
Reducing Harm and Improving Practices
Calcium cyanide does solve real problems in each of these sectors, but the margin for error stays razor-thin. Workers receive more training now than ever before. Factories and mines use closed handling systems to shrink the risk of exposure. Regular equipment inspections and environmental monitoring are routine, not optional, for sites working with calcium cyanide.
Local residents and landscape groups have become powerful voices in pushing for better reporting, stricter regulation, and more research into safer alternatives. The drive to swap out high-risk chemicals for greener, less toxic options keeps picking up steam. This pressure from the outside and inside has convinced many operators to look for technology and chemistry that put people and ecosystems first, without letting up on productivity.
Industries depending on calcium cyanide share a responsibility. They’ve seen what can go wrong, and the tools to do better are sitting within reach.
What are the environmental risks associated with calcium cyanide?
Understanding Calcium Cyanide’s Impact
Calcium cyanide gets used mostly in mining for gold and silver extraction, but its story doesn’t end with industry. The real problem starts after the stuff leaves storage. If a spill occurs, the chemical reacts quickly with moisture—turning into hydrogen cyanide gas. This gas moves quickly and can cover a wide area, putting people and animals in danger. In a community near a mining site, a friend once told me they started noticing dead fish floating hours after a spill. The speed at which this chemical can do damage caught even the experienced folks off guard.
Waterways Take the Biggest Hit
Most mining operations sit close to rivers or lakes for practical reasons, but calcium cyanide increases the risk when it gets into water. Even tiny amounts can kill fish and crustaceans within minutes. Ammonia and simple cyanides break down with sunlight and microbes, but calcium cyanide sticks around. Studies from the US Geological Survey found high cyanide levels downstream from gold mines in Nevada. These spots saw a big drop in aquatic life—lots of fish, amphibians, and invertebrates just disappeared.
Drinking water supplies can get contaminated too. Once cyanide seeps into groundwater, the cleanup rarely goes smoothly. It's a nightmare for rural towns that don’t have much budget for water filtration. Living in a mining area taught me how quickly an environmental accident becomes a public health crisis.
Air and Soil Aren’t Safe Either
Cyanide fumes drift quickly. Agricultural fields near mining sites often end up with lower yields because cyanide disrupts plant growth. Local farmers have reported yellowing leaves and stunted stems after accidents. Soil bacteria suffer too, and that slows down natural detoxification.
A report by the EPA showed that even a short-lived cyanide leak can poison the soil for months. Cleaning it out takes energy and money that small towns usually lack. Children are especially at risk if they play near contaminated soil.
Wildlife Faces Greater Dangers
Raptors, scavengers, and even songbirds can die after eating poisoned prey. Cyanide doesn’t just stay put—it moves up the food chain. Research in Australia uncovered the bodies of wallabies and other native animals near old mining pits. The same thing shows up in reports from South Africa and South America. Each loss ripples out—the ecosystem starts to unravel.
Searching for Safer Solutions
Mining companies can install stronger containment barriers, monitor chemical use more tightly, and treat wastewater right at the source. Modern detox systems use ozone and hydrogen peroxide with far better results than older setups. Replacing calcium cyanide with less toxic reagents would help, but not every mine moves that quickly.
State inspectors should get more funding for unannounced visits and stricter fines. Technology like continuous monitoring sensors, which I’ve seen work wonders in municipal water plants, can spot leaks before they become disasters. Communities that push for transparency and stay involved end up safer. Mines closing open tailings ponds—covering them and planting grasses—cut down on wind-blown dust.
Holding companies accountable for cleanup, funding better technology, and listening to local voices could take us a long way toward protecting lands and lives. Environmental harm from calcium cyanide never feels abstract if you’ve watched your local river change color or worried your well water might not be safe tomorrow.
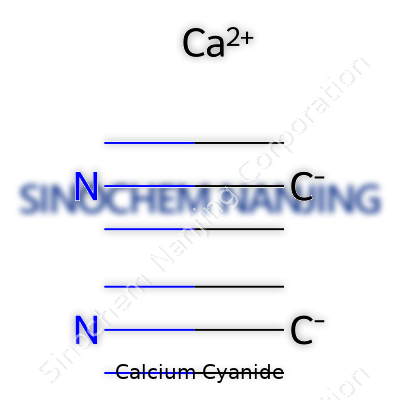
| Names | |
| Preferred IUPAC name | Calcium dicyanide |
| Other names |
Black cyanide Calcium dicyanide Calcium cyanide (Ca(CN)2) Cyanogen calcium |
| Pronunciation | /ˈkæl.si.əm ˈsaɪ.ə.naɪd/ |
| Identifiers | |
| CAS Number | 592-01-8 |
| Beilstein Reference | 1207036 |
| ChEBI | CHEBI:51821 |
| ChEMBL | CHEMBL1201587 |
| ChemSpider | 20209 |
| DrugBank | DB11557 |
| ECHA InfoCard | ECHA InfoCard: 027-001-00-5 |
| EC Number | 209-804-0 |
| Gmelin Reference | Gmelin Reference: **83350** |
| KEGG | C01698 |
| MeSH | D002117 |
| PubChem CID | 10797 |
| RTECS number | EZ9450000 |
| UNII | C1WWW1KQG4 |
| UN number | 1588 |
| Properties | |
| Chemical formula | Ca(CN)₂ |
| Molar mass | 94.11 g/mol |
| Appearance | White or grayish-white solid |
| Odor | almond-like |
| Density | 2.25 g/cm³ |
| Solubility in water | Moderately soluble |
| log P | -3.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | 4.04 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 116.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -298 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -771.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB18 |
| Hazards | |
| Main hazards | Toxic by ingestion, inhalation, or skin absorption; releases highly toxic hydrogen cyanide gas when in contact with acids or moisture; fire and explosion risk when in contact with acids or oxidizers. |
| GHS labelling | GHS02, GHS06, GHS09, Danger, H260, H300, H410 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Precautionary statements | P220, P223, P260, P264, P273, P280, P284, P301+P310, P302+P350, P304+P340, P305+P351+P338, P306+P360, P320, P330, P361, P363, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3 3 2 W |
| Autoignition temperature | > 200°C (392°F) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD₅₀ oral (rat): 11 mg/kg |
| LD50 (median dose) | LD50 (median dose): 11.3 mg/kg (oral, rat) |
| NIOSH | BIC |
| PEL (Permissible) | 5 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit): 5 mg/m³ (as CN) |
| IDLH (Immediate danger) | 50 mg/m³ |
| Related compounds | |
| Related compounds |
Calcium carbide Calcium cyanamide Sodium cyanide Potassium cyanide |

