Calcium Chlorate: A Spotlight on Its Place in Chemistry and Industry
History and Development
Sometimes, real progress comes out of patient effort and no shortage of trial and error. Calcium chlorate, a chemical often overlooked in favor of better-known relatives, has quietly shaped many corners of industrial chemistry. Its story reaches back to the late 19th century, when the pursuit of better oxidizers for agriculture and related processes led to methods for producing large quantities from basic raw materials. Farms and industry workers once depended on less stable chemicals, so calcium chlorate earned attention by sticking around without turning into dust or sludge at the first opportunity. Early chemists managed to push calcium chlorate past laboratory curiosity status through improvements in electrolysis and chemical extraction. Like a lot of chemical mainstays, it didn’t get famous overnight, but its reliability and performance eventually gave it a seat at the table.
Product Overview
Calcium chlorate doesn’t carry the name recognition of some oxidizers, but anyone who works with herbicides or specialty chemicals gives it plenty of respect. It keeps a white, crystalline form and performs steadily as an oxidizing agent. Some industries look to it for its predictable decomposition and straightforward chemistry; others select it to sidestep regulatory headaches that follow certain alternatives. Despite a shift in the regulatory landscape over time, especially with changes in how persistent chemicals are viewed, calcium chlorate has weathered plenty of transitions by finding niche uses that hinge on its chemical stability and decent solubility in water.
Physical and Chemical Properties
The compound’s properties make it a workhorse in situations calling for controlled bursts of oxygen release. Having worked as a chemical technician long enough to get a feel for handling different powders and granules, I can say calcium chlorate ranks high on the “manageable” scale: it doesn’t cake excessively or degrade unexpectedly under dry storage. With a molecular formula of Ca(ClO3)2, it dissolves well in water, delivering a clear solution rather than a milky mess or residue-laden soup. Some oxidizers pick fights with almost any organic they meet, but calcium chlorate’s reaction risk increases mainly with heat or strong reducing agents, not every passing scrap of matter, which makes it a little easier to handle with care and focus on proper mixtures.
Technical Specifications & Labeling
Producers of calcium chlorate communicate purity, crystal size, water content, and sometimes stabilizer content straight on the label, because even slight impurities or particle size differences can seriously alter its function. I remember reading technical bulletins that caution against open-air exposure, since it can collect moisture and clump, losing its clean flow in process machines—a detail production managers always double-check. Its safety and storage labels warn about the potential for explosive mixtures with organic powders, which keeps it on the radar for chemical safety reviews, even if it’s less volatile than some competing products.
Preparation Method
Calcium chlorate comes together most reliably through a reaction between calcium hydroxide and sodium chlorate. My time in the lab taught me that batch purity depends heavily on keeping contaminants out and not rushing filtration or crystallization steps. Traditional synthesis by reacting calcium hypochlorite solutions with chlorine gas is less common in commercial settings due to strict emission controls. Most industrial plants stick with solid-phase replacement, followed by purification, since this method drops production costs and makes recycling the byproducts practical.
Chemical Reactions & Modifications
On the reaction front, calcium chlorate stands out for its readiness to hand over oxygen. It accelerates combustion in herbicide formulations, and it can react with concentrated acids or strong reducers to make mixtures too unstable for most hands-on work. Chemists often introduce structural tweaks by blending calcium chlorate with buffering salts to reduce volatility in sensitive applications. Some research labs chase new modifications for soil sterilization and slow-release oxidizers, where tweaking cation ratios shifts not just stability, but also environmental persistence.
Synonyms & Product Names
Around the globe, calcium chlorate pops up under several aliases, reflecting both local language and chemical naming conventions. "Calcium salt of chloric acid" and "chloric acid, calcium salt" get used in regulatory filings, while trade names sometimes lean toward combinations like "Herbichlor" or older agricultural monikers. Anyone working across borders needs to double-check these synonyms, since regulations often track one version or another, causing confusion if labels don’t match paperwork.
Safety & Operational Standards
Working with calcium chlorate keeps safety inspectors on their toes. The compound won’t self-ignite, but it turns energetic fast when mixed with flammable materials like sawdust, sugar, or oil. I’ve always seen its storage isolated from all organic supplies and solvents, a lesson reinforced after hearing stories of fires started when a bit of sweepings or a loose bagged chemical landed in the wrong bin. Handling regulations usually require PPE—goggles, gloves, dust masks—and good ventilation. Emergency protocols call for dry cleaning of spills and banning open flames in the area. Any process using calcium chlorate in volume should rely on closed feeders and ventilation systems. Some companies go so far as to install chemical sensors in bulk rooms just to flag unsafe dust levels long before trouble even thinks of starting.
Application Area
Agriculture historically welcomed calcium chlorate as a herbicide, knocking back stubborn weeds like morning glory and Johnson grass. Municipalities used it for defoliation, vegetation control along railways, and in some cases for regulated root-killing in sewer management. Occasionally, water treatment labs toyed with its strong oxidizing power for purifying water or destroying organic contaminants, although rising environmental scrutiny began to limit these uses. Special effect artists and pyrotechnics technicians look to it for colored flame production and oxygen-releasing mixtures, provided they stick to rigorous safety plans. Research into alternative soil treatment chemicals keeps calcium chlorate interesting for agricultural science teams looking for ways around resistance to conventional herbicides.
Research & Development
Academic and industrial labs continue to dig for safer, more sustainable ways to deploy calcium chlorate’s oxidizing strength. Environmental chemists investigate reaction byproducts in soil and water, hoping to map out degradation timelines and long-term effects, especially as regulations squeeze out persistent chemicals. Modern research efforts aim to encapsulate or bind calcium chlorate within polymers, trying to create slow-release formulations that demand less frequent application and minimize groundwater leaching. More than once, chemists have targeted the development of stabilized blends, attempting to balance effectiveness against a shorter environmental half-life, a research frontier that demands patience, chemical intuition, and a deep bench of experimental failures.
Toxicity Research
Calcium chlorate poses health risks easy to underestimate, mostly because early field workers lacked reliable protective gear and got exposed through dust or spills. Swallowing the chemical delivers a powerful oxidizing jolt to internal organs, so researchers have repeatedly called for fresh studies on chronic exposure, especially as some water and soil remnants stick around longer than predicted. Animal studies log kidney and blood impacts at doses far below those considered immediately dangerous, which keeps toxicologists alert for new risks. Over the years, hazard awareness increased, both in Europe and North America, and product labels became much clearer and stricter. It’s no surprise researchers in public health circles keep pressing for continued monitoring of both occupational and environmental exposures, knowing that long-term data only now start filling gaps in human health risk.
Future Prospects
Future use of calcium chlorate won’t stick to the same paths. Stricter environmental review and the search for cleaner, more controllable forms of oxidizers steer both manufacturers and end users toward innovation. If research efforts bear fruit, slow-release or encapsulated formulations could open doors for more targeted agricultural treatments and even specialty uses in controlled remediation projects. Modern chemical engineering teams try to design applications with near-zero byproducts and minimal reactivity with native soils. Though regulations carve out some traditional roles, calcium chlorate still sits on the roster for those who value its strong and fairly predictable chemistry. The next generation of scientists and engineers who work on oxidation chemistry might view calcium chlorate as both a relic and a promising foundation for safer, smarter, and more sustainable chemical solutions.
What is calcium chlorate used for?
Looking at Real-World Uses and Challenges
People often forget how many chemicals serve as the backbone for building and growing things. Calcium chlorate rarely gets attention, but behind the scenes, it fuels some serious jobs. Not long ago, I spent afternoons in greenhouses and fertilizer sheds, seeing the mess and caution signs that come with basic agricultural salts. Calcium chlorate looks harmless as a powder, but it packs a punch that can change harvests—or cause headaches for anyone careless enough to ignore its properties.
Key Uses: Beyond the Textbook
Historically, calcium chlorate showed up in the weed control playbook. It takes out unwanted plants fast, strong enough to leave fields and railroad tracks clear even after heavy rain. Before synthetic herbicides took over, farmers reached for this stuff because it worked. The residue could stick around and prevent regrowth for months, meaning less labor over a growing season. This made a difference for small farms with thin profit margins. Now, most people lean toward newer chemicals, but a few still prefer the old way if local laws allow it.
Fireworks manufacturers also know calcium chlorate by name. Because the compound gives up oxygen quickly, it helps colors and effects stand out in pyrotechnic shows. Some smaller factories and hobbyists use it to get deeper reds or brighter bursts. School chemistry teachers and science clubs experiment with it to show basic oxidation and combustion reactions, which makes lessons more than dry theory. These uses connect to real curiosity, and sometimes, pure excitement.
In water treatment efforts, especially for emergency situations, calcium chlorate stands in as a disinfectant and oxidizer. It fights off some bacteria and breaks down organic waste in small-scale operation. I’ve seen a few remote field stations carry it as a backup for their usual chlorine, just in case shipping delays or storms keep them from restocking supplies. No one wants to gamble with water quality in those places.
Risks and Room for Improvement
Working around calcium chlorate means watching your step—literally. It catches fire with everyday things like sugar or paper. Accidents happened decades ago when factories overlooked dust and mixed it with sawdust from pallets. I met an old plant manager who still checks labels because of the scars this stuff left behind. Strict rules help, but cutting corners brings big costs.
Drinking water and runoff carry traces into soil and rivers if disposal skips proper treatment. Even low levels can push native plants out, and some water-borne creatures don’t handle exposure well. These problems matter because they stick with a community for years, long after the field or plant shuts down. Strong regulations and worker training lower these risks, but cost often slows change on small operations or in countries with limited oversight.
Looking Toward Safer Practices
Safer substitutes come with investment and retraining. Companies can switch to less reactive herbicides, or automate mixing tanks to control spills. Emergency responders use online databases now to identify chemical hazards quickly and organize cleanup. Local leaders hold workshops to teach storage and handling, using real stories—not just dry technical bullet points. I see promise where old-school experience meets new tools.
Calcium chlorate isn’t the most high-profile chemical out there, but the way people handle and regulate it tells a story about balancing progress with safety. From fields and fireworks to water plants, the lessons stick around for the next generation who’ll face similar decisions—how to get results without cutting corners or ignoring long-term harm.
Is calcium chlorate safe to handle?
The Substance Behind the Labels
Calcium chlorate doesn’t come up much outside a lab or a factory. It finds its main use in herbicides, explosives, and sometimes pyrotechnics. But its chemistry holds more than meets the eye. This isn’t table salt. Calcium chlorate mixes a strong oxidizing punch with a deceptively plain look. That means regular gloves and goggles suddenly matter a lot more.
Personal Experience on Shop Floors
Years ago, I spent some time in an agricultural warehouse, where folks unpacked and repacked drums of chemicals all day. Workers hand-wrote fresh labels on every tin, and dust would get everywhere. The safety training made us sweat in our gloves and double-check our goggles, but the stories stuck with me. Chlorates caught fire with almost no warning—one spark, or an oily rag in the wrong bin, and things could go very bad. Even a stray sweep of fertilizer dust mixed with a chlorate could set up an unpredictable risk.
What the Science Says
Chemists have documented that calcium chlorate reacts with organic material. Paper, wood, even just a dusty shirt could become hazardous if chlorate dust settles on it. The chemical won’t always signal danger with smell or smoke. Sometimes, it just waits for friction or a tiny drop of oil. Results from safety bodies back this up. The National Institutes of Health and European Chemical Agency both report accidents from careless handling. Burn injuries often turn out worse because the reaction keeps feeding itself oxygen from the chlorate, making it difficult to put out.
Health Hazards Beyond Fire
Calcium chlorate can do harm without flames. Inhalation irritates the nose and lungs. A handful of cases push people into days of coughing or lightheadedness. Even skin contact isn’t safe; burning, redness, and sometimes blisters show up after accidental spills. Tough gloves—preferably nitrile or neoprene—make a difference. Lab coats protect arms. Respirators or good ventilation keep tiny crystals out of the air.
Regulation and Common Sense
Countries don’t agree on regulations. The European Union restricts public use, while industrial supply houses in the United States require proof of professional intent. Online retailers have tightened purchasing rules after several incidents. This patchwork leaves room for mistakes, especially among hobbyists or small farm operators. People sometimes trust a bit of internet advice, skip the protection, or dump unused product with regular trash.
Practical Steps Forward
Safe handling always starts with practical habits. Keep containers sealed. Store calcium chlorate away from flammable or organic material, including paper, wood, and fertilizer. Only bring out what you will use. Dispose of unused product at a chemical waste facility. No shortcuts—never combine it with household cleaners, acids, or fuel. If an accident happens, the fastest response is isolation, then reaching emergency services.
The Human Cost and a Better Approach
Accidents rarely feel real until someone gets hurt. My time on the warehouse floor showed me how quickly a complacent day turns chaotic. Over the years, I’ve seen safety checklists grow fatter, but the basic rules remain the same. Be present, respect the label, and don’t get creative with chemicals. Calcium chlorate will not forgive mistakes.
How is calcium chlorate stored?
Safety at the Center of Chemical Storage
Calcium chlorate doesn’t get much mainstream attention, but anyone who has worked with oxidizing agents knows just how quickly things can go wrong. At its core, calcium chlorate brings a double punch: it reacts with plenty of other substances, and it kicks up oxygen, making fires burn hotter and faster. The core lesson here is that a little carelessness turns a warehouse into a news headline. What often gets overlooked is just how much the details of storage can prevent disaster.
Recognizing Real-World Hazards
Stories go around among lab workers and farmers—someone left a bag near some oil or stored it in a wet corner, and things went sideways fast. Calcium chlorate isn’t forgiving. Moisture breaks it down. Heat sparks problems. Mixing with organic material—like sawdust or even spilled fertilizer—sets up perfect conditions for combustion. The explosion risk ramps up if anyone thinks “out of sight, out of mind” is a storage strategy.
Practical Steps, Not Just Fine Print
Securing calcium chlorate calls for more than reading a safety manual. A solid, well-ventilated space with a roof that keeps out rain matters more than a shiny floor. I’ve seen facilities where rusty drums collect water, and the results aren’t pretty. You want sealed containers—think heavy-duty plastic or steel, nothing that corrodes easily. Seals and lids make a difference; airborne dust or water creeping in leads to caking and dangerous decomposition.
Temperature swings spell trouble. Hot rooms accelerate breakdown and raise the likelihood of combustion. If the storage space doubles up for other chemicals, the risks multiply in ways that don’t become obvious until it’s too late. Keep calcium chlorate far from acids, reducing agents, and anything flammable. It sounds simple, but in rushed workspaces, things get mixed up. Dedicated storage, labeled shelves, and regular checks catch mistakes before they snowball.
Fire safety gets personal for chemical workers, and it goes beyond rules on a poster. Automatic suppression systems with the right extinguishing agents—not just water—give crews a fighting chance during an emergency. Training saves more than property. Workers who know how to handle a spill, or spot a leaking container, prevent accidents others might never hear about.
Looking Beyond the Labels
Mistakes with calcium chlorate often start with the idea that “it’s just another bulk chemical.” Knowledge, not just liability forms, forms the barrier between routine and disaster. Inventory tracking sounds dull, but if a container disappears or some material goes missing, you want to notice fast. Real transparency—knowing where every bag and barrel sits—reduces the odds of careless stacking or accidental blending with other risky compounds.
Building Safer Workplaces
Manufacturers and distributors play their part. If containers break down during shipping, no set of warehouse rules can undo the harm later. Sourcing from reputable suppliers and insisting on robust packaging pays off in the long run. On-site inspections, audits, and straightforward communication about what’s coming in and out set everyone up for safer handling.
What matters most in storing calcium chlorate isn’t a checklist—it’s a culture of paying attention. Keeping this compound safe means treating every step as if the next mistake could be the one everybody remembers. The right locker, label, and fire plan strangely enough, make the difference between a routine Tuesday and sirens in the distance.
What are the hazards associated with calcium chlorate?
What People Should Know about Calcium Chlorate
Calcium chlorate stands out as a powerful oxidizer. For folks in agriculture or anyone dabbling in old-school chemistry sets, it looks like an ordinary salt—white, grainy, nothing too surprising. But don’t let that fool you. This compound reacts fiercely with organic material and flammable substances. Over the years, serious fires traced back to misplaced calcium chlorate. It takes just a bit of oil, dust, or paper to spark off trouble.
Fire and Explosion Risk
The biggest mark against calcium chlorate comes from its readiness to feed fires. Unlike common table salt, sprinkling this around oily rags or sawdust practically guarantees disaster. In home experiments gone wrong, tossing it into a trash can once led to an explosion that shattered windows. Local news covered warehouse fires caused by improper storage of oxidizers like this compound. The risk cannot be just brushed aside in small shops or storage units. Even a small spill can make a cleanup task a potential emergency, especially where organic material gathers—think flour mills, paper plants, anywhere dust hangs in the air.
Toxicity and Health Hazards
Calcium chlorate poses a health threat aside from its fire risk. Once mixed with acids, it produces toxic chlorine gas, a substance that gave soldiers lung damage in the trenches a century ago. Soil cleanup crews and chemical workers run into this hazard; one mistake, and the air becomes unsafe in seconds. Smaller exposures also matter. Dust from the powder irritates eyes and lungs. Chronic exposure eats away at red blood cells, making folks tired and weak. In my own experience with high school chemistry, a runny nose lasted a day after handling related oxidizers with sloppy technique—a small taste of what chronic exposure could look like on the job.
Impact on the Environment
Soil and water contamination bring long-lasting trouble. Calcium chlorate doesn’t just disappear after use. Some farming areas still show stunted plant growth years after chlorate-rich weed killers. Water runoff hurts fish populations, with small amounts changing the survival odds of native species. I’ve spoken with farmers who saw fields fall barren, all traced back to a single spill near an irrigation ditch. Local councils faced hefty cleanup bills as a single chemical mishap poisoned more than a stream or two.
Reducing the Risks
Safer habits go a long way. Storing calcium chlorate far from anything flammable or reactive keeps most trouble at bay. Workers need solid training, not just a stack of safety sheets. Simple steps like locking up oxidizers, using sealed containers, and regular, honest inspections of storage rooms cut risk. If a place works with acids or bases, no shortcuts can exist; even a small mistake with forgotten residue can generate chlorine gas. Emergency spill kits, eye wash stations, and strong ventilation matter more than they might seem.
Governments and companies need to keep an active eye on chlorate use. Fines help, but straightforward conversations among staff, supply managers, and regulators build trust and better safety overall. Switching to less dangerous alternatives—sometimes just a smarter solvent or weed killer—can spare entire communities from future health and environmental damage. In my own small garden, sticking to vinegar over harsh chemicals was all I needed to wipe out weeds and sleep easier about runoff.
What is the chemical formula for calcium chlorate?
Getting Straight to the Chemistry
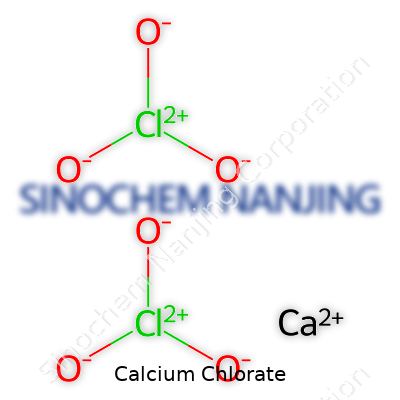
Calcium chlorate’s chemical formula is Ca(ClO3)2. That means every molecule contains one calcium atom and two chlorate ions. Looking at it, the symbol says a lot about how calcium, a pretty steady alkaline earth metal, pairs with the chlorate ion, known for its strong oxidizing properties. Someone taking high school chemistry might memorize the formula for their next quiz, but there’s a bigger story here that often slips by unnoticed.
Why This Compound Gets Attention
Calcium chlorate isn’t something everyone thinks about on a walk to the grocery store. In the labs and fields, though, it’s a different world. Farmers have counted on compounds like this in herbicides and defoliants. Chlorates hit weeds hard, clearing fields faster, giving cash crops a fighting chance. It’s also used for fireworks and explosives. Tossing big words aside, the simple formula Ca(ClO3)2 signals a substance with clout, and not just in theory.
Fact Check: Safety and the Environment
A lot of people might not give much thought to what happens after a chemical has done its job. Calcium chlorate’s ability to break down organic material raises tough questions for water and soil safety. Leaching and runoff can bring residues straight to rivers. Some countries have banned or restricted certain chlorates. The World Health Organization and the Environmental Protection Agency both watch for issues like contamination and toxicity. A study by the EPA dug deep into how chlorate ions disrupt thyroid function, not just in fish or livestock, but in humans, too.
Choosing Wisely: Balancing Production and Responsibility
No farmer wakes up hoping to damage drinking water or wipe out local aquatic life. Still, the temptation to use powerful compounds lingers, especially when yields and profits ride on the outcome. Trust gets built when clear communication happens between scientists, farmers, regulators, and the public. Maximum residue level rules, better testing, and real investments in alternative weed controls help slow the rush to use these potent salts. A balanced approach often leads to using less harmful options and updating practices on the ground.
Personal Take: More Than Just Science—It’s About Daily Life
I’ve seen scientists map contaminants from fertilizers in groundwater, connecting dots between what gets sprayed in fields and what pours from kitchen taps. My own garden saw improvement by using less aggressive treatments—fewer harsh chemicals, more patience with nature doing its work. Calcium chlorate’s formula stays the same, but the way society handles these compounds continues to shift. Most people want clean air, fresh water, and healthy crops, not just efficient chemistry on paper. Looking at the facts, digging past headlines, and thinking about consequences brings an answer every community can stand behind.
Seeking Solutions
Public education stands as the first step. Simple guides and community discussions about what goes into local soil and water tend to make everyone pay closer attention. Support for research into safer alternatives needs to move ahead. Regulation only works if everyone knows the reasons behind new rules and can track real results. The chemical formula Ca(ClO3)2 isn’t just a science class answer. It signals an everyday chemistry lesson: every decision made upstream changes the world downstream, one atom, one action at a time.
| Names | |
| Preferred IUPAC name | calcium chlorate |
| Other names |
Calcium chlorate(VII) Calcium salt of chloric acid |
| Pronunciation | /ˈkæl.si.əm ˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 10137-74-3 |
| Beilstein Reference | 1711395 |
| ChEBI | CHEBI:63046 |
| ChEMBL | CHEMBL502022 |
| ChemSpider | 14222 |
| DrugBank | DB15640 |
| ECHA InfoCard | 19b3fcba-1bdb-47ce-ab3e-d9e2fbc185c0 |
| EC Number | 231-847-6 |
| Gmelin Reference | 16172 |
| KEGG | C18798 |
| MeSH | D002119 |
| PubChem CID | 24598 |
| RTECS number | FJ9625000 |
| UNII | N6YO1M75X7 |
| UN number | UN1453 |
| Properties | |
| Chemical formula | Ca(ClO3)2 |
| Molar mass | 206.98 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 2.71 g/cm³ |
| Solubility in water | Very soluble |
| log P | -2.62 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.23 |
| Magnetic susceptibility (χ) | -40.0·10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -795.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -795.6 kJ/mol |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes serious eye irritation, may cause fire or explosion if heated. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS03, GHS05 |
| Signal word | Danger |
| Hazard statements | H272, H302, H318, H410 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-1-1-OX |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 707 mg/kg |
| LD50 (median dose) | LD50 (median dose): 926 mg/kg (oral, rat) |
| NIOSH | BKW |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Calcium Chlorate: Not established |
| REL (Recommended) | REL (Recommended): Not established |
| IDLH (Immediate danger) | IDLH: 20 mg/m³ |
| Related compounds | |
| Related compounds |
Calcium hypochlorite Calcium chloride Sodium chlorate Potassium chlorate |

