Calcium Bisulfite: A Deep Dive Into Its Past, Present, and Future
Historical Development
The journey of calcium bisulfite spans back to the early 19th century, when chemists searched for reliable solutions to tackle food preservation and industrial problems. It grew out of the push for new preservation agents and found its place during a time when winemakers looked for compounds to prevent spoilage and oxidation. Over time, papermaking and other industries started picking up on its potential. Sulfite-based agents like this grabbed attention because of their strong reducing power, which helped manage several chemical reactions in batch processes and fermentation. Calcium bisulfite didn’t remain just a bit player; it found a home in diverse sectors, and the journey from humble beginnings to broad industrial use shows how science adapts to the needs of the time.
Product Overview
Calcium bisulfite is a salt commonly made as a solution, typically recognized by its slightly yellowish, sometimes cloudy appearance and a sharp, sulfurous odor. It stands out as a preferred option due to its solubility and the way it interacts with other compounds in water-based processes. In commercial markets, manufacturers often package it in liquid form because it degrades on exposure to air. Most work involving this chemical revolves around dechlorination, bleaching, and as a reducing agent, but it still finds use in smaller operations where alternatives don’t fit. Products appear under names like “calcium hydrogen sulfite” as well, showing up in industrial catalogs, municipal water supply chains, and some food processing environments.
Physical & Chemical Properties
Looking at the physical nature of calcium bisulfite, it doesn’t stand out as flashy. It tends to present as a colorless to lightly yellow solution when fresh. The chemical makeup includes a calcium cation paired with two bisulfite anions, giving it the formula Ca(HSO3)2. This structure means that it dissolves freely in water, forming a mildly acidic solution. There’s a mild sulfur dioxide scent, a telltale sign of partial decomposition. Stability remains a real challenge; it works best when kept sealed or under inert conditions since oxygen exposure starts turning it to calcium sulfate and releasing sulfur gases. Handling in laboratories often means measuring pH levels (typically between 4-5) and verifying an active sulfite content using titration because product performance can slip quickly if left unchecked.
Technical Specifications & Labeling
Spec sheets for calcium bisulfite often quote grade purity as a percentage of available SO2, sometimes between 58% and 62% for liquid solutions. The presence of impurities, such as sulfate, iron, or insoluble matter, often comes into focus in technical settings. Industry requires clear labeling, especially for hazardous materials compliance. On the label, you’ll find names like “Calcium hydrogen sulfite,” hazard warnings for corrosivity, and recognized numbers for transport. In the U.S., there’s a UN number and hazard class marking; in Europe, specific risk phrases and safety advice accompany each shipment. Transport and shipping must follow these guidelines because of the reactivity and risk of chemical burns or inhalation hazard. Acid-resistant storage tanks and plastic piping avoid metal corrosion, and strict record-keeping forms a key part of operational routines.
Preparation Method
Making calcium bisulfite takes nothing more exotic than passing a stream of sulfur dioxide gas through water containing suspended calcium carbonate, like limestone or chalk, at low temperatures. The gas dissolves, reacting with both water and the calcium carbonate to form calcium bisulfite. This whole process gets tweaked depending on the intended purity level. Manufacturers often favor the wet scrubbing variant, where controlling pH prevents runaway reactions and excessive acidification. In some plants, excess sulfur dioxide improves yield, but it can cause calcium hydrogen sulfite to start breaking down. The whole operation demands well-sealed systems with good ventilation, since accidental exposure to sulfur dioxide isn’t something crews forget anytime soon. Finished solutions move directly to tankers or drums, and time-sensitive delivery avoids drop-offs in SO2 strength.
Chemical Reactions & Modifications
Calcium bisulfite doesn’t just sit in storage. It steps into reductions, where chemical engineers use it to quench chlorine or hypochlorite in water treatment plants. Here, it reduces these oxidizers safely and cleanly to chloride ions. Another routine task comes during pulping processes, where it helps break up bonds in wood chips, leading to softer, more manageable pulp. In some laboratories and specialty industries, chemists tinker with its reactivity, adjusting pH or temperature for fine control over downstream reactions. It reacts with strong acids to give off sulfur dioxide, sometimes intentionally released to control wild fermentation in winemaking. Mix it with oxidizers and the exothermic reaction can turn hazardous quickly, so training and tight protocols remain essential. Overdosing this compound creates its own set of secondary pollutants, driving ongoing efforts to monitor outflows or apply recovery steps downstream.
Synonyms & Product Names
Companies rarely use a single name for this material. You’ll run into synonyms and trade names—calcium hydrogen sulfite, bisulfite of lime, or even E227 (as a food preservative code). In some technical literature, variations with "solution" or "technical grade" appended can appear, marking the product for a specific end use. Customers in food industry circles may ask for "preservative E227" while industrial buyers stick to "calcium bisulfite solution" in procurement forms. Sometimes you’ll see calcium bisulphite (British spelling) or names translated into multiple languages. Such variation can create confusion if buyers or handlers aren’t aware, so documentation should avoid ambiguity, especially in multi-lingual regions or in transportation settings where clarity helps avoid accidents.
Safety & Operational Standards
Lab and plant teams learn early on that calcium bisulfite isn’t a casual chemical. Its fumes, chiefly sulfur dioxide, sting the eyes and burn the lungs with short exposures. Direct skin or eye contact causes lasting burns. Respiratory protection and rigorous glove and goggle rules stand in force every time a drum opens or a line breaks for cleaning. Local exhaust ventilation, emergency showers, and rapid eyewash stations become everyday fixtures. Safety Data Sheets detail the dangers—mix it with acids and you get a rush of choking gas; leave a spill unchecked and metal pipes corrode. In the U.S., OSHA sets permissible exposure limits for sulfur dioxide, and companies working with this compound must monitor air quality and worker health. Training never becomes a checklist item; ongoing drills and emergency planning make a difference between near-miss and catastrophe. Modern facilities tie chemical tracking into digital systems for quick access to stock and safety records, and regular audits help spot small issues before they become larger hazards.
Application Area
Few chemicals turn up in as wide an array of fields as calcium bisulfite. The pulp and paper industry uses it for wood pulping, softening fibrous material during the manufacture of paper and cardboard. Water utilities dose it into drinking water and effluent to neutralize excess chlorine after disinfection, which prevents toxic residue from reaching the tap or natural streams. Brewers and winemakers reach for it to halt wild fermentation and avoid spoilage, giving them more precise control over flavor and quality. There’s a smaller, but persistent, place in food preservation, especially for dried fruit and some pre-cut vegetables, since its antimicrobial nature stops discoloration and spoilage. Even smaller roles exist in the tanning of hides and as an additive in some specialty textiles, where its reduction capabilities come into play. Environmental cleanup companies use it to detoxify waste streams, neutralizing harmful oxidizers in industrial runoff before they cause harm downstream.
Research & Development
Research into calcium bisulfite rarely stands still. Scholars and engineers look into reducing the environmental burden by capturing released sulfur gases or by converting byproducts into safer, usable materials. In pulp mills, the move toward closed-loop systems includes projects that recycle spent bisulfite liquors, extracting valuable lignosulfonates for use in dispersants or concrete additives. University teams analyze the breakdown products, looking for efficient ways to trap or transform residual sulfur for agricultural or energy applications. Research groups examine its effects in new fields—such as using it in bioprocessing or as part of integrated nutrient removal systems. With tightening regulations on workplace safety and wastewater, innovation thrives around safer formulations and process controls. Practicality stays central: smarter sensors, automated dosing, and robust waste treatment systems keep labs and factories running within legal and ethical limits.
Toxicity Research
Toxicity always draws close scrutiny, especially when a chemical reaches food production or water supplies. Calcium bisulfite can irritate skin, eyes, and the respiratory tract. Chronic exposure to low doses of airborne sulfur dioxide, its principal fume, links to lung function decline. In food, doses remain low, but for sensitive individuals, even these traces may provoke asthma or allergic reactions. Researchers track metabolic paths, mapping the way sulfites affect the human body and the environment. Reports from regulatory agencies suggest food-safe limits, and continuous debate weighs its utility against potential risks. In schools and hospitals, risk assessments often push for alternatives where possible. Those trends shape policy, leading to labeling requirements and consumer warnings, especially as evidence mounts around sulfite sensitivity. Wildlife ecologists also track spills and accumulation in the wild, seeking a fuller picture of long-term ecological impacts.
Future Prospects
Looking ahead, calcium bisulfite faces big questions and fresh opportunities. Lower-emission manufacturing processes may unlock new roles in pollution control. Pulp and paper makers turn toward cleaner chemistry, demanding better lifecycle management from raw materials to plant effluent. In food and beverage, natural preservatives make inroads, but sulfites won’t disappear overnight—they’re still cost-effective and well understood. Technologies that better detoxify outflows and recover trace chemicals could expand its safe use. Policy movement across Europe and Asia points at tighter oversight and new documentation, nudging companies to rethink handling, storage, and worker protection. As automation and digital monitoring gain steam, risk management steps up its game too, using smarter alarms and real-time tracking to head off trouble. Collaborative efforts between labs, industry, and regulators push experimentation into new blends and uses, chasing efficiency and a reduced health burden, all while keeping an eye on the shifting landscape of global chemical safety.
What is Calcium Bisulfite used for?
What Does Calcium Bisulfite Actually Do?
Calcium bisulfite shows up in a handful of places that might surprise folks outside chemistry or water treatment. My first brush with it came while helping an older neighbor sort out a problem with sulfites in his well water. Digging into its usage, I saw just how underrated this compound can be—especially where clean water or safe food matter most.
Water Treatment: A Quiet Workhorse
Municipal water treatment plants rely on calcium bisulfite to scrub out chlorine from drinking water supplies. That dechlorination step doesn’t always grab headlines, but it removes taste, smell, and potential health risks left by chlorine. There are alternatives, sure—sodium bisulfite might be more recognized in some labs—but calcium bisulfite has a solid track record for being less hazardous and easy to store. According to the U.S. Environmental Protection Agency, dechlorination with bisulfite compounds remains vital before water reaches rivers or returns to nature. That prevents unnecessary harm to aquatic life.
Food Preservation and Winemaking
Anyone into winemaking or fruit canning probably knows about sulfites. Calcium bisulfite acts as a preservative, playing a big part in stopping spoilage and browning. In my own kitchen experiments, especially during apple harvests, drops of calcium bisulfite solution kept fruit salad looking fresh for hours. The U.S. Food and Drug Administration recognizes it as Generally Recognized As Safe (GRAS) for certain applications, though only at low levels. This approval follows decades of research and consumer use. But people with sulfite sensitivity still need clear labels and low-sulfite options—a challenge the food industry keeps working on.
Pulp and Paper Manufacturing
Factories that make paper and wood pulp lean on calcium bisulfite to help break down wood fibers. This process, called sulfite pulping, separates cellulose needed for clean, white paper from the sticky lignin that glues trees together. While the industry keeps looking for greener, less chemical-heavy options, this approach works at massive scale because it’s reliable. Problems pop up in waste management—sulfur dioxide odors and byproducts aren't welcome neighbors—which pushes companies to innovate with recycling and emissions controls.
What’s the Risk?
Anytime chemicals touch food or water, questions about health and the environment should follow. Most folks, myself included, want transparency. Calcium bisulfite can irritate airways if mishandled, and anyone sensitive to sulfites can react badly if it sneaks into food without clear labeling. Long-term environmental effects tie back to industrial waste, which needs more focus from regulators and companies. Stricter controls, improved technology, and clean-up strategies have already dropped the odds of problems, but vigilance pays off.
Moving Toward Safer, Smarter Use
It’s easy to overlook compounds like calcium bisulfite unless you see the value firsthand. In the right hands, it stops contamination, keeps food safe longer, and supports big industries. In the wrong setting, it can create headaches for health or the planet. Smart, careful handling—along with research into safer alternatives or better waste processing—ensures this unsung helper keeps doing its job without outstaying its welcome. For anyone worried about exposure, keeping an eye on labels and understanding how everyday products get made remains the best defense.
Is Calcium Bisulfite safe for consumption?
Calcium Bisulfite and Its Place in Modern Food Processing
Calcium bisulfite pops up in food processing as a preservative and bleaching agent. It prevents spoilage, sits among the “sulfites,” and works by slowing down the growth of bacteria and molds. Many people eat foods treated with sulfites every day without a thought. You’ll spot calcium bisulfite in dried fruit, wine, and some canned vegetables. Food scientists lean on it because it keeps color and freshness without cutting down on cost or shelf stability.
Addressing Safety: What the Experts and Science Tell Us
The U.S. Food and Drug Administration (FDA) lists calcium bisulfite as “generally recognized as safe” for most people. Regulatory agencies in Europe and other countries stack up behind similar views. They place limits on how much can go into food. When these limits are respected, the science says you’re unlikely to run into health problems. Tests look for reactions among large, diverse groups, making these guidelines feel solid to anyone overseeing food quality or working on a family meal.
Personal experience plays into the story. I’ve enjoyed dried apricots, wine, and other fruit carrying this preservative and never felt a thing. Friends and relatives in the food business echo the same. Research lines up with daily experience for most people—small doses of calcium bisulfite pass through the body without effects.
Spotlight on Sulfite Sensitivity: Who Needs to Watch Out?
Here’s where issues can sneak up. A small slice of the population reacts badly to sulfites, including calcium bisulfite. The Asthma and Allergy Foundation of America points out that roughly one in a hundred people with asthma can have real trouble after eating foods with sulfites. Symptoms can go from hives to trouble breathing. For the rest, mild headaches or flushing sometimes crop up after large intakes, but these are rare at the levels found in food.
Regulations step in to protect sensitive folks. Food packages must list sulfites if they hit or go above a certain level—10 parts per million in the United States. This rule lets people with allergies or asthma keep an eye out and make choices. In my own circle, one friend carries an inhaler everywhere and glances at labels just like someone with a nut allergy does at a bakery.
Building a Safer Food System
Industry leaders and those working in smaller food businesses keep up on best practices. They run tests, stick to approved levels, and keep records. Regular training sessions remind everyone on the line why exact measurement matters. These steps safeguard public health while still bringing consumers products that last and taste appealing.
For anyone concerned, reading labels goes a long way. Choosing fresh produce and asking questions at restaurants helps, too. If a reaction has ever popped up, your doctor or an allergist can give clarity and tips. Technology keeps moving things forward, with new preservatives being studied for safety and benefits. If better solutions show up, food regulations and habits will shift along with new evidence.
Calcium bisulfite stays on the food supply list for now because it works and passes safety tests for most. Its story points to how food science, regulations, and community feedback all work together—striving for safe, enjoyable meals with options for everyone.
What are the storage requirements for Calcium Bisulfite?
Getting Storage Right Keeps People and the Product Safe
Anyone handling calcium bisulfite knows the stakes can be high. This chemical’s main job falls in water treatment, paper bleaching, and some food processes. The right storage routine doesn’t just guard inventory. It protects employees, keeps the environment out of trouble, and steers clear of surprise expenses.
Watch Out for Moisture and Air
Calcium bisulfite likes to react with air and water. Sitting in open air, it tends to lose punch, breaking down into calcium sulfate and releasing sulfur dioxide gas. Dampness makes it even worse, so a dry space is critical. Year-round, low humidity keeps the chemical intact and stops leaks or unwanted fumes. I’ve seen how even a little water tracked in from rain can start a smelly mess that sticks around for days, and that’s never good for morale or reputation.
Containers Make or Break the System
Steel drums lined with plastic, HDPE containers, or similar corrosion-resistant bins keep this chemical happy. Metal without protective linings or thin plastics invite rust, leaks, or deforming. Lids must seal tightly. In the real world, workers are tired and in a rush, so labeling and color coding help keep everybody using the right bin. That means fewer mix-ups and less risk of incompatible materials mingling and causing trouble.
Avoiding Heat and Direct Sun
Heat speeds up chemical changes. Keeping calcium bisulfite in a cool spot preserves its quality and helps avoid dangerous gas release. Indoor storage out of the sun works far better than outdoor racks, which catch every temperature swing. Even budget-conscious outfits eventually learn that extra ventilation or air conditioning in storage pays off.
Safe Distance from Acids and Oxidizers
Nearby containers of acids, strong oxidizers, or even spilled cleaning agents spell disaster. If calcium bisulfite touches these, reactions can get out of hand: toxic fumes, fire, or fast breakdown. Setting a designated zone just for this chemical, clear from incompatible materials, lowers accident risk. It also speeds up emergency response — everyone already knows where to look, unlike in a cluttered mixed-chemical space.
Tracking Inventory and Shelf Life
Over time, even well-stored calcium bisulfite can lose its punch. Log inventory with batch numbers, delivery dates, and rotation. Older stock gets used up first. I’ve witnessed places lose entire shipments because the team lost track of dates and the product spoiled silently, hidden behind newer cases. Good records stop waste and save money.
Emergency Routines Build Trust
Spills and leaks will happen sooner or later. Emergency kits, training sessions, and easy-to-follow procedures help the crew keep calm and act fast. Clear signs, eyewash stations, and plenty of ventilation keep small mishaps from turning into catastrophes. Safety isn’t just ticking boxes — it means everyday habits that guard workers and the surrounding area.
Practical Improvements—Not Just Regulations
Following storage guidelines for calcium bisulfite isn’t just about meeting legal rules. It makes real sense at every step, protecting people, investment, and reputation. Regulators provide minimum standards, yet those who invest in better storage see fewer accidents, less waste, and stronger teamwork. Prevention pays off more than damage control.
How should Calcium Bisulfite be handled in case of a spill?
Facing a Spill: Practical Realities
Calcium bisulfite plays an important role across water treatment and chemical industries. It pops up in everyday products and processes, trusted for its ability to remove chlorine and act as a preservative. But that trust only goes so far. Spilled on the floor, suddenly it stops being helpful and starts causing problems.
Anyone who’s worked around chemical storage understands the urgency of proper handling. A slip-up with calcium bisulfite isn’t like spilling sugar or salt. This chemical reacts with water and air to form sulfur dioxide, which stings the nose and irritates lungs. Left unaddressed, it poses harm not just to workers, but also to the environment outside those factory doors.
Immediate Actions Speak Volumes
Trusting your training and responding with a clear mind makes a huge difference. Every responsible facility operator keeps spill kits visible and ready to grab. Time isn’t a luxury, so the first move should involve isolating the affected area. If I ever see a spill, instinct takes over—I get people out and close doors where possible. Industrial exhaust fans or open windows clear the air, but only if it’s safe. Proper gloves, goggles, and chemical aprons protect skin and eyes. It’s not overkill—those burns and blisters aren’t worth the risk.
Containment works best with absorbent materials. Granular clay or vermiculite soaks up the liquid, keeping it from spreading. Never sweep it around or rinse it down a drain. My own worst mistake involved hosing down spilled chemicals early in my career. That water carried waste right into a drain, and it took weeks to prove the impact didn’t reach the local river. These days, I steer clear of drains at all costs and bag collected waste for proper disposal.
Health and Environmental Stakes
Getting calcium bisulfite on your hands can cause redness and pain. Inhaling the gas it releases irritates your throat and chest, and some people with asthma notice it most. If someone’s exposed, remove them from the area and check for breathing trouble. Emergency showers and eye wash stations stay stocked for moments like these.
Beyond the walls of a factory, a careless spill puts nearby streams and soil at risk. Sulfur compounds leaching into groundwater can stick around far longer than most folks realize. That’s why training sessions and regular drills matter as much as the equipment itself. People feel more confident making quick, smart decisions during a real spill if they’ve already practiced the routine a few times.
Reducing Risks Long Before a Spill
Safer storage helps avoid this mess altogether. Tightly-sealed containers, clear warning labels, and dry, temperature-controlled spaces go a long way. Managers who walk the floor every week spot cracked lids and leaky tanks before they turn into problems. In the bigger picture, using smaller containers for regular handling shrinks the risk zone if something slips.
Practical Solutions for Safer Workplaces
Real safety comes from a blend of habits and rules. Frequent training, easy access to protective gear, and strong communication mean everyone moves together during emergencies. Local authorities and emergency services expect detailed plans, not just checklists filed away in a drawer. Companies that value trust and community share reports and lessons learned in staff meetings and industry workshops alike.
Mistakes can cost more than downtime on the shop floor. They affect community trust and hurt the environment. Sensible, quick responses backed by hands-on training keep accidents from defining a company’s reputation. In my experience, lessons from cleaning one sticky spill stick around long after the mess is gone.
What are the potential health hazards of Calcium Bisulfite exposure?
What You’re Really Dealing With
Calcium bisulfite isn’t a name you hear much outside of factories or drinking water plants. It ends up in a surprising number of places, especially where people treat water or use preservatives. It helps stop things from rotting or reacting in ways we don’t want. At work, I’ve clocked time in labs and seen this chemical used to combat chlorine or keep food from turning brown.
Most warnings about this substance come from what it does in the air and on your skin. The fumes smell sharp, like sulfur or burning matches. Inhaling the dust or mist can leave you coughing, with a tight feeling in your chest. Even if you wear gloves, long exposure dries out skin and might trigger rashes. Workers splashing it around risk burned eyes that sting all day. I remember seeing folks who didn’t wear proper gear deal with red skin and watery eyes for the rest of their shift. It’s hardly just a mild nuisance.
What Science Tells Us About the Hazards
The Institute for Occupational Safety and Health lays out the effects in plain language. High enough concentration in the air mixes up the respiratory tract, sparking symptoms like shortness of breath, headaches and wheezing. Some people get asthma attacks from much smaller doses — especially those with a history of allergies or breathing problems. The World Health Organization recognizes sulfites, including calcium bisulfite, as potential asthma triggers. Kids and anyone with sensitive lungs have even more trouble.
The chemical can also get through to people without them knowing, since it’s common in water treatment. The Environmental Protection Agency sets tight limits for sulfite presence in public water because regular sips over time could upset stomachs or make sensitive folks downright sick. Repeated, unwitting exposure means sore throats, nausea or diarrhea. That kind of thing won’t knock out a healthy person in a day, but the risk sticks around for certain groups.
Why Some Groups Face Bigger Risks
Sulfite-sensitive individuals experience stronger reactions, from simple breathing discomfort to full-blown anaphylaxis. Older adults, children and people with asthma make up most reports of bad outcomes. The CDC notes that even small quantities slip past labeling in restaurant food or processed drinks. People wind up in hospitals after simply trusting public water or eating factory-prepped meals. I’ve seen family friends with asthma suffer attacks after eating out, connecting the dots only after a doctor traced the reaction back to hidden preservatives.
What Actually Helps Prevent Exposure
Oversight doesn’t only fall on big industry. Workers in plants need real training and consistent protective equipment — gloves, goggles, and ventilation systems that actually work. Warn folks clearly about the risks with proper labeling on water, processed foods, and cleaning products. The FDA mandates such labels, but they often get overlooked or written in codes most shoppers ignore. My years in food safety showed that even managers need regular reminders that “sulfite free” is not the same as “safe for everyone.”
Health departments can encourage restaurants to post allergen warnings, not just hide ingredient lists in the back office. If you work around this stuff, wash your hands before eating and change out any clothes you got dirty at work. Educating the people processing foods about sulfite risks goes much farther than hoping old policies will carry everyone through.
Living and working close to chemicals like calcium bisulfite forces everyone — employer, employee, and consumer — to respect how easily invisible exposure turns into a health crisis. No one stays safer just by guessing; real, simple precautions backed by research matter the most.
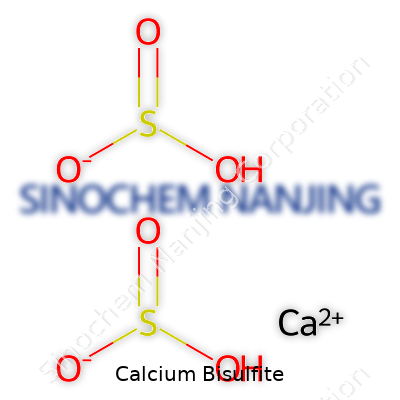
| Names | |
| Preferred IUPAC name | calcium hydrogen sulfite |
| Other names |
Calcium hydrogen sulfite Calcium bisulphite Calcium hydrogen sulphite |
| Pronunciation | /ˈkæl.si.əm baɪˈsʌl.faɪt/ |
| Identifiers | |
| CAS Number | 13780-03-5 |
| Beilstein Reference | 1204229 |
| ChEBI | CHEBI:49699 |
| ChEMBL | CHEMBL1201732 |
| ChemSpider | 27341028 |
| DrugBank | DB11398 |
| ECHA InfoCard | 03bf4fa7-8a33-4a2c-89b7-45892473e7e6 |
| EC Number | 233-265-8 |
| Gmelin Reference | 12262 |
| KEGG | C01730 |
| MeSH | D002118 |
| PubChem CID | 24456 |
| RTECS number | EW5250000 |
| UNII | X73F93E9D7 |
| UN number | UN2693 |
| Properties | |
| Chemical formula | Ca(HSO3)2 |
| Molar mass | 120.17 g/mol |
| Appearance | White or yellowish crystalline solid |
| Odor | Sulfur dioxide odor |
| Density | 1.3 g/cm³ |
| Solubility in water | 72g/100mL (20 °C) |
| log P | -3.49 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 5.8 |
| Basicity (pKb) | 7.11 |
| Magnetic susceptibility (χ) | -5.3×10⁻⁶ |
| Refractive index (nD) | 1.332 |
| Viscosity | 10-30 cP |
| Dipole moment | 3.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 161.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1029.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, releases toxic sulfur dioxide gas. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. H314: Causes severe skin burns and eye damage. H400: Very toxic to aquatic life. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P301+P330+P331, P304+P340, P305+P351+P338, P310, P330, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2 0 1 |
| Explosive limits | Non explosive |
| Lethal dose or concentration | Lethal Dose/Concentration: **LD50 (oral, rat): 2,000 mg/kg** |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 2,000 mg/kg |
| NIOSH | WFJ99430W |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 500 mg/m³ |
| IDLH (Immediate danger) | 250 mg/m3 |
| Related compounds | |
| Related compounds |
Calcium sulfite Calcium sulfate Sodium bisulfite Potassium bisulfite |

