Calcium Arsenite: A Grounded Look at a Toxic Compound
Tracing the Roots: Historical Development
Calcium arsenite arrived on the scientific scene in the late 1800s, following a wave of research into metallic arsenic compounds. Back then, chemists dug deep into mineralogy and agricultural chemistry, searching for effective pesticides and pigments to meet growing market demands. Most early uses of calcium arsenite can be traced to borax and pigment makers, who used arsenite mixtures for wood preservation and manufacturing green paints, particularly the infamous “Scheele’s Green.” Farming communities, desperate to guard crops from blight, also adopted this compound into orchard sprays, despite recognizing its considerable risk. Historical laboratory notebooks and patent records tell a story of trial, error, and sometimes tragic consequence, with workers and consumers alike exposed to its dangers before regulations caught up.
Product Overview
Calcium arsenite doesn’t show up on store shelves or even in bulk chemical catalogues the way more benign salts do, but it was once a staple for manufacturers of pigments, insecticides, and fungicides. The product typically presents as a white to light greenish powder, sometimes appearing crystalline depending on its manufacturing route. Its main appeal in the past stemmed from a potent toxicity—useful in killing insects and fungi—though this same potency turned out to be its downfall, as regulators became alarmed about human and environmental hazards.
Physical & Chemical Properties
Forms of calcium arsenite often exhibit a pale, powdery texture, shifting toward green shades if impurities from arsenic trioxide or other metallic ions have crept in. Chemists cite its density near 3.7 g/cm³, with poor solubility in cold water but partial dissolution in hot water and acids. As for chemical behavior, calcium arsenite can react vigorously with strong acids, releasing highly toxic hydrogen arsenide gas, making it especially hazardous in mishandling scenarios. The compound’s temperature stability keeps it solid under normal conditions, though strong heating risks decomposition into arsenic trioxide and lime. This reactivity brings both promise and peril, depending on application and control.
Technical Specifications & Labeling
Any legitimate labeling for calcium arsenite needs to highlight concise safety warnings, including GHS symbols for toxicity, environmental hazard, and acute oral toxicity. Technical data sheets usually specify chemical purity above 98% for research-grade material, listing residual levels of arsenic trioxide and calcium oxide. Particle size analysis occasionally features in specifications if the batch is intended for industrial blending. The United Nations hazard class for this substance is 6.1—poisonous material—and strict transport guidelines follow, such as covering packaging requirements, secure drums, and removal from public transit. Barcodes and QR codes point back to safety data sheets online, making sure handlers know what they’re dealing with at every step.
Preparation Method
Manufacturing calcium arsenite takes a two-step approach. First, industrial arsenic trioxide reacts with calcium hydroxide slurry under controlled temperature and mixing, usually in a glass-lined reactor to avoid corrosion and leaching. The reaction gives rise to precipitated calcium arsenite, which is then filtered, washed, and dried to remove excess soluble arsenic and lime. Any residue left in the reaction mix receives neutralization treatment to destroy lingering arsenic before disposal, reflecting tougher environmental rules that came into play through the 1900s. Early operators lacked modern environmental controls, and runoff polluted local waters—a grave lesson that shapes today’s processes.
Chemical Reactions & Modifications
Chemists have identified a handful of reactions that give calcium arsenite some versatility, but also pose risk in the lab or factory. Mixed with hydrochloric or sulfuric acid, the compound quickly decomposes, forming soluble arsenic acids and liberating heat and fumes. Combining this arsenite with oxidizers doesn’t end well either, as explosive gases can evolve from contaminated batches. Over the years, teams tried doping calcium arsenite with metallic ions or polymer carriers to reduce its leaching from paints and wood treatments. These efforts sometimes slowed the migration of arsenic into the air or soil but rarely solved the problem entirely.
Synonyms & Product Names
Trade names rarely featured calcium arsenite outright in consumer products, but technical bulletins and scientific literature often list synonyms such as “calcium meta-arsenite,” “calcium arsinate,” and the plain language “white arsenic of lime.” Historic pesticide manuals sometimes used proprietary codes or product numbers, making it tough to track supply chains or recall batches tied to poisonings. Ingredient labels on legacy paints refer to “arsenite colorant” or simply “arsenic pigment,” masking the true identity from the public. This cloak of synonyms kept regulatory eyes away for decades until stricter chemical disclosure laws arrived.
Safety & Operational Standards
No one treats calcium arsenite casually. Exposure routes—mainly inhalation, ingestion, and dermal contact—lead to severe health crises, including organ failure and certain cancers. Factory setups handling this compound require sealed ventilation, chemical-resistant suits, and strict handwashing procedures, enforced by workplace safety officers familiar with hazardous materials. Storage takes place in tightly-lidded containers, labeled with skull-and-crossbones, away from water sources and incompatible chemicals. Emergency showers, eye-wash stations, and detailed decontamination plans stand by for spills or accidental contact. Modern guidelines from agencies like OSHA and NIOSH set airborne arsenic exposure limits at microscopic levels, pushing facilities to invest in continuous air monitoring and rigorous medical surveillance for their staff.
Application Area
Old literature brims with references to calcium arsenite in orchard dusts and sprays for apple, grape, and citrus crops, aiming to wipe out leaf curl and moth infestations. Paint companies and wood treatment firms relied on it for its colorfast properties and resistance to fungus, filling demand for durable exterior coatings and preserved railway ties. It also found a home among early glassmakers and textile dyers who wanted intense green tints that stayed bright under sunlight. As safer replacements like copper-based fungicides or synthetic biocides took hold, industries abandoned calcium arsenite, pushed by both scientific warnings and consumer activism.
Research & Development
Interest in calcium arsenite faded fast once chronic toxicity hit the headlines, but pockets of research persist. Analytical chemists use the compound as a reference standard for arsenic speciation studies in soil remediation and waste management. Environmental scientists monitor sediments near abandoned industrial sites, charting arsenic residue and studying potential means of immobilization or extraction. Some universities still analyze calcium arsenite’s legacy in heritage buildings and old orchards, piecing together the compound’s environmental journey using advanced mass spectrometry and historical mapping. Lab teams keep refining ways to detect and safely neutralize accidental releases, learning from past disasters to avoid future harm.
Toxicity Research
Few chemical substances have earned the kind of infamy that arsenic compounds—especially calcium arsenite—hold in the public and medical record. Toxicology studies uncover links between even low-level exposure and elevated risk for lung, skin, and bladder cancers, along with acute poisoning symptoms like vomiting, anemia, and death. Animal studies echo these warnings, with repeated low doses leading to neurological and developmental harm. Environmental health surveys track arsenic entering waterways, leaching from contaminated soils, and moving up the food chain into fish, birds, and, ultimately, people. Policy-makers cite this large body of evidence to justify the strictest bans, and today it stands as a cautionary tale for anyone developing next-generation biocides.
Future Prospects
The future of calcium arsenite sits almost entirely with historians and environmental engineers, not manufacturers or farmers. Regulations worldwide limit use to highly controlled research, with disposal aimed at complete destruction, not recycling or reuse. Still, the legacy remains—contaminated farm fields, aging green-painted homes, and brownfields that keep chemists and toxicologists busy for decades. Society faces the challenge of cleaning up these past mistakes, developing affordable, scalable methods for arsenic removal from soils and water supplies. Some researchers eye bioremediation with fungi or plants, others refine chemical stabilization agents that lock down arsenic migrations. At every step, the enduring lesson draws from history: smart innovation can’t overlook safety, even in pursuit of the next agricultural or industrial breakthrough.
What is Calcium Arsenite used for?
Why Farmers Once Turned to Calcium Arsenite
Calcium arsenite stands out in the history of farming for its place among older, hard-hitting insecticides and fungicides. For folks working citrus groves or vineyards in the first half of the 20th century, pest outbreaks could wipe out both yield and income in a single season. Pests like moths, beetles, and scale insects moved in quickly, and choices for fighting them used to be slim. Calcium arsenite provided a strong chemical solution that, at the time, knocked down infestations faster than hand-picking or natural approaches.
The logic was simple: limit damage, save the harvest, and keep fruit or grape quality up. With few regulations on pesticides, the use of arsenic-based products was mostly driven by immediate survival on the land. While some growers might have noticed damage to soil or nearby plants, priorities put pest control above all else. Stories from old family-run farms remind me that people often had to choose between a risky chemical and a ruined crop. That’s real, everyday experience shaping farming decisions.
Looking Closer at the Risks
Science gradually made the dangers of calcium arsenite clearer. Arsenic compounds, even in small amounts, can linger in soil and water. During long, hot summers, the drift from sprayed fields floated into the air, settling onto nearby plants, wells, or even into the lungs of folks tending the fields. Chronic exposure has well-documented links to skin problems, lung issues, and certain cancers. The World Health Organization warns against arsenic in drinking water, yet for decades, these risks weren’t widely known or discussed.
Watching my own town change as regulations increased brings home the point—changing what’s considered “safe” always takes time, and sometimes feels slow. Yet learning from neighbors who dealt with lingering chemical residues shaped new public health rules. Real data from infected wells, sick livestock, and declining rural health pushed countries around the world to phase out products like calcium arsenite. The shift wasn’t overnight, and some older folks still talk about the struggles to find alternative products that work as well—without the cost to health.
Modern Alternatives and Farming Today
Farmers now deal with more oversight, yet they have better options. Biopesticides, natural predators, and improved crop management mean pest control no longer needs to carry the same risks. Knowledge has grown, and with access to research, anyone running a farm today can look up the basics, check recent findings, and get recommendations for non-arsenic treatments. Soil monitoring keeps contamination in check, so families and workers stand a better chance at a safe working environment.
For communities facing lingering arsenic from decades past, solutions come in the form of cleanup programs, soil amendments, and monitoring water. While money and resources for rural recovery don’t always stretch far, community organizing and persistent advocacy help move things forward. Sharing honest stories about health and farmland recovery highlights the real costs of using short-term solutions with long-term consequences.
My own experience pulling weeds, picking fruit, and talking to growers has convinced me that farming always involves tradeoffs. Understanding what calcium arsenite did—both good and bad—reminds us to keep asking questions and to value both yield and safety in equal measure.
Is Calcium Arsenite hazardous to health?
Common Uses and Where Exposure Happens
Walk into any old orchard, and there’s a fair chance the ground has felt the touch of calcium arsenite at some point. This compound once promised to control pests better than mother nature’s birds, and so it landed in sprayers across farms. Back in the day, you could find it in pesticides for fruit trees, grapevines, and even in some wood preservatives. People used it often with gloves on and heads down, not always knowing the real story behind the dust.
Digging Into the Hazards: What Science Says
Researchers tell us that calcium arsenite contains arsenic, a heavy hitter in the world of toxic elements. Arsenic isn’t just another dirt mineral; it’s known for causing serious trouble in the human body. People who breathe in or swallow dust with arsenic over time can develop skin problems, nerve issues, or even certain types of cancer.
Being around arsenic compounds means risk sticks around too. Inhaling or getting calcium arsenite on your skin can lead to poisoning. The symptoms don’t always rush in—sometimes they sneak up slowly, showing up as stomach pain, thickening skin, breathing problems, or tiredness that just won’t quit. In a report by the U.S. Environmental Protection Agency, long-term arsenic exposure is strongly linked to skin, lung, and bladder cancer, as well as developmental issues in children.
There’s also the environment to think about. Calcium arsenite can wash into streams, sink into soils, or even ride the wind into new places. Wildlife feels these effects, building up arsenic in food chains, which can circle back to people’s dinner plates.
Personal Experiences and Lessons Learned
Growing up near farm fields, I watched the shift from dusting crops with white powders to using other pest control methods. Some older neighbors talked about headaches, mouth sores, and peeling skin in their younger years, long before doctors linked those troubles to calcium arsenite or other arsenic-based sprays. I knew folks who covered up with rags and hats, but that didn’t always keep dust out.
Stories like theirs helped push laws that phased out most arsenic-based pesticides. Within my state, leftover barrels of calcium arsenite sometimes turn up in barn corners, treated like ticking time bombs until hazardous waste teams haul them away. That’s not just talk—one leak can lead to contaminated wells, which sparks big worries for rural families relying on groundwater.
What Can Actually Help?
Solutions don’t just mean swapping one chemical for another. Stronger regulations have put pressure on companies and farmers to look for less risky pest control. Biological methods, like releasing helpful insects or using natural plant-based sprays, gained ground. Education helps too; growers now check safety labels, wear real respirators (not just old bandanas), and keep water testing part of their routine.
Old stockpiles, though, remain a problem. Safe disposal programs must keep rolling. Funding for hazardous waste pickup, along with public info hotlines, makes a difference in preventing accidents.
What people have learned about calcium arsenite stands as a signal: ask where chemicals go, who gets exposed, and what happens years down the road. Trust isn’t just about rules—it’s about listening to those who’ve worked the land and faced the fallout first-hand.
What is the chemical formula of Calcium Arsenite?
Understanding Calcium Arsenite
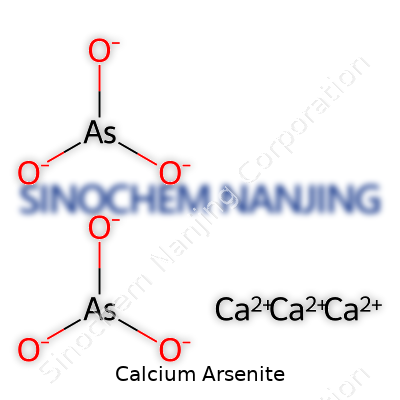
Calcium arsenite falls under that curious list of compounds that aren’t talked about much, probably because most folks think “arsenic” and tap out. The chemical formula for calcium arsenite comes down to its makeup, which isn’t as tricky as it sounds at first glance. It goes by CaAsO2—with one calcium atom, one arsenic atom, and two oxygen atoms. Sometimes, you’ll spot another version written as Ca3(AsO3)2 depending on the context and the ratio involved in certain applications.
Why It Matters
I’ve noticed that the conversation around chemicals often glances over the real-world impact these formulas can have. Calcium arsenite’s story stretches well beyond lab books. This compound used to sneak its way into pesticides—especially in orchards—thanks to its strong fungicidal ability. Farmers wanted a tool to keep their harvest safe and productive. It was cheaper than other options, so it made sense from a budget point of view.
Problems only started to show up much later. Long-term exposure to arsenic compounds, even those sporting a “less toxic” profile like calcium arsenite, raises big red flags. I’ve read studies from the EPA and WHO that connect arsenic-based pesticides with everything from cancer to cardiovascular and neurological damage. Nobody wants those kinds of risks in the produce aisle or groundwater.
Personal Reflections and Broader Lessons
Growing up near farmland, I watched neighbors blend chemicals for their crops. Actions seemed routine until more reports of health issues began surfacing. Those stories changed attitudes fast. Families who never thought much about chemistry began asking tough questions about what went into food and water. It’s a reminder that science isn’t just an abstract subject—it reaches right into dinner tables and doctor’s offices.
The landscape has definitely shifted. Governments banned a bunch of arsenic-based pesticides in large parts of the world. Synthetic alternatives aren’t perfect, but they moved the needle forward on safety. The calcium arsenite case gives a lesson in why understanding chemical formulas isn’t just about memorizing symbols. Every compound sitting on a shelf has a ripple effect beyond the classroom and the laboratory.
Balancing Science and Public Health
Plenty of folks still deal with contaminated soils years after these chemicals were phased out. It turns into a cleanup problem that costs time and money. I’ve seen community groups advocate stronger soil testing and cleanup efforts, pointing out that short-term decisions can create long-term headaches if safety takes a back seat. The EPA, for instance, has posted clear guidelines for managing arsenic pollution. Progress builds on these lessons, making future risks easier to spot and reduce.
Education drives the smartest outcomes. As more people understand the nuts and bolts of compounds like calcium arsenite, they’re better positioned to advocate for both innovation and caution. It comes down to not repeating avoidable mistakes. Chemistry isn’t just formulas; it’s choices, actions, and the lives affected on the other end.
How should Calcium Arsenite be stored safely?
Understanding the Risks
Calcium arsenite comes with a heavy reputation. This compound brings together calcium’s common presence with arsenic’s infamous toxicity. Many people remember growing up learning about dangers hidden in old paints or pesticides. Arsenic sat at the center of those warnings, and for good reason. Inhaling or ingesting small amounts can cause lasting harm or even prove fatal. Having handled chemical substances in school labs and on factory tours, the sight of anything labeled arsenic still pulls up caution flags in my mind.
Workplaces and labs that rely on calcium arsenite must focus on serious safety steps. A lapse doesn’t just threaten trained staff; accidents can reach families and neighborhoods. The toxic dust from this pale yellow solid drifts invisibly. A single poorly sealed jar or a knocked-over bag could release invisible problems into an entire workspace. The risk can’t be shrugged off as remote or unlikely. It just requires diligence every step of the way.
Choosing the Right Storage Environment
Dry, cool, isolated—these aren’t just buzzwords when it comes to this substance. Humidity will break down the solid and release arsenic compounds into the air. Sunlight and heat speed up chemical changes nobody wants in a storeroom. For extra safety, dedicated chemical lockers fitted with tight-sealing lids provide a hard line of defense. These storage cabinets should lock, supported with bold warning labels, and kept off the beaten path where only authorized staff tread.
Glass or high-quality plastic containers work better than the cardboard or thin bags sometimes found in older supplies. Seals must always fit snugly. For added protection, try double-containment: putting sealed jars inside another sealed bucket or leakproof box. If one barrier fails, the next holds the line. Having seen cases where improper containers corroded away, letting powder escape into forgotten corners, this double layer makes a lot of sense.
Ventilation and Hygiene: Breathing Room Isn’t Optional
Never store calcium arsenite near snacks, drinking water, or personal items. Even a little cross-contamination crosses a dangerous line. Separate chemical-only zones in storage rooms help avoid mix-ups, and a dedicated airflow system sweeps any escaping dust or fumes away to filters, not into staff lungs.
Regular cleaning with specialized equipment, not brooms or vacuums, keeps dust from spreading. Wet wiping and HEPA-rated vacuums trap even the tiniest particles. If a spill does happen, chemical spill kits made for arsenic compounds should never be out of reach, and staff should stay up to date on how to use them—training isn’t something you do once and forget for years. In my own experience, drills and hands-on sessions make genuine confidence grow, way more than reading manuals ever could.
Accountability, Not Just Rules
No matter the setting, confident handling and secure storage only build up from a culture of accountability. Inventory logs and sign-in sheets force attention to detail. Double-checking labels and practicing rigorous handwashing routines become part of the routine, not special occasions.
Government agencies like OSHA and the EPA keep tight watch on chemicals like this, and with good reason. Following their guidelines stops harm before it can start, not just to workers but to anyone who spends time around the facility. From gloves to goggles to full-face respirators depending on the job at hand, personal protective gear steps in to close the safety circle.
Failing to Take Safety Seriously Brings Real Consequences
Many of us remember news reports about unsafe chemical handling leading to community evacuations, illnesses, or long legal battles. Taking shortcuts or trusting fate with something as serious as calcium arsenite does not end well. Far better to set tough rules, follow them without exception, and treat this compound as the threat to health and safety that it plainly is. Safe storage isn’t just about the law—it protects real people, real communities, and the future of workplaces everywhere.
What are the environmental impacts of Calcium Arsenite?
Understanding Calcium Arsenite
Calcium arsenite, a compound once used in pesticides and fungicides, brings real concerns wherever it turns up. Talk to anyone familiar with old agricultural sites and they’ll probably recall stories of long-abandoned pesticide drums rusting behind barns. Arsenic-based compounds, including this one, stick around for a long time in soil and water. They carry trouble beyond the fields they were meant to protect.
Soil and Water: Where the Damage Hits Hardest
Once calcium arsenite touches soil, it doesn’t go away quickly. Arsenic binds to soil particles, which means these chemicals settle into the ground, sticking around for decades. Rain can eventually move arsenic deeper, sometimes straight into water tables. Wells in farming communities from California to Bangladesh have shown dangerously high arsenic levels, threatening crops, livestock, and people. Long-term exposure brings higher rates of skin, lung, and bladder cancers, along with nerve damage and developmental problems in children.
Back when these products were common, few folks checked groundwater. Now, more modern testing paints a clearer picture. High arsenic in soil makes food crops hazardous too — rice, a staple for billions, takes up arsenic easily. The poison ends up in kitchens and schools. If you’ve ever wondered how food grown far from big factories can turn risky, contaminated soil and water point to one answer.
Damage to Ecosystems and Wildlife
Streams and lakes near application sites pick up runoff after rainy seasons. Fish, amphibians, and insects all take up arsenic; it travels up the food chain, building in birds and mammals. Deer and small mammals grazing local vegetation get sick or die, even far away from the original source. Insects, which play a massive role as pollinators and as food for other creatures, also take a hit. Loss of these vital species breaks natural cycles, making it tough for any ecosystem to bounce back.
Human Health: Not Just a Distant Issue
No one likes thinking about arsenic in their water or food, but millions deal with it daily. Studies trace high rates of cancer, diabetes, and cardiovascular problems to arsenic exposure. Just washing produce won’t clear the risk, since arsenic gets inside the plants themselves. Governments set "safe" levels of arsenic in drinking water at 10 parts per billion, though many wells overshoot that number. If you visit rural areas with a legacy of old pesticide use, water filters and regular testing become a way of life.
What Can Be Done?
Solving arsenic contamination starts with stopping further use of arsenic-based compounds. Over the years, many countries have banned or restricted these chemicals, but plenty still remain in old sheds and forgotten corners. Safe disposal programs make a big difference, helping farmers and landowners get rid of the stuff without just burying or dumping it.
Next up comes remediation. Technologies like phytoremediation, which uses special plants to "suck out" arsenic, show promise. Soil washing and chemical treatments clean up the worst spots, though costs hold back widespread use. Portable water filters and community wells provide cleaner water, protecting families while bigger cleanups get underway. Real progress always means collaboration between scientists, communities, and governments.
A Personal Note
Growing up in a farming community, I saw the effects of soil and water pollution firsthand. Protecting clean air, land, and water matters not only for the land you stand on, but also for future generations trying to grow food, raise families, and stay healthy. The legacy of calcium arsenite shows short-term fixes sometimes create bigger problems down the road. Thoughtful action and persistent cleanup offer the best path forward.
| Names | |
| Preferred IUPAC name | calcium arsenite |
| Other names |
Calcium(II) arsenite Calcium(2+) arsenite Calcium metaarsenite |
| Pronunciation | /ˈkæl.si.əm ˈɑː.sɪˌnaɪt/ |
| Identifiers | |
| CAS Number | 7784-41-0 |
| Beilstein Reference | 136144 |
| ChEBI | CHEBI:86337 |
| ChEMBL | CHEMBL34297 |
| ChemSpider | 23542100 |
| DrugBank | DB11379 |
| ECHA InfoCard | 100.013.862 |
| EC Number | 1303-20-8 |
| Gmelin Reference | 126712 |
| KEGG | C18722 |
| MeSH | D002119 |
| PubChem CID | 159410 |
| RTECS number | CAS03288 |
| UNII | 7VQ0I63V8G |
| UN number | UN1546 |
| Properties | |
| Chemical formula | CaHAsO3 |
| Molar mass | 262.07 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 3.52 g/cm³ |
| Solubility in water | Insoluble |
| log P | -0.098 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 2.56 |
| Magnetic susceptibility (χ) | −44.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.58 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 86.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1337 kJ/mol |
| Pharmacology | |
| ATC code | V03AW04 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; may cause cancer; may cause heritable genetic damage; environmental hazard. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P314, P330, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-Acute Tox |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD₅₀ oral rat 40 mg/kg |
| LD50 (median dose) | LD50: 20 mg/kg (oral, rat) |
| NIOSH | GG8925000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Calcium Arsenite: 0.01 mg/m³ (as As) |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Calcium arsenate Sodium arsenite Lead arsenate Arsenic trioxide Calcium phosphate |

