Calcium Arsenate: A Close Look at an Industrial Chemical
Historical Development
Taking a look back, calcium arsenate entered the agricultural world in the early 20th century. Farmers wanted strong pesticides to protect cotton, potatoes, and apple orchards from fierce insect infestations. University studies in the United States brought calcium arsenate to the frontline as an answer, riding the wave of chemical solutions that defined the "dust bowl" decades. The U.S. Department of Agriculture officially recommended its use by about 1922. For decades, this powdered material poured from hand-cranked spreaders. Growers valued its punch against weevils, bollworms, and other damaging pests at a time before synthetic organophosphates and carbamates arrived. By the 1950s, toxicity concerns and more potent alternatives started to steal its thunder. Yet in parts of the developing world, legacy stockpiles still pop up on farms that can’t afford pricier, modern solutions.
Product Overview
Calcium arsenate usually appears as a fine white or grayish powder, though manufacturers started to pack it into pellets and tablets for easier handling by mid-century. While some look at it only as a relic of hard-edged pest control, the compound also serves as an intermediate in the manufacture of arsenic-containing products used across industry: ceramics, glass, and even military applications. It is not a household product. Simple labels show a stark message: "Danger. Poison.”
Physical & Chemical Properties
In the lab, Ca3(AsO4)2 stands out with its distinctive white color and low solubility in cold water. Once in contact with moisture, a minor fizz or effervescent release points to mild reactivity. This powder kicks up dust quickly, which led to safety concerns long before strict environmental laws. Calcium arsenate’s pH hovers just above neutral, but the threat doesn’t lie in acid or base—it’s in the arsenate ion’s grip on living systems. The melting point pushes beyond typical workshop temperatures, so normal use keeps it in solid form. Ambient air won’t break it down, although strong acids risk freeing up arsenic acids and further hazards.
Technical Specifications & Labeling
Shipping barrels and bags of calcium arsenate come with distinctive diamond-shaped hazard symbols marking toxicity. Technical sheets list arsenic weight content, generally sitting between 38–41%. Particle size can make or break proper field application, with too-fine dust placing farmworkers at risk. Labels sometimes carry multiple languages: “Caution,” “Toxic,” and references to local code TPS standards. U.S. and EU states both require lot numbers, batch dates, and directions for disposal. Authentic manufacturers tout purity based on Good Manufacturing Practice (GMP) certificates. Long shelf life—when dry—relies on airtight storage above humidity and out of sunlight.
Preparation Method
Production comes down to reacting arsenic acid (or arsenic trioxide dissolved in acid) with calcium hydroxide. The procedure cracks out large volumes of fine powder, followed by filtration, washing, and slow drying. Modern chemical plants automate the precipitation step, pump off the liquid, and dry the remains in rotary kilns. Some older facilities fall behind on dust controls, releasing emissions that raise environmental flags. Chemists keep a close eye on the arsenic level in both raw materials and finished lots, since just a fraction of excess metal can tip waste over into the hazardous category, raising landfill and shipping costs.
Chemical Reactions & Modifications
Although fairly stable at room temperature, calcium arsenate breaks down in acid, liberating arsenic acid and leaving behind calcium salts like gypsum. Calcium arsenate changes form under strong heat, sometimes driving off arsenic oxides as vapor. In large reactor vessels, mixing with phosphate fertilizers creates blends for specialty applications, but these practices lost favor once less toxic and just as effective alternatives came to the market. Little chemical modification work happens today since researchers push industry toward substitutes and remediation more than novel arsenate uses.
Synonyms & Product Names
Industry catalogs mention names like “Basic calcium arsenate,” “Tricalcium arsenate,” and “Arsenate of lime.” Across regulatory documents in the United States, one finds it listed as “Fowler’s Dust” or “Leadless arsenate.” Catalog numbers appear in company records, but product codes vary wildly by manufacturer and country. No matter the name, the skull-and-crossbones label remains a clear signal for caution.
Safety & Operational Standards
Strict safety codes surround both handling and disposal. Workers must rely on gloves, fitted respirators, and full skin coverage to keep dust off hands and lungs. Regulatory bodies like OSHA and ECHA cite permissible exposure limits as vanishingly small—often less than 0.01 milligrams per cubic meter. Facilities require forced ventilation and negative-pressure rooms during packaging. Spill protocols call for vacuuming with HEPA filters and separate disposal channels. Washing stations sit near every loading bay. Used containers need triple washing and puncturing before landfill, as the tiniest residue can poison water tables. Safety Data Sheets warn operators to stay clear of storm drains, and runoff from production facilities faces regulatory monitoring with part-per-billion detection.
Application Area
In its mid-century heyday, calcium arsenate dust swept across Southern cotton fields, American apple orchards, and peanut plantations in India. Commercial operators saw it as a go-to for root-boring and leaf-chewing pests. Apart from agriculture, certain parts of the ceramics industry used it as a fluxing agent for specialty glass, and the pigment trade once toyed with arsenic minerals for rare color effects. Today, regulatory bans keep it away from food crops in most developed countries. Any legal use draws scrutiny, demanding explicit approval and paperwork.
Research & Development
Research now circles around safe disposal more than imaginative uses. Environmental chemists study how calcium arsenate moves in soil, chasing the fate of arsenic around groundwater and food systems. Remediation projects at contaminated sites in the southern U.S. combine soil washing, solidification, and bioremediation. Toxicologists continue to test old stockpiles stored in rusting barns. A few legacy projects search for low-cost ways to convert calcium arsenate to less bioavailable forms before landfill. Most recent R&D points toward analytical detection, not application.
Toxicity Research
No escaping the poison label: calcium arsenate is carcinogenic and highly toxic when inhaled or ingested. Even small-scale exposure damages the liver, kidneys, and nervous system, with chronic intake leading to skin cancers and cardiovascular disease. Epidemiological studies in rural cotton regions pointed fingers at increased cancer risks among field workers. Animal models confirm lasting damage, while in vitro tests show how arsenate disrupts cell metabolism and gene expression. The EPA lists it as a Group A carcinogen, meaning no safe exposure level has been measured in people. Groundwater monitoring near historical application sites still finds arsenic leaching years after farm use stopped. Every toxicologist I’ve ever read stresses that even minor residues present a problem on both land and water.
Future Prospects
Few see a mainstream revival for calcium arsenate. International bodies like the Stockholm Convention press for phase-out and destruction of all remaining reserves, and manufacturers slot in more sustainable alternatives. Yet the story isn’t over. Vast acres of farmland across the southern U.S., China, and Latin America still carry traces from mid-century use. Cleanup contractors gear up for decades of soil washing or phytoremediation. Researchers look to the microbial world, hunting for species that digest or immobilize arsenic. New sensor technology pledges to catch even trace contamination before it hits crops and water supplies. In the future, the central task is containing and remediating the environmental legacy, not expanding the footprint. Scars from calcium arsenate remind us that no short-term farm victory trumps generations of health and land.
What is calcium arsenate used for?
Digging Into the Story
Calcium arsenate has a name that makes even people who know nothing about chemistry pause. This compound shows up mostly in discussions about pesticides and the legacy of the industrial era. It's long been tossed around U.S. soil, sprinkled on fields of cotton, tobacco, and fruit orchards, with the promise that it would wipe out boll weevils, grasshoppers, and other bugs threatening farmers’ hard work. These days, you won’t see garden centers advertising it—that’s no accident.
Why It Got Popular
The years between the world wars saw massive growth in the use of agricultural chemicals. Farmers wanted cheap, effective ways to protect their crops. Calcium arsenate answered that call, slashing pest populations and bumping up harvest yields. The government offered little in the way of regulation then. Agronomists and rural communities put their trust in results, not research.
The dust didn’t settle right away. For decades, fields got showered with calcium arsenate. Some growers swear even now that it was the only thing that saved the season from ruin. Old-timers stand by it, recalling the headaches of hand-picking infested crops before chemical shortcuts became available.
Health and Environmental Consequences
It’s hard to ignore the price that came with those bigger harvests. The same compound that wrecked insect nervous systems got into drinking water, air, and eventually, human bodies. Arsenic, a key ingredient in calcium arsenate, is a known carcinogen. Reports began stacking up—farmers sickened, children exposed, soil left unable to grow anything but weeds. The Environmental Protection Agency flagged it early as a hazardous substance.
The EPA, Centers for Disease Control, and state health officials all attach decades of health warnings and bans to this pesticide. If a product leaves behind arsenic in water and soil, modern science doesn’t have many good ways of getting rid of it. Unlike fuel spills or bacterial contamination, arsenic lingers for generations. No farmer I know would want this kind of inheritance.
Solutions Looking Forward
Today’s farmers rarely ask about calcium arsenate unless they’ve inherited land that once ran on it. Cleaning up contaminated ground takes time, money, and a lot of faith. Activated carbon, careful crop rotation, hauling topsoil, phytoremediation with special plants—every fix has trade-offs and limits. Governments have started identifying old arsenic contamination zones, but full-scale projects cost millions and disrupt farming families.
Most of us want to see farms thrive without taking on chemical risks. Crop scientists push for safer options—biological controls, integrated pest management, and newer, less persistent pesticides. Some growers choose to go organic, relying on habitat management and natural predators. These tactics demand more time in the field and a bigger learning curve, but they don’t leave a toxic trail for future generations.
Learning about calcium arsenate means accepting that shortcuts often create their own emergencies. Farmers deserve tools that won’t poison their neighbors. People need to keep asking hard questions before putting a substance into the land, no matter how tempting the promise of bigger harvests or faster solutions.
Is calcium arsenate toxic to humans and animals?
Understanding the Danger
Calcium arsenate once showed up everywhere from Southern cotton fields to suburban manicured lawns. Farmers and homeowners spread it generously across the ground, chasing after bugs, fungus, and tough weeds. The reason? This powdery compound works fast and wipes out a range of pests. But the deeper story shows far-reaching risks, especially for people and animals.
Health Risks for People and Pets
Dust from calcium arsenate curls up from old barns, drifted soil, and sometimes even antique garden products stored on garage shelves. A child running barefoot across a contaminated yard or a dog pawing through dirt can pick up more than a dirty foot—calcium arsenate enters the body through the skin, tiny cuts, or inhalation. Poisoning does not take much. Even at doses too small to see or taste, arsenic starts to build up in the body, sticking to kidneys, liver, skin, and nerves. People exposed regularly to arsenic dust or residues—farm workers, home gardeners, and children playing in old fields—face higher rates of cancer, nervous system damage, and chronic skin problems. It does not stop with people: livestock grazing on treated pastures or pets that roll in the grass end up just as vulnerable.
Science links arsenic compounds with lung, bladder, and skin cancers. The World Health Organization does not mince words; arsenic is a serious carcinogen. Unsafe water laced with arsenic leaches from contaminated fields into wells, spreading the risk to those just drinking their morning glass of water.
Environmental Fallout
Rains hardly dilute the threat; instead, they help the poison hitch a ride downhill, seeping into water tables and streams. Small fish, frogs, insects, and birds take the first hit, picking up arsenic through water or soil. It climbs the food chain, landing on the dinner table in meat, eggs, or vegetables. Studies confirm arsenic accumulates in crops grown on old, contaminated land.
Decades-old use still echoes in the ground. The contamination is stubborn. People living near former calcium arsenate-treated lands sometimes see wells test positive for arsenic long after anyone remembers why the soil turned dangerous in the first place.
Better Choices and Safer Practices
Plenty of places now ban or severely restrict calcium arsenate, favoring options that break down quickly and leave the land safer for kids, pets, and wildlife. Anyone digging in an old garden or farmland ought to check up on the history of the property and run a soil test. Homeowners with historic pesticides tucked away in sheds should drop them off at hazardous waste facilities.
Switching toward integrated pest management works—using natural predators, rotating crops, improving soil health—to keep bugs in check without risking human or animal health. For fields already touched by old pesticides, experts use soil amendments and phytoremediation (planting arsenic-absorbing plants) to clean up contamination. These steps cost time and money, but they keep dangerous chemicals out of food and water, and away from vulnerable communities.
The Case for Caution
My own experience tending a family garden on land once treated in the 1940s taught me not to trust the soil until it’s tested. Our local university lab flagged arsenic levels way above safety limits. Simple changes—raising fresh beds off the ground, choosing non-contaminated compost, trading chemicals for hand-weeding—brought peace of mind as well as healthier vegetables.
Calcium arsenate belongs in the past. Its dangers reach farther than expected, hurting people, animals, and the food chain for generations. Keeping it out of gardens, fields, and pantries is more than a smart move. It’s a responsibility.
How should calcium arsenate be handled and stored safely?
Understanding the Risks
Calcium arsenate showed up on farms and orchards decades ago, promising to tackle tough insects. Old barns and storage sheds still hold leftover bags marked with faded warnings. It's important to realize the risk stays real. This powder does a number on pests, but it also exposes people to arsenic poisoning. A little exposure over time chips away at health, throwing the kidneys, skin, and even nerves into a mess. Everyone working near this chemical needs real protection, not just a quick dust mask or ordinary gloves.
Building Barriers—Not Excuses
Walking into a storeroom with poorly closed calcium arsenate bags invites trouble. I remember someone once grabbing a bag with bare hands, ignoring the warning. Days later, he felt a persistent throat ache and rash on his arms. That moment stuck with me. Gloves made from thick rubber, goggles that hug the face, long sleeves, boots—these keep the dust away from skin and eyes. N-100 or P-100 respirators grab almost all airborne particles. Some may think a regular surgical mask will do, but those give a false sense of safety.
Where Storage Makes All the Difference
Walls, not tarps, belong between people and pesticides. Thrown together with paint cans or rakes, calcium arsenate becomes a ticking hazard, not just for workers but for kids or pets who might wander in. A separate room with a locked door makes sense. The air inside should flow well, drifting fumes out and not back in. Dryness matters, since even small leaks make these powders cake up or leak into the floor. Label everything with big, bold writing. Stack containers tightly, with nothing above them that could fall and burst a bag or jar.
Simple Habits—Big Impact
Eating or drinking anywhere near calcium arsenate counts as a gamble. One friend picked up his sandwich right after mixing powder into a sprayer. It didn’t cross his mind—the arsenic had settled under his fingernails. Wash hands every single time before breaks, and ditch work clothes before heading home. If any gets on skin, rinse right away—no delaying for the end of a shift.
Disposing—Don’t Mix Old with New
Never pour leftovers into a drain or toss them onto a compost pile. Local waste authorities know the correct disposal channels. Their guidelines may feel like a headache, but they come from hard lessons from spills and poisonings in the past. Leaving old containers empty or half-used for the next person adds risk, not thrift.
Learning from Tragedy—Safer Together
Incidents of arsenic poisoning tell the same story: warnings ignored, shortcuts taken, damage done. It's not just about following textbook rules; it's about caring for yourself and those around you. Talking about safe handling during regular team meetings keeps everyone alert. Training isn't a one-and-done thing—it grows with new people and as old risks show up in fresh ways.
Checking for Better Solutions
Alternatives have emerged over the years. Whenever possible, choose safer, less toxic products. Some farms and municipalities hold onto old stocks out of habit or cost concerns. They often find, with a little curious research, that new tools do the same job, minus the constant worry. Keeping calcium arsenate around out of convenience puts community health on the line.
What are the environmental risks of using calcium arsenate?
A Pesticide from the Past Still Lingers
Calcium arsenate isn’t a name most people hear in everyday conversation, but on farms and fields, it carried a strong presence in the past. Used to combat boll weevils in cotton and other pests, this white powdery chemical promised relief and higher yields. Walk through older farming towns in the southern United States and folks still recognize its legacy—and its threat. My family grew up near such fields. Stories filtered down of poisoned wells and sudden fish kills after a heavy rain. Those experiences become hard facts you can’t ignore.
The Problem with Arsenic in Soil and Water
Arsenic, the toxic element in calcium arsenate, does not vanish. It seeps into earth and stays for generations. Once applied, rain pulls it deeper, reaching roots and groundwater. Animals grazing nearby, even decades later, risk picking up arsenic from plants or surface water. The Centers for Disease Control underline links between arsenic exposure and cancers of the skin, bladder, and lungs. Fish and aquatic life also show stunted growth or sudden die-offs. The soil in affected fields may carry warning levels of toxicity for many years, and some local farmers refuse to use land they know got dusted with calcium arsenate by their grandparents.
Human and Wildlife Health Remain at Risk
Arsenic doesn’t just threaten crops—it follows people. Drinking water near old application sites sometimes tests too high for safety, leading families to haul bottled water every week. Symptoms like stomach pain or numbness in hands can show up in people exposed to even small amounts over time. In my county, a few wells have been closed because tests showed arsenic lingering from old cotton fields. No one should risk their family’s health just to live where roots run deep.
Spreading Beyond the Field
Runoff from fields flows into rivers after storms, carrying arsenic downstream. Fish populations drop, and residents notice fewer frogs or birds. It’s tough seeing how pollution from old chemicals still shapes ecosystems. The U.S. Geological Survey found arsenic hotspots in watersheds that traced straight back to agricultural land where calcium arsenate was once king.
Facing the Problem—Not Brushing It Aside
Ignoring the risk only invites bigger problems later. Some solutions start with mapping hotspots, then capping or remediating old fields. Farming can heal its legacy by switching to less toxic controls and leaning into organic practices on old arsenic-laden soils. Local governments can help by testing more wells and providing clean water to affected homes instead of putting the burden on families. Land conservation groups in my area have also taken on projects to restore contaminated wetlands.
Moving Forward for Safer Communities
The story of calcium arsenate is one of learning from past mistakes. We now have the proof that ignoring heavy metals in agriculture leaves scars on the land, wildlife, and people. Stories from neighbors, hard lessons from science, and common sense all point to one thing: we need to heal the places harmed by legacy chemicals like calcium arsenate—and keep future generations from repeating the same errors.
Are there safer alternatives to calcium arsenate for pest control?
Understanding Calcium Arsenate’s Legacy
Calcium arsenate used to be a popular pesticide, especially in cotton-growing areas in the U.S. It worked against boll weevils and slugs, but it left behind a toxic legacy. Farmers, farm workers, and communities near treated fields paid a heavy price. Studies linked calcium arsenate to cancer, nerve damage, and contaminated land. In school, I learned how fields treated with these arsenic-based products sometimes grew nothing but disappointment for years.
Risks Outweigh Rewards
Keeping crops healthy matters, but people started asking if toxic chemicals are worth the risk. The Environmental Protection Agency and scientists didn’t need to look far for answers. In my own reading, cases in rural Texas and Mississippi convinced regulators to take calcium arsenate off the market for most uses. Groundwater tests picked up arsenic traces, and former users spoke about unexplained illnesses. The pattern is pretty clear — this chemical sticks around, and getting rid of arsenic isn’t easy.
Exploring Safer Alternatives
Pests don’t take a break just because a chemical gets banned, so growers looked for new tools. Biological controls stepped up. These days, ladybugs, lacewings, and parasitoid wasps find their way into many organic and conventional farms. I once visited an apple orchard in northern California where they used pheromone traps and sticky barriers to control pests. It sounded too simple at first, but yields stayed strong, and the air felt cleaner.
For crops where biological methods fell short, growers turned to less toxic synthetic chemicals. Products containing spinosad, derived from soil bacteria, gained approval from many scientists. Studies show spinosad breaks down quickly in sunlight and doesn’t stick around in soil. I’ve met gardeners who switched to neem oil, made from tropical neem trees. Neem messes with insect hormones but doesn’t pose serious risks for people or wildlife when used right. These alternatives carry their own trade-offs, but so far, the track record looks good.
Integrated Pest Management: A Smarter Approach
One solution that keeps popping up in agricultural meetings is integrated pest management, or IPM. This involves mixing physical, biological, and chemical tactics, tailoring responses to actual pest pressure. Farmers watch their fields for pests before applying anything, which saves money and reduces exposure to any chemical. In Iowa, I talked with a farmer who rotates corn and soybeans and lets natural grass strips grow between fields. He swears by fewer sprays and better soil.
Encouraging Changes Through Policy and Education
Switching away from calcium arsenate took push from government regulations and local action. But real progress doesn’t happen without education. Cooperative extensions, workshops, and government cost-share programs gave farmers the tools and knowledge to change. It worked in the past, and it's still working. Whenever growers learn about safer options and see the evidence in their own fields, more join in.
Looking Ahead
Nobody wants to go backward to the era of calcium arsenate. Healthier fields and healthier communities show that safer alternatives exist and succeed. Continued investment in research, extension programs, and farmer support can keep pest control moving in a safer direction. Plus, listening to the experiences of workers and communities affected by past practices can help everyone avoid making the same mistakes again.
| Names | |
| Preferred IUPAC name | calcium diarsenate |
| Other names |
Ledeen Purasit TL 115 Tricalcium arsenate |
| Pronunciation | /ˈkælsiəm ɑːˈsɛneɪt/ |
| Identifiers | |
| CAS Number | 7778-44-1 |
| Beilstein Reference | 377229 |
| ChEBI | CHEBI:33342 |
| ChEMBL | CHEMBL1201587 |
| ChemSpider | 20228413 |
| DrugBank | DB11075 |
| ECHA InfoCard | 100.007.785 |
| EC Number | 007-004-00-9 |
| Gmelin Reference | 169344 |
| KEGG | C18641 |
| MeSH | D002119 |
| PubChem CID | 24413 |
| RTECS number | CG3325000 |
| UNII | 6TC78V60Q1 |
| UN number | UN1557 |
| Properties | |
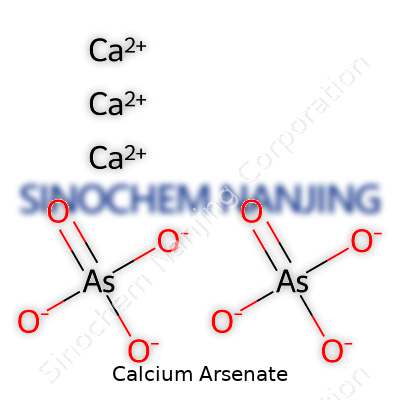
| Chemical formula | Ca3(AsO4)2 |
| Molar mass | 398.07 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 2.4 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | −0.847 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 1.99 |
| Magnetic susceptibility (χ) | \-49.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.642 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1862 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3720 kJ/mol |
| Pharmacology | |
| ATC code | SAC08 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or absorbed through skin; carcinogenic; may cause damage to organs through prolonged or repeated exposure; very toxic to aquatic life. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H331: Fatal if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P270, P271, P272, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P320, P330, P361, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-ACID |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LDLo oral human 143 mg/kg |
| LD50 (median dose) | 6.3 mg/kg |
| NIOSH | SN38500 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Calcium Arsenate: 0.01 mg/m³ (as As) |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Lead arsenate Magnesium arsenate Zinc arsenate |

