Calcium Amide: Perspective on a Niche Chemical with Growing Importance
Historical Development
Calcium amide entered the world of industrial chemistry as something of a byproduct in the evolving quest for cheap sources of ammonia and nitrogen-based fertilizers. Its story traces back to the early 1900s. As folks tinkered with the old Haber-Bosch process and ways to fix atmospheric nitrogen, the idea of reacting calcium carbide with ammonia soon caught on. This wasn’t just some academic exercise. Early calcium amide found its place in agriculture, and you would spot it in the dusty ledgers of fertilizer plants that tried to squeeze every useful compound out of the then-revolutionary air-to-fertilizer process. Older chemists sometimes remember the pungent smell and the powdery residue left over from reactions involving this material.
Product Overview
These days, calcium amide pops up most often in the chemical industry as an intermediate. You can usually recognize it by its grayish-white, granular appearance, something that doesn’t look especially remarkable but carries a considerable punch in certain reactions. Unlike pure calcium compounds, it brings its own reactivity thanks to the amide group. Fertilizer makers keep it on deck, but so do those working in pharmaceutical precursors and other specialized syntheses. The material arrived at its status partly because of its affordability, but mostly thanks to how it steps up as a strong base and nitrogen donor.
Physical & Chemical Properties
In your hand, calcium amide feels gritty—chalky, almost like powdered cement. The chemical formula, CaNH2, paints the picture: one calcium atom bonded to a couple of amide groups. It's not especially resistant to water. Exposure triggers a release of ammonia gas that escapes with that sharp, unmistakable smell. The powder burns eyes and nostrils, and a whiff is enough to remind anyone—respect the compound. Heat it up, and it shows stability up to around 600°C, but the real key is how hungry it is for protons, which lets it break down a whole range of other chemicals.
Technical Specifications & Labeling
Suppliers usually designate calcium amide by nitrogen content and physical granule size. Labels warn against moisture exposure with good reason: ammonia release is no joke, and contact with skin can leave burns. Classification often falls under “corrosive” and “dangerous for the environment.” The product ships in tightly sealed bags or drums, and regulations insist storage happens well away from acids or moisture. You won’t find it on hardware store shelves, so procurement usually routes through established chemical suppliers with licenses and proper documentation.
Preparation Method
Making calcium amide starts with calcium carbide and dry ammonia gas under a nitrogen atmosphere. The reaction’s basically this: pass ammonia over hot calcium carbide, anywhere from 350–400°C. Solid calcium amide forms, while hydrogen gas floats away. Some older methods tried direct reactions with calcium metal itself, but those faded for safety and cost reasons. The entire setup demands anhydrous conditions and a sealed reactor—otherwise, you lose not only yield but also pose some nasty risks to workers while breaking glass and corroding equipment.
Chemical Reactions & Modifications
Chemically, calcium amide is something like its cousin, sodium amide, but with a few quirks. It strides into alkylation and amination reactions, especially where organic chemists want to avoid extra sodium. You see it convert nitriles to amidines, split up esters, or help form deaminated products. It reacts fiercely with water, acids, and halogenated hydrocarbons. That gives formulators an edge in niche syntheses but turns any spill into a mess that must be neutralized quickly. For more tailored applications, calcium amide sometimes blends with alkoxides or other metallic reagents to nudge selectivity in certain reactions.
Synonyms & Product Names
You might see calcium amide labeled as “calcium diazanide” or “calcium azanide” in older literature, but the industry standard name sticks to calcium amide. Internationally, some call it “ammonide of lime” or “calcium nitramide,” a reflection of the old practices before IUPAC names held sway. Different brands carry it under their own product codes, but on a shipment manifest, it’s almost always “calcium amide, CaNH2.”
Safety & Operational Standards
There’s no skirting around the hazards. Calcium amide is especially rough on unprotected skin, eyes, and respiratory membranes. Workplaces make gloves, goggles, and face shields standard when handling it. It can form explosive mixtures with organic materials or emit toxic ammonia if it meets water. Storage protocols everywhere treat the product with healthy respect, calling for dry, cool, well-ventilated warehouses and strict access controls. Training is not optional; it’s part of onboarding for new lab techs or plant workers. Safety data sheets outline what to do if anything goes wrong—neutralize spills with dilute acid, never water, and evacuate if ammonia clouds up.
Application Area
While fertilizer use has shrunk as cheaper, easier-to-handle options took over, calcium amide didn’t disappear. Its true value lies in specialty organic synthesis where strong basic conditions are needed, especially for industries producing pharmaceuticals, dyes, and certain plastics. Some metal processing operations use it to introduce nitrogen into melts, and even a few environmental cleanup projects bank on its reactivity to break down persistent contaminants. That versatility keeps it in circulation among industrial buyers with advanced technical chops.
Research & Development
Chemical researchers haven’t turned away from calcium amide, even as other reagents become trendy. Ongoing work pushes its reactivity, tweaking reaction conditions to open new synthetic pathways or cut down on byproducts. A few efforts look at how calcium amide might facilitate greener processes for nitrogen introduction, especially when companies work to shrink carbon footprints and chemical waste. Others keep an eye out for new uses in catalysis or battery tech, given how strongly the amide group interacts with transition metals and organic frameworks.
Toxicity Research
Exposure studies show calcium amide goes beyond mere irritation. Chronic inhalation or repeated skin contact can have long-term impacts, particularly in unventilated spaces. Researchers spent years digging into the toxicological profile because fertilizer workers fell ill before proper standards arrived. The compound’s effect comes mostly from ammonia release and local caustic burns. Thankfully, the body of data means current industrial hygiene keeps exposure far below danger levels, but each new study reinforces the message: don’t underestimate it.
Future Prospects
From a broader view, the future for calcium amide hinges on specialty demand rather than mass consumption. As green chemistry goals dig deeper into industry, calcium amide’s strengths—high reactivity, strong nucleophilicity, ability to serve as a nitrogen source—to keep it relevant for those willing to manage its hazards. There’s real hope that improved handling techniques or encapsulated forms might expand its footprints, especially in energy storage and environmental restoration. While the scientific world gives some compounds a quiet retirement, calcium amide continues to attract innovators looking for reliable ways to drive tough chemical reactions.
What is Calcium Amide used for?
Tapping Into the Power of a Simple Compound
Calcium amide doesn’t show up much in everyday conversation, but step inside a fertilizer plant or a chemical factory and you’ll notice this grayish powder finds plenty of work to do. Its chemical formula, CaNH2, sets it up for a starring role in soil treatment, chemical synthesis, and gas removal. Most folks might not know it by name, yet what it contributes makes quiet waves throughout several industries.
How Farmers Benefit From Calcium Amide
Fields full of hungry crops need nitrogen, and this compound delivers just that. Spread across a field, calcium amide slowly releases ammonia when it comes into contact with water and soil microbes. Crops like wheat and corn soak up that ammonia and transform it into proteins. Some fertilizers promise quick results but bring downsides—leaching, burning roots, or harming helpful bacteria. Calcium amide provides a more controlled, steady ammonia trickle, reducing environmental losses and keeping crops fed through key growing phases.
From my own experience growing vegetables on a small plot, managing nitrogen helps keep yields strong and disease at bay. Too much at once, and you get leafy growth with no fruit. The gentle feed from this fertilizer sort keeps things balanced. It’s good to know there’s an option that lets you work with nature instead of against it.
Key Uses in Chemical Manufacturing
Step into a chemical processing lab, and you’ll see calcium amide used for making dyes, plastics, and even pharmaceuticals. The strong base properties of this compound let it grab onto unwanted acids. Chemists rely on it to synthesize all sorts of chemicals because it acts as a strong deprotonating agent, pulling hydrogen atoms off other compounds so new bonds can form. That’s a fancy way of saying it makes other reactions possible—like a team member that cleans up the workspace so everyone else can get their job done.
In making sodium cyanamide, an important step for producing certain herbicides and even pharmaceuticals, calcium amide joins the process and keeps reactions moving forward. Safety matters a great deal here. Unlike some alternatives, it won’t easily explode or release toxic gases at room temperature, which means people working around it can stay safer with proper handling.
Clearing Out Gases—Hydrogen Removal
Hydrogen gas builds up during large-scale chemical production, causing headaches for equipment and safety. Tossing calcium amide into the mix pulls hydrogen out of the system. Inside vacuum tubes or sealed reactors, it reacts directly with hydrogen and locks it up in a more stable form. This keeps reactions running smoothly and helps prevent pressure build-up that can lead to accidents or shutdowns.
Safer Handling Means Less Risk
Calcium amide carries its share of risk—moisture can set off a reaction, so storage in dry, airtight containers matters. Workers need gloves and eye protection, and facilities have to keep air humidity low. Still, compared to alternatives like sodium or potassium amide, which can catch fire simply by sitting out, calcium amide proves less volatile. That brings peace of mind for workers and plant owners alike.
Potential for Greener Solutions
The world craves better ways to feed people without polluting rivers, and to run chemical plants with fewer risks. Research has started looking at calcium amide-based blends that don’t just fertilize but also help control pests—or add organic material back into the soil. If regulators and industry groups work hand in hand, updated training plus better gear could make its use even safer. Investments in handling systems and worker education shape a safer, cleaner industry.
What is the chemical formula of Calcium Amide?
Understanding Calcium Amide
Calcium amide doesn’t get much attention compared to some of its cousins in the chemical world, but anyone who’s set foot in an agricultural setting or spent time dealing with specialty industrial synthesis recognizes its value pretty quickly. The formula is simple on paper: Ca(NH2)2. That’s one calcium atom paired up with two amide groups. Doesn’t look complex. The significance runs much deeper, though, especially if you look past textbooks and into the real world.
Everyday Impact in Agriculture and Manufacturing
Farmers rely on fertilizers and soil amendments to manage crops and get a better yield. Calcium amide slips into that process as a source of both calcium and ammonia, two ingredients plants crave. Out on the field, it reacts with water and soil, breaking down into products that can help balance pH and supply nitrogen. This isn’t just theory. Corn and wheat producers, looking for consistent output, turn to this compound, knowing it works in slightly more acidic soil conditions where other amendments fall short.
Handling Calcium Amide
No one can talk about this chemical without talking safety. The industrial production process usually means reacting calcium carbide with ammonia. Ever worked with either material? Both demand respect. Before you even open the sack or drum, you make sure your gloves and goggles aren’t missing: calcium amide can react with moisture, giving off ammonia—never a pleasant surprise. Stories of minor accidents in remote storage sheds remind everyone why chemical literacy is not a luxury but a necessity, especially outside the lab.
Role in Synthesis and Industry
Factories use calcium amide for more than just fertilizer. Industry taps it for organic synthesis, picking it for jobs where a strong base can get things moving. For chemists prepping dyes or pharmaceuticals, sometimes it provides the right punch, making difficult reactions possible. These fields don’t always make headlines but form the backbone of the products on store shelves. From my time talking to chemical engineers, there’s a common appreciation for calcium amide’s reliability—especially for cost-effective processes involving large batch runs.
Challenges and Room for Improvement
Using calcium amide doesn’t run free of issues. Storage in humid environments leads to caking or release of ammonia. I’ve seen shipping containers treated with extra sealant, only to open them and catch the unmistakable whiff of released gas. That’s a real headache for producers and end-users. Smarter packaging and improved labeling practices can cut down the risk. Training goes a long way too. Regular review sessions focusing on chemical handling have made a real difference in businesses I’ve worked with.
Where Calcium Amide Stands Today
Balancing the chemical’s usefulness with its hazards demands honest, practical approaches. Whether you’re spreading it on a field or using it in manufacturing, knowledge and caution play just as important a role as the actual material. Those who work with calcium amide don’t waste effort on theoretical debates—they want safe, effective, and timely results. The chemical formula, Ca(NH2)2, tells you what you’re dealing with, but experience shows you how to get the best out of it—safely and efficiently.
How should Calcium Amide be stored?
Understanding the Real Risks
Calcium amide turns heads in labs and industry for its reactiveness. Folks who have handled this stuff know it doesn’t play around. Moisture causes it to react, and that reaction can throw off ammonia and heat. Mix water and calcium amide, and you’re courting danger: fires, explosions, not only chemical burns. It’s tempting to toss such details into a safety data sheet and move along, but I watched a minor oversight in storage trigger a full week of cleanup in an old fertilizer plant. If you think a “dry place” means a corner of a storeroom, think again.
Keep It Dry, Keep It Separate
A warehouse manager once told me that storing reactive chemicals is more about respecting their quirks than following a template. Calcium amide wants an arid, sealed space. Moisture hangs in the air, wicks in through cracks, and even escapes from loosely fitted lids. Even basic humidity spells trouble. A properly sealed drum, stored under a dehumidifier or in a climate-controlled spot, avoids all sorts of unwanted surprises. Back in the day, we checked storage temperatures as carefully as we checked shipment schedules.
No Shortcuts: Choose the Right Container
Nothing spoils a week like opening a container of calcium amide and finding corrosion or caked-up powder. Plastic drums or well-lined steel containers hold up. Any compromise—one dent, a tiny breach—means moisture gets in. I’ve heard of someone skimping with a reused fiberboard barrel. That lesson lasted years, since nobody forgot the resulting ammonia stink. Safe storage leaves zero room for improvisation. Good locks and clear labels cut confusion, especially if you use shared facilities. Nobody wants the night shift mixing this up with something else.
Less Is More: Avoid Overstocking
A small operation, thinking it made sense to buy in bulk, paid for that mistake when excess stock went bad. Calcium amide doesn’t age gracefully, so keep on hand only what you need for the short term. Distributors appreciate buyers who understand risk management. Smaller, well-sealed containers beat half-empty drums with mystery powder at the bottom. If you think you’re saving money by buying big, budget future disposal and possible emergency services.
Training, Not Just Signage
I’ve sat in rooms listening to safety briefings while half the folks doodled in their notebooks. You want everyone handling calcium amide to know why the storage rules exist. Not just the science—what can actually go wrong, what it smells like when trouble starts, and who to call. Real training, with real examples. Emergency access to spill kits and ventilation plans should be as familiar as the location of the coffee machine. If you’ve never smelled burning ammonia, trust me, that lesson sticks.
Sensible Steps Forward
Every bit of storage advice around calcium amide comes from lessons learned the hard way. Respect for the material, containers that actually keep out moisture, and zero tolerance for shortcuts make the difference between safe operation and a headline nobody wants. Focus on true knowledge, strong habits, and a work culture that sees safety as a given, not a cost.
Is Calcium Amide hazardous to health?
Getting to Know Calcium Amide
Calcium amide, known to chemists as Ca(NH2)2, pops up in applications like synthetic fertilizers, chemical manufacturing, and sometimes in laboratory research. Its strong reactivity comes from its ability to break down or combine with other substances—qualities that make it useful but also tough to handle safely.
Why Handling Calcium Amide Demands Care
A handful of farmers or workers in chemical plants already know the harsh bite of calcium amide dust. Breathing in its powder or getting it on your skin isn’t like touching table salt. The compound reacts with water—including sweat and humidity in the air—to release ammonia, which has a sharp, choking odor. Prolonged exposure burns nasal passages and lungs. Eyes sting. Skin can break out in rashes or even suffer chemical burns.
Research shows calcium amide can trigger respiratory issues in workers where dust becomes airborne. The Centers for Disease Control and Prevention (CDC) notes short-term symptoms: severe irritation of eyes, skin, and mucous membranes. With accidents, burns might form. Cases documented over years in fertilizer factories reveal that repeated skin contact often leads to dermatitis—a fancy term for stubborn, uncomfortable rashes.
What Science and Experience Reveal
A few labs study the compound for unique chemical properties, but not many household users come across it outside agriculture or industrial settings. Looking at my own experience, spending time in soil science research, there’s a lesson that echoes: the more unfamiliar a chemical seems, the easier it is to underestimate risks. Calcium amide doesn’t act like the stuff under your sink. Chemicals like this don’t advertise danger with bold colors. Accidents result from a casual swipe of the hand or glove worn a little too long.
Ammonia gas, the byproduct, destabilizes indoor air quality fast. Short exposure can cause coughing fits; longer spells land people in urgent care. Besides burns and breathing trouble, the chemical poses a fire hazard since it can react with water and certain organic substances, raising the risk for dangerous accidents.
Ways to Keep Safe Around Calcium Amide
Most of the problem boils down to workplace safety. Adequate training and clear communication about chemicals rank just as important as any fancy equipment. Gloves, eye protection, and face masks should be on hand, not locked away in a cabinet. Facilities need air ventilation systems that don’t let dust linger.
Signs and Safety Data Sheets need to stay posted and up to date. Employers and supervisors can schedule regular safety drills or checklists. It isn’t about paranoia. Chemical burns and breathing issues hang heavy for those unlucky enough to get exposed, sometimes leading to long-term lung conditions. It helps to foster a culture where people feel comfortable warning about unsafe conditions or reporting faulty gear.
Finding Smarter Solutions
Switching to less hazardous alternatives pays off where possible. Some industries can swap high-risk chemicals for options that don’t carry such a hard punch. Technology allows for better dust extraction, plus sensors now track air quality nearly in real time. Government agencies roll out stricter regulations and inspections after learning the hard way from older case studies and industrial accidents.
It’s easy to overlook how serious calcium amide exposure gets until you talk with someone who’s endured chemical burns or trouble breathing from a workplace mishap. Staying informed and taking small measures—like a new pair of gloves or an extra five minutes on a job site checklist—means coming home safe is much more likely.
What are the safety precautions when handling Calcium Amide?
Why Calcium Amide Calls for Respect
Calcium amide doesn’t announce itself with a bang, but it packs plenty of risks in the lab or out in the field. I remember my first encounter back in chemistry class: the instructor barely touched a jar before launching into warnings about “violent reactions.” He wasn’t exaggerating. This white powder reacts fiercely with water and acids, spinning off ammonia gas and heat so hot it can set things on fire. This isn’t just a classroom concern; if you spill some or get it on your skin, the effects aren’t minor or subtle. Chemical burns, respiratory issues, irritated eyes—these aren’t what anyone wants to come home with after a shift.
Protective Gear Isn’t Optional
People sometimes roll their eyes at goggles and gloves. Big mistake with calcium amide. Safety goggles need to cover the sides of your eyes, not just the front. Ammonia doesn’t respect the limits of your eyewear. Skin contact means serious alkali burns, so thick, chemical-resistant gloves make the difference. Long sleeves and closed shoes keep splashes off your arms and feet. A lab coat or apron just gives another layer between you and a very bad day.
Good Ventilation Matters
I once saw what happens if calcium amide gets damp in a poorly ventilated space: clouds of ammonia, strong enough to flood your eyes with tears and choke your lungs. Always handle this stuff in a fume hood. If you’re not in a lab, find a well-ventilated area and use portable fans that blow air away from you. Don’t trust a regular office room’s airflow—ammonia can linger in dead corners and find people who wander in later.
Water and Acids: Keep Them Far Away
Water and calcium amide should stay on opposite sides of your workspace. Mixing the two gets hot fast and forms ammonia gas. If you keep your materials organized, you’re a lot less likely to have an accidental spill. Acid containers should never stand near calcium amide. All it takes is one careless reach and the reaction kicks off. I’ve heard stories of storage closets turned into clean-up sites, just because someone left incompatible chemicals on the same shelf.
Spill Response: No Rushing
If there’s a spill, don’t grab a wet paper towel and swipe at it. That’ll make things worse. Use a dry, chemical-safe absorbent and carefully scoop up the solid. Dispose of it according to local hazardous waste rules—don’t dump it down the drain. If any dust gets in the air, leave and let the ventilation clear it out before coming back. I’ve seen well-meaning managers tell workers to sweep without thinking. Training and preparation save time and keep people out of the hospital.
Training and Constant Vigilance
No one should handle calcium amide without a clear rundown of the risks and a walkthrough of the emergency plan. Include ammonia detectors where practical, especially if quantities get large. Make sure everyone knows where the eyewash and emergency showers are. In my experience, people remember what they’ve practiced, especially under stress. That’s why regular drills beat lectures every time.
Safer Alternatives Where Possible
Sometimes chemistry allows for less hazardous choices. If the job can get done with something safer, take that option. Check in with chemical suppliers and ask about substitutes. Many companies, especially after tougher safety laws, have started to rethink their need for risky reagents. Sticking to calcium amide only when truly necessary lowers the risks for everyone on site.
Careful Storage and Clear Labels
Store calcium amide in airtight containers made for corrosive substances, far from anything it could react with. Labels should stand out, not fade into the shelf. Emergency contacts, hazard warnings, and instructions go right on the container. If containers get old or labels rub off, replace them fast. Good housekeeping keeps problems from snowballing when people get busy or distracted.
Handling calcium amide safely takes attention on details and respect for the risks.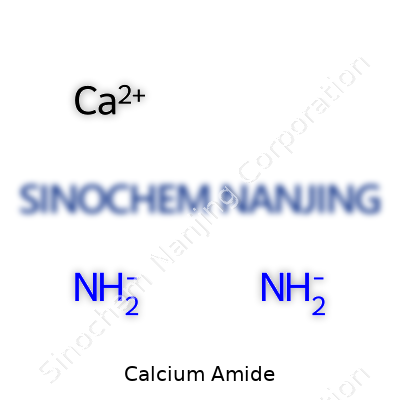

| Names | |
| Preferred IUPAC name | Calcium diazanide |
| Other names |
Calcium nitride Calcium diazanide Calcium azanidylazanide Calcium amide (Ca(NH2)2) |
| Pronunciation | /ˈkæl.si.əm ˈæm.aɪd/ |
| Identifiers | |
| CAS Number | 643-26-7 |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:31343 |
| ChEMBL | CHEMBL1201647 |
| ChemSpider | 87660 |
| DrugBank | DB01328 |
| ECHA InfoCard | ECHA InfoCard: 029-002-00-4 |
| EC Number | 215-137-3 |
| Gmelin Reference | 12004 |
| KEGG | C01892 |
| MeSH | D002119 |
| PubChem CID | 6093251 |
| RTECS number | EW2700000 |
| UNII | 8LJ08GX3K8 |
| UN number | UN1402 |
| Properties | |
| Chemical formula | CaNH |
| Molar mass | 64.09 g/mol |
| Appearance | White or greyish-white powder |
| Odor | Odorless |
| Density | 1.98 g/cm³ |
| Solubility in water | Decomposes |
| log P | -1.3 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 35 |
| Basicity (pKb) | 11.2 |
| Magnetic susceptibility (χ) | -30.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.973 |
| Dipole moment | 1.55 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 69.5 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -66.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -356.6 kJ/mol |
| Pharmacology | |
| ATC code | V03AB15 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H260, H314, H400 |
| Precautionary statements | P223, P231+P232, P280, P335+P334, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 3-1-2-W |
| Autoignition temperature | > 590 °C (1094 °F; 863 K) |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 960 mg/kg |
| LD50 (median dose) | 960 mg/kg (rat, oral) |
| NIOSH | MG0175000 |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | **60 mg/m3** |
| Related compounds | |
| Related compounds |
Calcium cyanamide Calcium hydride Sodium amide Potassium amide |

