Cadmium Sulfate: Substance, Risk, and the Path Forward
Historical Development
Cadmium sulfate made its first mark in the chemical world not only as a curious byproduct of zinc extraction but quickly gained ground for its unusual properties. Early chemists saw the brilliance of cadmium pigments and the potential in the metal’s unique chemistry. The story took shape in the industrial era, when mining and metallurgical processes ramped up supplies of cadmium, sparking research into practical uses. Over decades, industries leaned into this compound, using it in electroplating, as part of battery chemistry, and even as a pigment. Its use came with little early concern for health, but as factory workers and researchers experienced contamination, the world woke up to cadmium’s toxic side. This mix of excitement and care set the tone for how the world now treats cadmium sulfate today—with a blend of respect for its abilities and caution about its risks.
Product Overview
Cadmium sulfate isn’t a chemical most folks encounter day-to-day, unless they're working in specific industrial settings. It shows up as a colorless salt, usually found in solid, hydrated form. Labs and manufacturers rely on its solubility in water and its ability to supply cadmium ions where needed. Even though its uses have narrowed over the years due to toxicity, electrolytic cadmium plating and specialty batteries still depend on it. People in my line of work have seen how a substance like this, which starts off as a tool for progress, can shift in reputation as science uncovers new facts about danger to humans and the environment.
Physical & Chemical Properties
Cadmium sulfate crystallizes as a white solid with a slight solubility kick that allows easy integration into aqueous solutions. Under the microscope, its crystals may appear inviting, but handling them demands care. With a melting point that sits comfortably for many chemical applications, it melts before it boils, giving off toxic fumes long before vaporization. This compound dissolves readily, dissociating into cadmium and sulfate ions, making it useful as a solution for precise chemical controls. Yet, its very chemistry that draws out its utility also leaves it risky, since those same ions bring heavy-metal toxicity when entering biological pathways.
Technical Specifications & Labeling
Law and regulation drive how cadmium sulfate gets labeled and handled, and the days of vague warning stickers are long gone. Storage happens in tightly sealed containers, out of reach for most, with clearly defined hazard statements. Labels must flag toxicity and environmental risk, including acute and chronic health dangers. Concentrations in products require tight measurement, and every container lists not just the concentration, but hazard pictograms and safety advice in plain sight. Technical data sheets, which I’ve pored over many times, chart out exposure limits, required protective equipment, and disposal steps—minimizing the risk of careless use. The details on a label are not just for regulatory compliance; they serve real human protection, reinforcing lessons that past industrial accidents have made painfully clear.
Preparation Method
Producing cadmium sulfate usually links back to mining and refining zinc ores. Manufacturers obtain cadmium as a minor trace metal, then turn it into a solution of cadmium sulfate by reacting pure metal with sulfuric acid. Industrial setups run this reaction under careful control, keeping emissions and spills contained. Waste streams require strict monitoring, since the byproducts, if uncontrolled, damage water sources and soil. During my time consulting with chemical production facilities, even minor upgrades in filtration and containment made noticeable reductions in off-site risk. The method hasn’t changed much fundamentally, but attention to containment, automation, and worker protection has grown sharply in new plants.
Chemical Reactions & Modifications
Out in the field or back at the lab, cadmium sulfate becomes a starting point for an array of cadmium compounds. Adding various reagents leads to the formation of cadmium carbonate, cadmium oxide, or even cadmium selenide—used in quantum dots and various electronics. Each transformation requires care, not simply for chemical yield but due to persistent risks of heavy-metal exposure. Most chemists will tell you: handling cadmium compounds ranks high among tasks requiring personal protective equipment and fume hoods. These transformations drive research, but they also underscore how tightly cadmium’s value and toxicity are wrapped together. Each step demands not only chemical precision but a safety-first mindset.
Synonyms & Product Names
Across trade, cadmium sulfate appears under a raft of names—cadmium monosulfate, sulfuric acid cadmium salt, even specific nomenclature linked to its hydrated or anhydrous forms. This confusion can trip up even experienced buyers or warehouse workers. In practical terms, the lesson is simple: every shipment and bottle, whatever the name, deserves a double-check on the chemical identity and hazard profile. Forgetting this step has cost companies dearly—both financially and in health incidents—so clear naming and rigorous verification stick as essential habits in industry.
Safety & Operational Standards
Cadmium sulfate regulations run wide and deep, and for good reason. Handling protocols call for everything from gloves and goggles to air monitoring and fume extraction. I’ve spent time in workshops where policies didn’t match practice, and the health outcomes speak for themselves: cadmium builds up in the body and can trigger long-term illness without obvious short-term symptoms. Training and regular audits now stand central to any responsible site working with cadmium compounds. Waste management plays a major role as well, since improper disposal not only breaks the law but threatens local groundwater and, by extension, the community’s health.
Application Area
Industry demand for cadmium sulfate narrowed with new awareness of toxicity. In fact, a lot of companies started phasing it out, but there’s still a place for it. Electroplating remains one key use, offering corrosion-resistant coatings. In batteries, especially certain types of nickel-cadmium cells, this compound still shows up, though alternative chemistries are gaining ground. It sometimes contributes to pigment production and specialty glassmaking, though substitutes are more common now. Academic research taps cadmium sulfate for synthesizing other chemicals, and despite its dangers, some processes still have no perfect replacement for the properties cadmium offers. Each application brings an ongoing risk-benefit calculation, and in my view, the safest path keeps searchlights focused on substitution and engineering controls wherever the compound still finds use.
Research & Development
Chemists and materials scientists keep chasing new ways to replace or neutralize cadmium compounds like cadmium sulfate. Years spent in academic and private labs show the back-and-forth between the hunt for high-performance materials and the push to shed toxic substances. Battery researchers, for example, lean heavily into lithium, sodium, or other chemistries, while alternative coatings try to match the corrosion protection of cadmium without its downsides. Some new developments look at capturing and recycling cadmium out of waste streams, cutting environmental release. For many in the research space, the challenge now is not just finding a substitute that works, but one that brings no new health hazards and can scale at a price industries will pay.
Toxicity Research
Evidence around cadmium sulfate’s health dangers piles up year after year. It enters the human body most often by inhalation or ingestion, and the metal accumulates in kidneys, bones, and other organs. Chronic exposure links to kidney damage, weakened bones, and in worst cases, cancer—the sorts of risks no business can take lightly. Epidemiological studies traced higher disease rates in populations living near heavy cadmium use, especially where environmental controls lagged. Even low-level exposure, repeated over long periods, can add up. Solutions depend on strict process controls, regular health monitoring for workers, and substitution in all possible end uses. Once damage occurs, reversing it proves difficult, so prevention remains the smarter investment by far.
Future Prospects
Looking ahead, demand for cadmium sulfate will likely keep shrinking, churching out only in those corners where no ready replacement works. As greener battery technologies and safer coatings come to market, pressure will only grow for older chemistries to retire from regular use. Policy changes, stricter waste controls, and public awareness all point the needle toward restriction rather than expansion. For those still using cadmium sulfate, transparency about safety, real investment in protective technology, and constant vigilance on environmental release stand as minimum expectations. Innovation can take two directions: some will refine the safe handling of cadmium, others will reroute research into materials that ditch cadmium altogether. That’s where the greatest long-term promise lies: in building a world where industrial progress and human health, rather than conflict, can run side by side.
What is Cadmium Sulfate used for?
What Cadmium Sulfate Brings to Industry
People don’t usually talk about cadmium sulfate at the dinner table, but this compound plays a quiet role in modern life. Folks working in battery manufacturing, electroplating, and some specialty pigment production keep crossing paths with it, even if the general public rarely gives it a thought. I first came across cadmium sulfate during my time in a small plating shop; its sharp warning labels underscored just how much care goes into handling something both useful and risky.
Trusted in Electroplating
Walk into any plant where workers coat steel with thin, high-performance metals, and cadmium sulfate often serves as a go-to ingredient. The electroplating bath owes much of its utility to cadmium sulfate. It helps form a tight, corrosion-resistant layer over metals—something aerospace technicians and marine engineers keep in mind. While steel can rust out from simple rain, a cadmium-plated part can shrug off saltwater or chemicals far longer. The end result is better reliability for airplane landing gear or fasteners that won’t freeze up after a month at sea.
Supporting Battery Technology
Years ago, before the lithium-ion age, nickel-cadmium (NiCd) batteries powered everything from walkie-talkies to emergency exit lights in office buildings. Cadmium sulfate stays relevant in this field. The process that creates the positive plate in these batteries relies heavily on cadmium compounds. NiCd batteries still find a home in power tools, aviation, and backup systems where reliable performance makes more sense than chasing the latest tech fad.
Playing a Role in Pigment Production
Sometimes chemistry paints a brighter world, literally. Cadmium sulfate counts itself among the starting points for yellow-orange cadmium pigments, which end up in ceramics, plastics, and artist-grade paints. These hues stand out for their intensity and long-lasting color, helping artists and manufacturers achieve vibrant results. In my own experience experimenting with glazes, nothing quite replaces the boldness that cadmium brings to ceramics, although it takes strict ventilation and safe cleanup to prevent exposure.
Weighing the Risks and Seeking Solutions
Not everything that cadmium sulfate touches ends up for the better. This compound, like other cadmium salts, carries well-documented health risks. Long-term exposure links to kidney problems and cancer; the same durability that makes it valuable can also make cleanup tricky. Countries enforce restrictions for a good reason: unsafe processes put workers and nearby communities at risk.
A handful of researchers and startups push towards safer alternatives. Zinc-nickel, for example, now finds a place in some plating baths, reducing reliance on cadmium without sacrificing too much in corrosion protection. Battery chemists continue chasing improved nickel-metal hydride and solid-state chemistries. These efforts take time and coordination among industry, regulators, and scientists. For now, those who use cadmium sulfate must blend know-how with serious respect for lab safety and environmental rules.
Striking a Practical Balance
Nobody can ignore the skills and insight built up in workplaces that handle cadmium sulfate. Workers need clear training, strong local ventilation, and ready access to safety equipment. Regular inspection and thoughtful waste handling keep the compound in its useful lane, out of water supplies and people’s homes. From my perspective, progress means blending real-world needs with the best available science, and listening to the voices of both skilled tradespeople and health researchers. Only a practical balance keeps both value and safety in sight.
Is Cadmium Sulfate hazardous to health?
Understanding The Risks From Real-World Places
Walk into a battery plant or a pigment factory, and there is a good chance someone nearby keeps an eye on cadmium sulfate. This substance often pops up in industries that make rechargeable batteries, electroplating solutions, or certain kinds of dyes. Yet, long before the bright colors dry or batteries land on shelves, it’s the people handling cadmium sulfate who run into the biggest dangers. Even outside the plant gates, it’s easy to forget how much these risks can travel—right down to the drinking water or fields growing crops.
How Cadmium Messes With Health
Breathe in cadmium dust, and there is a real shot of it sticking around in the body for decades. Years ago, I sat in a midwestern town watching trucks roll out of a plant. A scientist explained to me how even small leaks or clouds leave a mark. Cadmium sulfate gets inside through the lungs or the gut, then the kidneys bear the brunt. It packs into bones too, sometimes leading to painful bone disease or brittle fractures. Kidney damage builds up slowly, with symptoms hiding out until function drops.
The World Health Organization has warned that long-term exposure can lead to lung cancer and kidney failure. Smoking just adds more cadmium, since tobacco soaks it up from the earth easily. Children living near certain factories have shown higher levels of it in their blood, which links to learning problems and lower bone density. This isn’t a theory. It’s been measured in real neighborhoods.
What Makes Cadmium Sulfate Hard To Dodge
Part of the danger comes from how sly cadmium can be. It doesn’t smell, and the powder rarely looks threatening. Some workers use gloves and masks, but strict rules and oversight often slip. It only takes a tiny amount in the air or tucked in workplace dust for trouble to start stacking up. Many wastewater systems can’t filter it out well.
Crops in places with contaminated soil absorb cadmium, which then sits quietly in common foods like rice, potatoes, and leafy greens. One Japanese region in the 1960s saw entire villages sickened because rice fields soaked up polluted water. Neighbors stumbled around, bones snapping, and kidneys failing, long after the cause seemed invisible. That story pushed governments to clamp down, but regulation is still patchy in parts of the developing world.
Steps That Make A Real Difference
It’s not all doom. Some countries force manufacturers to limit how much cadmium sulfate gets out into the air and water. Workers in these places rely on regular health checks, smart ventilation, and training programs that actually stick. Upgraded filtration at factories makes a big dent in airborne dust.
The wider fix lands with better soil testing and public education. People in farming regions deserve to know if their crops are picking up toxins. Labs can test food samples on a routine basis—think of it like checking for lead in paint. Curiosity from local journalists has exposed gaps in regulations, pressuring officials to patch up old pipes and storage sites. These changes take guts and money, but skipping them never saves anything in the long haul.
Tough Choices, Real Stakes
No one wants to shut down jobs or tank an entire industry. The real work comes from making health costs visible, not tucked away in medical bills or overlooked in far-off villages. When news about cadmium sulfate rolls into the spotlight, it serves as a warning: what settles out of smokestacks or drains from pipes doesn’t just disappear. The best results come from not treating health warnings as background noise but as urgent reasons to act now.
What is the chemical formula of Cadmium Sulfate?
Breaking Down Cadmium Sulfate
Anyone who’s spent a little time in a lab or thumbed through a periodic table likely knows cadmium doesn’t get the best press. It belongs to the group of heavy metals, which often raises a red flag. If you google up cadmium sulfate, you land on its formula: CdSO₄. Simple, but there’s more going on below the surface. The chemical formula tells you that for every one cadmium atom, there’s a sulfate ion carrying four oxygen atoms and a sulfur atom, bonded together. It might look dry on paper, but this compound pops up in all sorts of industries.
What’s at Stake With CdSO₄?
Having handled both research samples and industrial materials, I've noticed that people rarely talk about the nitty-gritty until there’s a spill or a misplaced shipment. Cadmium sulfate gets a real workout in the electroplating business. Folks turn to it for its ability to lay down a tough, shiny metal finish, often on aircraft parts or electronics. You’ll also find it in pigments, and batteries, especially older nickel-cadmium versions. Its applications reflect why knowledge about the chemical formula matters—knowing the ratio of elements isn’t just a dry fact, it helps control reactions, manage waste, and build safe workplaces.
Health, Safety, and Real Risks
Cadmium isn’t something to shrug off. It's well-studied in toxicology circles. Breathing in dust or vapor for too long or letting it slip into water supplies leads to trouble, from lung problems to kidney damage. The formula CdSO₄ helps chemists and safety managers keep tabs on just how much cadmium is floating around. Being specific with formulas becomes crucial when setting up ventilation or choosing protective gear. Organizations like OSHA set limits for cadmium exposure because this stuff sticks around in the body.
What Can Be Done About the Hazards?
From experience, the most direct solution starts with education. People working around cadmium sulfate need hands-on safety training, not just binders full of regulations. Providing clear labeling and up-to-date instructions on handling and disposal keeps everyone sharp. Keeping good ventilation and personal protective equipment on hand isn’t just a checklist item—it really drops exposure. These practical solutions build a safer working environment, whether it’s a massive factory or a high school chemistry lab.
Bigger Picture
On the environmental side, regulations and responsible management matter. Local authorities and international bodies keep a watch on sites that use or dispose of cadmium compounds. Tracking quantities, following disposal laws, and cleaning up after accidents play an important role in protecting communities. Science has handed us facts, and it's up to companies and individuals to roll up their sleeves. Small changes, like using alternatives when possible or recycling batteries, add up over time.
Trust Built on Accuracy
Getting the chemical formula right matters for everyone from the chemistry teacher, to the factory technician, to the policymaker. Mistakes in formulas aren’t just wrong; they can be dangerous. My years in the lab have taught me that accuracy earns trust—among colleagues and regulators. Sharing verified data and leaning on trusted sources ensures smart decisions and keeps everyone a bit safer around potentially risky materials like cadmium sulfate.
How should Cadmium Sulfate be safely stored and handled?
Understanding the Hazards on a Practical Level
Cadmium sulfate demands respect from anyone who works around it. Breathing even a little dust or touching the powder can lead to serious health problems. Science links cadmium exposure to kidney damage, lung issues, and an increased risk of cancer. Lab workers and those in manufacturing see regulations tighten every year as more studies surface. My own time in industrial environments showed me how easy it is to let safety protocol slide—until someone gets sick. Complacency costs health, and sometimes, lives.
Why Proper Storage Makes the Difference
Leaving cadmium sulfate containers on the floor or near a heat source might not seem risky during a busy workday. But improper storage lets the compound absorb moisture, spill, or even react with other materials. I’ve seen storerooms where dust gathered near open drums. Those shortcuts put everyone nearby at risk. Guidance from the Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) stresses secure, dry, and isolated storage for good reason.
Practical Steps: How Workers Handle the Real Risks
Well-labeled containers: Labels with clear warnings in plain language help everyone, from entry-level staff to experienced chemists, know exactly what’s inside. In one facility, language barriers led to a close call—labeling in multiple languages quickly became standard.
Securing a dry and cool environment: Keeping this substance away from humidity, sunlight, and any form of heat stops unwanted reactions. Store it above floor level, on shelves meant for chemicals, without crowding flammable or food-related items nearby. I’ve seen chemical cabinets in schools crowded with snacks and cleaning products—a recipe for trouble if rules go ignored.
Locked cabinets for toxic chemicals: Every workplace stores substances that threaten health. Locks and restricted access ensure only trained hands come near the stockpile. An unlocked door gave a curious visitor access in a lab where I worked; the situation could have spiraled without careful intervention.
Personal Protection Should Never Be an Afterthought
Personal protective equipment blocks most routes of exposure—goggles, gloves, dust masks, or respirators. Even “safe” jobs where exposure seems unlikely benefit from consistent PPE. In my experience, it’s easy for workers to trust luck over safety gear, especially in high-turnover jobs. Supervisors have to remind and train, not just hand out supplies.
Training and Emergency Plans Save Lives
Regular drills and clear procedures set apart responsible workspaces. Quick access to material safety data sheets (MSDS), eye-wash stations, spill kits, and clear air must always exist nearby. A single spill can throw a whole shift into panic if workers don’t know where to find the right gear or whom to call. In my experience, people remember hands-on practice more than a stack of safety manuals.
Long-Term Solutions and Accountability
Trust builds up as employers show regular audits, education, and honest conversations about chemical risks. Trying to “get by” without investing in safety only creates bigger problems. Real leaders inspect storage frequently and invite workers to speak up about unsafe corners. Modern businesses thrive when health isn’t sacrificed for speed. Good storage and handling habits protect lives, careers, and companies from disaster.
What are the physical properties of Cadmium Sulfate?
Understanding the Basics
Cadmium sulfate doesn’t often make headlines, but anyone who’s spent time in a chemistry lab or environmental field knows it comes up in more ways than you’d expect. Recently, cadmium sulfate has caught the eye of regulators and manufacturers because of the risks it presents and its ongoing role in industry. Before talking about solutions or risks, it makes sense to start with some hard facts about its physical qualities.
What Cadmium Sulfate Actually Looks Like
Cadmium sulfate shows up as colorless to white crystals if it sits in a pure form. It appears soft and sometimes powdery to the touch. This salt pulls moisture from the air, a trait called hygroscopicity. Small spills can leave a damp patch because cadmium sulfate isn’t shy about attracting water, which often makes storage a careful job. The compound’s crystalline shape adds to its solubility in water. Over the years, I’ve watched it dissolve rapidly in beakers, which means it can slip into water systems before anyone has time to respond.
Temperature, Melting, and Stability
Solid cadmium sulfate doesn’t melt at room temperature, but turn up the heat and it breaks down instead of melting cleanly. This decomposition means fumes, not just a harmless liquid puddle. Handling cadmium sulfate in heated environments—whether in a factory or during a fire—shifts the concern from powder control to fume inhalation. This is a detail that often goes unnoticed until it’s too late. In the right (or wrong) conditions, those fumes carry real health and environmental consequences.
Solubility—The Double-Edged Sword
Cadmium sulfate dissolves in water with ease. That seems like a small factor for people outside labs, but it puts safety at the forefront for anyone near storage sites. If rainwater hits raw cadmium sulfate, watch the runoff closely—local streams can become contaminated fast. This property also allows its use in electroplating and battery production, where quick dissolution gives it an edge. Despite the benefits, there’s a catch: cleanup after spills isn’t straightforward. A solution spreads and seeps, complicating any remediation effort.
Practical Safety Issues and Solutions
The reality about cadmium sulfate’s properties tells a sharp story about risk. Hygroscopic compounds demand dry, sealed containers, making them costlier to store. Liquid leaks mean faster environmental exposure; even a teaspoon escaping into the wrong spot brings weeks of paperwork and soil testing. I’ve worked cleanups where teams in gloves and respirators spent full days tracking every crystal, knowing that regulatory eyes are keen.
Industry faces two main choices: improve containment or reduce use. Secondary containment—think double-walled tanks or specialized handling rooms—has become standard in places relying on cadmium salts. Tougher labeling and regular inspections make accidental exposure less likely. Many research centers test alternatives to cadmium sulfate for the same reasons—they want less risk and easier disposal. Investing in closed-loop systems, where everything from storage to waste handling takes place in sealed units, cuts accidents to a minimum.
The Bigger Picture
Cadmium sulfate’s physical traits aren’t just tidbits for textbooks—they drive real-world decisions every day. Anyone handling the compound should demand clear labeling, secure storage, and training for spills. These aren’t just bureaucratic hoops. People gain safety, communities avoid pollution, and businesses cut costs linked to accidents. Physical properties shape regulations, set storage costs, and push the search for better materials one step further.
| Names | |
| Preferred IUPAC name | Cadmium sulfate |
| Other names |
Cadmium sulphate Sulfuric acid, cadmium salt Cadmium(II) sulfate |
| Pronunciation | /ˈkæd.mi.əm ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 10124-36-4 |
| Beilstein Reference | 1732854 |
| ChEBI | CHEBI:31379 |
| ChEMBL | CHEMBL1201737 |
| ChemSpider | 54626 |
| DrugBank | DB14544 |
| ECHA InfoCard | 100.233.039 |
| EC Number | 233-331-6 |
| Gmelin Reference | Gm.293 |
| KEGG | C15652 |
| MeSH | D002108 |
| PubChem CID | 24609 |
| RTECS number | GF8575000 |
| UNII | QF9351L90M |
| UN number | UN2570 |
| Properties | |
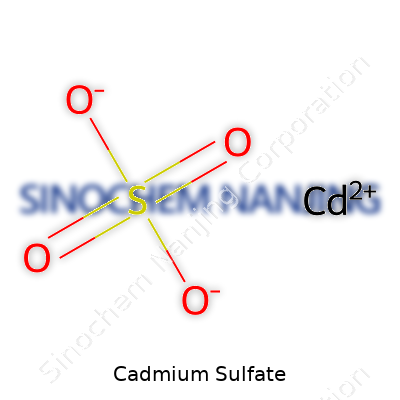
| Chemical formula | CdSO4 |
| Molar mass | 208.47 g/mol |
| Appearance | White crystalline solid |
| Odor | odorless |
| Density | 4.691 g/cm³ |
| Solubility in water | 76.9 g/100 mL (20 °C) |
| log P | -2.18 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.37 |
| Magnetic susceptibility (χ) | −2.7·10⁻⁵ |
| Refractive index (nD) | 1.619 |
| Dipole moment | 0 |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 107.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -828 kJ/mol |
| Pharmacology | |
| ATC code | V03AB57 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; causes damage to organs through prolonged or repeated exposure; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H350: May cause cancer. H360: May damage fertility or the unborn child. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | Contains the following 'Precautionary statements' as a string: "P201, P202, P260, P264, P270, P272, P273, P280, P302+P352, P304+P340, P308+P313, P312, P314, P320, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-0-NA |
| Lethal dose or concentration | LD50 oral rat 2,080 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 138 mg/kg |
| NIOSH | 'TLV: 0.01 mg(Cd)/m3 as TWA; 0.002 mg(Cd)/m3 as respirable fraction; IDLH: 9 mg/m3 (as Cd)' |
| PEL (Permissible) | 0.05 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | 9 mg/m3 |
| Related compounds | |
| Related compounds |
Cadmium chloride Cadmium carbonate Cadmium nitrate Cadmium acetate Cadmium oxide |

