Cadmium Nitrate: From Historical Routes to Modern Applications
Historical Development
Chemists have experimented with cadmium compounds since the early 1800s, after Friedrich Stromeyer discovered cadmium by accident during an analysis of zinc carbonate. Cadmium nitrate soon entered laboratory stockrooms as researchers learned to harness its intense solubility and reactivity. Early industrial chemists took advantage of its use as a source of cadmium ions, especially in analytical chemistry and pigment creation. The push for stronger, more stable pigments for art and textile work highlighted cadmium nitrate’s role as a stepping-stone to brilliant cadmium colors, even before the dangers of cadmium exposure were widely understood. Behind many advances in coordination chemistry, batteries, and materials science, this compound played a quiet but vital role, helping shape the landscape of modern inorganic research and application long before widespread safety awareness took hold.
Product Overview
Cadmium nitrate appears as a colorless, crystalline solid or as granules that dissolve quickly in water. Laboratories usually keep it in tightly closed containers to keep out moisture. Most people working with it notice its slight, metallic acrid smell. This compound serves as a valuable starting material for a range of cadmium-based chemicals. Small-scale pesticide research, electroplating, and niche areas of the dye and photography industries all pull from the same stockroom bottle. Reliable chemical suppliers offer pure, analytic-reagent-grade cadmium nitrate for research, ensuring trace metals fall far below threshold levels so that experiments match expectations and results remain repeatable.
Physical & Chemical Properties
The solid form of cadmium nitrate melts quickly around 50 °C and readily absorbs water from the air, forming hydrates with ease. That deliquescent nature often leaves a damp patch on benchtops if left open too long. Water absorbs the salt fast, yielding a clear, acidic solution, and those solutions corrode most metals except designated compatibility materials like glass, PTFE, or certain high-nickel alloys. Chemists working with it rarely forget its strong oxidizing power, which can set off violent reactions if mixed thoughtlessly with organic substances. Its reliability in releasing cadmium ions makes it versatile and, for those who have to clean up spills, a touch troublesome.
Technical Specifications & Labeling Practices
Cadmium nitrate, usually marked with its chemical formula Cd(NO3)2·4H2O, demands close attention to specification sheets. Analysts pay attention to purity levels, metal contamination, moisture content, and hydrate form. Regulatory standards require labels displaying hazard pictograms for acute toxicity, which appear on every commercial drum and bottle. I’ve seen suppliers include details such as batch, purity (often >99% for research), storage precautions, and recommended handling procedures. Safety Data Sheets and transport regulations all reference the substance’s UN number, GHS codes, and details on accidental release precautions. Transparent specification is non-negotiable for legal compliance and lab safety.
Preparation Method
Preparation starts with dissolving pure cadmium metal or basic oxide in nitric acid at controlled temperatures. That reaction yields a concentrated solution of cadmium nitrate, with brown nitrogen dioxide fumes given off—a reminder to always keep reactions well-ventilated or under fume hoods. Filtration clears out particulate contaminants, and evaporation produces the crystalline product. One can often spot crystals forming as the solution cools. Further drying using desiccators or gentle heat prevents any water retention that might skew measured quantities. Scrap zinc containing cadmium sometimes enters the process in recycling pathways, but purity takes a hit unless further refined.
Chemical Reactions & Modifications
Cadmium nitrate reacts with sulfides to form insoluble cadmium sulfide, a familiar yellow pigment called cadmium yellow. It releases oxygen when heated above 200 °C. In the right hands, this strong oxidizer can drive the synthesis of organocadmium compounds, coordination complexes, or act as a precise cadmium ion donor for analytical chemistry. Researchers passionate about material science continue testing it as a precursor for cadmium oxide thin films and specialty semiconductors. Mixing solutions with common anions (such as chloride or sulfate) quickly yields visible precipitates, which serve as a useful teaching tool in old-fashioned qualitative analysis labs.
Synonyms & Product Names
Cadmium nitrate pops up under varying trade names and chemical synonyms: nitric acid cadmium(2+) salt, cadmium dinitrate, and its hydrate descriptors like cadmium nitrate tetrahydrate or simply “cadmium nitrate, purified.” Its CAS number—10022-68-1—serves as the universal code for procurement, inventory, and hazard reporting. Catalogues tie these names together so end-users don’t order the wrong hydrate, risking bad chemistry or safety violations.
Safety & Operational Standards
Anyone handling cadmium nitrate in a research or manufacturing setting soon learns strict safety habits. The compound ranks as very toxic by inhalation, ingestion, and skin contact, with recognized links to cancer and organ damage. About a decade ago, a colleague with poor ventilation and no gloves developed chronic rashes—an expensive, life-altering lesson. Storage zones use spill trays, locked and vented cabinets, and air sampling when used regularly. Respirators, gloves, and lab coats aren’t enough without robust training and emergency plans. Environmental regulations bar disposal into drains; instead, spent solutions get gathered for hazardous waste handling and licensed incineration or chemical neutralization. Compliance with regulations like OSHA, REACH, and the Globally Harmonized System keeps everyone awake to the ongoing risks.
Application Area
Manufacturers and researchers both reach for cadmium nitrate for different reasons. In pigment production, it bridges the gap from basic cadmium compounds to high-purity yellows and reds favored by artists and industrial dye makers. Electroplaters exploit its solubility, producing tight cadmium coatings for corrosion protection in certain aerospace and marine parts where alternatives fail. By supplying clean cadmium ions, it helps produce advanced battery electrodes. Cats-eye road paint, quantum dot research, and early phase photovoltaic explorations all draw on cadmium nitrate’s chemical reliability. Many applications now fade due to toxicity concerns, spurring innovation elsewhere.
Research & Development
Research into cadmium nitrate covers diverse ground, from refining synthetic routes for better yields to exploring its suitability as a precursor in nanomaterial chemistry. Material scientists use it to grow cadmium chalcogenide crystals or deposit thin metal layers on semiconductor substrates. In analytical chemistry, it aids in titrations, spectroscopic standards, and calibration. Modern instrument calibration often depends on certified reference standards based on cadmium compounds. Calls for substitutes drive inquiry into safer, less toxic reagents. Yet, for certain high-end experimental needs, few replacements deliver the combination of reactivity and reliability.
Toxicity Research
Scientists have thoroughly probed cadmium nitrate’s impact on humans, animals, and the environment. Research shows it accumulates in soft tissues—mainly the liver and kidneys—leading to chronic effects like renal dysfunction, bone fragility, and elevated cancer risk. From the mid-20th century, studies linked industrial exposure to serious occupational illness. Those findings produced modern legal exposure limits of less than 0.01 mg/m³ in air for cadmium compounds, as set by agencies like OSHA. Researchers have shown it enters aquatic ecosystems through improper disposal, persisting in sediments and entering the food chain. The weight of evidence leaves no room for lax handling.
Future Prospects
As awareness grows about heavy metal hazards, researchers and industry shift away from legacy applications. Some see future value in tightly controlled research into quantum materials, green photoconductors, or niche batteries where no substitutes exist. New generations of scientists look for ways to recover, recycle, or replace these compounds for safer chemistry. Upcoming regulatory shifts and breakthroughs in environmentally benign materials will likely limit new cadmium nitrate production to only the most pressing experimental and industrial needs. As technology changes how we work with toxic elements, cadmium nitrate stands as both a warning and a challenge—a relic of scientific progress with a complicated future.
What are the main uses of Cadmium Nitrate?
Understanding the Role of Cadmium Nitrate
Many people might walk past a chemistry lab, see a yellowish powder, and hardly realize the kind of influence the contents inside—a chemical like cadmium nitrate—can have on real-world industries. This compound doesn't sit on shelves just to look pretty. Its reach goes deep because of its unique chemical properties, often sparking debate, caution, and strict handling rules. As someone who’s seen both the excitement of chemical innovation and the fallout from industrial accidents, I can say most conversations about cadmium nitrate should go beyond technical definition and touch on health, ethics, and true need.
Industrial Color and Coating Uses
Cadmium nitrate gets popular in the world of pigments, especially for artists, ceramics, and glassmakers. Its role in making vibrant reds, oranges, and yellows gave rise to signature pottery and glassware—objects many folks may even have sitting on their mantle or dinner table. Cadmium-based pigments handle high heat and sunlight without fading, a quality hard to match with safer alternatives. Manufacturers also reach for cadmium nitrate when making specialty paints for aircraft or machinery that need lasting color under tough conditions.
Electronics Manufacturing
In tech factories, cadmium nitrate helps in forming cadmium oxide films, which show up in electronic sensors, thin-film resistors, and a handful of battery types. Electronic devices depend on predictable performance from their materials, and cadmium-based compounds keep up under demanding environments. In my own experience tinkering with old electronics, I learned the hard way about the fine line between improved device lifespans and the hazards these compounds can bring. Many engineers love the reliability, but some now push for safer upgrades due to health concerns.
Research and Analytical Chemistry
Labs pull cadmium nitrate off shelves for chemical analysis, educational demonstrations, and synthesis of other cadmium-based salts. Students and chemists use it in controlled reactions, catalysts, or when they need a reliable source of cadmium ions for experiments. This chemical sees plenty of action in research, feeding into development of new materials and observation of reaction pathways. Yet lab safety takes center stage, as accidental spills or improper disposal create headaches for staff and the environment alike.
Risks and Environmental Impact
The drawbacks with cadmium nitrate come clear the moment you talk to anyone in environmental science or public health. Its toxic nature means even small spills, improper storage, or careless disposal threaten soil, water, and all living things in the area. In communities near factories, cadmium compounds have caused serious health scares, leading to tougher regulations around handling, transportation, and waste management. I’ve listened to stories from affected residents—their trust in local industry, sometimes gone for good because of poor safeguards.
Can We Replace It?
Switching away from cadmium nitrate isn't always easy. Artists still crave the true color that synthetic pigments can’t usually give. Electronics makers count on its durability. Even so, scientists and engineers search for greener substitutes and invent new procedures to minimize risks. Training workers, using closed systems in factories, and better education in labs all give odds for safer use while we keep looking for replacements that actually meet performance needs. Stronger rules, full disclosure, and concern for the people handling these chemicals—those matter as much as any technical advance.
Is Cadmium Nitrate hazardous to health?
What’s in the Air and on Your Skin
Most folks have never heard much about cadmium nitrate, but this chemical doesn’t fade into the background when it comes into close contact. Used in labs, dye-making, and even in fireworks, cadmium nitrate grabs attention with its bold red color. The concern begins the moment it turns to dust or dissolves in water. When breathed in, those tiny particles travel straight to the lungs, and the story isn't promising from there. Coughing, shortness of breath, and chest pain are just the beginning, according to worker safety documents from OSHA. Nose and throat irritation often show up quickly.
If the chemical touches bare skin or gets splashed into eyes, burning and redness can arrive fast. In some cases, blisters and ulceration follow. I've worked across a few old manufacturing sites, and the advice always sounded the same: handle cadmium compounds with gloves, goggles, and serious respect.
Piling Up Inside the Body
Swallowing cadmium nitrate by accident or exposure through poorly washed hands hits the digestive system. Nausea, vomiting, and stomach cramps are all reported symptoms, and none of them are minor. What most people don’t realize is cadmium stays in the body, not just in the moment but for years. Over time, kidneys and livers build up the metal, and that leads to long-term damage. Chronic exposure cuts down kidney function and increases the risk of high blood pressure and fragile bones.
The World Health Organization pulled together decades of studies and found workers most exposed showed higher rates of kidney damage and certain cancers. Evidence links cadmium compounds to lung and prostate cancers after years of breathing in the dust. I’ve looked at old data from battery plants in the Midwest, and the trend is tough to ignore—higher illness rates track with jobs handling cadmium.
Everyday Exposures and What Can Be Done
Most people won’t bring pure cadmium nitrate into their homes. The story grows trickier in places with pollution from factories or recycling plants. In those areas, dust and water can carry trace amounts outside plant gates, building up in the soil or water. Even home gardeners risk exposure if their dirt picks up industrial residues. Washing hands, cleaning produce, and using gloves become vital habits—not old wives’ tales, but practical steps from environmental health experts.
I’ve met workers who wore masks and gloves every day on the job and still had concerns. The real key is personal protective equipment backed up by regular air and surface monitoring. Factories that take shortcuts put employees at risk faster than any warning sign can fix. Federal rules demand sealed containers, fume hoods, and medical screening for good reason.
Lessons from the Field
Decades of industrial progress have taught a tough lesson: once cadmium nitrate leaves the lab, cleaning it up takes more than mops and soap. Training, monitoring air quality, and building a safety culture work far better than just posting warnings. For workers and neighbors, knowledge pays dividends. Staying informed—reading safety sheets, not ignoring weird symptoms, and speaking up if practices seem sloppy—makes the biggest difference.
Nobody wins by downplaying cadmium hazards. Following the facts, listening to experienced voices, and respecting both science and common sense keeps communities safer and workplaces healthier.
What is the proper storage method for Cadmium Nitrate?
Seeing Cadmium Nitrate for What It Is
Cadmium nitrate isn’t some everyday chemical you keep under the sink. Its bright red crystals grab your attention and so do its hazards. This stuff gives off toxic fumes if it heats up and it sneaks into your body through skin or air. The danger is real—breathing cadmium compounds links to kidney damage, bone problems, and cancer. Full personal protective gear makes sense for even quick handling. From my days working alongside science teachers, a bottle of cadmium nitrate created expected tension: nobody skipped the paperwork, nobody took shortcuts on labeling or double-checking containers.
Choosing the Right Place: Away From the Busy Corners
A smart science department stores cadmium nitrate in a reinforced cabinet, locked and reserved just for hazardous inorganics, nowhere near acids or combustibles. Not every lab respects these boundaries, so you might spot a bottle in a general chemical closet or shoved next to a solvent. That’s an accident waiting to happen. In practice, I’ve seen seasoned lab managers separate cadmium nitrate from flammables on its own leak-proof tray—because spills are more common than people admit, and nobody forgets the one time a loose lid ruined everyone’s afternoon.
Why Moisture and Light Matter
Factories and labs lose product and health when chemicals meet humidity. Cadmium nitrate draws in moisture, clumps up, and eventually breaks down. Leaving a jar open gives decay a head start and can cause leaks or corrosion that sneak up slowly. Keeping the container sealed, using only original packaging or tightly-capped chemical glass, blocks the air and saves both the chemical and the surfaces it touches from sticky, caustic residue. No one enjoys chiseling caked nitrate off a shelf during a surprise safety inspection. Good lighting helps you spot problems fast, but sunlight encourages chemical decay, so storage goes in dark or opaque cabinets.
Clear Labels: Accidents Disappear With Good Habits
Labels do half the work. A hand-written tag easily fades or rubs off under real conditions, so professional, water-resistant labels matter. That way, years later, nobody plays guessing games with old powder. Labeling with the date, full chemical name, pictograms, and supplier gives anyone walking in a fighting chance to do things right.
Ventilation and Emergency Preparedness
Cadmium nitrate doesn’t give off clouds unless it burns, but small leaks and broken bottles can still lead to exposure. Decent ventilation, either through an exhaust fan or even a fume hood when handling larger amounts, keeps the room safe. In all the schools and small labs where I’ve taught, eye-wash stations and spill kits nearby gave us backup when mistakes happened—which they always do. Supervisors who actually run drills, rather than just posting charts, build a culture that never freezes during a spill or unexpected reaction.
Disposal and Inventory Check
Nothing lasts forever. Old cadmium nitrate loses purity, and forgotten jars crowd storage space. Managing a sharp inventory system flags expired material and keeps total supply down. Any waste or expired cadmium compound goes straight through certified hazardous waste disposal; pouring it down the drain is both illegal and dangerous, something I’ve heard whispered but never seen anyone smart try.
Solutions That Actually Stick
Smart storage isn’t just an item on a checklist. The best labs run regular training and inspections, not as punishment, but as a normal part of working together. People remember lessons from real problems, so sharing past near-misses keeps stories alive and standards high. It all comes back to respect: for the chemical, for the room, for yourself and your team. That’s the path to a safe workspace—one habit at a time.
How should Cadmium Nitrate be disposed of safely?
Recognizing the Danger
Cadmium nitrate sits on the list of chemicals you don’t want floating around in the trash or seeping into water supplies. Beyond its official safety sheets, stories out of labs and manufacturing plants all point in the same direction—this compound brings real danger to health and the environment. It causes kidney and lung problems after even low-level exposure, and it doesn’t break down easily. Public health agencies like the EPA and OSHA warn against mishandling these kinds of chemicals for good reason.
Especially when working in a lab or a factory, even small spills lead to cleanup headaches and worry among staff. This isn’t a substance anyone wants to encounter outside carefully controlled conditions. I’ve seen entire storerooms locked down for a single broken ampoule, and the stress that comes with tracking potential exposure can’t be overstated.
Practical Steps to Keep Things Safe
Companies, schools, and researchers all have a shared stake in making sure no cadmium compound ends up in regular waste streams. Federal and state rules classify cadmium salts as hazardous waste. You can’t pour a solution of it down the drain or toss a container in the dumpster. Fines for improper disposal sting, but the real impact comes if someone gets hurt—either right away or after long-term contamination builds up.
Every place with a chemical waste policy should spell out storage and disposal routes for cadmium nitrate. Leakproof, labeled containers go a long way in stopping accidents before they happen. Secure cabinets, away from acids and incompatible substances, shield people from accidental mixing and potential releases. Equipment and surfaces that touch this compound don’t just need a wipe-down—thorough cleaning and routine monitoring catch lingering residues before they spread further.
Trusted Waste Handlers Lead the Way
Licensed hazardous waste firms handle the heavy lifting. These folks have the gear, training, and paperwork needed for moving and treating cadmium salts. Their trucks have strong seals and double-checks. They take chemicals to fully engineered disposal sites, incinerators, or treatment plants that capture heavy metals instead of letting them reach landfills or waterways. EPA Resource Conservation and Recovery Act (RCRA) guidelines cover every handoff, right down to manifest and receipt procedures.
I remember trying to save money by waiting until the end of a project to call in a chemical waste pickup. That kind of logic quickly falls apart when storerooms fill up or when safety officers spot expired or mismarked containers. Regular pickups keep inventories down and cut risk for everyone involved. Besides, local environmental authorities often have tips on reputable disposal vendors and programs, especially for smaller labs or teaching facilities.
Better Habits for the Long Run
Replacing cadmium salts with safer alternatives stands as a wise move wherever possible. Green chemistry isn’t just a buzzword—schools and forward-thinking manufacturers now favor less toxic materials to keep staff and students out of harm’s way. Transparent safety training goes beyond just ticking boxes. Workers and students remember drills that actually walk through spill response or mock pickups. Clear protocols breed confidence and cut panic—something I’ve learned matters most in high-stress situations.
Cadmium nitrate’s risks don’t just hit one lab or factory. They affect whole communities if mismanaged. Adopting a careful, methodical approach to chemical disposal speaks volumes about a group’s commitment to both health and environmental protection.
What is the chemical formula and appearance of Cadmium Nitrate?
The Basics: Chemical Formula
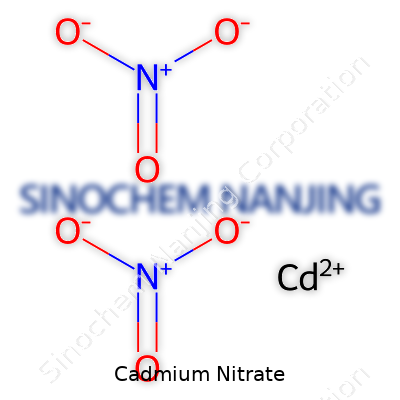
Cadmium nitrate has a straightforward chemical formula: Cd(NO3)2. You’ll spot cadmium on the periodic table with the symbol Cd. It teams up with two nitrate ions, each holding the formula NO3-. The structure reflects what’s going on at the molecular level: one cadmium ion, two nitrate groups. It’s not exotic, but, as anyone who’s spent time in a lab knows, the impact and risk ride beyond its simplicity.
What Cadmium Nitrate Looks Like
Most researchers and industrial users have seen cadmium nitrate in the form of small, colorless crystals. The solid chunks catch the light, looking almost like table salt or sugar, but nobody should get confused about its purpose or safety. The substance dissolves easily in water, forming a clear solution. This solubility helps in various applications such as pigment manufacturing, dye fixatives, and even in some photography processes.
Handling this material puts you in direct contact with an industrial backbone. I remember working in a research facility where chemicals like this required locked storage and training just to check out a tiny sample. This tells you all you need about the seriousness surrounding cadmium nitrate. The crystals start off colorless, but they have a sharp, slightly acrid note, the kind of chemical scent you expect in a well-equipped lab.
Health and Safety: Not Just a Footnote
Although cadmium nitrate looks harmless at first glance, experience and history underline its dangers. Cadmium compounds rank among the more toxic substances found in laboratories. Touching or inhaling them can damage kidneys, bones, and lungs. The International Agency for Research on Cancer (IARC) classifies cadmium compounds as carcinogenic to humans. Laboratory staff and factory workers who spend their days near these materials face real health risks if safety slips. The law rightfully restricts its handling, for good reason.
Years back, an accident at a metal plating company in my city made the local news. A small spill, ignored in a dark corner of the plant, lead to long-term exposure for cleaners who didn’t receive proper training or protection. The company ended up paying heavy fines, but it’s hard to put a value on health lost through chemical exposure. Safety training and strong oversight on chemical storage serve as the best shields for people on the front lines. That’s where companies excel or fail—by what they do to protect real people, not just balance sheets.
Addressing the Hazards
Reducing risks linked to cadmium nitrate involves serious buy-in from everyone working with or around it. Wearing gloves, goggles, and protective clothing doesn’t stand as an empty rule; it saves lives. Fume hoods keep vapors out of lungs, and chemical-specific storage cabinets stop leaks and cross-contamination. In most teaching labs, cadmium compounds get replaced by safer substitutes whenever possible. More companies now test for air and surface contamination, investing in detection kits and employee training programs. Green chemistry initiatives keep pushing for alternatives, which points the way forward.
Chemicals like cadmium nitrate don’t shape the news often, but their quiet influence in industry and research continues. Knowing their formulas and how they look is just the first step—the challenge, and the responsibility, lies in handling them with the care they demand.
| Names | |
| Preferred IUPAC name | Cadmium dinitrate |
| Other names |
Nitric acid, cadmium(2+) salt Cadmium dinitrate Cadmium(II) nitrate Cadmium nitrate tetrahydrate |
| Pronunciation | /ˈkæd.mi.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10325-94-7 |
| Beilstein Reference | 1207930 |
| ChEBI | CHEBI:32541 |
| ChEMBL | CHEMBL3422380 |
| ChemSpider | 14637927 |
| DrugBank | DB14544 |
| ECHA InfoCard | 100.028.761 |
| EC Number | 233-710-6 |
| Gmelin Reference | Gm. 793 |
| KEGG | C00449 |
| MeSH | D002165 |
| PubChem CID | 10156 |
| RTECS number | AF8375000 |
| UNII | SWQ31855XK |
| UN number | UN2570 |
| Properties | |
| Chemical formula | Cd(NO3)2 |
| Molar mass | 236.42 g/mol |
| Appearance | Colorless crystals |
| Odor | Odorless |
| Density | D = 2.45 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.59 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | +45.0e-6 cm³/mol |
| Refractive index (nD) | 2.45 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 231.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -467.1 kJ/mol |
| Pharmacology | |
| ATC code | V03AB58 |
| Hazards | |
| Main hazards | Oxidizer, toxic if swallowed, toxic if inhaled, may cause cancer, causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | **"Danger; H301, H410, H350, H330, H341, H372, H315, H319, P261, P273, P280, P301+P310, P305+P351+P338, P501, GHS06, GHS08, GHS09"** |
| Pictograms | GHS03,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331, H350, H410 |
| Precautionary statements | P210, P220, P221, P280, P302+P352, P305+P351+P338, P308+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 2-2-3-OX |
| Autoignition temperature | 250°C (482°F) |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | TLV: 0.01 mg(Cd)/m3 as TWA; 0.002 mg(Cd)/m3 as STEL; IDLH: 9 mg/m3 |
| PEL (Permissible) | 0.05 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | 9 mg/m3 |
| Related compounds | |
| Related compounds |
Cadmium chloride Cadmium sulfate Cadmium acetate Cadmium oxide |

