Cadmium Fluoroborate: Tracing the Arc from Discovery to Future Challenges
Historical Development
Cadmium fluoroborate didn’t arrive on the scene with much fanfare, but its journey through the chemical world offers up a study in how nuanced compounds can come to play a big role in technological progress. The early days of borate chemistry, going back to the nineteenth century, mostly circled around research labs experimenting with the properties of boron’s quirky tendency to bond in unusual ways. Cadmium, on the other hand, has its own complicated reputation—celebrated in pigments and coatings, scrutinized for its toxicity, and sometimes flat-out banned in the wrong places. By mid-twentieth century, as the drive for more specialized electrochemical processes picked up, chemists started combining cadmium salts with borofluoride complexes to chase stability and new functionality. These were years shaped by trial and error, documentation painstakingly assembled in research journals and patents. In the end, what started as curiosity in the lab became a niche workhorse in several industries, with real staying power thanks to the properties that this unlikely pairing delivered.
Product Overview
Cadmium fluoroborate is a salt, usually encountered in the form of a white to colorless crystalline substance, sometimes as a solution. It’s not the sort of compound one would stumble across in everyday life, but its uses punch well above its weight. The material tends to find a place in specialized electroplating processes, electrochemical surface treatment, and occasionally in analytical chemistry settings. It may sound esoteric, but the story of this compound has as much to do with legacy industrial applications as it does with cutting-edge battery research.
Physical & Chemical Properties
Looking at its chemical structure, Cd(BF4)2 teams up a cadmium ion with the tetrafluoroborate anion. The chemical stability of borate structures, especially under conditions where you want low reactivity with water and decent ionic conductivity, gives this material its edge in the right application. Solubility sits high on the list of priorities for users in solution-based processes, and this compound complies, dissolving readily in water, generating a clear, nearly neutral solution. Its crystalline nature, coupled with high solubility and ionic mobility, helps it stand out, especially when reviewing candidate compounds for electroplating baths or other similar technology. One has to give thought to cadmium’s toxicity as well—this isn’t a substance to treat lightly or ignore in the workplace or the environment.
Technical Specifications & Labeling
The standards for production and transportation are strict, for good reason. Regulatory agencies in North America, Europe, and Asia pay close attention to purity levels and impurity limits, keeping a close watch on any material containing cadmium. Labelling has to be crystal clear, noting the risks and the content to avoid accidental mishandling. Specifications covering minimum purity percentages, moisture content, and the absence of contaminants such as lead, arsenic, or unreacted starting materials often shape quality checks at both the supplier and consumer end. The importance of rigorous labeling and specification reporting climbs even higher with shipping, as regulations push for unambiguous documentation during transport and storage, hoping to reduce the risk of accidental exposure.
Preparation Method
The synthesis of cadmium fluoroborate involves reacting cadmium oxide or carbonate, which acts as a source for the metal ion, with fluoroboric acid or its salts. The reaction gives a relatively straightforward path—not much heat or exotic equipment required, but a keen hand since both cadmium compounds and borofluoric acid demand proper ventilation, fume extraction, and chemical safeguards. Filtering and crystallization steps allow the compound to be dried and then stored under conditions preventing moisture pick-up or accidental human contact. The overall process requires adherence to both chemical safety and responsible waste handling, due to the challenges of cadmium disposal.
Chemical Reactions & Modifications
What sets cadmium fluoroborate apart is its ability to fit within a narrow slice of the chemical world—stable enough to survive mild acids and bases, yet reactive enough to function in electrochemical cells and plating solutions. Mixing it with various solvents or additives changes the deposition characteristics on metal substrates—a handy trick for engineers customizing surface finishes. Adjustments to the solution chemistry, whether through the addition of complexants or secondary salts, bring about tweaks to deposit density, smoothness, or even metallic luster. Researchers still look for improvements, aiming for enhanced operational flexibility without compromising on safety or environmental standards.
Synonyms & Product Names
A handful of names follow this compound into the marketplace and research literature. The simplest reference, cadmium tetrafluoroborate, appears in western journals, while several Asian texts use the term cadmium fluoroborate without modifying the anion. Chemists sometimes shorten it to Cd(BF4)2 in their notes. Sellers might use their own proprietary brand names, but for regulatory filings, the IUPAC-derived terminology tends to prevail.
Safety & Operational Standards
Cadmium comes loaded with challenges—you don’t work with it casually or skip the respirator. Chronic exposure links to kidney damage, bone weakening, and a real risk of cancer; this reality shapes every operational protocol. Standard guidelines require proper containment, full labeling, and frequent air monitoring wherever cadmium salts are handled. Waste streams contaminated with cadmium fluoroborate face close scrutiny, often heading for controlled treatment and disposal, never general sewer systems. Workers’ training, regular medical screening, and up-to-date material safety data inform the daily routine for anyone in a plant or lab using this compound.
Application Area
The scene where cadmium fluoroborate earns its place most clearly is in electroplating—particularly where decorative, corrosion-resistant, or electrically conductive layers get laid down on metals. Automotive parts, aerospace fittings, and select components in electronics all lean on the reliable, bright surface that electrodeposited cadmium leaves behind. In some places, fluoroborate-based solutions help replace older cyanide-based systems, which carry their own batch of hazards. The operational characteristics—quick dissolution, readiness to transfer ions, predictable behavior in the plating bath—keep it in service. Over time, new research continues to push for alternatives that can offer the benefits of cadmium electroplating without its toxicity baggage.
Research & Development
Plenty of recent R&D still focuses on making electrodeposition cleaner, less wasteful, or more adaptable to high-tech requirements. Technologists working in battery research have also noticed cadmium fluoroborate—though nickel-cadmium batteries dominate headlines, researchers investigate whether modifications in salt composition can give better cycle life or enhanced safety margins. Advanced surface engineering looks for ways to lessen environmental impact, exploring chelating agents or bath additives that trap free cadmium ions and prevent their escape into the workplace or discharge streams. Some academic labs try to synthesize analogs with less toxic metals, but getting the same deposition quality remains an unsolved puzzle for now.
Toxicity Research
The elephant in the room always stomps back to toxicity. Studies on cadmium compounds keep finding the same message: accumulation in the body does long-term damage. Datasets from manufacturing workers over decades show that lower exposure thresholds don’t fully eliminate risk; kidney function, lung health, and carcinogenic pathways remain compromised even at low levels. Animal models only reinforce these findings. Part of the burden now lies in biomonitoring populations potentially exposed to cadmium in the workplace and keeping environmental release close to zero. The need for real-time air sampling and rapid blood/urine screening isn’t just a regulatory box to check, but a real safeguard.
Future Prospects
Cadmium fluoroborate finds itself at an inflection point. Demand from sectors unwilling to give up on its deposition strengths still exists, but political winds and regulatory heat keep blowing, with strict bans or phaseouts looming in many jurisdictions. Alternatives—ranging from zinc or tin-based plating solutions to fully non-metal protective films—continue to advance, sometimes capturing market share but often struggling to match the all-around performance of cadmium-based baths. The challenge for future chemists lies in finding ways to maintain industrial capabilities without compromising worker health or public safety. As greener technologies develop, there’s potential for a gradual shift away from cadmium in all but the most application-critical spaces, but getting there takes sustained research, real investment in alternative methods, and stronger global coordination. At stake is not just business continuity, but the health of communities and ecosystems exposed to the stubborn legacy of toxic metals. The arc of cadmium fluoroborate, from obscure salt to keystone input and maybe back to obsolescence, deserves careful attention as industry and science weigh the costs and benefits of keeping it around.
What is Cadmium Fluoroborate used for?
The Workhorse in Electroplating
Growing up around machine shops, I sometimes spotted bottles labeled with challenging names. Cadmium fluoroborate always seemed like something for chemists in lab coats, but it plays an overlooked role in our daily lives. In plating shops, it's used for one thing above all: making tough, corrosion-resistant cadmium coatings. Parts like bolts, switchgear, and aviation connectors often owe their durability to coatings made with the help of cadmium fluoroborate baths.
Factories choose cadmium-based coatings to stop rust in extreme spots—out at sea, inside aircraft, around chemical plants. Not every metal holds up against salt or industrial smoke as well as cadmium does. Electroplating baths built with cadmium fluoroborate become a key ingredient because they let workers apply thin layers without sacrificing smoothness or adhesion. The stuff works well in pulse-plating: short bursts of current create sleek deposits without pitting or rough patches. It saves on wasted material because coatings come out just the way engineers specify.
Trouble in the Toolbox
Even with benefits, cadmium fluoroborate brings baggage. Breathing in cadmium dust, or letting it seep through the skin, can hurt lungs or kidneys. The U.S. Environmental Protection Agency flagged cadmium compounds as known human carcinogens. Plating shops that ignore these dangers endanger working families and may end up in fierce legal fights. Avoiding leaks and spills calls for rigid safety rules, as well as careful training for anyone handling the chemical.
On the disposal front, cadmium never truly goes away. Waste left behind can slip into soil, water, and the food chain. Regulators across the globe keep tightening the screws, demanding lower exposure limits and stricter waste handling. A few years back, Europe’s REACH rules pushed automakers and electronics companies to cut back on cadmium for consumer-facing goods. Old batteries and e-waste still confuse recyclers about what’s safe to salvage.
Finding an Alternative Isn’t Simple
Some engineers have switched to zinc-nickel or tin coatings, but cadmium still handles brutal conditions better than most. Helicopter rotors, landing gear, and hardware used in deep-sea drilling stick with cadmium-backed solutions. Researchers keep hunting for replacements that don’t give up performance or cost too much, but big leaps have proved slow. For many, it’s not about ditching cadmium entirely; it’s about using it with maximum care.
Safer Practices Bring Real Rewards
I’ve seen small shops follow best practices—using sealed tanks, good ventilation, and thorough training—to lower risks. Digital monitoring and tighter recordkeeping started as big-investment items but made it easier to spot leaks before trouble grows. By re-using rinse water and recycling metal-rich sludges, some operations cut hazardous waste bills and cleaner technology wins customer trust. Whenever big industry players share successful techniques, everyone along the supply chain stands to gain.
In the end, cadmium fluoroborate isn’t going anywhere just yet. As long as metal parts take a beating from the weather or chemicals, it will keep its place as a hard-working—if controversial—tool. Care, innovation, and strict safety measures remain the best ways forward.
Is Cadmium Fluoroborate hazardous or toxic?
What Makes Cadmium Fluoroborate Worth Discussing?
Cadmium fluoroborate is not used in most homes, but folks in industries like electroplating or battery manufacturing might spot it on the shelf. Its catchy name doesn’t reveal much, but the moment cadmium gets mentioned, alarms start ringing. My background in chemical safety pushes me to dig deeper—some chemicals, while useful in small circles, come packaged with a heavy set of risks.
The Real Dangers Behind the Name
Science does not play around with cadmium. Medical literature and workplace safety guides treat it as a major health hazard. The main trouble comes from cadmium’s knack for staying in human organs once it gets inside. The World Health Organization lists cadmium as a human carcinogen. Reports from the National Institute for Occupational Safety and Health (NIOSH) echo this, laying out dangers from inhaling dust or fumes during usage. That means people handling cadmium fluoroborate can face bigger health threats than they might expect from a warehouse chemical.
Toxic exposures do not always happen in obvious ways. A lab coat and gloves can only help so much if folks get careless about air filtration, spills, or skin contact. Workers exposed to cadmium compounds may face symptoms that pop up later: kidney trouble, weak bones, and sometimes even lung cancer. For someone who worked around chemical warehouses, those health issues are not just words on a safety manual—they’re stories from people forced to leave the industry early.
Why Regulations Exist—And Sometimes Fall Short
Regulations exist for a reason. The Environmental Protection Agency strictly limits cadmium emissions, and the Occupational Safety and Health Administration (OSHA) sets rules for workplace exposure. Still, mistakes happen. In my time on-site, I’ve seen small spills overlooked, short cuts taken to save time, and waste disposal handled without enough planning. Even smart, trained people get burned out after long shifts, so it’s easy for standards to slip.
Disposal can turn murky, too. Flushing cadmium-containing waste into regular drains causes bigger headaches for communities. Cadmium builds up in soil and water, so even low-level, repeated leaks can affect crops and wildlife, not just workers in plants or labs.
Building a Safer Work Culture
Pushing safety training isn’t just about following rules—it’s about protecting lives. Regular air quality checks, good exhaust systems, and strict enforcement of storage rules keep people from learning safety lessons the hard way. Engineers and lab managers who invest in better ventilation and personal protective equipment protect not only their teams but everyone living near their plants.
Choosing substitutes can help, too. Engineers can look for less toxic materials for industries that still use cadmium fluoroborate. If switching isn’t possible, extra steps—like automating risky tasks, mandating frequent health screenings, and limiting time spent handling raw chemicals—can bring down risks. My experience hints that solutions that work well for one factory or lab do not always fit everywhere, but being willing to adapt means fewer people get sick.
Final Thoughts on Responsibility
Cadmium fluoroborate remains hazardous. Those in the industry carry a responsibility that stretches far beyond factory walls. Putting safety first, respecting regulations, and watching for better alternatives keep workers healthy and the wider community safe from invisible risks. My years of talking with those in the trenches taught me that raising awareness is not just smart, it saves lives—and that’s worth the effort every time.
What is the chemical formula of Cadmium Fluoroborate?
Understanding Cadmium Fluoroborate
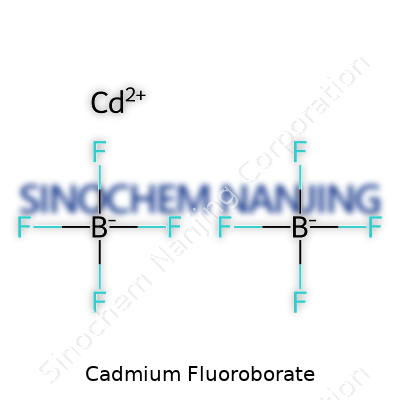
Chemical compounds don’t make headlines, but their impact quietly traces the backbone of countless processes we rely on every day. Cadmium fluoroborate carries its own unique profile in the chemistry world, holding the formula Cd(BF4)2. This isn't just a jumble of letters and numbers. It’s a combination that helps shape electroplating, battery development, and laboratory research.
Factoring In the Elements
Breaking it down, the formula includes cadmium, a heavy metal known for both its usefulness and toxicity, and the tetrafluoroborate ion, which counts among the handy anions in modern synthetic chemistry. The combination means you get a salt that dissolves well in water and introduces specific ionic properties: something I’ve run across often in electrochemical setups. While working in a university lab on metal finishing processes, I saw this compound's ability to deliver cadmium ions efficiently—something other salts couldn't match in the same way.
The Importance of Chemical Accuracy
Getting the formula right doesn’t serve just as a test question in a classroom. In research labs and manufacturing plants, accuracy in chemistry spells the difference between a process running smoothly or going dangerously off track. Errors with compounds like cadmium fluoroborate can lead to workplace exposure, environmental discharge, or outright product failure. Cadmium-based salts aren’t forgiving: mistakes bring consequences, as I’ve seen during glovebox incidents where a careless error in chemical calculation led to a frustrating cleanup and a stern review from the safety officer.
Risk, Responsibility, and Ongoing Choice
Cadmium’s natural toxicity makes the proper use and disposal of its salts a matter of real concern. The World Health Organization and the Environmental Protection Agency have highlighted this risk for decades. The regulatory landscape reflects the need to balance utility with health: laboratories and plating shops dealing with cadmium fluoroborate must keep waste streams contained and surfaces clean. Wastewater streams from such facilities are tightly monitored, not just by law but by the understanding that even trace amounts can harm aquatic and human life.
Looking for Safer Alternatives
There’s a push within the industry and the academic world to find substitutes that match the performance of cadmium fluoroborate without the baggage. Innovations in functional plating have pointed toward less hazardous metal salts and organic conductors. I spent months working on alternative electrolytes in grad school, testing compounds like zinc- and tin-based salts for similar applications. Each came with its own headaches—lower efficiency, higher impurity risks—but the drive to reduce the toxic load kept us tackling those challenges. Progress might be slow, but the direction serves a clear purpose.
The Path Forward
Cadmium fluoroborate’s formula is a compact symbol for all the choices we keep making in science and technology. Understanding what goes into a substance, how we use it, and what happens when we’re done with it isn’t just about memorizing facts for an exam. Every detail matters—safety, performance, and responsibility all rely on accuracy. Future scientists, engineers, and policymakers will need not just the right formula, but the discipline to ask: is this the best choice for the job at hand?
How should Cadmium Fluoroborate be stored?
Why Safe Storage Isn’t Just a Checklist
Walk into any lab supply room, and you can spot what sets careful chemists apart from the rest. Lined along labeled shelves, bottles with names like cadmium fluoroborate tell a story of both scientific curiosity and responsibility. This stuff plays a big role in industries—especially electroplating—where a slip-up could cost someone their health or job. It’s not enough to just put chemicals in a cabinet and lock the door. Storing cadmium fluoroborate calls for real attention to detail and a respect for its hazards.
The Risks Make the Rules
Cadmium compounds have a bad reputation for good reason. Breathing in dust or coming into contact with these chemicals leads to kidney, lung, and bone damage, and even increases cancer risk. Inhaling or ingesting fluoroborates throws in potential for fluorine-based toxicity on top. These facts aren’t just points from a data sheet—they’re the truth for anyone who handles this salt in a job or research setting.
Solid Containers, Smart Placement
I’ve seen too many labs where space runs out and bottles end up wherever they fit. For cadmium fluoroborate, glass or high-density polyethylene containers with tight seals do the job because they block air and moisture, both of which lead to product breakdown and toxic gas release. Shelves for these chemicals stay far from acids, foods, and heat sources. Everyone thinks “separate from what?” The answer—acids, bases, anything with water. This rule isn’t about some esoteric chemical knowledge; it’s about real-world reactions that hurt people.
Climate Control Isn’t Just for Comfort
Humidity and temperature changes ruin chemical stock. At one point in my life, I checked a shared university lab fridge and found a sticky mess from improperly capped cadmium salts. Once moisture hits this compound, you’re not just seeing shelf-life reduced—hydrolysis can release hydrofluoric acid gas, which burns skin, damages lungs, and corrodes metal. Storage rooms stay dry, below 25°C, and airflow stays steady to keep unexpected vapors from pooling. That’s a lesson nobody wants to learn the hard way.
Forget Out-of-Sight, Out-of-Mind
Keeping dangerous chemicals hidden doesn’t make them safe. Every bottle needs a clear label with chemical name, hazard symbols, and date received. Expiry dates aren’t just for compliance—they stop accidental use of degraded, unstable product. MSDS sheets don’t belong in a binder across the hall; they should hang right next to the shelf, ready for anyone, including the firefighter or EMT who races in during an emergency.
Routine Checks, Not One-Offs
Weekly inspections saved my old team from serious headaches. We checked seals, labels, any signs of leaks, and made sure nothing got shoved into the wrong cabinet during a rushed cleanup. If you spot crust around the lid or discoloration, don’t think twice—quarantine and call in hazardous waste professionals. Don’t cut corners on personal protective equipment either. Gloves, eye protection, and lab coats stay on, even for a “quick look.”
Training Goes Further Than a Poster
Too many new hires learn chemical safety from a poster in the hallway. Real learning sticks when supervisors show how storage works in practice. Involving people in active drills, running through spill containment steps with the actual compounds, and explaining why every step counts leaves a lasting impression. When new staff see their mentor model good habits, the risk of accidents drops.
Solutions for Real-World Safety
Locking up dangerous chemicals isn’t enough. Labs and shops with cadmium fluoroborate benefit from keeping up-to-date digital inventories and automating reorder and inspection reminders. Point-of-use cabinets with local exhaust keep dust contained. Feedback loops—where techs report near-misses without fear of blame—improve every storage system over time. Standards come from books, but safety culture comes from people who care enough to do things right, every time.
What are the handling and safety precautions for Cadmium Fluoroborate?
Respecting the Dangers of Cadmium Compounds
No one working with Cadmium Fluoroborate can afford to be casual about safety. Cadmium rises near the top of toxic metals, and regular exposure—whether through dust, fumes, or accidental ingestion—invites a long list of health problems. Respiratory trouble, kidney failure, and even cancer show up in research looking at workplace exposure. These aren’t theoretical risks. Studies from organizations like OSHA and NIOSH make it clear: chronic cadmium exposure shortens lifespans and creates lifelong medical issues.
Personal Protective Equipment: Non-Negotiable
Gloves, lab coat, safety glasses—these belong on anyone even opening a bottle of Cadmium Fluoroborate. Add a chemical-resistant apron or full-face shield if splashes seem likely. A standard surgical mask falls short; the right answer is a certified respirator designed to block particulates and vapors. Ventilation matters. A fume hood, checked and maintained, needs to run before any bottle opens or weighing paper hits the balance.
Labeled Storage and Contamination Control
Every bottle needs solid secondary containment, clear hazard labeling, and a spot on a dedicated shelf or cabinet. I’ve seen labs save hassle by keeping cadmium compounds locked up and away from anything used for food or drink storage, to lower the chances of cross-contamination. Anyone handling these chemicals should avoid eating, drinking, or smoking in the same area and always scrub hands and arms afterward. A spill kit sits near the workbench, and staff actually know how to use it—not just as a ticked box for audits.
Spill Response and Waste Management
A small spill never just gets wiped up with a paper towel and tossed in the regular trash. Anyone who’s cleaned up cadmium knows to grab disposable gloves, a proper mask, and absorbent pads or inert materials. Cleanup debris goes straight into a labeled, sealed hazardous waste container, not the regular garbage. Larger spills trigger an emergency plan—evacuate, ventilate, call in the chemical safety officer, not just hope for the best.
Training Saves Lives
No amount of equipment replaces experience and habit. Safety training doesn’t just rattle off a material safety data sheet; it roots practical knowledge. I recall a colleague who, thanks to annual refreshers, calmly shut down a potentially dangerous situation with Cadmium Fluoroborate. Proper handling only works when everyone knows why these steps matter and understands the consequences of skipping them.
Prevention Works Better Than Treatment
Routine medical monitoring shows its value. Simple blood and urine tests catch warning signs of cadmium poisoning before symptoms hit. In my experience, labs that bake medical screening into their routines face fewer emergencies and health scares. Regular audits and updated protocols also catch gaps before someone gets hurt.
Better Alternatives and Waste Reduction
Safer alternatives sometimes exist—using less hazardous chemicals, substituting materials, or changing processes can cut risks in half. Process engineers and chemists keep an eye on new research and emerging substitutes for cadmium salts. Even small changes in procedure can shrink waste and lower exposure.
Standing Up for Safer Labs and Workplaces
Responsibility for safe handling belongs to more than the lab tech holding a pipette. Supervisors, procurement managers, and safety officers all hold pieces of the puzzle. Creating a culture where questions about procedures, risks, and emergency actions are met with real answers saves lives. No one regretted spending an extra minute on safety protocol—plenty of people regret cutting corners with Cadmium Fluoroborate.
| Names | |
| Preferred IUPAC name | Cadmium tetrafluoridoborate |
| Other names |
Cadmium tetrafluoroborate Cadmium(II) fluoroborate Cadmium borofluoride |
| Pronunciation | /ˈkæd.mi.əm ˌflʊr.oʊ.bəˈreɪt/ |
| Identifiers | |
| CAS Number | 13812-64-5 |
| Beilstein Reference | 2813106 |
| ChEBI | CHEBI:91251 |
| ChEMBL | CHEMBL4296937 |
| ChemSpider | 22718749 |
| DrugBank | DB14633 |
| ECHA InfoCard | 03ca09b7-b1f9-4c13-ab27-0f8fd9f7e7f0 |
| EC Number | 232-222-0 |
| Gmelin Reference | 6087 |
| KEGG | C18657 |
| MeSH | D017734 |
| PubChem CID | 159410 |
| RTECS number | FO5950000 |
| UNII | X1928C0W16 |
| UN number | UN3265 |
| Properties | |
| Chemical formula | Cd(BF4)2 |
| Molar mass | 238.22 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 2.7 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.1 |
| Vapor pressure | 3 mmHg (25°C) |
| Acidity (pKa) | 10.5 |
| Basicity (pKb) | -4.1 |
| Magnetic susceptibility (χ) | −54×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.433 |
| Dipole moment | 6.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1043 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause cancer. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H350: May cause cancer. H360Df: May damage the unborn child. Suspected of damaging fertility. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P264, P280, P302+P352, P304+P340, P312, P321, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD₅₀ Oral Rat 215 mg/kg |
| NIOSH | TM8225000 |
| PEL (Permissible) | 0.005 mg/m3 |
| REL (Recommended) | No REL established. |
| Related compounds | |
| Related compounds |
Cadmium borate Cadmium fluoride Cadmium bromide Cadmium chloride |

