Cadmium Cyanide: A Closer Look at History, Properties, Uses, and Future Directions
Historical Development and Discovery
Cadmium cyanide stands out in the long chemical timeline. Its discovery and industrial use can be traced back to the era when the push for new materials sparked explosive growth in inorganic chemistry. Early records show researchers experimenting with cyanide compounds to push boundaries in electroplating and pigment production. Laboratory synthesis of cadmium cyanide evolved from the classic reaction of cadmium salts with potassium or sodium cyanide, a method that goes back nearly a century. As industrialization picked up, the spotlight on cadmium expanded. Chemists scrutinized it for its striking electroplating characteristics, vaulting it into an important role in niche metal finishing processes. This wasn’t just an academic exercise—the need for better corrosion resistance and precision coatings in manufacturing made cadmium cyanide more than just another chemical curiosity.
Product Overview and Synonyms
Cadmium cyanide, often carrying the term “cadmium dicyanide,” appears in chemical texts under several names. Common synonyms include cadmium(II) cyanide and cyanocadmate. Manufacturers and researchers also refer to it using shorthand terms like Cd(CN)2. Its product footprint isn’t the widest, but where it shows up, it tends to make an impact. People working in metal finishing, scientific research, and chemical synthesis recognize cadmium cyanide for its specialized roles.
Physical & Chemical Properties
Physically, cadmium cyanide settles as a white, crystalline powder—a look it shares with many other cyanide compounds. It dissolves readily in water and forms complex ions. The compound’s solubility plays a role in its handling, especially when used as an electrolyte in electroplating baths. Chemically, it carries the familiar punch of the cyanide group: strong reactivity, particularly toward acids, and an ability to generate toxic hydrogen cyanide gas. Melting and decomposition trigger release of those gases, demanding careful control in industrial settings.
Technical Specifications & Labeling
Product packaging warns clearly about its acute toxicity. Labels bear hazard and precautionary statements. Regulatory frameworks in the US, EU, and other regions recognize cadmium cyanide as a controlled substance, guiding storage, handling, and worker training. Reading the fine print on any container, one finds specifics around purity, recommended handling temperatures, and PPE requirements. All these technical standards reflect hard-learned lessons, both in the lab and in real-world applications.
Preparation Method
Lab preparation of cadmium cyanide usually proceeds by reacting soluble cadmium salts (often the sulfate or chloride) with a solution of potassium or sodium cyanide. The reaction throws down a white precipitate of cadmium cyanide, which can then be filtered, washed to remove excess alkali, and sometimes recrystallized for higher purity. It’s a straightforward process, but rarely risk-free. Generating toxic byproducts and handling highly poisonous reactants pushes chemists and technicians to stick to rigorous safety protocols. There’s a strange sense of trust and tension with every batch, as history has proven what can happen when things go wrong with cyanide chemistry.
Chemical Reactions & Modifications
Cadmium cyanide serves as a platform for different chemical transformations. Exposing the compound to acids forms hydrogen cyanide gas, which ranks as one of the more dangerous outcomes. In aqueous environments, it hydrolyzes, interacting with water and atmospheric carbon dioxide to give cadmium carbonate or other cadmium salts. Researchers have pushed its boundaries through complex formation with ammonia and other ligands, showing the versatility of cadmium coordination chemistry. These modifications underpin its use in selective extraction or plating baths, helping fine-tune metal deposition—essential for industries stressing precision.
Application Area
Few chemicals tread more carefully than cadmium cyanide in terms of where and how they’re used. It claimed an early niche in electroplating, giving jewelry, aerospace components, and critical industrial parts resilient coatings against wear and corrosion. Its ability to lay down smooth, adherent layers of metallic cadmium gave materials extra life where failure could carry a high price. Chemists lean on its reactivity for creating cadmium-containing compounds needed in research and analysis, including certain pigments and sensors. More recently, as regulations tighten, the chemical’s use narrowed, with substitutes growing more common. Still, it hangs on in research circles, where controlled conditions and specialized requirements justify the risks.
Safety & Operational Standards
Handling cadmium cyanide demands respect for its hazards. Both cadmium and cyanide knock hard on the body’s defenses—cadmium as a heavy metal toxin and cyanide for its ability to disrupt cellular respiration. Workers and researchers gear up in full PPE, rely on tested ventilation systems, and keep emergency response plans at the ready. Training isn't just a checkbox; it means a deep grasp of hazardous materials procedures, not only for themselves but to protect the wider workplace and environment. Regulatory oversight includes strict tracking of quantities, secure storage with secondary containment, and constant air and surface monitoring in facilities that still use the compound. This culture of vigilance stems from a long record of industrial accidents and chronic health issues in the past.
Toxicity Research
Cadmium cyanide’s toxicity record is thoroughly documented. Acute exposure leads to poisoning symptoms ranging from shortness of breath and chest pains to seizures and quick collapse if not treated. Chronic exposure to even low levels of cadmium, and similar organometallics, stacks the odds for kidney damage, lung disease, and cancer. Cyanide’s rapid interference with oxygen use leaves no margin for error, especially around acids and heat. Toxicologists have spent decades mapping out dose limits, pathways of exposure, and effective antidotes—not just for industrial users but for accidental public exposures. This body of work informs stringent standards and fuels ongoing campaigns to phase out unnecessary use.
Research & Development
In my years following the shifting landscape of hazardous materials, research around cadmium cyanide always surfaces in discussions of heavy metal regulation and alternative technologies. Chemistry departments and government agencies pour resources into developing cleaner, safer options—both for plating and for chemical synthesis. At the same time, analytical chemists test new detection and remediation methods that target trace cadmium residues in soil and water. Much of the R&D here centers on risk reduction: new encapsulation strategies, better personal monitoring tools, and methods for neutralization before environmental release. In the lab, exploration into alternative complex agents and lower-toxicity cadmium sources aims to keep the benefits of precise electrochemistry while dropping the health risks.
Future Prospects
Legislators, public health experts, and industry veterans all agree: cadmium cyanide’s best days are likely behind it. Environmental and occupational health regulations grow tighter every year. Costs for compliance, disposal, and liability keep climbing. Where substitutes perform nearly as well, manufacturers make the switch. Yet, there’s always a stubborn space for chemicals like cadmium cyanide in cutting-edge research, specialized industry corners, and historical study. Ethical stewardship, rigorous process controls, and continual monitoring keep the hazard in check. The future looks to better training, stronger regulatory frameworks, and investment in alternatives—driven by a clear-eyed view of past harm and current necessity. The push isn’t only about eliminating risk entirely, but about living up to higher standards in science, industry, and public health.
What is cadmium cyanide used for?
Pushing Metal Finishing Forward
Growing up, I watched my uncle work in his machine shop, transforming plain metal into parts that glinted bright in the light. Ever since, I’ve been fascinated by what makes that smooth, silvery surface possible. It turns out, underneath that shine, chemistry is doing the heavy lifting. Cadmium cyanide sits right in the middle of one of these processes: electroplating. Most folks never encounter this compound directly, but its impact stretches from airplanes to electronics.
Cadmium cyanide entered my radar back in a college lab where we dipped basic bolts into an electric bath and ended up with something far more durable. In industries where gears and switches must last, engineers rely on cadmium because of its wear resistance and ability to keep rust at bay. Its cyanide salt—the version combined with cyanide ions—makes the metal easy to dissolve into that plating bath, which means a stronger, more reliable coating.
Why Use Cadmium at All?
Take aircraft hardware, for instance. Tiny screws on landing gear might not grab headlines, but one rusty bolt can ground a plane. Cadmium coatings prevent this, helping protect steel from the corrosive grind of rain, oil, and salt. Automotive makers once chose cadmium plating for a similar reason—fighting corrosion in harsh environments. In electronics, thin cadmium layers shield parts against both moisture and unwanted electrical currents. The results stand out: smoother assembly, extra protection, and equipment that doesn’t quit early.
Chemistry Brings Challenges
Any mention of cyanide calls for caution. Manufacturing with cadmium cyanide takes serious oversight. Stories from those who’ve worked in plating shops make this clear—the air, equipment, and runoff all demand attention. Cadmium can build up in the body and harm kidneys or bones after long-term exposure, and cyanide spells danger on its own. Even small spills or poor ventilation lead to big risks for workers and nearby communities.
In the United States, agencies like OSHA and the EPA set strict rules on how companies handle cadmium and cyanide. Workers need training, regular health checks, and reliable protective gear. Wastewater from plating lines cannot go straight back into rivers or sewers. Plants have to catch, treat, and neutralize these toxins before anything leaves the building. Skipping any of these steps courts disaster and costs trust.
Rolling Forward: Safer Plans
Cadmium cyanide isn’t the only tool for metal finishing anymore. Zinc-nickel and phosphate coatings are finding their way into more factories as companies try to balance performance and safety. I’ve seen some shops shift to these alternatives for parts that don’t face as much wear, but some applications—jet engines and old military hardware—still rely on cadmium’s unique mix of traits. Still, every year, the push for cleaner, less hazardous technology grows stronger.
Decades of real-world experience and scientific research show that awareness and accountability pay off. Companies that run clean operations and offer options for recycling old metal outshine their competitors. People working in those factories deserve environments where the only thing they bring home is a sense of pride, not lasting health worries.
Cadmium cyanide opened doors for stronger, longer-lasting hardware, but new ideas and tougher rules can push industry toward a future where safety and durability go hand in hand.
Is cadmium cyanide toxic or hazardous?
Common Uses Bring Real Risks
Factories and labs keep jars and tanks of chemicals like cadmium cyanide because this compound helps make things like batteries and coatings for electronics. Trouble is, it’s not some everyday chemical you sweep off the workbench and forget about. Anyone who’s spent time in a plating shop recognizes that sharp, almost metallic smell and the anxious respect it commands. The stuff is no joke.
Where the Danger Sits
Cadmium cyanide takes hazards seriously. The cadmium in it carries enough weight by itself—once it gets into the body, it stirs up major trouble. The kidneys end up bearing the brunt, leading to damage most people never see coming until it’s too late. Breathing in dust, touching powders, or mixing solutions can lead to absorption, and there’s decades of research proving cadmium isn’t something bodies know how to get rid of. That metal piles up, with chronic exposure tracking closely in studies with higher rates of cancer and bone disease. The World Health Organization calls cadmium a human carcinogen, and reliable government data from the U.S. and Europe matches that assessment.
The other half of this duo, cyanide, is infamous for its ability to shut down vital functions in a matter of minutes. There’s a reason it’s the main character in poison novels. Cyanide vapor blocks the body’s cells from using oxygen—on the shop floor or in an accidental spill, even a short whiff can start headaches, confusion, pounding heart, and worse. The effects spike fast because cyanide slips through skin and lungs with ease.
The Realities on Shop Floors and Labs
Discussions about toxicity often feel distant unless you’ve seen a supervisor rush workers out of a room after a spill. Standard respirators, gloves, and splash-proof aprons help, but nobody lets their guard down. Most workspaces handling cadmium cyanide pipe waste water into treatment tanks, using chemical tricks to break down cyanide before it reaches the drain. Some older plating lines still struggle with leaks, especially in regions without strict chemical laws.
Even outside the factory, the threat lingers. Landfills and accidental dumping push cadmium compounds into soil and water. Farmers and families a couple miles down the road might never know where the faint metallic taste in well water comes from. Studies in mining regions and towns near battery plants draw lines between cadmium exposure and health issues generation after generation.
Reducing the Risk Is Everyone’s Business
It’s not just on the workers or managers to prevent problems. Companies have updated their engineering controls—ventilation, scrubbers, and storage rules—after watching hospitalizations and lawsuits pile up cost. Regular blood checks, better training, and clear labeling add layers of defense. Governments helped by setting strong limits for workplace exposure and demanding companies document every barrel and drum.
Using safer alternatives helps too. Some industries now swap cadmium cyanide for less toxic plating solutions or switch to mechanical parts that don’t need these dangerous finishes at all. For those who can’t avoid it, clear emergency plans and education cut down risk. Areas that clean up old waste sites or support communities with testing and care make a massive difference for families who never expected to share a backyard with something so dangerous.
How should cadmium cyanide be stored and handled safely?
Cadmium Cyanide: A Hazard that Demands Respect
Cadmium cyanide doesn’t sit on a lab shelf like a harmless powder. The risks are real—breathing dust or vapors exposes people to both cadmium and cyanide toxicity. I still remember being taught in my first chemistry job that just a small bit of exposure could mean headaches, dizziness, or worse. The bottom line: this is a chemical for professionals who understand the stakes.
The Storage Environment Isn’t an Afterthought
A lot of people think locking up a dangerous chemical solves the problem, but buildings shift, humidity creeps up, and a forgotten bottle on a shelf can change everything. Cadmium cyanide thrives in dry, cool places—think 15°C to 25°C, away from heat, sunlight, and any hint of moisture. The slightest bit of water can trigger a reaction, so anything near sinks, pipes, or steam lines is a big mistake. Sturdy, sealed containers made of compatible material (usually high-quality glass or certain plastics) work best. No metal shelves or rusty cabinets—contact with metals can start a chain reaction you want no part of.
Why Segregation Matters
In college, our storeroom had clear sections: oxidizers here, cyanides there, acids somewhere else. This wasn’t just about being organized. Cadmium cyanide, if kept close to acids, can let out hydrogen cyanide gas—even a whiff of that stuff is deadly. No point risking lives for the sake of convenience. I always double-check labels, and encourage others to keep incompatible substances on opposite ends of the storeroom.
Handling with Focus and Respect
Handling cadmium cyanide means stepping into a strict routine. Nitrile gloves, eye protection, and sturdy lab coats aren’t optional—they form the baseline. Inhaling dust causes big trouble, so anyone working with the substance should use a chemical fume hood, not just a cracked window. Cleaning up spills calls for special neutralizers—no sweeping under the rug or makeshift solutions. I’ve seen situations where improper cleanup led to expensive, stressful remediation. The cost of a little vigilance up front is nothing compared to fixing a bad mess.
Smart Disposal Prevents Accidents Down the Line
Dumping remains out back or into a sink has never been an acceptable option. Hazardous waste handlers follow rules laid out by regulatory agencies, usually involving specialized containers and paperwork. Solid or liquid leftovers wait for pickup in double-sealed, clearly labeled barrels. I’ve filled out chain-of-custody sheets before, and I can say it slows you down—but you do it because some risks aren’t forgivable. Just one mistake and suddenly someone downstream pays the price.
Training Makes the Difference
People hold the real power to keep labs and workplaces safe. I’ve seen experienced chemists take nothing for granted, running drills and asking younger staff to practice emergencies. Safety data sheets are more than fine print—they’re roadmaps to staying healthy. Peer supervision and regular retraining mean smaller chances of complacency. In my own work, any sense of routine never erases the basic fact that what sits in those bottles can change lives in the blink of an eye.
Possible Steps Forward
More engineering controls and automated handling could move people farther from risk, but education never takes a back seat. Digital tracking keeps storage conditions right and flags issues in real time. Regular third-party audits help spot cracks before they spread. These extra steps can seem like overkill on a smooth day, but I’ve learned that you only get to regret a shortcut once.
What is the chemical formula of cadmium cyanide?
Understanding the Essentials
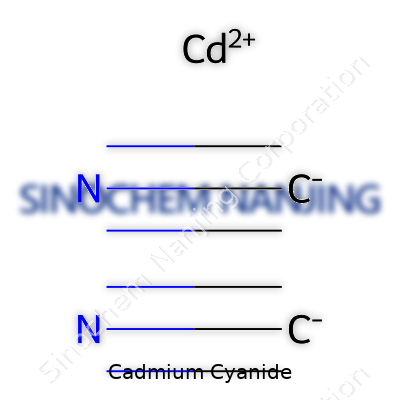
The chemical formula for cadmium cyanide is Cd(CN)₂. At first glance, that might look like trivia from a chemistry textbook, but this compound carries weight in several fields. Some people think about metals and immediately picture heavy machinery or jewelry. Cadmium cyanide isn’t something you see every day, yet it plays a quiet role behind the scenes—particularly in electroplating and chemical research labs. Recognizing its formula unlocks an understanding of its properties and why safety conversations around it feel urgent.
Safety and the Human Factor
Cadmium, the element in the formula, lands on toxic substance lists for a reason. Breathing in its dust or fumes spells trouble for kidneys and disrupts bone health, according to the Centers for Disease Control and Prevention (CDC). Cyanide compounds, like the CN⁻ group here, add another layer of concern. Knowledge about the formula pushes science professionals to handle this compound with guarded respect. In smaller labs, folks sometimes assume a little extra caution will do, but the reality is that both cadmium and cyanides can cause serious damage even at low doses. Mishandling leads to lifelong health consequences or worse. Just naming the elements in Cd(CN)₂ should prompt anyone to reach for gloves, fume hoods, and clear signage.
Application and Industry Impact
Cd(CN)₂ finds its use mostly in industrial electroplating. This method coats objects—often steel or copper—with a thin layer of another metal. In this case, cadmium plating protects against corrosion, especially for components in aircraft, electrical connectors, or fasteners. Some of my own student days in a university lab included assisting in research that dealt with metal complexes, including cyanides. It took real discipline to keep safety routines strict. In industry, that discipline shapes daily routines, worker training, and emergency protocols.
Transparency and Responsible Use
The importance of understanding what makes up cadmium cyanide goes beyond chemistry class. It calls for collaboration across science, labor, and health agencies. The U.S. Occupational Safety and Health Administration (OSHA) requires posting safety data and limits worker exposure. Still, incidents happen. That’s why open access to chemical information, including formulas and health risks, supports everyone involved—from shop floor workers to environmental officers.
Environmental Concerns and Community
Cd(CN)₂ production generates waste that can linger in soil and water. Both cadmium and cyanides disrupt aquatic life. Once, in a research project, we tracked spill responses at small plating plants in our city. Heavy rains flooded an old site, and legacy waste with these compounds reached a creek downstream. It took years for local wildlife populations to recover. If communities near industrial centers push for transparency and regular environmental testing, they play a part in keeping the land healthier for the long run. Local councils and city public works can help by funding clean-up projects and monitoring programs—actions that might prevent future harm.
Looking Forward: Solutions for Safer Practices
Science keeps searching for safer substances to replace cadmium cyanide. Some companies now test zinc-based or nickel-based alternatives, though trade-offs always exist. The best solutions often come from open dialogue between chemists, workers, regulators, and the surrounding neighborhoods. As research uncovers safer chemicals and new plating technologies, updating guidance and practices matters as much as knowing the formula itself. Community involvement, strong regulations, and responsible business decisions work together to lower the risks tied to harmful compounds like Cd(CN)₂. Understanding the formula becomes the starting line for keeping people and the planet safer.
What precautions are needed when disposing of cadmium cyanide?
The Real Risk of Cadmium Cyanide
Cadmium cyanide doesn’t grab headlines like some toxins, but that doesn’t make it any less dangerous. Workers in plating plants and chemical labs see this compound up close. It delivers a double punch—cadmium and cyanide—both toxic on their own and even worse together. Skin contact, inhaling dust, or swallowing even a small amount can wreck the kidneys, poison the lungs, and trigger neurological damage. When it escapes into the environment, the damage spreads far beyond factory walls.
Why Tossing It Out Isn’t Enough
Some folks say, “Just neutralize it.” That sounds simple on paper. Neutralization doesn’t mean dumping it down the drain and hoping for the best. If even a little cadmium or cyanide slips through, fish, plants, and people catch the consequences for decades. Cyanide destroys aquatic life fast. Cadmium takes its time—building up in soil, sliding into food chains, and crawling into drinking water. Japan’s Itai-Itai disease stands as proof: cadmium-contaminated water from industry led to widespread kidney disease and bone disorders that haunted families for generations.
Solid Steps For Safe Disposal
The best defense starts with a good offense. If goggles, gloves, smart ventilation, and tight storage aren’t in place, accidents can turn routine disposal into a crisis. Proper gear keeps workers safe during every step, not just the final toss.
Chemical treatment ranks high for breaking down cyanide—using oxidizers like hypochlorite or hydrogen peroxide under controlled conditions. After splitting the cyanide, the process moves on to handling the cadmium. Precipitation, where chemicals pull cadmium out of liquid, forms solid waste that won’t seep as easily into the ground. That’s not the endpoint. Before sending anything off-site, companies should test for any trace amounts left in the water or sludge.
Transport matters too. Loaded barrels rolling down the highway hold no less risk than those in the lab. Trucks carrying cadmium cyanide require clear labeling, tight seals, and careful routes. State and federal regulations demand paperwork and tracking, and for good reason. Missing containers or careless spills have shaped superfund clean-ups that taxpayers shoulder for years.
Working With Certified Professionals
Few shops handle hazardous waste in-house anymore. Certified, audited disposal companies own this responsibility. They spend years training staff to avoid shortcuts, manage emergencies, and stay up to speed with shifting regulations. Partnering with experienced providers increases the odds of catching problems early and prevents fines and environmental lawsuits.
Better Options for the Next Generation
Everyone benefits when upstream solutions shrink the need for cadmium or cyanide. Alternatives exist for many plating uses, although they cost more at first or require new equipment. Cutting usage and recycling safely not only saves money in the long run but eases the burden on landfills and water systems.
Transparency Builds Trust
Neighbors and workers deserve honesty when it comes to dangerous waste. Publishing yearly waste reports, showing cleanup goals, and involving local communities in safety plans foster trust. Mistakes happen, but companies that keep communication open see fewer conflicts and build better working relationships all around.
| Names | |
| Preferred IUPAC name | cadmium dicyanide |
| Other names |
Cadmium dicyanide Cyanogendicadmy Cyanure de cadmium |
| Pronunciation | /ˈkæd.mi.əm saɪˈæ.naɪd/ |
| Identifiers | |
| CAS Number | 544-92-3 |
| Beilstein Reference | 14620 |
| ChEBI | CHEBI:35479 |
| ChEMBL | CHEMBL3980467 |
| ChemSpider | 21170473 |
| DrugBank | DB14566 |
| ECHA InfoCard | 12bbd5a1-c532-4de1-97c2-381b9fa5eea5 |
| EC Number | 208-806-4 |
| Gmelin Reference | Gmelin Reference: **CdC6N2 2** |
| KEGG | C02353 |
| MeSH | D002150 |
| PubChem CID | 10197494 |
| RTECS number | GF8575000 |
| UNII | M3D90473M9 |
| UN number | UN1680 |
| Properties | |
| Chemical formula | Cd(CN)₂ |
| Molar mass | 240.484 g/mol |
| Appearance | White crystals |
| Odor | Odorless |
| Density | 2.46 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.47 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.10 |
| Magnetic susceptibility (χ) | +89.0·10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 81.3 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -168.5 kJ/mol |
| Pharmacology | |
| ATC code | V03AN01 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; causes damage to organs; may cause cancer; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P262, P264, P273, P280, P284, P301+P310, P302+P352, P304+P340, P308+P311, P314, P320, P330, P361+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-☠ |
| Lethal dose or concentration | LDLo oral rat 25 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 25 mg/kg |
| NIOSH | GL2275000 |
| PEL (Permissible) | 0.05 mg/m3 |
| REL (Recommended) | 7H |
| IDLH (Immediate danger) | **9 mg/m3** |
| Related compounds | |
| Related compounds |
Cadmium nitrate Cadmium carbonate Cadmium hydroxide Zinc cyanide Mercury(II) cyanide |

