2,2'-Azobis(2-Methylbutyronitrile): Progress, Impact, and Continuing Questions
Shaping the Field: Historical Development
Back in the early days of organic synthesis, chemists looked for reliable sources of free radicals to spark polymerization and other chain reactions. The family of azo compounds, known for their ability to split and create radicals upon heating, started to gain real attention. 2,2'-Azobis(2-methylbutyronitrile), which often goes by abbreviated versions of its chemical name, emerged from work on structural modifications designed to improve safety margins and performance over earlier azobisisobutyronitrile (AIBN). Research labs favored these materials for their consistent thermal decomposition, a property that remained valuable as polymer chemistry expanded in the twentieth century.
Product Overview: Applications Begin at the Bench
What pulls me toward this compound is how it has changed the approach to certain synthetic schemes. At its core, 2,2'-Azobis(2-methylbutyronitrile) acts as a radical initiator, turning the engine of free-radical chemistry in both research and industrial reactors. By heating this white to off-white solid, labs generate a steady source of radicals. Too often, a chemist can get tripped up with unpredictability, but this compound brings a welcomed element of control—something I value when working on precise molecular architectures. Polyacrylonitrile production, for instance, depends on radical initiation, and this is one of the molecules making the process tick.
Physical & Chemical Properties: Characteristics That Matter
Let’s get past the textbook look of things for a second. Holding this powder, I notice it’s not much different by eye from common lab reagents—fine, nearly odorless, a bit waxy. What stands out in application is its melting point, which hovers between 100 and 103°C. It dissolves comfortably in organic solvents like ethanol and acetone, but water doesn’t get along with it. Heat is the straightforward trigger: once past its decomposition temperature, it splits into nitrogen and highly reactive radicals. This trait lets it drive clean reactions without flooding the environment with wild byproducts. Chemists who have been through enough experimental headaches recognize how valuable that predictability is for reproducible synthesis.
Preparation Method: Sourcing the Building Blocks
Crafting this azo compound calls for attention to both safety and yield. The method most researchers use involves the condensation of the right nitrile precursors with hydrazine derivatives, followed by oxidation. Temperature and pH monitoring matter at each stage, as the intermediate hydrazides can be touchy. Small lapses in control might scare off those unfamiliar with handling nitrogen-rich chemicals. I remember the first time synthesizing an azo initiator—donning all the gear, double-checking glassware, grateful for each quiet, uneventful step. These are reactions nobody rushes. Getting a pure batch checks off several boxes: reliable decomposition profile, good solubility, and, most importantly, a product that won’t start decomposing on the shelf.
Chemical Reactions & Modifications: Opportunities and Limits
What’s remarkable about 2,2'-Azobis(2-methylbutyronitrile) is not just how it initiates reactions, but how chemists can use it as a launchpad for more complex molecular frameworks. It splits to make cyanoalkyl radicals, handy for grafting reactions or step-growth processes in making specialty plastics. This functional group sets it apart from other azo initiators that lack useful handles for further transformation. Despite this, the chemistry rarely ventures far from its radical-forming role; once it releases nitrogen, the molecule’s fate is more or less sealed. What matters then is how those radicals spark the rest of the synthesis. Years spent in the lab teach you quickly: a little bit of predictability in radical chemistry means a lot, especially for polymers where chain length and branching can make or break a product.
Technical Specifications & Labeling: What End-Users Watch For
Nobody in the chemical world wants surprises with hazardous solids. Good suppliers package 2,2'-Azobis(2-methylbutyronitrile) in airtight, opaque bottles, with lots flagged about safe temperatures: the label says keep below 40°C, and there’s good reason. The risk of spontaneous decomposition stacks up quickly with exposure to heat. Labels mention purity, typically 98% or better, and impurities are listed because they might steer unwanted side reactions. From an end-user perspective, high-purity lots mean smoother reactions and easier purification downstream. Having handled plenty of chemicals that degrade or polymerize in the bottle, I value companies that actually stick to tight labeling and storage standards.
Synonyms & Product Names: Navigating Chemical Language
Chemists rarely talk in long strings of numbers outside formal documents. The compound sometimes pops up as AMBN or 2,2'-Azobis(2-methylbutyronitrile), and older literature calls it by other variants rooted in its systematic name. On the bench, most people reach for the acronym, churning through abbreviations to keep things moving. Synonyms help track down relevant papers, but in practice, the structure matters more than the name unless you're ordering it by the caseload.
Safety & Operational Standards: Getting It Right
Anyone who’s worked with azo initiators knows the story: heat, rough handling, or a leaky vial will create problems. Unintentional decomposition can release a lot of gas and heat, which might cause explosions or, at the least, spoil a workday. Safety guidance puts a strong emphasis on avoiding sparks and keeping cool. Personal protective gear isn’t optional—goggles, gloves, and fume hoods define safe handling. Disposal brings its own challenges, as any unused material still holds potential energy. I’ve seen labs get a rude awakening from letting residue sit too long in a warm corner. Regulators draw a hard line: storage below a critical temperature, specific secondary containment, clear hazard labeling. The trouble comes when budget or training falls short, especially in smaller facilities.
Application Area: Why it Shows Up on the Lab Shelf
Polymer chemists prize this initiator for starting polymer chains in acrylonitrile-based plastics and specialty copolymers. Its reliability means that batch-to-batch consistency stays high, which trickles down through the supply chain to consumer products. I remember working with research teams where even small inconsistencies in initiators would throw off polymer properties, causing hours of troubleshooting. 2,2'-Azobis(2-methylbutyronitrile) also finds roles in controlled radical polymerization, giving researchers flexibility to design functionalized plastics and study kinetics in detail. At commercial scale, its predictable decomposition rate means fewer surprises on the reactor floor, which keeps production costs down and safety risks manageable.
Research & Development: Where Curiosity and Utility Meet
With new polymers popping up every year, radical initiators don’t fall out of fashion. Recent work focuses on modifying the basic structure to tune decomposition temperature and rate, aiming for an even safer or more controllable product. In academic circles, labs still dig into the nitty-gritty—can this initiator open new doors for block copolymer assembly or surface modification? On the applied side, engineers look for ways to recycle or recover unused agents, cutting down on waste and improving process economics. My experience shows that the most valuable advances come from unlikely places—a new solvent, a cleaner synthesis, or a slightly different radical chain transfer protocol.
Toxicity Research: Safety Beyond the Lab Bench
As with all nitriles and azo agents, toxicity studies matter. Researchers investigate both acute and chronic effects. Early data point to skin and respiratory irritation, and animal studies push for caution with repeated exposure. Breakdown products, especially volatile nitriles, demand extra scrutiny as long-term inhalation can take a toll on health. The lack of detailed data on long-term human exposure to this compound stands as a call for more aggressive research. Having worked near a few too many open bottles years ago, I learned firsthand the sting of poor ventilation. Regular medical surveillance for workers and proper engineering controls can close a lot of these gaps, but gaps still appear in developing regulatory frameworks and public awareness.
Future Prospects: Improvements and Questions Ahead
2,2'-Azobis(2-methylbutyronitrile) won’t dominate headlines, yet its role behind the scenes shapes everything from advanced polymers to smart coatings. The quest for even cleaner-decomposing, less hazardous radical sources drives ongoing investment in both industrial and academic labs. Improvements in sustainability—better synthetic routes, non-toxic breakdown products, strategies for post-reaction cleanup—might cement this compound’s position in more eco-friendly chemistry. Pressure from both government regulation and end users will likely demand safer alternatives. But, if the past half-century in polymer science has taught anything, it’s that reliable, well-characterized reagents like this will remain in demand, provided stakeholders invest in continued research, transparent labeling, and robust safety practices.
What is 2,2'-Azobis(2-Methylbutyronitrile) used for?
Real Use in Radical Polymerization
Some people might look at the name 2,2'-Azobis(2-Methylbutyronitrile) and turn their heads, but chemists who spend time in the lab get what it can do. This compound lives in the world of free radical chemistry. It enters the scene most often as an initiator—basically, it starts chemical reactions that need a kick to get rolling.
You’ll find this stuff in action during the production of plastics and synthetic rubbers. I remember standing in a university lab, watching a batch polymerization begin once we dropped in our initiator. It felt like opening the floodgates. Once this chemical breaks down, it produces radicals that trigger the formation of large, chain-like molecules. The resulting polymers aren’t just theoretical. They’re the backbone for durable items, flexible seals, and all sorts of molded goods.
Why Industries Want These Features
2,2'-Azobis(2-Methylbutyronitrile) keeps getting attention because of its reliability. Chemists know how fast it breaks down at different temperatures. They can design recipes to match the speed of production and the properties they’re chasing. Not all initiators deliver such repeatable results, and that level of predictability keeps the manufacturing line moving without surprises.
This chemical also handles high temperatures better than some other options. That matters on a crowded shop floor where temperature control isn’t perfect. Think about the need for a compound that stays stable enough not to kick off a reaction at the wrong time, but active enough to get the job done quickly once the process starts. That’s the kind of thinking that keeps production managers, engineers, and researchers coming back to this azobis compound.
Looking at Safety and Environmental Impact
Anyone who works with nitrile-based azo initiators knows to respect them. They can decompose to release nitrogen gas and some potentially toxic byproducts. Careless handling invites risks to health and safety. I’ve seen colleagues spend hours drafting safety protocols and setting up ventilation systems just to account for these possibilities.
There’s another angle here. Synthetic chemistry keeps running up against environmental concerns. Some byproducts from compounds like this can stick around in the environment if companies don’t manage waste streams properly. Waste management isn’t only about following rules. Responsible labs and factories create detailed plans, from scrubbing exhaust to using well-monitored collection tanks, so these chemicals don’t cause trouble downstream.
What Makes a Real Difference
If you ask people who’ve done this work in person, they’ll tell you that better training saves both materials and people. Handling potent initiators means spending time with mentors, not just manuals. Proper labeling, personal protective gear, and emergency drills never fall out of fashion when dealing with strong radical sources like 2,2'-Azobis(2-Methylbutyronitrile).
Some companies go a step further by exploring less hazardous alternatives. Research keeps pushing toward “greener” chemistry. These options may reduce persistent organic pollutants without leaving manufacturers behind in terms of performance. For now, this compound remains in wide use—partly out of inertia, partly out of necessity—but the push for safer, cleaner chemistry gains ground each year.
What are the safety precautions when handling 2,2'-Azobis(2-Methylbutyronitrile)?
Risks Don’t Take a Break
I’ve seen plenty of lab safety posters in my time, and every so often, there’s a chemical that gets more red flags than the rest. 2,2'-Azobis(2-Methylbutyronitrile) is one of those. People use it as a free-radical initiator in polymer chemistry and organic synthesis, but it comes with sharp risks: sensitivity to heat, shock, and friction, as well as toxic effects if it escapes into the air or lands where it shouldn’t. Anyone who values their skin, lungs, and eyes needs to pay close attention here.
PPE Isn’t Just a Buzzword
I always start with the basics: gloves that actually hold up, not those thin ones that rip after a few minutes. Nitrile gloves offer solid protection. Goggles shield your eyes from dust or splashes, and a full lab coat keeps your clothing and skin out of harm’s way. A face shield adds another layer when weighing out powders or pouring from bottles. A fume hood is a must — this chemical loses its charm when inhaled, so breathing it in should never even be an option.
Heat and Friction Invite Trouble
Polymer initiators like this one have a reputation: they decompose if things heat up too much, sometimes causing fires or explosions. I’ve seen stories where someone left the bottle near a sunny window, or didn’t bother checking if equipment was sparking. It’s not just lab folklore. The lesson is simple — keep it in a cool place, away from direct sunlight or heat sources. Use gentle tools for transferring or weighing. No rough glass rods or metal spatulas that can create sparks.
Respect the Labels
SDS sheets often seem long and boring, but I’ll never forget handling a chemical with a missing label years ago. It’s shaky ground. Clear labeling cuts confusion and helps everyone on a team know what’s in front of them, what to do in an emergency, and where to find the emergency shut-off. Store 2,2'-Azobis(2-Methylbutyronitrile) in tightly sealed containers, and keep it far from acids, peroxides, or oxidizers. More than one disaster started with bottles shelved side by side in the wrong place.
Housekeeping Isn’t a Nice-to-Have
Spills with organic powders can go from zero to hazardous in seconds. Always clean as you go. Scraps or flakes on a bench can stick to hands, find their way onto door handles, or worse, into lunchrooms. Spatulas, balances, and weighing boats need a good cleaning after use. Waste disposal is another place where corners get cut — follow hazardous-waste rules so this chemical doesn’t just end up in the standard trash.
Emergency Lessons Stick
I once saw someone ignore a minor splash — just a dot on the sleeve. Several hours later, skin irritation made it clear even a tiny bit matters. Quick rinsing under running water isn’t an overreaction; it’s protocol for a reason. If anyone breathes in dust or fumes, fresh air and immediate medical help come before filling out any forms.
Training: The Habit That Saves Lives
New lab hires sometimes get a whistle-stop tour, but thorough training makes a real difference. Hands-on demos stick much better than booklets. Knowing where spill kits, eyewash stations, and fire extinguishers live, and how to use them, pays off in an emergency. Experience — yours or a colleague’s — keeps safe practices from getting cut for the sake of speed.
Respect earns safety. Every time 2,2'-Azobis(2-Methylbutyronitrile) comes off the shelf, every eye on the room counts. The margin for error is thin, but diligence makes the process smooth — and lets you walk out with health and peace of mind.How should 2,2'-Azobis(2-Methylbutyronitrile) be stored?
Why Storage Isn’t Just a Backroom Job
A good friend once told me, “Don’t cut corners on things you barely understand.” Handling chemicals fits that rule. I walked into a small polymer lab job years ago, eager to impress. My first day, the manager pointed at a shelf lined with white bottles, each one labeled in black marker. “Pay attention to these,” she said. That lesson stuck, especially when we worked with tricky compounds like 2,2'-Azobis(2-Methylbutyronitrile), known for kicking off reactions by tossing out nitrogen and freeing up radicals. But its wild side isn’t fun if you ignore storage rules.
Why This Stuff Matters
Some chemicals carry their own risks, but organic peroxides and certain azos sit near the top. 2,2'-Azobis(2-Methylbutyronitrile) isn’t something you toss on a shelf in the break room. Its reputation for thermal decomposition means it can turn dangerous out of nowhere. Heat, light, and even friction have flipped safe storage labs into disaster scenes. If you want to avoid that, pay attention to these details, especially if you work in research, plastics, or even specialty inks.
Cool and Consistent Always Wins
The molecule prefers a cold, dark home, far from labs running at a lazy room temperature. In practice, this means tucking it into a refrigerator or cold room at about 2-8°C (36-46°F). I’ve seen labs run at the low end, near 2°C, if the stuff sits for months. The point here isn’t just “keep it cold”—what you want is to stop any chance of runaway decomposition. Refrigerators with temperature logs help. If the power hiccups, staff spot the spike right away. Every reputable chemical supplier marks this on the bottle—but I’ve seen enough accidents start with someone skipping the obvious.
Stay Dry, Stay Safe
Humidity shifts can play havoc over time. Any moisture sneaking into a container accelerates breakdown and can trigger odd reactions. I still remember a colleague cursing at clumped powder during a summer storm. Tight lids mark the difference between a safe shelf and a chemical headache. Desiccant packs in the fridge can keep the microclimate dry.
Containers and Compatibility
Original, airtight containers win every time. The less contact with air, the slower any oxidation or contamination. Transfer to glass or approved plastic jars if the original package gets damaged, but never toss extra into metal tins or unfamiliar plastics. Labels should show the date opened. That way, nothing quietly ages in the back corners, forgotten until the cleanout day.
Segregation and Inventory: No Shortcuts
Some chemicals turn violent when mixed, even by accident. 2,2'-Azobis(2-Methylbutyronitrile) shouldn’t neighbor acids, bases, or anything else that could tip its temper. Avoid stacking it near sources of ignition or sunlight. One day I opened a cabinet to find an old canister sitting right beside a leaky acid jar—an accident set on a slow burn.
How to Keep People (and Labs) Out of Trouble
Clear signage on storage units matters. Safety officers—real or self-appointed—ought to drive regular inventory checks. Detailed records let labs spot any slip before it grows. When the container runs low or hits the supplier’s expiration date, arrange proper disposal. Skipping this step lets all those little risks stack up, and suddenly it’s not just paperwork anymore.
Why It’s Worth the Effort
Accidents cost more than lost lab time. I once watched a full project delayed by a single heat-triggered bottle rupture. The cleanup chewed up months. Mindful storage isn’t just science; it’s respect for coworkers and the lessons other labs have learned the hard way.
What is the chemical structure and formula of 2,2'-Azobis(2-Methylbutyronitrile)?
The Story Behind a Specialty Chemical
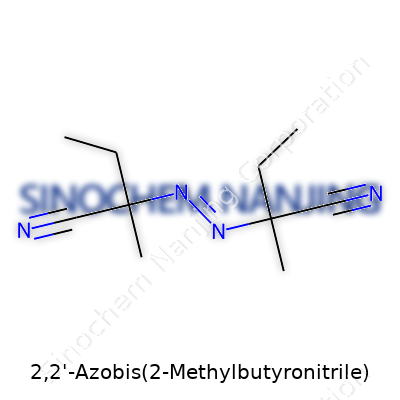
2,2'-Azobis(2-methylbutyronitrile) doesn’t show up in grocery stores or on the shelves of your local hardware shop, but it plays a big role behind the scenes in a number of industries. I remember the first time I saw its name on a safety data sheet in a college lab. The full chemical formula jumped off the page: C10H16N4. The formula gives a snapshot—ten carbon atoms, sixteen hydrogens, and four nitrogens—but the structure digs much deeper.
The Details of Its Structure
Chemists always seem to light up when talking about azo compounds like this one. The defining feature here is the azo group, which means two nitrogen atoms connected by a double bond (N=N). On each side, that azo group connects to a carbon chain:
- Each chain has a nitrile group (C≡N) hanging off it. If you picture it, you see two nearly identical arms stretching out from the midpoint (the N=N group), each one ending in a nitrile. That’s key: it shapes how this molecule behaves in chemical reactions.
- Each carbon chain is built like this: starting at the azo group, you pass through a methyl (–CH3) group and then reach the rest of the butyronitrile skeleton. So, chemically, you’re looking at two 2-methylbutyronitrile units, both anchored at the second carbon position to the central azo group. The whole structure comes together as (CH3)2CHCH2CH(N≡C)-N=N-CH(CN)CH2CH(CH3)2.
Why Its Structure Matters
Azo compounds aren’t rare, but 2,2'-Azobis(2-methylbutyronitrile) carries a special punch as a source of free radicals. In polymer chemistry labs, I’ve seen this molecule help kick off chain reactions that stitch together plastics or rubbers. Its structure, with the symmetrical arms and azo core, makes it a strong choice for this job. The N=N bond can break apart cleanly under the right conditions, usually heat, to make radicals on each side.
The design means you can add it to a vat of monomers, heat things up, and watch as it quietly helps build polymers used in everything from car parts to medical devices. The methyl group branching off those arms helps keep the radicals stable enough so you don’t get runaway reactions or unwanted side products—something anyone who’s ever run a messy polymerization will appreciate.
Looking at Safety and Handling
Working with compounds that generate radicals always puts an extra layer of responsibility on chemists. The molecule breaks down energetically, so it demands respect in storage and handling. Safety sheets flag the dangers of heat and sparks, and that matches what I observed in lab protocols: keep the bottle cold and away from ignition sources, handle small amounts, and always use proper shielding.
Supporting Innovation with Smart Choices
Plenty of chemicals can make radicals, but the right initiator cuts costs and reduces waste downstream. Researchers look for reliable reaction starters that don’t break the bank or make a lot of byproducts that are hard to clean up. The structure—and formula—of 2,2'-Azobis(2-methylbutyronitrile) support its place in the toolkit for modern manufacturing. By understanding both its shape and behavior, you get repeatable results and safer processes, which matter far beyond the laboratory.
Is 2,2'-Azobis(2-Methylbutyronitrile) hazardous to health or the environment?
Understanding the Chemical
2,2'-Azobis(2-Methylbutyronitrile) catches attention in labs and industrial sites for its work as a free-radical initiator. This compound plays a role in making plastics and modifying polymers, which often means it turns up somewhere behind the scenes. Deciding if it puts people or the planet at risk isn’t just a matter of reading a label. Experience with real chemicals—seeing what a splash or a fume does—teaches more than a safety sheet ever could. So what concerns really come with this one?
Health Hazards Deserve Respect
I remember once in a polymer lab, someone opened a bottle of a related azobisisobutyronitrile (AIBN). Everybody could smell this sharp, odd scent, and the ventilation kicked into high gear. These azo compounds, including 2,2'-Azobis(2-Methylbutyronitrile), break down easily, letting off nitrogen and creating a storm of free radicals. This makes them powerful but also risky.
If inhaled or touched, these nitrile-bearing compounds go through skin and respiratory tracts. Most sources agree they can irritate, and that cyanide-like breakdown products can show up. Once these get into the system, things can turn ugly fast—they affect the nervous system, cause dizziness, or worse. Chronic exposure brings up headaches, fatigue, even potential for organ harm. Not something you want to gamble with, especially not outside a fume hood and without gloves and goggles.
Environmental Impact
Once down the drain, these azo chemicals persist. Wastewater treatment plants don’t always knock them out. That raises red flags, because fish and aquatic insects lack defenses against one-two punches like this: the azo part turns to toxic amines, and the nitriles linger. Scientific reports show small crustaceans and fish get hit hard by these substances, affecting growth and reproduction. The breakdown products can build up over time, showing up in sediments or even entering drinking water sources. That’s not just lab talk—concerns like this have pushed government agencies to nail down disposal and reporting rules. The European Chemicals Agency (ECHA) and the US Environmental Protection Agency (EPA) both keep watch lists.
Safer Handling and Better Solutions
Wearing gloves and goggles doesn't cut corners here. Good practice also means solid ventilation, smart containers, and real spill plans. Having seen a spill soak into an unprotected bench, I know that cleanup isn’t just about wiping—it’s about stopping contamination from spreading into drains or landfill. Companies should train teams properly, and researchers need real, hands-on hazard sessions. Sharing knowledge and mistakes helps everyone avoid surprises.
On the positive side, green chemists push for alternatives. Researchers look for initiators that don’t drop toxic nitriles or stubborn amines after use. Sometimes, newer photoinitiators or oxygen-based starters step in, changing the chemistry game for the better. Switching old habits for newer, less hazardous materials needs encouragement—from regulators, from managers, and definitely from the folks on the ground using these chemicals every week.
Looking Forward
Staying aware keeps risk under control. People in labs and factories need real facts, not vague warnings. Regulators can tighten disposal routes, give incentives for safer alternatives, and make sure communities near chemical plants get honest answers. It’s about staying sharp—and making sure the tools that build products don’t quietly chip away at the well-being of people or the environment.
| Names | |
| Preferred IUPAC name | 2,2'-Azobis(2-methylbutanenitrile) |
| Other names |
AMBN Azobis(2-methylbutyronitrile) 2,2′-Azobis(2-methylbutyronitrile) Vazo 67 |
| Pronunciation | /ˌtuː tuː ˈæz.oʊ.bɪs tuː ˌmɛθ.əl.bjuːˈtraɪ.oʊˌnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | '13472-08-7' |
| Beilstein Reference | 91361 |
| ChEBI | CHEBI:87254 |
| ChEMBL | CHEMBL156720 |
| ChemSpider | 124788 |
| DrugBank | DB04174 |
| ECHA InfoCard | 03e3b893-ef49-46bd-8ffe-7d8a6b860cce |
| EC Number | 2589-64-2 |
| Gmelin Reference | 92077 |
| KEGG | C19619 |
| MeSH | D002450 |
| PubChem CID | 126877 |
| RTECS number | UF5950000 |
| UNII | 7G8N6X8W97 |
| UN number | UN3236 |
| CompTox Dashboard (EPA) | DTXSID7020902 |
| Properties | |
| Chemical formula | C10H16N4 |
| Molar mass | 136.21 g/mol |
| Appearance | White crystalline powder |
| Odor | Odor: mild characteristic odor |
| Density | 1.06 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.8 |
| Vapor pressure | 0.45 hPa (25 °C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | 14.53 |
| Magnetic susceptibility (χ) | -7.23 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 2.3 mPa·s (25 °C) |
| Dipole moment | 3.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 209.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −50.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7265 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H241 |
| Precautionary statements | P210, P261, P264, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2,2=3-3-2-W |
| Flash point | 87 °C |
| Autoignition temperature | 250 °C |
| Explosive limits | 1.8-10.7% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 860 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 485 mg/kg |
| NIOSH | SN 19200 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.2 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Azobisisobutyronitrile Azobis(cyclohexanecarbonitrile) |

