Butylphosphoric Acid: A Down-to-Earth Look at a Complex Chemical
Tracing its Roots: The Evolution of Butylphosphoric Acid
Butylphosphoric acid has a history tied closely to the rise of organophosphorus chemistry during the early to mid-20th century. Years ago, researchers in Europe and North America raced to understand and modify phosphorus-based acids for both military and civilian uses. Butylphosphoric acid came about as chemists reacted butyl alcohols with phosphorus oxychloride, seeking a balance between reactivity and stability. Interest spiked during the expansion of solvent extraction processes, particularly in nuclear energy and metallurgy, where the compound offered useful extraction properties and allowed for collection and separation of rare metals. Even though people outside research hardly discuss it, inside chemical circles, butylphosphoric acid represents a story of practical adaptation, a link between laboratory curiosity and industrial muscle.
What’s in the Bottle: Overview of the Substance
Butylphosphoric acid, often described by its molecular formula C4H9OPO(OH)2, stands out among organophosphorus acids due to its mix of hydrophobic and hydrophilic character. The acid commonly appears as a viscous, colorless or faintly yellowish liquid, with a slightly pungent smell. Many chemists like it because this blend of organic butyl with the more “inorganic” phosphoric acid section brings a range of solubilities and binding capacities, making it valuable as an extractant. Its synonyms include n-butyl phosphoric acid and mono-n-butyl phosphoric acid, names that crop up in research papers and procurement catalogs alike. One can’t ignore the compound simply because it’s tough to pronounce—its impact on chemical separations and syntheses gives it lasting value.
What It’s Made Of: Physical and Chemical Qualities
Butylphosphoric acid boils at higher temperatures and sits with a density that puts it below water, thanks to the butyl chain. It mixes partly with water, more fully with most organic solvents, swinging between polar and nonpolar worlds. The acid’s reactivity focuses around its phosphorus-oxygen core. Phosphoryl groups, known for making strong connections with metals, give it an edge in extraction work. Acidic hydrogen atoms allow it to donate protons and hook onto bases, so one sees it playing roles in both acid-catalyzed reactions and as a ligand in coordination chemistry. Its stability endures under moderate heat, but it reacts around strong oxidizers.
The Nitty-Gritty: Technical Specs and Labeling
In my experience, you rarely see butylphosphoric acid without several safety warnings about corrosiveness. Regulatory labels instruct on eye and skin protection and point out proper ventilation. Physical parameters—such as its specific gravity, acid content, and purity—matter to anyone running a process, since small impurities sometimes change extraction performance. Technical data usually includes color index, water content, and residual phosphorus oxychloride, which can cause problems if overlooked. Chemical manufacturers supply information about storage, shipping, and shelf-life, but users tend to focus on whether it does the job for a process or application rather than obsessing over fine print.
From Lab Bench to Scale: How It’s Made
Most butylphosphoric acid starts its journey as butyl alcohol, getting mixed with phosphorus oxychloride or phosphorus pentoxide in the controlled conditions of a chemical reactor. Workers add the phosphorus compound slowly to the alcohol, often under cooling to keep reaction rates safe. Acidic byproducts appear and need careful handling. After the primary reaction, a water or dilute acid wash purifies the final liquid, pulling off leftover chlorides or phosphoric acid. Large producers may run extra distillation or solvent treatment steps to reach higher purity, since applications in nuclear separation, for instance, leave little room for contaminants. Looking at the manufacturing floor, one realizes both chemical skill and respect for safety keep the process running.
Getting Down to Chemistry: Reactions and Tweaks
Butylphosphoric acid likes to react at both its butyl group and its phosphoryl center. Chemists sometimes substitute more or fewer butyl groups, creating di-butyl and tri-butyl variants, which behave differently in extraction systems. The acid can form esters with alcohols or react with bases to give corresponding salts. Its phosphoryl oxygen draws in metal ions, making the acid a frequent guest in experiments on selective metal uptake. Some researchers try grafting the molecule onto polymers, hoping to take its extraction ability into new technological fields. Chemical modification offers more than academic fun—it aims at improved target selectivity or easier processability, both vital in major industrial settings.
The Many Names: Synonyms and Product Labels
People in the field refer to butylphosphoric acid by a surprising number of names. Mono-n-butyl phosphoric acid, butyl acid phosphate, and MBP all point back to the same family of molecules. These names often reflect degree of substitution, so it’s worth clarifying exactly which molecule sits in a bottle before running experiments or ordering supplies. Some older literature might use slightly different variations, but the chemistry roots all these synonyms firmly in the organophosphorus world. Anyone navigating the marketplace or research landscape learns to double-check the chemical structure, not just the label.
The Rules of the Game: Safety and Operations
Working with butylphosphoric acid demands real caution. Its acidic nature can burn unprotected skin, and the liquid gives off vapor that irritates eyes and lungs. In facilities where I have worked, the rulebook calls for gloves, goggles, lab coats, and fume-hood ventilation. Chemical spill kits and neutralizers stay close by, since nobody wants a splash going unchecked. Storage away from basic materials, oxidizers, and open flames lowers the risk of accidents. On the regulatory side, shipping and handling laws in many countries classify it as a hazardous substance, so all paperwork better be airtight. Training goes beyond video modules—seasoned lab workers often pass on practical tricks that avoid costly or dangerous mistakes. Mistaking its safety profile leads to real injuries, so respect remains both cultural and enforceable.
Where It Goes: Real-World Uses
Butylphosphoric acid becomes especially valuable in industries that need fine control over metal separation. Its strong binding to uranium and rare earth elements brings it into the nuclear fuel cycle, where extracting actinides from waste streams means cleaner processes and higher yields. Metallurgists reach for it during copper and cobalt extraction, cutting down steps and improving selectivity. In the lab, it serves as a reagent in organic synthesis and as a catalyst in esterification. Some specialty chemical makers test its potential as a flame retardant or as an intermediate in surfactant production. Its use stretches beyond research—real industrial players rely on its chemistry to save money and boost production.
Stirring the Pot: Research and the Road Ahead
Research teams constantly probe butylphosphoric acid’s ability to bind metals, hoping to push the technology into cleaner rare earth element production, recycling, and even water treatment. The market for green extraction technologies, pushed by rising prices for nickel, cobalt, and lithium, draws attention to this and related acids. Chemists want new derivatives with higher selectivity, better solubility, or easier biodegradability. The search for safer formulations, with fewer worker health risks and lower environmental persistence, spins off dozens of projects around the world. People focus not just on yield but also process safety and environmental compliance, responding to stricter rules and sharper public scrutiny.
Digging Deeper: Toxicity and Risk Research
The question of toxicity comes up for any phosphorus acid, and butylphosphoric acid is no different. Early studies warned about skin and eye irritation, but newer reports look much harder at chronic exposure and breakdown products. The acid doesn’t rank as severely toxic by ingestion under most circumstances, but laboratory animals exposed to high doses show possible effects on liver and kidney function. Its environmental breakdown remains only partly mapped out, with some worry about waterborne transformation into more persistent phosphates. Regulators and company labs lean hard into understanding exposure thresholds, especially in water treatment and metal recycling facilities. My own work with similar chemicals convinced me that real diligence on containment and ventilation beats responding to an emergency after-the-fact. For now, the consensus treats it with care, recognizing that knowledge gaps call for future research.
Future Outlook: Where Things May Head
Butylphosphoric acid looks set for a broader future, especially with surging demand for metals tied to renewables and electronics. Connection to the nuclear sector and critical mineral recovery means any breakthrough—safer manufacturing, smarter extraction, or eco-friendly disposal—gets global attention. Ongoing research promises new compounds for hydrometallurgy, plus greener, more biodegradable variants. The road ahead puts a premium on reducing worker exposure and finding better ways to handle waste and spent reagents. Industry players and research labs move toward improvements that keep people safe, cut down costs, and adapt to tighter environmental standards. Based on conversations across fields, chemists watch for this substance to play a bigger role in both old industries and emerging clean-tech applications.
What is Butylphosphoric Acid used for?
Digging Into What Butylphosphoric Acid Really Does
Ask anyone who’s ever worked near industrial labs or spent time with chemical engineers, and you’ll probably hear stories about oddly named compounds doing heavy lifting behind the scenes. Butylphosphoric acid is one of those chemicals. Not exactly a household name, but make no mistake—the stuff matters, and most folks have no clue how much it impacts everyday products and industries.
Key Uses in Metal Extraction
I come from a family where several folks worked in mining and metallurgical plants. Growing up, I heard terms like "extraction solvent" tossed around over dinner. Butylphosphoric acid is one of those unsung workhorses in metal refining. Operations use it to separate valuable metals from ore. For instance, the acid forms complexes with metals like uranium or rare earth elements, making it easier to pull them out and purify them for use in electronics, green tech, or even in health care devices. Many of the tools and devices you use—your phone, laptop, medical scanners—owe their performance to rare metals purified with the help of compounds like this one.
Building Blocks for Better Chemistry
Butylphosphoric acid plays a role in making chemicals that act as softening agents and specialty additives. In the past, companies focused on cheap mass production, but harmful byproducts and flimsy materials raised health and safety concerns. These days, chemists turn to substances like butylphosphoric acid for creating more stable and effective agents in detergents, crop protection and water treatment. These additives keep detergents working better under hard water conditions, or help pesticides cling longer to crops while breaking down more safely in the environment.
Challenges with Handling and Safety
One thing I learned early is that every handy chemical brings trade-offs. Butylphosphoric acid has drawbacks—it's corrosive and toxic if mistreated. That means anyone working with it wears layers of personal protective equipment. Sites handling this acid take special care with spill management and waste disposal for both worker safety and community health. Incidents with leaks or poor storage—especially in the past—left some communities with water or soil issues that took years to resolve. Companies today need to keep tighter control and invest in worker education, since a single mistake can ripple out far beyond the plant gates.
Searching for Greener Alternatives
Looking at the bigger picture, society wants cleaner, safer chemical processes. Labs continue to search for options that do the same job as butylphosphoric acid, but break down faster in nature or come from renewable sources. Some researchers are experimenting with plant-based extraction aids or tweaking the structure of existing acids to improve biodegradability. That’s no small task; every chemical swap has knock-on effects throughout production lines. It takes honest conversations between researchers, manufacturers, and regulators to balance production needs, worker safety, and long-term environmental health.
Why Awareness Matters
So much about daily life relies on precise, powerful chemicals most people never see. Butylphosphoric acid stands as a reminder: high-tech gadgets or clean water often depend on complex chemistry under serious safety controls. If we want a future that’s both innovative and sustainable, paying close attention to these foundational compounds—and supporting better practices for using or replacing them—will make a difference for everyone’s health and well-being.
What are the safety precautions when handling Butylphosphoric Acid?
Why It Matters
Butylphosphoric acid isn’t a household name, but in industries like chemicals, mining, and nuclear fuel processing, contact with it happens daily. I’ve seen operators work with this stuff, and it’s clear there’s a right way and a wrong way to go about it. Being careless with butylphosphoric acid can lead to burns, breathing trouble, and long-term health problems. There’s a reason every seasoned technician pays close attention to safety rules here.
The Gear Makes a Difference
The first step is the gear. Nitrile gloves stand out on the shop floor. Latex won’t cut it—one splash, and the acid chews through. Splash-resistant goggles cover the eyes, and anyone who has gotten even a tiny bit of this on exposed skin learns quickly that a cotton lab coat isn’t enough. Chemical-resistant aprons, boots, and sometimes even a face shield offer a solid line of defense. I know people who double up on protection after seeing what a careless slip can do.
Keeping Fumes Out of Your Lungs
Fumes from butylphosphoric acid can creep up on you. There’s a certain smell in the air—sharp, almost electric—and once you notice it, you’re likely too close. Always working under a fume hood or in well-ventilated spaces cuts risk way down. In rooms with limited airflow, the acid vapor settles at nose level. Wearing a proper respirator with acid gas cartridges isn’t optional in these spots. Emergency eyewash stations and safety showers can’t just exist for show. I’ve watched someone rush to the shower after an accident, grateful it was less than ten feet away.
Handling Spills and Storage
One careless pour can mean a nasty spill. Keeping neutralizing agents—like sodium bicarbonate—on hand helps clean up small messes before they spread. For bigger spills, everyone clears the area, and a trained crew jumps in. No one picks up a mop without gloves and a face shield. Storage matters just as much. Butylphosphoric acid finds a home in dedicated chemical cabinets with secondary containment trays. Storing it away from anything reactive—especially bases and oxidizers—prevents chemical fireworks you definitely don’t want. Labels must stay clear and updated. I’ve spent time searching for a mislabeled bottle and saw how easy it is to make a mix-up.
Training and Communication
Staying safe isn’t about a list taped to the wall. It comes down to training. Every new worker gets a walkthrough of what the acid does, what signals mean trouble, and how to react if things go south. Regular drills help the team stick to the plan without panicking. Open communication between shifts means whoever’s coming in next knows what they’re working with. I’ve seen issues arise when someone forgets to mention a leaky valve or a near-miss spill, only for someone else to run into trouble because of it. Plain talk and real stories keep the risks real.
A Culture of Attention
Protecting yourself and your team from butylphosphoric acid isn’t only rule-following. It’s about respect for what the substance can do. If something looks off, no one hesitates to stop work. Good habits—like washing hands after handling, not eating in the work area, and keeping gear in top shape—turn into routines that save skin and lungs. From what I’ve seen, workplaces that treat safety as a shared responsibility end up with fewer injuries and more trust among workers.
What is the chemical structure of Butylphosphoric Acid?
Understanding Butylphosphoric Acid—From Lab to Life
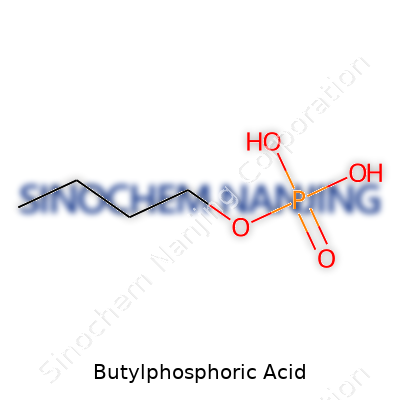
In the world of chemistry, butylphosphoric acid isn’t exactly a household name, but its structure reveals a lot about its uses and why people in both industry and research pay attention to it. The formula sits at C4H11O4P. At the core, a phosphorus atom links up with four oxygen atoms, and one of those oxygens connects to a straight butyl group—the telltale carbon chain with four atoms.
What Sets Its Structure Apart
Each phosphoric acid derivative tweaks something about its behavior. Adding a butyl group brings out changes everywhere from solubility to reactivity. Studying butylphosphoric acid, what jumps out is the P-O-R configuration, where R is the butyl chain. The backbone stays anchored by a phosphorus atom sharing double bonds with one oxygen—imagine that giving the molecule a stiffer attitude compared to a basic alkyl group. The three other oxygen atoms, with at least one as a hydroxyl (-OH), introduce sites for both hydrogen bonding and further modification.
Holding this structure, the butyl group sticks out, giving the molecule a broader footprint. It acts as a handle for chemists who want to connect the acid to other molecules or use it as a bridge in complicated extraction processes. In some applications, chemical properties that normally stay hidden in phosphoric acids wake up once you attach that four-carbon tail.
Real-World Relevance in Industry and Science
Walking into laboratories working on metal extraction, the name pops up at the extraction interface. Butylphosphoric acid brings phosphorous’s knack for holding on to metals like uranium or rare earth elements, but its butyl group lets it dissolve in common organic solvents. In nuclear chemistry, where safety and selectivity mean everything, these molecules help separate dangerous materials with less fuss. The solubility twist comes straight from that butyl tail, which pulls the whole molecule into the organic phase, letting it grab metal ions that normally stay stuck in water.
From my own perspective, having handled organophosphorus compounds in grad school, you remember the sharp, almost sour-fuel smell, and the need to respect gloves and ventilation. While they might look pretty basic on paper, small changes in their tails—ethyl, propyl, butyl—can make or break a process or an entire project budget. Phosphoric acid itself acts almost like water, but add those carbon atoms, and you have a molecule balancing between hydrophilic and hydrophobic, an edge that's crucial for chemists fine-tuning an extraction or a chemical synthesis route.
Issues and Safer Paths Forward
The hands-on side of using butylphosphoric acid brings up some thorny issues. Like many organophosphorus compounds, toxicity and environmental persistence pose real risks. Once spilled, these molecules resist breakdown, and they can mess with aquatic life. Legislation and best practice from major chemical agencies call for tight controls over their use and disposal.
Safer alternatives draw a lot of attention in industry. Green chemistry pushes for less toxic substitutes, and a lot of work centers on designing extraction agents that break down harmlessly after use. Chemists also scale back concentrations in processes or opt for closed systems, keeping vapor and waste out of the environment. These approaches don't just plug regulatory gaps—they help research labs and companies limit long-term liabilities and protect workers’ health.
Understanding the structure of butylphosphoric acid means knowing more than just lines on a page. That butyl chain isn’t window dressing; it’s the key that makes this molecule part of some of the most challenging chemical separations happening today.
How should Butylphosphoric Acid be stored?
Reality of Handling Chemical Hazards
Butylphosphoric acid rarely makes headlines, yet it earns respect in every lab that deals with extraction and separation. I’ve spent days wrestling with jars, labels, and the burly shelves of supply closets, realizing that careless storage can bulldoze any sense of safety. It pays to stay sharp, since this acid pulls no punches when handled the wrong way.
What Happens If You Don’t Store It Right?
This acid brings real risk. Vapor escapes lead to headaches or worse. If the liquid drips or spills, hands or eyes suffer. Get it near incompatible chemicals and you can create a runaway mess. Ignoring its needs sets up staff and expensive equipment for unnecessary harm.
A spilled bottle once nearly ruined a costly synthetic run in my colleague’s lab. He opened the cabinet, caught a whiff, and scrambled for the eyewash station. We discovered the container had sweated and leaked—not just bad luck, but bad shelving habits and a hint of laziness with sealing the lid.
Choosing the Right Spot and Container
Butylphosphoric acid deserves a container that actually earns its keep—high-density polyethylene or glass, both with tight-fitting, corrosion-resistant caps. I keep my chemicals in cabinets marked by discipline, not hopeful guesswork. Chemicals never share tight quarters with bases, oxidizers, or anything flammable. That’s not a rule to fudge. Separate spaces, clear labeling, and upright bottles cut risk to the bone.
Shelving must support the load, with trays below to catch drips before they creep to lower shelves or floors. I’ve seen labs regret cheap plastic shelves or skip secondary containment. Budget gear turns into cleanup duty when the acid chews through a weak spot.
Taming Temperature and Humidity
Stable rooms make for stable chemicals. Warmth nudges volatile liquids towards trouble, and humidity creeps into loose caps. Controlled environments matter—think room temperature, not the back corner by the whirring vent. Extra-cool storage almost never helps and can sometimes push concentrated acids into a more hazardous state.
I place a digital thermometer and humidity gauge right by the cabinet. If the numbers start to wander, that’s a signal, not a suggestion, to check insulation or ventilation.
Clear Labels and Fresh Eyes
Nothing protects like certainty. Every bottle wears a label with the full name, concentration, hazard pictograms, and a date. Unlabeled containers tempt disaster. Staff briefings aren’t just for new hires; I call everyone together once per quarter for a walk-through and quick quiz to keep knowledge sharp.
Older stock drifts to the front and gets checked for degraded seals or unexpected color. I set a reminder to inventory every two months, and I trust memory much less than I trust a pen and clipboard.
Good Practices Build Trust, Not Routine
Regulators give plenty of guidance—OSHA, NIOSH, and the chemical manufacturers all chime in with tough language for a reason. If a spill or exposure happens, the cost stacks up with every minute spent wishing for tighter procedures. For me, confidence comes from practiced care, clear routines, and the refusal to cut corners, no matter how many other lab tasks fight for time.
With chemicals like butylphosphoric acid, strict storage pays off every day. Knowing what’s at stake hands you all the reason you’ll ever need to do things right.
Is Butylphosphoric Acid hazardous to the environment?
People and Nature: The Real-World Effects
Butylphosphoric acid does a job in many industries. Mining uses it to pull rare earth metals out of ores. Chemistry labs rely on it to move things like uranium or plutonium from one place to another. On paper, it sounds like a clever tool. Out in the real world, the story gets complicated. This chemical, if not handled right, can leave a mark that is tough to clean up.
I’ve seen how waste from industrial sites sometimes ends up in nearby soil or waterways, even with good intentions. Regulations say to keep things contained, but leaks and spills still happen. Once butylphosphoric acid gets out into a stream or underground water, trouble follows. This acid isn’t just strong ― in water it breaks down slowly. Fish and insects exposed to small amounts often stop reproducing or growing right. Birds or mammals drinking from contaminated water start to show health issues. People living near these sites sometimes notice odd tastes in their water or changes in crops and plants nearby. In places without strong chemical oversight, I’ve watched entire stretches of river lose most of their life after an incident with acids like this.
Why the Science Matters
Chemical safety studies show that organic phosphoric acids, like butylphosphoric acid, can break up cell membranes in plants and aquatic life. Small doses in the lab hurt algae and microcrustaceans. More concentrated leaks can scorch crops and knock back growth in garden plants. With ground and water contamination, people who fish or harvest nearby pick up residues that might pass along the food chain.
When mining companies use this acid in solvent extraction, residuals sometimes head out with waste streams. In low-oxygen soils, bacteria won’t break down this stuff quickly. It hangs around, raising risks over time. Phosphorus compounds, in general, can cause algae blooms in lakes and rivers if enough ends up in the water. Too much growth cuts off oxygen for everything else, tipping the whole ecosystem out of balance.
Choosing Better Ways Forward
Clean production takes work from everyone involved. In my experience, the most effective plants invest in robust spill containment — double-walled storage, regular inspections, real-time leak monitors. Workers get serious safety training, with drills for what to do if a pipe bursts or a truck tips over. Efforts to recycle and neutralize waste streams rather than just dumping them tend to pay off. Companies that talk openly with nearby communities build more trust and fix mistakes faster.
Some researchers at universities look for replacements with less environmental punch. Chelating agents based on amino acids or sugars sometimes do a similar job without lingering as long or hurting wildlife. The mining and nuclear sectors slowly test these, though change takes time and serious investment.
For those of us living near industrial sites, good water testing and public reporting mean a lot. If neighbors get involved and ask questions, problems show up sooner and solutions arrive faster. Environmental groups frequently run their own independent sampling, keeping everyone honest. These actions don’t block progress or industry — they help ensure mistakes get caught early and cleaned up before they turn into disasters.
Looking back, I’d rather see industries spend money upfront keeping chemicals from escaping than spend even more later trying to reverse the damage. The cost of lost rivers, ruined farmland, or broken community trust never shows up on the first budget, but everyone pays in the end. A safer approach protects both business and the basics people need to live well.
| Names | |
| Preferred IUPAC name | Butyl dihydrogen phosphate |
| Other names |
Butylphosphoric acid Dibutylphosphoric acid DBP Phosphoric acid, dibutyl ester Dibutyl hydrogen phosphate |
| Pronunciation | /ˌbjuː.tɪl.fɒsˈfɔːr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 1070-08-4 |
| 3D model (JSmol) | `load =C1CCOP(=O)(O)OC1` |
| Beilstein Reference | 1367938 |
| ChEBI | CHEBI:131873 |
| ChEMBL | CHEMBL2000241 |
| ChemSpider | 81483 |
| DrugBank | DB02199 |
| ECHA InfoCard | ECHA InfoCard: 100.013.726 |
| EC Number | 207-527-5 |
| Gmelin Reference | Gmelin Reference: 100350 |
| KEGG | C14340 |
| MeSH | D001223 |
| PubChem CID | 95449 |
| RTECS number | TC8750000 |
| UNII | BY6M9VQTCO |
| UN number | UN3265 |
| Properties | |
| Chemical formula | C4H11O4P |
| Molar mass | 186.15 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Odorless |
| Density | 1.06 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.92 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 1.54 |
| Basicity (pKb) | 1.50 |
| Magnetic susceptibility (χ) | -7.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.438 |
| Viscosity | 60 mPa·s (20 °C) |
| Dipole moment | 3.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 242.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1250.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3933 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V09IX05 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Flash point | 108 °C |
| Autoignition temperature | 340 °C |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 820 mg/kg (rat, oral) |
| NIOSH | WA6475000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 2500 mg/m3 |
| IDLH (Immediate danger) | IDLH: 15 mg/m³ |
| Related compounds | |
| Related compounds |
Methyldiphenylphosphine oxide Tributyl phosphate Trioctyl phosphate |

