Butadiene Dioxide: Framing the Story Behind a Complex Compound
Looking Back: How We Got Here
The roots of butadiene dioxide run deep in the history of industrial chemistry. Chemists first began exploring the world of diene oxides in the mid-20th century, driven by a hunger to manipulate double bonds and oxygenation in new ways. Early researchers realized that such compounds could serve as powerful intermediates, not just for experimentation but for applications touching plastics, synthetic rubbers, and even pharmaceuticals. The growth in petrochemical processing after the war years gave many chemicals their big break, and butadiene dioxide was no exception. Lab curiosity gave way to factory-scale interest, with engineers eager to chase every advantage in polymer stability and reactivity.
What Sets Butadiene Dioxide Apart
Butadiene dioxide isn’t the sort of compound most folks run into at the hardware store. This molecule stands out with its tight ring structure, formed by adding two oxygen atoms across a four-carbon backbone that used to be a diene. That little chemical twist sets off a host of interesting properties and challenges, giving it a distinct flavor compared to regular butadiene. The presence of two epoxide groups packed onto the same framework makes it more than a curiosity—it’s a chemical workhorse for scientists looking for reactive yet manageable intermediates.
The Heart of Its Physical and Chemical Nature
Chemically, butadiene dioxide presents a fascinating mix of reactivity and stubbornness. Its dual epoxide rings make it highly strained, so it often wants to react with all sorts of nucleophiles. In the bottle or flask, it's no stranger to forming colorless liquids with a faint, somewhat sweet odor. Boiling and melting points sit close to those of similar low-weight epoxides, but its ring strain can lead to quirky behavior during heating or in the presence of acids. A splash won’t burn your skin off, but direct exposure draws a sharp response from your eyes and lungs. People working in labs get plenty of warning smells and learn pretty quickly not to underestimate these “simple” molecules.
Standards and Technical Nuances
Labeling for butadiene dioxide, like most industrial epoxides, relies heavily on proper hazard pictograms and clear instructions for storage. Regulatory bodies demand detailed breakdowns of purity, water content, and handling rules. A bottle out of spec doesn’t just mean wasted money; it can mean unpredictable chemical behavior or ruined experiments. In modern setups, chemists use thin-layer chromatography and NMR signals to confirm both purity and structure. The technical standards reflect lessons learned over generations about what can go wrong in storage, shipping, or everyday use.
The Craft of Synthesis
Factory and lab synthesis for butadiene dioxide usually tracks back to well-tuned oxidation reactions, starting from 1,3-butadiene or one of its simple derivatives. People often use peracids like peracetic or m-chloroperbenzoic acid to coax both double bonds into epoxide forms in a single step. The chemical yield hinges on controlling temperature and moisture—let things stray, and you wind up with a messy batch of unwanted side products. Purification takes patience, using distillation or column separation to wash away impurities that can haunt downstream applications. These days, even famously inventive chemists work hard to improve yields and ease of manufacture, with some driving toward “greener” peroxides or enzymatic alternatives.
Core Chemical Reactions and Tweaks
Butadiene dioxide pulls its weight in a broad sweep of chemical modifications. Its dual epoxide rings open up in the presence of acids, bases, or most nucleophiles—turning into diols, amino alcohols, or glycols, depending on what you throw at it. This means it fits perfectly for ring-opening polymerizations or as a handshake molecule for building bigger, fancier chemicals. The fact that you can target either one or both epoxide groups brings a lot of flexibility. For the old-school organic chemist, that’s the sort of functionality that makes a compound worth a second look.
Aliases in the Lab and in Papers
If you pull up research articles or older patent filings, you’ll find butadiene dioxide hiding under a few different names: 3,4-epoxy-1,2-epoxybutane, 1,2:3,4-diepoxybutane, and simply diepoxybutane. The International Union of Pure and Applied Chemistry tends to favor systematic names, but old habits die hard, so chemists bounce between these labels depending on where they learned their trade. Rather than trapping us in endless footnotes, this jumble of synonyms just shows how science moves forward by building on what’s come before.
Staying Safe in Practice
Any professional working hands-on with butadiene dioxide respects the strict safety standards put forward by regulatory authorities. You don’t approach this molecule in shorts and a T-shirt. Respirators, chemical-resistant gloves, and tight-fitting safety goggles count as minimum protection in research or industrial settings. Storage involves tight seals and cool environments, since slow leaks or spills can irritate airways or spark unwanted reactions. Training programs focus on spill control, first aid for splashes or inhalation, and ways to minimize fire risk. The legacy of chemical safety rules—often written in blood—reminds everyone that even “small” molecules can become major headaches if taken lightly.
Where It Sees Action: Real-World Uses
Butadiene dioxide is more than a laboratory toy. It’s turned up as a specialty cross-linking agent for creating advanced resins and polymers. In synthetic chemistry, it acts as a building block for drugs, agrochemicals, and specialty fibers, offering unique geometric features that beef up the physical properties of finished materials. Some formulations use it to fine-tune the mechanical resistance of plastics, leveraging the added oxygen atoms for better durability or reduced flammability. Its reactivity creates both opportunity and danger in research settings, with scientists taking full advantage of its double epoxide nature for crafting complex molecules or probing disease mechanisms.
Digging Deeper: The R&D Environment
Researchers have spent years probing the deeper chemistry behind butadiene dioxide. The emphasis has shifted lately toward finding gentler, cleaner routes for synthesis—green chemistry isn’t just a buzzword, but a necessity, especially as environmental standards toughen. Teams in well-funded labs use advanced computational modeling to predict new reactions and unravel side products. Biochemists harness butadiene dioxide as a tool for DNA crosslinking and mutagenesis research, taking advantage of its double epoxide structure to disrupt genetic material in highly controlled settings. That research, in turn, brings the compound into the orbit of toxicologists and policy makers.
Facing Up to Toxicity: What Science Tells Us
There’s no denying the concerns around butadiene dioxide’s toxicity. Multiple studies have established its potent ability to cause DNA damage—enough so that it’s often used as a positive control in mutagenicity tests. In rodents, chronic exposure leads to increased cancer risk, while in cell cultures, it triggers chromosome breakage. That’s enough evidence for public health authorities to flag it for strict regulation and call for exhaustive personal protection protocols. Industry users rely on closed systems and real-time air monitoring to keep workplace exposure far beneath legal limits. The public rarely encounters butadiene dioxide directly, but occupational safety rules and environmental monitoring stay on high alert; no one wants to repeat the mistakes of the past with unchecked chemical hazards.
What’s Coming Down the Road
The future for butadiene dioxide holds a blend of tough challenges and intriguing possibilities. Green chemistry approaches keep pushing for cleaner routes, aiming to replace hazardous reagents and cut down on waste. At the same time, the compound’s role in DNA research and cancer biology keeps it in the crosshairs of policymakers who have to balance its utility against the risks. Demand for tougher, lighter, and more sustainable plastics gives polymer chemists reason to keep the molecule in rotation. As industries adapt to tighter global environmental laws and public scrutiny, butadiene dioxide illustrates just how many layers a single chemical compound can hold—a story shaped by invention, caution, and a stubborn push for progress.
What is Butadiene Dioxide used for?
The Real-World Use Case
Butadiene dioxide doesn’t show up in daily conversation. Most folks haven’t heard of it, but this chemical quietly plays a role in the background of modern life. I remember my first job in a polymer lab, surprised by how everyday products come from materials that sound like science fiction. This compound helps craft synthetic rubbers and plastics, things we rely on every single day. Car tires, hoses, and protective gloves all owe something to it.
Why Manufacturers Rely on This Compound
Rubber production often hinges on the ability to control flexibility and resistance. That’s where butadiene dioxide comes in. Blending it with other chemicals can offer manufacturers some real control over how their products handle things like stress, heat, and abrasion. Stronger and longer-lasting consumer goods reflect this behind-the-scenes improvement. According to the American Chemical Society, adding specific compounds to the polymerization process tweaks the final product’s toughness and elasticity. Butadiene dioxide is no exception.
Looking at the Bigger Picture
Adding a chemical like butadiene dioxide isn’t just about better tires or rubber boots. It connects to larger issues— durability helps conserve resources. Fewer products tossed away means lower demand for raw materials and energy. Waste won’t pile up as fast. For anyone thinking about sustainability or circular economy, the benefit goes beyond performance. For example, in the automotive sector, more durable rubbers mean tires last longer before heading to the landfill. This backs up reports from the World Business Council for Sustainable Development, which emphasizes longer product life as a key factor in resource conservation.
Worker Safety and Handling
My experience in labs has taught me that working with chemicals rarely looks like a TV commercial. Butadiene dioxide, like many chemicals, requires careful handling. Exposure to volatile organics can cause trouble for workers’ health. Regulations around labeling and protective gear remain strict— you’ll see workers in goggles and gloves, not because it looks cool, but because mishaps hurt. The Occupational Safety and Health Administration (OSHA) has set clear standards for how to handle chemicals like this in industrial settings. Companies must train workers, provide the right protective clothing, and monitor air quality where this substance is used.
Better Practices and Safer Alternatives
One thing folks in the chemical industry don’t talk about enough: improvements come from both better chemistry and smarter safety rules. Green chemistry research looks for substitutes that give similar results without the health or environmental risks. For example, researchers study how alternative monomers can match traditional chemical results, aiming to sidestep possible long-term issues. The U.S. Environmental Protection Agency (EPA) encourages companies to track their emissions and seek out less hazardous inputs for rubber production.
The Path Forward
Making products tougher, stretching their useful life, and reducing waste all matter in a world trying to cut down on pollution. Butadiene dioxide, while technical in name, rolls right underfoot in everyday uses. The focus stays on protecting workers, boosting durability, and keeping an eye open for new materials that handle the job even better and safer. Every step forward in the lab or on the shop floor adds up to real progress out in the world.
What are the safety precautions for handling Butadiene Dioxide?
What Is Butadiene Dioxide and Why Handle With Care?
Most folks outside chemistry circles haven't heard of Butadiene Dioxide. It's one of those chemical compounds you bump into in labs or factories making synthetic rubber and certain plastics. Problems show up if you treat it like any harmless substance. It can irritate lungs and skin, and if you breathe in high concentrations, things go from uncomfortable to dangerous.
Personal Experience With Chemicals in the Lab
Years ago in my first year working at a chemical plant, I watched a coworker rush through a cleaning task without proper gloves. His hands itched and burned within minutes—he never skipped PPE again. People sometimes cut corners because they're familiar with the job, ignoring the risks until they pay for it. The discomfort from chemical irritation or inhalation isn't something you shake off easily.
The Right Gear Prevents Accidents
Any time Butadiene Dioxide makes an appearance at work, the rules are clear: gloves, goggles, face shields, and long sleeves. Heavy-duty nitrile gloves protect from splashes and accidental contact. Regular safety glasses aren’t enough—full goggles fit better and keep fumes and drops away from your eyes. Lab coats or proper coveralls keep the material off your skin and clothes. If you aren’t wearing chemical-resistant gear, you’re gambling with your health, plain and simple.
Breathing fumes is another big risk. In small spaces, ventilation matters as much as gloves and goggles. Fume hoods or strong extractor fans keep vapors away from your nose and lungs. On days when the air system lags, headaches or coughs serve as quick reminders. Some workers use properly fitted respirators if the material goes airborne, especially when mixing or pouring.
No Eating or Drinking Near the Action
No food, no drinks, and definitely no open water bottles near Butadiene Dioxide. It’s too easy for tiny drops to sneak onto your sandwich or coffee. I’ve seen people brush off “no eating” rules, but it’s not just about tidiness—it’s about cutting down accidental ingestion. Even washing your hands isn’t always enough if supplies or surfaces get splashed and aren’t cleaned right away.
Storage and Spill Basics That Make a Difference
Keep anything with Butadiene Dioxide away from heat, sparks, or flames. This compound doesn’t need much encouragement to turn hazardous. Lock up containers tight and store them on lower shelves, and never next to acids or oxidizers—some combinations send you running for the exits. Spill kits should stay close by. Kits should include absorbent pads, neutralizing agents, waste bags, and instructions. Spills do happen in labs and plants, and a fast and confident cleanup cuts down on injuries and panic.
Training Isn’t Just Red Tape
The best safety posters and checklists in the world won’t matter if folks don’t take training seriously. Regular hands-on sessions build habits, and they’re more useful than a stack of unread manuals. Emergency drills save lives, not because accidents happen every day, but because they teach people how to think clearly when stress makes most of us sloppy.
The Real Takeaway
Butadiene Dioxide won’t cause trouble if you respect it and work smart. Relying on the basics—protective equipment, good airflow, careful storage, and proper cleanups—makes all the difference. Companies can go further by building a culture where safety isn’t just a rule, but a daily expectation. After seeing firsthand what happens when folks skip steps, taking these precautions feels like common sense, not bureaucracy.
Is Butadiene Dioxide hazardous to health?
What Kind of Chemical Is Butadiene Dioxide?
Butadiene dioxide sounds like the sort of compound best kept in a lab. Most people won’t bump into it daily, but for folks working in industries tied to plastics and rubber, exposure risk creeps closer. This chemical comes up as a byproduct during certain synthetic processes, so it isn’t entirely rare behind factory doors. Evidence about how butadiene dioxide interacts with the body keeps showing us that it’s not the kind of thing anyone would want in their system.
What The Research Says
Scientific studies point to serious effects: researchers have flagged links between butadiene family compounds and increased cancer risk. Health agencies track this substance because animal experiments have turned up cases of tumor growth when they breathe in similar chemicals over time. Even low-level exposure in the air—especially in closed spaces—may lead to headaches, dizziness, and irritation of the eyes and throat.
A few decades ago, I worked for a tire plant where chemical safety was only half the priority it is now. Several of my older coworkers saw chronic respiratory symptoms, and many had spent years near vats and mixing tanks. We wore cotton masks. Modern labs and factories run with more oversight, and documented incidents over the last decade suggest the problem isn’t history just yet.
Who Should Worry About Butadiene Dioxide?
Factory personnel, lab technicians, and cleanup crews touch or inhale more of these chemicals than almost anyone else. Community members living near processing plants also wind up breathing trace chemicals when leaks or emissions slip past scrubbers. With large-scale operations, pollution can drift into neighborhoods.
The health effects raise alarms for vulnerable folks too—kids, elderly people, and anyone with lung problems. Chronic health issues don’t always show up right away, either. Continued exposure ramps up the odds of worse trouble down the road.
Regulations And Real-World Action
Government safety boards track chemical releases, but regulations have holes. U.S. Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency list strict limits for butadiene exposure. Many companies set in-house caps, but complaints from workers keep popping up. Sometimes equipment or monitoring just doesn’t keep pace with best practices. Part of the trouble comes from budget cuts and slow enforcement—not every plant sees regular inspections.
Communities near industrial sites have acted by monitoring air and pushing city leaders for stricter checks. Consumer groups share results to raise awareness. These grass-roots movements make it clear: people want upfront info about what floats through their air and sneaks into their water. Transparency and early action win trust.
What Makes The Difference?
Clean air and safe water shouldn’t hinge on which side of a plant fence someone lives. Installing high-efficiency scrubbers and better ventilation cuts down risk. Giving workers top-grade personal protective gear works, but training and culture matter, too. I’ve seen employees skip safety steps out of habit or hurry. Regular drills and open talk help drive home why it matters.
Routine health screenings spot symptoms early. Neighbors and watchdogs can press for plain-language reporting by local plants. Encouraging research into less risky alternatives for butadiene-related processes also pays health dividends.
Looking Forward
Community voices keep the pressure on companies and regulators. Making safety a routine part of the job and holding everyone—from management to regulators—accountable gives everyone a better shot at staying healthy. Respect for evidence, vigilance about rule-breaking, and smart investments in safer tech set the tone for real change.
What is the chemical formula of Butadiene Dioxide?
Getting to the Basics: What’s Butadiene Dioxide?
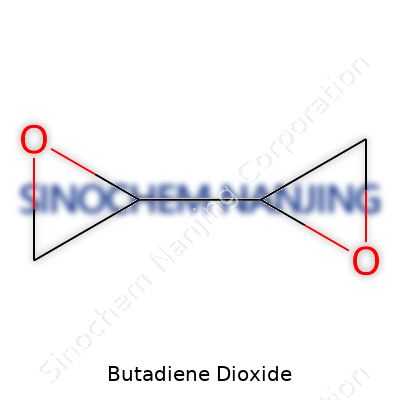
Walking through any industrial chemical catalog, you’ll see hundreds of unfamiliar names. Butadiene Dioxide stands out for anyone studying organic synthesis or working on polymers. This compound, sometimes called 3,4-epoxy-1-butene or 1,2-epoxy-3-butene, draws interest because it acts as both a building block and an intermediate in chemical manufacturing. The formula for Butadiene Dioxide is C4H6O2; its structure contains four carbon atoms, six hydrogens, and two oxygens.
Understanding the Structure: Why It Matters
C4H6O2 sounds simple, but the arrangement of atoms makes all the difference. In Butadiene Dioxide, those two oxygen atoms form epoxide groups. Epoxides react quickly and with all sorts of reagents, so chemists value them for making plastics, synthetic rubbers, and specialty coatings. In academic labs, that molecular structure becomes a way to teach students about reactions involving strained rings and reactive functional groups.
Why Butadiene Dioxide Isn’t Just Another Chemical
Some compounds grab attention because of their color or smell. Butadiene Dioxide grabs it because of reactivity. Having studied in an organic chemistry lab, I’ve seen the challenges and excitement it brings. Small molecules with epoxide rings often act like loaded springs—ready to react with nucleophiles and open up new chemical possibilities. The double bond and the epoxide feature provide two distinctive spots for transformation. This dual nature shapes how it gets used in synthesis. Chemists use the strained ring system to launch all sorts of chemical reactions that wouldn’t happen with a plain, open-chained molecule.
What’s the Big Deal in Industry?
Industrial chemists see Butadiene Dioxide as more than a teaching aid. They use it in processes where both reactivity and stability matter. Its presence in polymer research stretches its impact across tires, adhesives, and electrical insulators. Manufacturers depend on intermediates like this to develop more flexible materials or to fine-tune how quickly rubber hardens. In countries with strong rubber industries, having access to the right intermediates—like Butadiene derivatives—keeps product quality high and costs manageable.
Risk Doesn’t Take a Day Off
One thing about reactive chemicals: they tend to bite back if safety gets ignored. Butadiene Dioxide can irritate skin and eyes, and its vapors call for proper ventilation. The same features that make it attractive to researchers—small size, strained ring, high energy—make it dangerous in the wrong hands. My own experience has taught me the value of goggles and gloves, no matter how short the handling time. Chemical injuries take weeks to heal, and there’s no glory in shortcuts.
Room for Better Practices
Industry often chases efficiency, but safety deserves a permanent spot in that race. Good ventilation, strict labeling, and proper chemical storage don’t just save lives—they keep production running without interruptions. Regulations need to stay up-to-date to reflect new uses and hazards. Universities also play a big role: students who practice safe handling early don’t forget those lessons later. If industry, regulators, and academics keep communication open, handling chemicals like Butadiene Dioxide won't come down to luck.
Innovation Through Attention
Butadiene Dioxide’s formula stays the same, but its uses keep changing. Researchers find new catalysts, new polymerization techniques, and clever ways to add value. Every new discovery starts with understanding the basics and taking safety seriously. In a world where sustainability and efficiency matter more than ever, old compounds can find new jobs—if the right minds look at the formula and see more than just numbers on a page.
How should Butadiene Dioxide be stored?
No Room for Guesswork
Anyone with experience in chemical storage knows, comfort comes from confidence. Safety and quality control always take the front seat, especially with chemicals like butadiene dioxide. This isn’t a compound you stash just anywhere. Its volatility doesn’t invite shortcuts or improvisation. One misstep doesn’t just cost money—it risks health, supply chain integrity, and a company’s reputation.
Why Temperature and Ventilation Matter
Walk into any responsible facility, you notice how much emphasis sits on temperature control. Butadiene dioxide reacts to heat in a way that can rattle even seasoned managers. Even a moderate spike can set off degradation or pressure build-up. I’ve seen operations invest heavily in chilled, well-ventilated rooms for a reason. Poor airflow lets vapors accumulate, increasing the threat of spills and respiratory incidents. Reliable exhaust systems and environmental monitoring don’t just tick boxes for inspectors. They give peace of mind to teams who clock in daily. Even seasoned chemical handlers keep eyes on sensors and never rely on luck.
Containment—What Works, What Fails
There’s no wisdom in storing butadiene dioxide in generic off-the-shelf barrels. Stainless steel earns its place here. No one wants containers corroding from within or leaching harmful substances. My experience says to avoid vague sourcing: every supplier’s specs belong under a microscope. If a lid doesn’t snap shut tightly and show proof of rating, it invites trouble. Over years, I’ve watched companies lose tens of thousands trying to recoup from subpar container choices. Double-sealed drums with tight gaskets hold up the best and landfills see enough waste without throwing failed packaging onto the pile.
The Human Factor—Training Saves Lives
All the sensors and safeguards in the world mean nothing if people treat them like scenery. I’ve witnessed both ends: teams that run regular drills and those who learn protocols only after a near-miss. Annual hazmat updates make a difference. Workers who know how to spot container bulging or odor leaks react quickly and avoid injuries. Managers who walk the floor, ask questions, and encourage reporting foster a culture where warning signs never go unnoticed. Real trust grows from action, not policies on a wall.
Documentation Isn’t Just Red Tape
Some see logs as busywork. In reality, chemical inventory and audit trails solve problems before lawyers or regulators show up. Reliable records mean you can prove conditions remained stable and materials didn’t disappear. Systems that tie storage batches to lot numbers remove guesswork if an incident arises. Digital checklists and time-stamped entries become the backbone for continuous improvement—and workers sleep easier knowing each shift left a breadcrumb trail of responsible action.
Better Tech, Smarter Solutions
Technology keeps raising the bar. Real-time monitors hook into mobile alerts, so supervisors spot trouble early. Barcoding systems shave off time and mistakes, a benefit that pays off at every inventory check. Sensor-triggered containment shutoffs take the element of surprise out of chemical management. The message from the field matches what the experts say: investing in good tech upfront prevents costly clean-ups and regulatory fines down the road.
Responsibility Supports the Whole Chain
Storing butadiene dioxide safely connects personal responsibility to public trust. Disasters become headlines for years, staining industries and harming communities. A careful approach, built on training, technology, and common sense, protects workers, neighbors, and the bottom line. Experience teaches that cutting corners may save pennies today but ends up costing far more.
| Names | |
| Preferred IUPAC name | 1,2-dioxacyclobuta-3,4-diene |
| Other names |
1,2-Epoxybutene 1,2-Butadiene oxide 1,2-Butadiene dioxide 1,2-Butadiene epoxide |
| Pronunciation | /ˌbjuːtəˈdaɪiːn daɪˈɒksaɪd/ |
| Identifiers | |
| CAS Number | 106-59-2 |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:52255 |
| ChEMBL | CHEMBL116294 |
| ChemSpider | 77901 |
| DrugBank | DB04161 |
| ECHA InfoCard | 030000017924 |
| EC Number | 207-478-5 |
| Gmelin Reference | Gmelin 203020 |
| KEGG | C05930 |
| MeSH | Crotonolactone |
| PubChem CID | 62248 |
| RTECS number | FF0700000 |
| UNII | 19E9T2956N |
| UN number | UN1163 |
| CompTox Dashboard (EPA) | DTXSID1027916 |
| Properties | |
| Chemical formula | C4H6O2 |
| Molar mass | 86.0892 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 0.965 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.17 |
| Vapor pressure | 1.34E+4 mmHg (25°C) |
| Acidity (pKa) | 16.8 |
| Basicity (pKb) | 1.09 |
| Magnetic susceptibility (χ) | -52.0e-6 cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 2.94 mPa·s |
| Dipole moment | 1.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 294.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -72.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2417 kJ/mol |
| Pharmacology | |
| ATC code | C01EB10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H315, H319, H331, H335, H341, H350 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | **2-4-2-W** |
| Flash point | -76 °C |
| Autoignition temperature | Autoignition temperature of Butadiene Dioxide is 415°C |
| Explosive limits | Explosive limits: 2.5–12% |
| Lethal dose or concentration | LD50 oral rat 155 mg/kg |
| LD50 (median dose) | LD50 (median dose): 330 mg/kg (mouse, oral) |
| NIOSH | NIOSH: EI9100000 |
| PEL (Permissible) | 1 ppm |
| REL (Recommended) | 2 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Butadiene Vinyl ethylene carbonate |

