Bromotrifluoroethylene: More than Just a Chemical—A Commentary
Historical Roots: Pushing Boundaries in Chemistry
Bromotrifluoroethylene comes from a long line of chemical discoveries that have shaped entire industries over the past century. The substance popped up as chemists kept tinkering with fluorinated hydrocarbons during the mid-20th century, an era fueled by a relentless appetite for new plastics and specialty monomers. Digging into the earliest reports, researchers saw something promising in this clear, volatile liquid: the tricky bromine atom attached to a double bond alongside three electronegative fluorine atoms hinted at unique reactivity. Stories from the 1950s and 60s paint a picture of a niche material—never a household name, yet quietly pivotal in the development of advanced fluoropolymers and specialty coatings. Those who worked in academic and industrial chemistry departments saw bromotrifluoroethylene as a route not only to new products but also to a deeper understanding of how halogens and fluorine shift the electronic landscape of simple molecules.
Setting the Stage: What This Molecule Brings to the Table
Bromotrifluoroethylene catches attention in a laboratory because of its combination of volatility, reactivity, and stability in certain conditions. With a boiling point hovering just above room temperature, this clear, colorless liquid evaporates cleanly—a trait useful for controlled addition to reaction flasks but demanding focus on ventilation. Its structure—a vinyl group featuring bromine and three fluorine atoms—creates a high barrier to unwanted side reactions, a perk for those trying to build polymers with precise architectures. Chemists note its slight odor and high density, a physical clue to its halogen content, and they respect its aggressive behavior toward some metals and its susceptibility to decomposition under high temperatures or UV exposure. Measuring out a few milliliters, you can feel the weight of its promise and its risks.
The Inside Story: Physical & Chemical Personality
No one who has handled bromotrifluoroethylene soon forgets its physical quirks. At normal pressure, it remains liquid down to subzero temperatures but surges into vapor rapidly when things heat up. Its high electron density—thanks to the fluorine and bromine—makes it a logical candidate for nucleophilic addition reactions, and people who work with complex organometallic syntheses appreciate its ability to create strong carbon-fluorine and carbon-bromine bonds. The molecule resists oxidative degradation and can persist in an environment unless specifically targeted by reactive species. Unlike more docile hydrocarbons, bromotrifluoroethylene demands skill—its double bond draws in many reagents, leading to potentially vigorous, sometimes exothermic, reactions. This is the chemistry that rewards patience, knowledge, and sharp observation.
Getting Specific: Mixing Fact With Practice
The technical side of bromotrifluoroethylene often gets overlooked outside the circles of research and specialty manufacturing. People ask: What makes it tick? A look at its labeling in any well-stocked chemical storeroom reveals a set of warnings—its volatility makes secure sealing necessary, and you never store it alongside reactive metals or strong reducing agents. Its identification numbers matter for safe handling, but in the end, human memory serves best for repeated use. The chemical’s liquid state, sharp smell, and relatively high density serve as daily reminders of its halogenated backbone. Proper storage keeps it stable for months, yet once out in the open, time becomes a key factor. Every lab that uses this compound has its own in-house rules, fine-tuned by trial and error, taking general guidance from regulatory bodies but always going the extra mile with local know-how.
How People Make It: From Laboratory Benches to Industrial Vats
In my own experience, watching bromotrifluoroethylene form rarely feels routine. Most preparation methods rely on careful halogen exchange reactions, starting from simpler trifluorinated ethanes, often under tightly controlled temperatures and using solvents that won’t steal away the halogens. Skilled synthetic chemists know that the bromination of trifluoroethylene doesn’t leave much room for error—over-brominate, and you lose your product; under-react, and you wind up with a mixed bag of leftovers. This isn’t a substance you create on a whim: producers design reactors with corrosion in mind, keep a close eye on rising pressures, and use robust analytical monitoring to ensure a pure output. It’s the kind of process that rewards people who spend years tuning their craft, learning its quirks by heart, and respecting both the chemistry and the engineering challenges.
Reaction Patterns and Chemical Tweaks
Chemists enjoy the challenge of manipulating bromotrifluoroethylene’s double bond, which welcomes a wide slate of nucleophiles. The compound stands out in radical addition reactions as well as in typical halogenation or even polymerization sequences. Once, in a small research group, we watched it participate in a controlled reaction, serving up a fluorinated backbone perfect for a new generation of specialty membranes. Synthetic routes often involve adding organometallic reagents or experimenting with cross-coupling under palladium catalysis. The wide reactivity also means unwanted byproducts appear if something goes just slightly off plan—a reminder that working with such energetic molecules keeps you humble. Decades of publications map out these pathways, not just for academic interest but because every tweak opens new doors for materials science and industrial chemistry.
What Else Do People Call It?
If you dig through the literature, you’ll spot bromotrifluoroethylene under several aliases, including trifluorovinyl bromide and 1-bromo-1,2,2-trifluoroethylene. Sometimes, simple abbreviations like BTFV or BTFE pop up, another sign that those in the know find shorthand appealing. Newcomers need to keep an eye on synonyms in global patent filings and journal articles, avoiding confusion that can lead to errors in ordering or regulatory paperwork. This array of names testifies both to the chemical’s versatility and to the cross-border, multi-discipline collaborations that have touched its story.
Staying Safe: Doing Things the Right Way
Anyone who has cracked open a bottle of bromotrifluoroethylene knows safety doesn’t come from manuals alone. The chemical evaporates quickly, so fume hoods and adequate airflow turn from recommendations into must-haves. Splash-resistant gloves, chemical goggles, and sturdy lab coats form the standard uniform. In the event of a spill, workers limit spread by controlling vapor with absorbent pads, isolating contaminated zones, and breathing through certified filters only if all else fails. Long experience shows why no one ever leaves this compound unsupervised near open flames or heated surfaces: fire risks and the release of corrosive gases like hydrogen fluoride make you pay attention. Following documented standards—often written in the scar tissue of past mistakes—keeps students and researchers out of the ER and the building in one piece.
Why Industry and Academia Value It: The Application Angle
In the application sphere, bromotrifluoroethylene unlocks doors closed to less specialized compounds. Its most prestigious role comes as a monomer or co-monomer for advanced fluorinated polymers. Think membranes that resist swelling and chemical attack, wire coatings surviving in aerospace circuits, or gaskets holding up under corrosive fluids. Chemists in research labs take advantage of its reactivity to create building blocks for pharmaceuticals, agrochemicals, and functionalized surfaces. Its energy-rich double bond and halogen loadout have tempted generations of scientists to cook up new ligands, catalysts, and additives that do something traditional hydrocarbons simply can’t. This kind of versatility keeps it on order sheets even as stricter regulations arrive.
The Research Hunt: Finding the Boundaries
Ongoing research keeps carving out new possibilities for bromotrifluoroethylene. In my years following synthetic and materials chemistry, I’ve seen it pop up in projects ranging from lightweight composites to next-generation barrier films. In universities and corporate labs, teams measure subtle shifts in reactivity, seeking better catalysts and fewer byproducts. Every so often, a group stumbles on a quirky new property: perhaps a surprising compatibility with an exotic solvent, or a new way to link it into a repeating polymer chain. Much of today’s R&D focuses on pushing past performance limits, with an eye toward energy applications, microelectronics, and membranes for challenging separations. Investment in safer, greener synthetic routes shows that chemistry doesn’t just look back nostalgically—it adapts, often under regulatory and economic pressures, refining both the product and the process.
Toxicity: Accepting the Hard Lessons
Anyone who spends real time in chemical safety training hears about the hazards tied to halogenated ethylenes. Bromotrifluoroethylene, in particular, sits on lists for its acute toxicity, volatility, and stubborn persistence in some environments. Evidence from animal studies and accidental exposures points to risks for the lungs, nervous system, and skin, often through inhalation or splash contact. Overexposure can trigger headaches, dizziness, or even more serious respiratory symptoms. Experiences in industrial hygiene reinforce that proper containment, leak detection, and routine monitoring of air quality really do pay off. For decades, debate has swirled around long-term chronic exposure, so the sensible approach involves minimizing contact wherever possible. The trend favoring closed-system syntheses, automated transfers, and improved personal protective equipment isn’t just bureaucratic—it’s rooted in painful lessons that echo across the field. In my own circle, I’ve seen close calls, but those moments have always driven teams to triple-check procedures rather than shy away from valuable research.
Looking Forward: The Road Ahead
The story of bromotrifluoroethylene isn’t static. People are asking tough questions about sustainability, long-term toxicity, and the environmental fate of halogenated compounds. As regulatory frameworks tighten, especially in North America and Europe, those who depend on this chemical face increasing pressure to innovate—not just with the molecule itself, but in the ways they produce, handle, and recycle it. In my view, this pressure sparks better science: researchers push for catalysts that work under milder conditions, engineers develop safer process controls, and companies test greener starting materials. The push toward fluorinated materials with a lighter environmental footprint points to a new wave of research, where bromotrifluoroethylene may play a specialized but still important role. Students training today will shape how this chemistry moves forward, balancing curiosity with caution, and never losing sight of the need for both progress and responsibility.
What is Bromotrifluoroethylene used for?
What’s Bromotrifluoroethylene?
Most folks haven’t heard of bromotrifluoroethylene unless they’re buried in chemistry textbooks. In plain English, it’s a chemical compound with a pretty strong kick—it carries bromine and three fluorine atoms stuck to an ethylene framework. Its formula is C2BrF3. Not exactly a kitchen cupboard ingredient, but it’s found a place among chemists for good reasons.
How It’s Used
Bromotrifluoroethylene shows up most often as a building block for making special plastics and chemicals. Its main claim is in the world of fluoropolymers—a category of plastics that shrug off acids, heat, and sticking. Think of Teflon; it resists just about anything you throw at it, from frying eggs to strong industrial solvents. While Teflon comes from other cousins of bromotrifluoroethylene, this chemical steps up when companies need to make fluorinated films, custom coatings, or barrier layers that outlast and outperform regular plastics.
Manufacturers look to bromotrifluoroethylene when they need a source of both bromine and fluorine atoms in one package. Many commercial fluoropolymer applications, such as protective tapes, gaskets, or specialized tubing, spring from monomers produced with help from this compound. Electronics companies also value it: those thin, durable circuit boards and the insulators on data cables need just the right types of plastic to block heat, water, and static.
The Importance of Handling Safety
You probably won’t see this stuff in a local hardware store for good reason. Bromotrifluoroethylene is tricky—volatile, toxic if misused, and not a chemical to take lightly. Several government sources, including the U.S. Environmental Protection Agency and European safety agencies, flag it as hazardous if inhaled or handled without real training. Workers who have to use it get specialized safety gear, and labs tightly control its storage. Watching a chemist suit up for handling these chemicals brings home just how far the science community goes to keep everyone safe.
Too many accidents have happened in labs and factories where someone underestimated exotic chemicals. Companies are under strict regulations, and for good reason—one mistake can lead to long-term health issues, not to mention environmental problems if the stuff leaks into air or water supplies.
Finding Better Alternatives
The chase for greener chemistry is picking up everywhere. Researchers keep hunting for replacements with a lighter environmental touch. Some companies now design new monomers and polymers, hoping to cut back on persistent toxins and make recycling easier. Government incentives help chemistry teams develop processes that work with safer replacements and produce less waste.
Many folks in the field agree: the next step is high-performance plastics that break down cleanly or can be reclaimed with less hassle. Some startup labs even experiment with plant-based fluorinated compounds, something that would have sounded far-fetched ten years ago. People working with bromotrifluoroethylene today keep a close eye on these innovations, hoping for better ways to build tough, safe materials without the drawbacks.
Whether it’s tomorrow’s medical devices or the next generation of clean manufacturing, the pressure to improve doesn’t let up. Chemicals like bromotrifluoroethylene played a part in getting us here—now the goal is safer, smarter, cleaner solutions for everyone.
What are the safety precautions for handling Bromotrifluoroethylene?
Handling a Gas with Bite
Bromotrifluoroethylene isn’t something you’ll find in every chemistry classroom, but it shows up where specialized plastics or chemical synthesis come into play. This gas, clear and sharply reactive, can hit hard if you’re not careful. The sharp, sweet smell might be the first sign something’s leaked, but relying on your nose sets you up for trouble—good ventilation and monitoring equipment are far more reliable. OSHA and NIOSH both list it as hazardous, not just because of toxicity, but because it doesn’t take much to trigger nasty reactions when mixed with other compounds.
PPE Isn’t Just a Box to Check
I’ve seen folks trust a flimsy dust mask in places where full-on chemical respirators should be the rule. Bromotrifluoroethylene isn’t a risk you brush off with a pair of latex gloves. Splash-proof goggles protect your eyes since even brief contact irritates. Nitrile or PVA gloves act as a solid barrier. For any actual transfer of the gas, chemical-resistant aprons and sleeves (not just lab coats) close the gaps. It pays to double-check that respirator filters actually catch organic vapors—an oversight there invites chaos. Years ago, a rushed setup caused a hissing leak. Fumbling for a half-functional mask led to a lab evacuation and three trips to the eyewash.
Make Your Ventilation Count
Standard fume hoods aren’t a luxury here. Working out in the open invites low-level exposure that builds up—the classic case of a slow leak making people sick hours later. Good hoods pull vapors away fast and don’t just recirculate them. Monitoring tools—gas detectors with freshly calibrated sensors—let someone know if levels rise above safe limits. These tools can get expensive, but the cost tops out well below a hospital bill or facility shutdown.
Storage Calls for Respect
You can’t just tuck these cylinders away in a closet. Compressed gas rules apply, so locked cages, upright posture, and chain restraints become routine. Heat sources nearby? That’s asking for overpressure and a blown valve. Storing this material far from oxidizers, acids, or metals keeps runaway reactions at bay. Cylinder valves need a cap when not in use—commonly overlooked, but a lifesaver if something falls or bumps into the setup. Older labels fade or peel, so refreshing them ensures no one mistakes one gas for another during a busy shift change.
Response Plans Make the Difference
No matter how advanced the gear or strict the checklist, human error can sneak in. Quick exits and spill containment kits need clear labels and open access. The best teams run short drills so instincts kick in when real trouble hits. After witnessing a rushed tech fumble a valve and cause a cloud, it became clear: response is never just a theoretical exercise. Medical attention means bringing the right data sheet with exposure symptoms—paramedics move faster when they know exactly what they’re dealing with.
Building a Culture of Awareness
People trust safety protocols more after seeing how unforgiving these chemicals can be. Regular trainings, open conversations about mistakes, and leadership following their own rules all raise the bar. It isn’t just paperwork—it’s about keeping sharp so the next shift goes home intact. In a world tempted to cut corners, respect for the material always pays off, both in saved lives and kept reputations.
What is the chemical formula and structure of Bromotrifluoroethylene?
Getting to the Core Formula and Structure
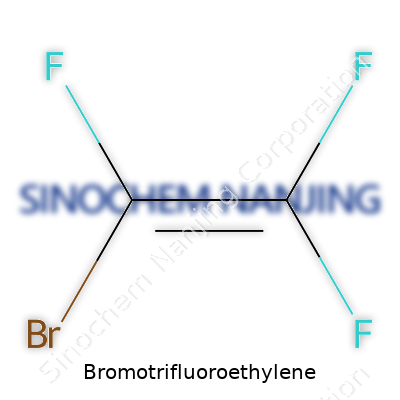
Bromotrifluoroethylene stands out with the chemical formula C2BrF3. To break that down, it holds two carbon atoms, three fluorine atoms, and one bromine atom. The structure isn’t some dense mystery out of a chemistry textbook. Picture the backbone of an ethylene molecule where hydrogen atoms get swapped out for more forceful partners—three fluorines and one bromine. This gives the structure Br–CF=CF2. So on a molecular level, you’ve got a carbon–carbon double bond, one carbon linked to a bromine and a fluorine, the other carbon tied to two more fluorine atoms. The double bond brings reactivity to the table, making this molecule both useful and risky, depending on who’s handling it and for what purpose.
Why Bromotrifluoroethylene Matters
Anyone working in chemical manufacturing knows the value packed into small, halogen-rich molecules like this. For industry use, bromotrifluoroethylene offers a platform for specialized reactions. The presence of both bromine and fluorine brings a unique punch—bromine provides a site for further functionalization, while fluorine delivers chemical stability and high electronegativity. This isn’t just academic posturing. In real-world applications, you see materials built from bromotrifluoroethylene showing resistance against harsh chemicals and remarkable staying power under heat or corrosive attack. That performance doesn’t come cheap or easy, so labs that work with this molecule respect its power and handle it accordingly.
Health, Safety, and Environmental Perspective
Chemists who’ve spent time with halogenated compounds know the downside—these molecules sometimes stick around in the environment, and their reactivity carries risks. Bromotrifluoroethylene is not an everyday material for DIY experimentation. This gas can irritate lungs and eyes, and inhaling it in any significant amount can cause trouble for the nervous system. People working around it hit the daily grind in gloves, goggles, and robust ventilation. Missteps in handling chlorofluorocarbons taught this industry harsh lessons, so extensive safety protocols exist for even a modest jar of bromotrifluoroethylene.
Beyond the lab, there’s another side—environmental stewardship. Perfluorinated compounds have already drawn scrutiny because they stick around and interact with our bodies and the broader environment. While the specific environmental quirks of bromotrifluoroethylene continue to get studied, its fluorine atoms signal long half-lives and potential as a persistent organic pollutant. Responsible companies recycle and destroy waste, instead of letting these gases drift into the atmosphere. Regulatory lines keep moving as research reveals the long view, but anyone working with bromotrifluoroethylene faces real-world pushback if corners get cut.
Improving Safety and Environmental Responsibility
There’s a push for routine safety training—real, hands-on drills, not just paperwork. Workers learn the limits for exposure and what to do if a leak occurs. Another piece is monitoring. Modern labs use sensors that sniff out leaks fast, triggering alarms before things spiral out of control. From an environmental angle, investment in scrubbers and closed-loop systems sees more uptake every year. Labs and factories working with bromotrifluoroethylene invest heavily in reclamation and thermal destruction technology, destroying any off-gassing at the molecular level to meet tough environmental standards. Chemical engineers and safety experts collaborate, combining deep knowledge with practical systems, which keeps both people and the environment on safer ground.
Experience at the Bench
I’ve seen the respect this molecule commands—whether it’s a wary glance when opening a cylinder or the extra checks on fume hoods before starting a reaction. For anyone in chemical research, learning the quirks and limits of chemicals like bromotrifluoroethylene sharpens the sense that science is both promising and unpredictable, and that the best chemistry comes from a place of practical caution as much as curiosity.
How should Bromotrifluoroethylene be stored?
Why Proper Storage Matters
Anyone who’s spent time in a lab knows chemicals rarely sit quietly. Bromotrifluoroethylene doesn’t break that pattern. With its reputation for volatility and the added trick of being a possible fire risk, storing it takes more than a dark shelf and a lock. It’s not paranoia—accidents involving unstable chemicals often start with sloppy storage, and nobody wants to read their facility’s name in the accident reports.
What Science and Experience Tell Us
Clear, hard facts help keep storage drama to a minimum. Bromotrifluoroethylene, with a boiling point around room temperature and low flash point, acts as a vapor creator at the drop of a hat. Stories float around about labs forced to air out entire rooms after a single poorly sealed bottle leaked. After working a summer job logging hazardous materials for an industrial supplier, I saw how a moment’s carelessness or a shortcut piles up future headaches.
Temperature control comes up constantly. Keeping this compound cool—usually below 10°C (50°F)—cuts down on pressure inside the container, which in turn helps avoid leaks or bursts. Regular refrigerators miss the mark because their thermostats swing too much and most don’t vent vapors. Flammable chemical refrigerators cost more, but you buy peace of mind with that price tag. Cold, consistent storage separates near-misses from disasters.
Specialized Containers and Labels
Glass bottles with PTFE-lined stoppers, or metal containers designed for pressure, become essential since plastics or soft caps don't hold up against aggressive fluorinated chemicals or creeping vapors. Clear labeling plays a bigger role than most give credit for. On a busy shelf, a missing hazard symbol or a faded label risks someone grabbing the wrong thing. Having seen close calls over barely legible bottles, adding clear hazard information and the storage date can make all the difference.
Ventilation and Emergency Precautions
Storing bromotrifluoroethylene in a dedicated chemical storage cabinet with active ventilation isn’t just overkill; it addresses invisible risks. Vapors hang out where they shouldn’t, and without fresh air exchange, small leaks become big problems. Facilities sticking to local fire codes and OSHA guidelines avoid many of the worst case scenarios, especially with periodic air tests and spill training. I once watched an otherwise airtight storage plan turn upside down from a bad ventilation fan that nobody checked in months.
Chemical spill kits, located close by, should offer everything needed to seal and clean up without scrambling for missing gear. These kits aren’t luxury—swift response means smaller incidents and less exposure.
Training Never Clocks Out
People treat chemical safety as something covered with a poster or a five-minute video, but proper storage needs reminders, drills, and walk-throughs, especially with something as twitchy as bromotrifluoroethylene. With turnover and new staff, frequent refreshers stop the drift toward shortcuts. In my early days, only constant reminders kept me from letting storage discipline slip.
Building a Safety-First Culture
Effort spent on secure, temperature-controlled, and well-labeled storage for bromotrifluoroethylene brings peace to everyone involved. Routine maintenance, twice-checked labels, and accessible spill equipment save more than time—they mean fewer emergencies and safer workspaces. Toxic vapor doesn’t forgive, so careful handling becomes a habit worth building every day.
What are the hazards and toxicity associated with Bromotrifluoroethylene?
Understanding the Chemical
Bromotrifluoroethylene shows up in specialty chemical labs and sometimes in larger chemical manufacturing setups. This compound draws interest because of its unique structure, but it can cause major risks if handled without care. Few people have even heard of it unless they've spent time in an advanced chemistry lab, and even fewer know about the dangers that come with it.
Direct Exposure Risks
Breathing in bromotrifluoroethylene gas or fumes attacks the respiratory system quickly. Immediate signs can include throat irritation, coughing, and chest discomfort. If a worker gets exposed for too long or without protective gear, the results head in a critical direction—breathing troubles, headaches, or worse. Accidental spills or releases in closed rooms have thrown me into some stressful situations in the past, forcing a full evacuate and emergency wash-downs.
Touching the liquid or vapor doesn’t go gently, either. Direct contact with skin may leave a nasty burn, and vapors sting the eyes. Chemical burns from this compound may linger far longer than the irritation from something like bleach or acetone. It also seeps through some gloves faster than you’d expect, making personal protection equipment checks a constant worry for lab managers.
Potential for Greater Health Impact
Long-term exposure can damage the liver and kidneys, based on animal studies and data supplied by occupational hazard sources. Fluorinated chemicals have a knack for sticking around in the environment and in the body, and bromotrifluoroethylene fits right into that category. Some reports suggest this compound may also disrupt the nervous system if inhaled at higher concentrations. Memory loss and problems with coordination don't always draw immediate links to chemical exposure, but toxicologists who dig deeper find patterns that point back to these sorts of chemicals.
Reactivity and Accidental Release
Heat and sparks turn bromotrifluoroethylene into a bigger issue. The chemical forms unstable peroxides over time—a fact confirmed by watching vials of this stuff slowly change on the shelf, even under lock and key. If left alone, pressure builds up and the risk of explosion rises. Spills into drains or open ground threaten water and soil far from the original site, especially since this class of compounds doesn’t break down easily.
Sensible Approaches to Safety
Fume hoods and strict airflow control dominate any lab routine dealing with this chemical. Double-gloving and frequent glove swaps become second nature for those who’ve learned the hard way. Emergency wash stations are kept next to storage locations—within reach, never across the hall. Clear labeling and real-time monitoring for leaks go hand-in-hand with training new team members, not once, but at every rotation.
Some labs use automated systems for container transport, aiming to keep humans at a safe distance. Others turn to closed-system synthesis, skipping any opportunity for fumes to reach air. Any leftover material needs neutralizing under tightly monitored conditions, with certified disposal professionals taking over the moment it’s sealed up.
Environmental and Community Impact
Concerns stretch beyond workers in labs and factories, touching neighbors and the ecosystem. Water near chemical plants has tested positive for related byproducts, raising alarms about long-term buildup and contamination. Local communities push for more transparency and accountability from companies using these kinds of chemicals.
Those who work with bromotrifluoroethylene every day understand that respect and vigilance matter as much as technical controls. New technology and better oversight may lower risks, but those risks never hit zero. The best chance for safety comes from staying alert, sharing experiences, and pushing for smarter, safer processes at every turn.
| Names | |
| Preferred IUPAC name | 1-Bromo-1,2,2-trifluoroethene |
| Other names |
1-Bromo-1,2,2-trifluoroethene 1-Bromo-1,2,2-trifluoroethylene BTF |
| Pronunciation | /ˌbroʊmoʊˌtraɪflʊəroʊˈɛθiːliːn/ |
| Identifiers | |
| CAS Number | '98-07-7' |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:51711 |
| ChEMBL | CHEMBL16251 |
| ChemSpider | 57316 |
| DrugBank | DB14095 |
| ECHA InfoCard | 100.995. |
| EC Number | 1.1.1.7 |
| Gmelin Reference | 184039 |
| KEGG | C18729 |
| MeSH | D001939 |
| PubChem CID | 6383 |
| RTECS number | KK5075000 |
| UNII | X73B3B873E |
| UN number | UN2418 |
| CompTox Dashboard (EPA) | DTXSID0049270 |
| Properties | |
| Chemical formula | C2BrF3 |
| Molar mass | 165.912 g/mol |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 1.831 g/mL at 25 °C |
| Solubility in water | insoluble |
| log P | 1.9 |
| Vapor pressure | 64.41 psia (25 °C) |
| Acidity (pKa) | pKa = -8.3 |
| Magnetic susceptibility (χ) | -29.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.252 |
| Viscosity | 0.631 cP (25 °C) |
| Dipole moment | 2.38 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 348.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −674.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1128.6 kJ/mol |
| Pharmacology | |
| ATC code | D08AX01 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H301, H331 |
| Precautionary statements | P210, P261, P271, P304+P340, P305+P351+P338, P309+P311, P403 |
| NFPA 704 (fire diamond) | 1-4-0 |
| Flash point | -20 °C |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Explosive limits | Explosive limits: 5.9–28% |
| Lethal dose or concentration | LD50 (oral, rat): 250 mg/kg |
| LD50 (median dose) | LD50 (median dose): mouse oral 4000 mg/kg |
| NIOSH | NIOSH = "LB8825000 |
| REL (Recommended) | 24 months |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Halocarbon Chlorotrifluoroethylene Trifluorochloroethylene |

