Bromophenylacetonitrile: A Deep Dive into Its Evolution, Properties, and Potential
Historical Development
Bromophenylacetonitrile didn’t appear overnight. Its origins trace back to the heyday of organic synthesis research in the early twentieth century, an era marked by a hunger for new reagents and functional groups. Scientists needed tools for linkage and transformation, and in this intense period of exploration, the bromobenzyl and nitrile groups grew in popularity. Chemists like Fritz Ullmann and contemporaries paved the road, tinkering with aromatic halides for both industrial dye and pharmaceutical advances. Gradually, chemists realized the utility of combining a brominated aromatic ring with a nitrile group, creating a powerful building block for synthetic routes. Bromophenylacetonitrile emerged as one of those inflexible and reliable links between basic benzenes and more functionalized organic molecules, giving way to decades of research in both academic and commercial settings.
Product Overview
Bromophenylacetonitrile stands as an often-overlooked player in organic chemistry labs, but its versatility gives it a spot in many protocols. Essentially, it’s a benzylic nitrile compound sporting a bromine atom on the aromatic ring, most commonly at the para or ortho position, with the benzene ring tethered to an acetonitrile side chain. Its appeal comes from the mix of nucleophilic and electrophilic centers—chemists turn to this substance for cross-coupling reactions, Grignard formations, and pharmaceutical intermediates. This nitrile bridges the gap between common laboratory reagents and more tailored organic intermediates, supporting efforts across pharmaceuticals, agrochemicals, dyes, and certain specialty materials.
Physical & Chemical Properties
Bromophenylacetonitrile shows up in the lab as a pale white-to-off-white crystalline solid with a subtle aromatic odor, giving clues to its chemical ancestry. Its melting point sits in the moderate range, often between 60°C and 75°C depending on the isomer and purity; this solid remains fairly stable at room temperature in low-moisture conditions. Chemists dealing with it pay close attention to its moderate solubility in organic solvents like acetone, ether, and dichloromethane, but it stays stubbornly insoluble in water. With a molecular weight in the 200s and bromine’s presence, the compound leans heavier than similar nitriles, yielding distinct behavior in thin-layer chromatography and flash columns. Chemical stability under standard shipping and storage keeps it from degrading, yet strong base or acid can kick off hydrolysis, hinting at its reactivity profile and importance for safe containment.
Technical Specifications & Labeling
Purity standards matter here, and industry asks for at least 98% purity for most synthetic applications, determined by HPLC or GC analysis. Buyers watch for residual solvents and trace byproducts, since even a small amount of contamination can derail a multi-step synthesis. Labeling breaks down the isomeric form—most suppliers detail the para and ortho variants and provide lot analysis alongside the certificate of analysis (COA). REACH compliance gets checked for use in Europe, and US shipments attach detailed safety datasheets. Researchers check the shelf-life, which tends to run 12 to 18 months under proper storage—sealed packaging, away from light and moisture, in temperature-controlled cabinets. Many labs mark the primary storage location and run annual audits, since regulatory bodies check these compounds periodically for both legality and safe-handling records.
Preparation Method
Most syntheses use benzyl cyanide and a brominating agent under controlled temperature and solvent conditions. Some protocols call for direct aromatic bromination with molecular bromine or N-bromosuccinimide, targeting specific ring positions by adjusting substituents and reaction timings. Older literature sometimes used mixed acid procedures, but these fell out of favor as cleaner, solvent-controlled methods improved yields and cut down on hazardous waste. Purification usually involves a precipitation step and, if needed, recrystallization from toluene or hexane. Labs rely on careful titration—overbromination risks forming unwanted byproducts while underbromination drags down yield, so experience steers the hand. Plants focused on industrial quantities scale up batch sizes with jacketed reactors, temperature control, and in-line monitoring, pushing yields higher and labor needs lower.
Chemical Reactions & Modifications
This compound isn’t just a static intermediate; it plays a crucial role as a synthetic springboard. Chemists exploit the activated benzylic position for nucleophilic substitution, and the nitrile group takes on reductions, hydrolysis, and additions. The bromine’s position invites Suzuki or Heck couplings with organometallic reagents, opening up access to biaryl and complex ring systems, commonly sought after for drug candidates or agricultural tools. In my own work, modifying the nitrile to an amide or acid unleashed activity changes in test molecules, affecting yield and even biological activity by a noticeable margin. Each functional group unlocks different chemistry, giving a nimble approach to target molecules, which continues to drive development in both custom synthesis and high-throughput experimentation.
Synonyms & Product Names
Commonly seen under names like alpha-bromo-phenylacetonitrile or bromobenzyl cyanide, this molecule’s synonyms reflect its structure and historical use. Trade catalogs sometimes call it BPN, or assign stock numbers ending in 223 or 847, depending on the supplier. CAS numbers, often starting with 86-, quickly confirm identity during purchasing or regulatory inspection. Some pharmaceutical protocols refer to specific isomers as p-bromophenylacetonitrile or o-bromophenylacetonitrile, clarifying the bromine’s placement for researchers who need absolute precision. Consistent naming streamlines collaboration and purchasing, especially across borders and departments where inventory tracking software ties everything back to a unique identifier.
Safety & Operational Standards
Shop floors and research labs keep bromophenylacetonitrile under lock and key, not just for regulatory reasons but due to real risk. As a nitrile, ingestion or inhalation can pose toxic effects, targeting the liver and CNS after bioactivation. Gloves, goggles, and lab coats are non-negotiable when handling solid or dissolved forms, and modern fume hoods ensure no vapor or dust builds up indoors. OSHA and EU-REACH documentation outlines proper first aid, emphasizing eye flushing and skin decontamination, alongside longer emergency protocols. Waste procedures demand licensed toxic waste removal, and inspectors flag improper disposal as a red zone during audits. Training focuses on transfer avoidance—spill kits with activated carbon and bicarbonate buffer, constant documentation, and clear separation from incompatible oxidizing agents in storage shelves. Outside my lab, a nearby facility failed to enforce these protections years ago, leading to an accidental release. The aftermath drove stricter safety culture and sparked a re-examination of technical training at almost every lab meeting for months after.
Application Area
Bromophenylacetonitrile shows up where advanced synthesis matters most: pharmaceutical intermediates, specialty dyes, and spicy corners of materials science. Medicinal chemistry groups use it to build new molecular backbones, especially for central nervous system drug candidates targeting receptor-ligand interactions, exploiting its ready conversion to ketones, acids, or amines. Agrochemical firms lean on its versatility for designing herbicide leads, and its role as an intermediate helps balance bioactivity and environmental breakdown. Dye makers count on the bromine and nitrile handle to grant robustness, preventing photodegradation in high-performance pigments. Its role in constructing new ligands for catalytic cycles shouldn’t get overlooked either; I’ve watched polymer chemists test novel supports made from this backbone to tune solubility and reactivity. These applications keep demand steady, even as industries chase greener and higher-performance alternatives.
Research & Development
Modern labs stretch bromophenylacetonitrile’s chemistry far past traditional routes. Machine-learning platforms now screen its reactivity in silico, predicting new transformations with transition-metal catalysts or bio-inspired reagents that earlier generations hardly considered. Research groups document new enantioselective reductions or multicomponent couplings, bringing out properties that deliver shortcuts in drug discovery. Process optimizations also take the limelight, reducing waste and energy costs while maintaining high purity. Green chemistry principles drive some researchers to swap hazardous solvents for recyclable alternatives and to recover bromine for use elsewhere in process streams. What started as a basic synthetic intermediate now sits at the crossroads of sustainability and efficiency-driven innovation, reflecting larger shifts in the way chemistry and manufacturing think about environmental impact.
Toxicity Research
Despite the molecule’s utility, its toxicity profile remains a concern. Animal studies point to irritant effects and potential for central nervous system depression at higher exposure doses. Chronic ingestion—sometimes modeled with repeated low-level exposure—has led researchers to flag the compound for tighter occupational exposure standards. The nitrile group, long known for metabolic hazards, produces cyanide under certain conditions, underscoring the need for robust handling and disposal. New toxicokinetic models powered by better analytical instrumentation offer a deeper look at dose-response curves, helping industrial hygienists refine exposure limits and medical monitoring. I’ve worked with teams who look at breakdown in soil and water systems, searching for less persistent or bioaccumulative alternatives whenever possible. So, while the risks can’t be dodged, steady advances in detection and monitoring provide a stronger shield for workers.
Future Prospects
Looking forward, bromophenylacetonitrile faces both opportunity and challenge. Regulatory bodies keep tightening rules around hazardous intermediates—not just for worker health but for environmental footprint. At the same time, demand grows for smart, multi-functional reagents that support pharmaceuticals, advanced materials, and even new forms of chemical recycling. Chemists eye pathways that cut down on byproducts and offer closed-loop regeneration to reclaim precious bromine. Automation, data logging, and real-time analytics open the door to broader adoption outside traditional settings, enabling both large and small labs to pursue synthesis that once felt out of reach. As more researchers harness these approaches, bromophenylacetonitrile’s footprint could shift, moving from niche chemical to a bellwether for safe and sustainable organic synthesis.
What is Bromophenylacetonitrile used for?
Chemistry that Touches Everyday Life
Bromophenylacetonitrile sounds like the kind of word that sends people running for a dictionary, but its uses help shape plenty of industries many of us rely on, even if we’ve never heard its name before. This compound often starts its journey in the lab, far away from most daily routines, yet the products and solutions it leads to ripple out into the world in clear, measurable ways.
How it Shows Up in the Real World
If you’ve ever wondered what gets put into motion before your allergy medicine or some over-the-counter pain reliever lands on a pharmacy shelf, this is the kind of chemical behind the curtain. Bromophenylacetonitrile steps in as a building block for many pharmaceutical agents. Scientists rely on its distinct structure to create more complicated compounds—sometimes medicines, sometimes diagnostic dyes. The story here is all about synthesis: making complicated molecules from simpler ones.
Think of pharmaceutical chemistry like a puzzle. The pieces have to fit and sometimes, a molecule like this locks everything together. Its usefulness goes beyond just creating new medicines. Research teams leverage its properties to develop imaging agents—those specialized molecules that help doctors see what’s going on inside a body more clearly.
Why Care about a Chemical Most Folks Don’t See?
It’s easy to ignore things that seem obscure—until mishandling or misuse hits the news. Handling chemicals like bromophenylacetonitrile should not be taken lightly. This one can irritate skin or mucous membranes, which means that anyone working with it in a lab needs proper training, access to protective gear, and clear protocols. All safety data points to one simple truth—caution isn’t just for show.
Recent years brought plenty of examples where lax standards or shortcuts led to health scares. I remember reading headlines about accidental chemical exposures or spills in facilities that stored compounds just like this. In each of those cases, transparency and training stood out as gaps needing to be closed. It’s not overkill to demand strict monitoring and reporting wherever these kinds of chemicals get handled. For everyone’s sake—at the end of the pipeline, a safe process means safe products.
Improving Oversight, Boosting Confidence
What helps here is a layered approach. Regulatory bodies keep an eye on things, but internal company policies fill in those day-to-day cracks. Keeping updated safety information, ongoing staff education, and clear labeling systems all make a difference. From where I stand, the idea isn’t just to avoid mistakes, but to give everyone—scientists, workers, and the public—a sense that the risk of slips has been taken seriously.
I’ve seen places where teams invest in routine safety drills, and keep extra emergency equipment on hand, not just because the law says to, but because every layer of precaution adds real value. That’s peace of mind in chem-lab form. Leaning on peer-reviewed research, manufacturers rely on data to refine every step, from synthesis all the way to shipment.
The Bigger Picture
Bromophenylacetonitrile plays its part quietly in the background, helping unlock new treatments and technologies. Knowing its risks, respecting its power, and using it with care turns it from a hazard into a helpful tool. That’s how innovation stays on the right track.
What are the safety precautions for handling Bromophenylacetonitrile?
Understanding Why This Chemical Deserves Respect
Bromophenylacetonitrile pops up in some advanced labs and chemical manufacturing. It’s not as well known as household names like bleach or acetone, but the risk it brings easily rivals more familiar hazards. Having spent years around labs, I've seen new researchers underestimate obscure compounds because they aren't loudly labeled as "dangerous." Risks like skin burns, lung irritation, and long-term toxicity don't always come with a cloud of dramatic smoke. Bromophenylacetonitrile, in particular, can sneak up with its acute toxicity and volatile nature. Touch or breathe in even small amounts, and you’ll have trouble on your hands—possibly on your skin or deep in your lungs.
Why Standard PPE Alone Won’t Cut It
Putting on a pair of gloves and goggles offers some peace of mind, but it’s not just about throwing on any old lab coat. Bromophenylacetonitrile can pass right through thin latex gloves and cheap face masks. Nitrile gloves with long cuffs, splash-resistant eye protection, and a sturdy lab coat built for chemical work become the basics. Most labs miss the mark by handing out the first box of gloves they see or using cracked safety glasses with warped frames. That choice might work with sugar and salt, not with this compound. I can count several stories where good scientists ignored glove selection and ended up with chemical burns that could’ve been averted.
Engineering Controls: Ventilation Is a Dealbreaker
Work in an open space, and you’re inviting trouble. Bromophenylacetonitrile demands a chemical fume hood that pulls vapors away before they hit your airways. One day, I watched a tired technician rush a prep on a benchtop instead of heading to the hood. The result? Twenty minutes of coughing and a mandatory medical check. Proper chemical work simply doesn’t happen without a hood; even the best respirators struggle if vapors build up. Fume hood airflow should get checked before starting work—taking shortcuts rarely ends well.
Storage and Compatibility Could Save Your Life
Storing bromophenylacetonitrile near acids, bases, or combustible materials is like stacking firewood next to a stove. Keep it in a sealed glass container, labelled clearly, in a flammables cabinet far from curious hands. It shouldn't live on an open shelf or with flasks of acid with faded labels. In hot and humid rooms, the compound might degrade, forming dangerous byproducts that no one bargained for. Every chemical storage shelf in the lab I’m in carries a chart—if you’re not using one, you’re gambling with safety.
What to Do if Something Goes Wrong
Accidents don’t send a calendar invite. Before even opening the bottle, know where the eyewash and shower stations are located. If you spill bromophenylacetonitrile on your skin, rinse for at least 15 minutes and call for help—don’t just wipe it off with a paper towel. Breathing in its vapors brings a rush of throat and lung irritation. Leave the area and get fresh air immediately, then see a medical professional; waiting until symptoms get worse is a proven recipe for regret.
Building Safer Habits
Training beats warnings printed on the side of a bottle. Walk through spill drills. Review what gets stored where. Share stories when mistakes happen. Experienced chemists remember the close calls more than the easy days, and sharing those stories keeps others from repeating them.
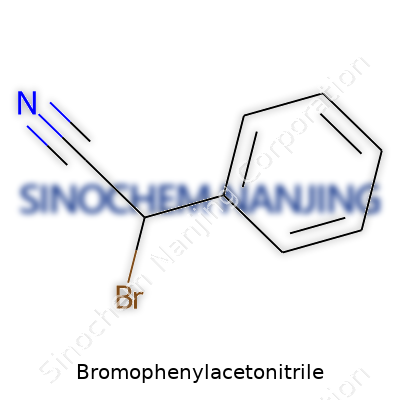
What is the chemical formula of Bromophenylacetonitrile?
The Formula That Matters: C8H6BrN
Bromophenylacetonitrile goes by a chemical formula: C8H6BrN. Stripping away the jargon, that means it carries eight carbon atoms, six hydrogens, a single bromine, and a nitrogen. One glance at the name, and you pick up on some of its main features—the ‘bromo’ flag tells you bromine is in the mix, and ‘phenylacetonitrile’ paints the rest of the picture about its backbone and functional group. The chemical world builds on these tiny differences. Change a single atom, and you don’t always end up with a useful chemical; you might land on something toxic or useless. That’s the puzzle and privilege of organic chemistry.
Why Its Formula Is Far From Trivial
A single bromine atom can change how a molecule reacts or fits into biological machinery. Bromophenylacetonitrile stands as a window into how chemistry influences society, mostly because of its place in law enforcement. This compound has seen use as a "riot control agent," delivered in aerosols or smoke. Unlike tear gas, it targets the body’s senses differently—causing pain in the eyes, mouth, and skin. The chemical structure, as simple as it looks on paper, was designed to maximize those effects while trying to avoid long-term injury.
Seeing the Impact Beyond the Lab
I still remember my time in an undergraduate chemistry lab. Safety goggles kept the stinging fumes away, no matter how nasty the chemicals smelled. But lab safety could never capture what it feels like for someone exposed to a chemical like bromophenylacetonitrile on busy streets. In the real world, this stuff turns chemical theory into hard reality, impacting people’s daily lives in protest situations. Activists and bystanders face health problems because of the way molecules like C8H6BrN work on the body—shortness of breath, coughing, burning eyes. That’s not abstract science; that’s someone’s daily struggle.
Understanding Risks and Safety
This chemical reminds us that the formulas written in textbooks don’t stay locked on the page. Misuse of riot control agents has stirred up real controversy, especially as research groups and medical journals report on lasting injuries. Organizations like the Centers for Disease Control and Prevention and World Health Organization advise using these compounds with caution. Data collected from emergency rooms show spikes in respiratory problems and skin burns after heavy police actions. Knowing the formula gives medical teams a fighting chance to track exposure and treat affected people. The simple breakdown—carbon, hydrogen, bromine, nitrogen— guides antidotes and helps researchers develop safer alternatives.
What Could Help Moving Forward
No simple fix solves the ethical and health issues tied to bromophenylacetonitrile. Lawmakers and health agencies can start with stronger regulation—requiring transparency on composition, limits on use in crowded areas, and standards for clean-up. Emergency responders need training based on solid chemical facts: knowing half-lives, vapor pressures, first-aid for skin exposure, respiratory support guidelines. International chemical safety programs could push for updated research, since a lot of current data on riot control chemicals is patchy and decades old. Scientists always chase safer formulas, weighing the balance between public order and safety—a pursuit only grounded in hard-won, practical chemical knowledge.
How should Bromophenylacetonitrile be stored?
Understanding What’s at Stake
Bromophenylacetonitrile shows up in organic synthesis labs and research spaces more often than most people realize. Its reputation as a respiratory irritant and its potential for environmental harm call for some respect. Forgetting just one detail in how it’s stored can lead to costly mistakes, exposures, or even dangerous reactions.
Straight Talk on Storage Conditions
I’ve worked in enough labs to know that a locked cabinet isn’t the whole story. A shelf full of reactive chemicals becomes less like a place of learning and more like a risky guessing game. For Bromophenylacetonitrile, cool and dry is the ticket. Elevated temperatures raise the risk of decomposition, which can release toxic fumes. Humidity? That might not sound threatening, but even a bit of moisture can nudge certain substances to break down or grow molds. A desiccator or a well-sealed container inside a climate-controlled storeroom keeps things under control.
Sunlight sneaking through a window can degrade sensitive chemicals. Bromophenylacetonitrile needs an amber glass bottle or a solid, opaque container, away from direct light. Light exposure over time chips away at the purity and reliability of any sample, and no one wants to redo a week’s work because the storage protocol got sloppy.
Safety Beyond the Label
I’ve seen careless mistakes turn a routine day into a scramble. This compound can irritate the lungs, so airtight containers with screw caps or PTFE liners help prevent fumes from leaking out. Most mistakes don’t happen because somebody doesn’t care, but because somebody thinks a flimsy lid on a beaker is “good enough.” Double-checking tight closure and labeling isn’t bureaucracy – it’s sanity insurance for everyone in the building.
Storing Bromophenylacetonitrile far away from incompatible materials like oxidizing or acidic agents makes sense. Shared shelves might breed laziness, especially late at night, but safe storage zones reduce accidents. If you handle this stuff regularly, you learn to scan for unmarked bottles or anything out of place, and you know what could go wrong.
Documentation Isn’t Just Paperwork
Documentation gives everyone on a research team or in a manufacturing facility access to the same knowledge. A chemical inventory with the specific location and the last check date can save time and lives. The accidents that stick in people’s memories often start with a missing label or a misplaced vial.
Anyone who’s lived through a lab audit knows handwritten notes only get you so far. Legible, updated logs—ideally digital—show due diligence if regulators come around. It also gets new team members on the same page faster.
Tough Lessons and Smarter Storage
Mistakes have consequences. Spilled Bromophenylacetonitrile won’t just cost money. The cleanup exposes more people, and none of the standard safety gear provides much comfort when things go wrong. Learning from near-misses—like someone briefly popping open the wrong container—motivates smarter handling in the future.
Securing a pound of this chemical behind a steel cabinet with a clear label and proper ventilation isn’t overkill—it’s smart science. For every person who’s ever worked late and wished they’d double-checked a lid, solid storage practice is as much about protecting each other as it is about following the rules.
Is Bromophenylacetonitrile hazardous to health or the environment?
A Chemical That Raises a Few Eyebrows
Bromophenylacetonitrile isn’t a household name, but it shows up in conversations about industrial chemicals—something I’ve crossed paths with as a science communicator. Used in making pharmaceuticals and dyes, this compound can slip under the radar, yet its effects deserve a closer look. Anyone who’s handled lab chemicals or reviewed Material Safety Data Sheets would recognize the tone of “hazardous” attached to it. That label is on the sheet for a reason.
Toxicity Isn’t Just an Academic Point
Short-term exposure can cause real problems. Eyes and skin feel the hit first. It doesn’t take much to start a rash or make your eyes sting. Inhaling its fumes causes throat irritation or coughing. If someone accidentally ingests it, the outlook is worse – nausea, headaches, even more dangerous symptoms depending on the dose. I’ve met researchers who suit up with gloves, goggles, and ventilators even before the bottle is opened. That level of caution speaks for itself.
On a deeper level, compounds like bromophenylacetonitrile can break down in the body to release cyanide ions. We’re not talking about routine handling, but significant spills, lab accidents, or carelessness step up the risk. For me, working with these substances always meant close attention to ventilation and emergency protocols. The body doesn’t recover from cyanide poisoning without rapid medical response—I’ve heard first-hand accounts from health and safety officers who’ve drilled this point home after close calls.
Wider Environmental Questions
Everything that washes down the drain or blows out a vent finds its way somewhere else. Bromophenylacetonitrile isn’t much different. It doesn’t just vanish after use. Released into water, it can harm aquatic life. Fish and invertebrates are more sensitive to certain chemicals than people assume. I spent a season working at a site where regular water sampling was mandatory. Runoff, even from careful labs, can throw off the health of streams and rivers.
Soil absorption is another piece of the puzzle. This chemical doesn’t stay put; it can spread through groundwater, lingering for quite some time. That persistence leads to worry about long-term effects on plant growth and microorganisms, which form the base of any ecosystem. Neighborhoods near factories or large research centers have a stake here—nobody wants contamination to show up years later in garden soil or well water.
Balancing Use and Safety
Chemicals like bromophenylacetonitrile pop up in pharmaceutical manufacturing, bringing clear benefits, but at a cost. Over the years, the push for tighter regulations has helped, but gaps remain. Up-to-date proper storage, good labeling, and transparent waste management matter as much as ever. It’s not just about what happens inside the facility—community right-to-know laws have made it possible for neighbors to learn what’s being used nearby.
Safe disposal is a recurring issue. I’ve seen projects where chemical waste sat in storage rooms because no one wanted to foot the bill for proper incineration. Some contractors take shortcuts, putting everyone at risk. Best practices mean funding collection and treatment, not hoping that risks fade with time. Labs and plants have gotten better—more audits, better accident reports, emergency plans ready to run at a moment’s notice. It still falls to people on the ground to pay attention, double-check ventilation, keep spill kits stocked, and speak up about unsafe conditions.
Moving Forward with More Responsibility
People tend to underestimate risk until something goes wrong. The lesson from bromophenylacetonitrile—and other industrial chemicals—is to treat safety as ongoing work. Rethinking how we handle, store, and track these substances helps protect both workers and the wider world outside the lab walls.
| Names | |
| Preferred IUPAC name | 2-(Bromophenyl)acetonitrile |
| Other names |
2-Bromo-2-phenylacetonitrile Benzyl bromocyanomethane alpha-Bromo-alpha-phenylacetonitrile 2-Phenyl-2-bromoacetonitrile |
| Pronunciation | /ˌbroʊ.məˈfiː.nɪlˌæs.ɪ.toʊˈnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | 769-90-6 |
| Beilstein Reference | 1718734 |
| ChEBI | CHEBI:91195 |
| ChEMBL | CHEMBL16230 |
| ChemSpider | 21573803 |
| DrugBank | DB08647 |
| ECHA InfoCard | 100.035.300 |
| Gmelin Reference | 82822 |
| KEGG | C18807 |
| MeSH | D015586 |
| PubChem CID | 72783 |
| RTECS number | UG5075000 |
| UNII | CJ6WT6QF1K |
| UN number | UN3439 |
| CompTox Dashboard (EPA) | DTXSID7045274 |
| Properties | |
| Chemical formula | C8H6BrN |
| Molar mass | 196.04 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | aromatic |
| Density | 1.24 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.00153 mmHg (25°C) |
| Acidity (pKa) | 19.0 |
| Basicity (pKb) | 11.89 |
| Magnetic susceptibility (χ) | -63.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.574 |
| Viscosity | 3.76 mPa·s (20°C) |
| Dipole moment | 4.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 226.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -37.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -340.8 kJ/mol |
| Pharmacology | |
| ATC code | D06AX04 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 114 °C (237 °F; 387 K) |
| Autoignition temperature | Unknown |
| Lethal dose or concentration | LD50 oral rat 210 mg/kg |
| LD50 (median dose) | LD50 (median dose): 210 mg/kg (oral, rat) |
| NIOSH | CN8575000 |
| REL (Recommended) | 2 mg/m³ |
| Related compounds | |
| Related compounds |
Benzyl cyanide Phenylacetonitrile Bromobenzene |

