The Journey and Challenges of Boron Trifluoride Dihydrate in Modern Chemistry
Tracing the Path from Discovery to Daily Practice
Boron trifluoride dihydrate doesn’t often headline mainstream science news, but its footprints in chemistry and industry run deep. Chemists started exploring boron compounds in the 19th century, wrestling with the extreme reactivity of boron trifluoride itself. Unlike its dangerous gaseous parent, the dihydrate form provided a safer bridge for laboratory and processing work, opening doors to practical use. In my own research years, the shift from dry, no-room-for-error BF3 handling to more manageable forms like the dihydrate stood out as a minor miracle. Laboratories that once braced for leaks and burns could breathe a little easier. Historical documentation from institutions like the Journal of the Chemical Society in the mid-1900s highlights moments when boron chemistry edged from curiosity to tool, often thanks to stabilizing water molecules attached in the hydrate.
Getting to Know What We’re Working With
Boron trifluoride dihydrate lands in labs as a clear to slightly cloudy liquid, giving off a strong acidic smell that hints at its urge to react. It dissolves readily in water, forming acidic solutions, and reacts fiercely with bases and certain organics. The chemical formula, BF3·2H2O, gives away its origin but only hints at its potential. It’s considerably less volatile than boron trifluoride gas, but mishandling still brings nasty surprises. The substance keeps a high density and behaves with a toughness that demands real respect. Fuming acid clouds mean you’re handling more than just another bottle in the reagent cabinet.
Preparation and Modifications in the Real World
Preparation needs more than a simple blend. Laboratories usually pass boron trifluoride gas through chilled water, triggering brisk exothermic reactions. Sudden temperature jumps create risks that lab veterans learn never to underestimate. Some technical guides advocate a staged, cooled process to prevent violent emission or glassware failure. That sort of hands-on process taught me to pay closer attention to safety reminders than to protocols written in a hurry. In my own early procedural trials, skipping a step or hurrying brought harsh feedback — not just from supervisors, but from near-miss splashes hot enough to etch glass or ruin a day for your skin. For modifying its reactivity, chemists blend the dihydrate with other solvents, sometimes changing acidity or unlocking different catalytic behaviors. These experimental twists fuel new patents and open routes to tailored reactions.
What Really Happens When Boron Trifluoride Dihydrate Meets Other Chemicals
In the day-to-day grind of synthetic chemistry and material processing, this hydrate acts as a strong Lewis acid. A common use lies in catalyzing oligonucleotide or polymer synthesis, helping to stitch molecules together faster and cleaner. The molecule’s craving for electron pairs means it can shift and shape carbon frameworks or coax out otherwise elusive products. I learned to appreciate its stubbornness during failed reactions: nothing hammers home respect for chemical selectivity like watching products decompose in a haze of fuming acid vapor. Many newer papers, especially from European organic chemistry circles, describe selective modifications for pharmaceuticals, energetic materials, and specialty plastics. For those willing to test its limits, even more nuanced transformations surface, sometimes beating older, dirtier industrial methods.
Finding Boron Trifluoride Dihydrate in the Wild — Or At Least in Catalogs
The compound goes by several alternative tags, including boron trifluoride bimolecular compound and boron trifluoride hydrate. More technical catalogs sometimes abbreviate it as BF3·2H2O or refer to it as a hydrated boron trifluoride acid complex. These synonyms help cross borders or industry silos, showing up in chemical indices worldwide, and avoid confusion when looking for compatible substitutes or MSDS sheets.
Staying Safe — Not Just a Paper Exercise
Anyone who has handled boron trifluoride dihydrate for real knows that safety can’t be just a classroom lesson. Its fumes sting the eyes and throat immediately, with even a drop on the skin calling for a cold water rush and fast action. Industrial sites and university labs alike set up strict protocols: chemical fume hoods, acid-resistant gloves, and eyewash stations within reach. Regulations from groups like OSHA and ECHA classify it under corrosives, mandating clear labeling and spill mitigation strategies. The strongest reminders come from scarred benches and persistent acid smells, a silent warning to skip shortcuts. I’ve seen too many bright students learn the hard way: minute lapses – one cracked pipette or an unlabeled flask – drive home why rules and redundancy matter.
Where Boron Trifluoride Dihydrate Really Works
Pharmaceutical exploration, advanced materials, and organic synthesis all depend on compounds with teeth. In process chemistry, boron trifluoride dihydrate powers alkylation, esterification, and oligomerization — backbone reactions for new drugs, high-tech coatings, or polymer precursors. During my time with research teams focused on cancer drug candidates, the compound showed up not in headlines, but in the scaffolding. The molecule’s sharp acid properties mean small mistakes snowball into lost batches or dangerous byproducts, but used with precision, it unlocks efficiency no substitute manages quite as cleanly. Advanced battery and microelectronics work benefits from its unique blend of strength and selectivity, hinting at a crossover into future energy storage markets.
The Push for Safer Use and Broader Innovation
Research circles often chase safer, greener alternatives to legacy reagents like boron trifluoride dihydrate. The toxicity profile, especially in confined or improperly vented settings, keeps ethics panels and regulators alert. Animal studies and case reports underline its corrosive effects on respiratory tissues and highlight severe danger to eyes and mucous membranes. Over the last decade, journals report on buffered or immobilized acid systems that aim to reduce accidental releases and lessen the impact from spills. I remember a coworker’s minor exposure quickly developing into a major accident, underlining how something so widely used can still demand everyone’s vigilance. Ongoing projects now explore encapsulation, in-situ generation, or remote-handling setups enhancing operational safety.
Looking Ahead — The Role of Boron Trifluoride Dihydrate in Tomorrow’s Chemistry
As chemists and materials scientists look for both performance and sustainability, the pressure mounts to either refine how boron trifluoride dihydrate is used or to replace it outright. Advances in reaction engineering — like flow chemistry setups and single-use reactors — provide hope for reducing both human and environmental risks. Ongoing work into non-volatile, solid acid catalysts could someday make liquid, fuming acids a thing of the past for many applications. I see room for growth in automated handling and real-time exposure detection, taking the human factor out of the most hazardous handling. For now, the compound remains essential to critical synthesis, even as oversight grows and green chemistry advocates search for alternatives. If history is any guide, boron trifluoride dihydrate won’t lose its place easily, but the trend points toward a safer, more responsible chemical toolbox.
What is Boron Trifluoride Dihydrate used for?
What Boron Trifluoride Dihydrate Does in Real-Life Labs
Boron trifluoride dihydrate doesn’t sparkle in headlines, but its impact in the lab is real. Every organic chemist who’s had their hands deep in reaction flasks knows the stuff as a strong Lewis acid. In plain terms: it’s great at pulling electrons and gets molecules to react in ways they wouldn’t do alone. I remember the first time I used it in grad school—slightly nervous, a little intimidated by its reputation—but the results left a mark. Good catalysts, like teachers, open new doors by making the impossible happen.
Making Medicine and Materials Better
Pharmaceutical companies rely on boron trifluoride dihydrate to pull off tough reactions. Take alkylation or polymerization—basic building blocks for heart medicine, antibiotics, and everyday plastics. Modern antihistamines or cholesterol-lowering drugs probably owe a step in their manufacture to this chemical. Chemists reach for it because the alternative processes cost more, waste energy, or generate toxic byproducts. Manufacturing safer and more affordable drugs means looking for shortcuts, and boron trifluoride dihydrate often provides the key.
Not Just in the Lab: Boron Trifluoride Dihydrate in Industry
Beyond small-scale experiments, factories pumping out resins or making additives for lubricants count on boron trifluoride dihydrate. Tetrahydrofuran gets made in thousands of tons each year using processes that count on boron trifluoride to keep the engines running. There’s nothing flashy here; the work is precise, and small slip-ups spell lost batches and broken equipment. I’ve seen teams obsess over the right storage containers, just to keep moisture levels balanced—because boron trifluoride dihydrate feeds on water chemistry and changes if left unchecked. Real-world experience teaches respect for chemicals that work hard and demand a steady hand.
Why Handling Safety Isn’t Optional
Boron trifluoride dihydrate doesn’t ask for much, but its strength cuts both ways. Direct contact can harm skin and lungs—something no seasoned worker forgets. Proper ventilation and personal gear matter, and there’s pride in keeping a shop safe. Chemical accidents, rare as they should be, end up costing time, health, and sometimes futures. I’ve witnessed close calls, and they underline one lesson: safety isn’t a box to check, it’s the rulebook you live by. Training goes beyond reading charts; it means building a culture where everyone looks out for each other.
Tackling Environmental Impact
Every industrial triumph carries a footprint. Boron trifluoride dihydrate, if flushed out or mishandled, can harm local water and air. Waste treatment plants handle harsh residues with multiple filters and neutralizers. Better methods and greener alternatives get attention as regulations tighten, and I see younger chemists pushing for process improvements that waste less and spill less. We owe future generations cleaner ways to make old standbys, and that drives research into recycling catalysts or swapping in less hazardous chemicals without losing that critical reactivity.
Building Trust in Chemical Manufacturing
Quality drugs, safe plastics, and reliable lubricants all start with choices made in the supply room. Cheap shortcuts cost more in the long run. Reputable sources check every barrel and batch, offering certificates that prove what’s inside. Scientists depend on this trust, especially as global trade speeds up. Knowing a chemical does what it’s supposed to do allows new products to hit shelves without headaches. Any manufacturer cutting corners puts everyone at risk—and I’d rather speak out than stay silent in a lab that gambles on safety and quality.
What are the safety precautions for handling Boron Trifluoride Dihydrate?
A Chemical with Bite: Know What You’re Dealing With
Boron trifluoride dihydrate isn’t your every day classroom chemistry set star. This stuff gives off sharp, acidic fumes and irritates the skin and lungs. My first close encounter with it happened early in my career, and I still remember the strong smell and the sudden prickling sensation on my exposed hands. Gloves hadn’t been optional then, but I learned that some materials barely slow these chemicals down.
Real Hazards, Not Just Red Tape
Plenty of people brush off safety rules until they see an accident. Breathing boron trifluoride dihydrate vapors can sting your throat, cause coughing, and burn your eyes. Even small splashes can eat away at skin or leave you struggling to see. The main issue? Folks often underestimate how quickly fumes fill a room when you open a bottle or pipette some out. In 2022, a study in “Chemical Health & Safety” highlighted serious injuries in labs that ignored eye and skin protection. Medical journals repeat the same: inhalation and skin contact send enough workers to the hospital each year.
Gear Up—No Cutting Corners
Respecting this chemical means covering up. Splash goggles, chemical-resistant lab coats, and gloves rated for acids—these feel bulky until you recall how quickly things go sideways. Not every glove stops boron trifluoride dihydrate. Fluoropolymer gloves top the list, but even then, swapping gloves right after spills matters. Folks sometimes use splash shields by default, but a sturdy fume hood works better: it grabs nasty fumes before you breathe them in. I’ve watched people get lazy about tying hair back or securing cuffs, only to wipe chemicals across their wrists absentmindedly.
Ventilation Isn’t Just a Box to Check
A working fume hood matters more than sparkling countertops. Gases from boron trifluoride dihydrate hang around unless you pull them away. Turn the hood on before you open the chemical, and double-check the sash height so your reaction stays protected. After one spill in my own lab, the room’s air system struggled for hours to clear that acrid reek. That day, we learned fume hoods need regular airflow checks—clogs can build up without anyone noticing.
Clean Up and Storage: Habits that Save Skin
After you finish up, don’t leave contaminated tools on the bench. Wipe surfaces with a spill kit or diluted sodium carbonate solution, then rinse with lots of water. Any towels or gloves go straight into a sealed disposal bag. Bottles of boron trifluoride dihydrate need storage in corrosion-resistant cabinets, away from bases, metals, and moisture. Too many accidents come from cracked containers or chemical reactions with metal shelving.
Training Isn’t Optional—It’s as Essential as Gloves
Every new lab member must practice mock handling before they get close to the real thing. Watching a video doesn’t cut it. Emergency showers and eyewash stations must always be within reach and in working order. Practicing spill drills with water or dye builds muscle memory, so you react fast when it matters.
Supporting Safer Chemistry Labs
Anyone handling boron trifluoride dihydrate should look over recent hazard data published in occupational safety bulletins—not just rely on faded labels. Open conversations about slip-ups or nearmisses can strengthen a lab’s safety culture. Well-defined checklists and clear access to protective gear cut down on small mistakes. None of this stops the work from getting done. It helps everyone walk out of the lab at the end of the day, healthy and ready to come back tomorrow.
How should Boron Trifluoride Dihydrate be stored?
Understanding the Risks in the Lab
People familiar with chemical labs know Boron Trifluoride Dihydrate carries some heavy responsibility. Its reputation as a strong Lewis acid comes with dangers: powerful reactivity, corrosive properties, and hazardous fumes. Even experienced technicians don’t take shortcuts around this stuff, not after seeing what careless storage can do—degraded containers, surprise leaks, burned fingers, and unhealthy air quality in tight rooms.
Why Isolation Matters
It’s tempting for newcomers to treat it like any other bottled acid. That attitude causes more problems than almost anything else in chemical storage. Boron Trifluoride Dihydrate should never end up next to combustibles, alkalis, or anything storing water. One spilled drop from a cracked cap can spark a violent reaction. If it gets loose, it chews up metal shelving or flooring and paints the air in the room with fumes that sting eyes and lungs. I’ve seen older storage closets where a careless bottle turned a steel shelf to rusty lace. Segregation keeps everyone from walking into a trap later on.
Shelving and Container Choice
Standard glass bottles don’t make the cut for this chemical. The material corrodes too quickly. Polyethylene or Teflon-lined containers hold up best. Screw tops need tight seals—leaks don’t just put labels at risk; they risk the whole shipment. In the stores I’ve worked, supervisors always lined up the bottles in corrosion-resistant polyethylene trays in case of drips or overfilling. Those trays pulled double duty by containing stray splashes during transfer, a lesson handed down after more than one all-hands cleanup.
Temperature: Not Just an Afterthought
Keep it cool, dry, and stable. High heat pushes up pressure in bottles, making small leaks turn into big headaches. Moisture and temperature swings always spell trouble. Ideally, bottles sit in a well-ventilated chemical store room, separate from offices or walkways. Refrigeration shouldn’t drop below freezing though, or else contents may crystallize or containers could stress and crack under expansion. I remember a shipment ruined by a mislabeled fridge setting—wasted product and hours of hazard calls.
Ventilation: Letting Fumes Out, Not In
Labs often run extraction fans, but a cramped, closed chemical locker turns vapors into a major risk. Good practice means choosing a ventilated storage cabinet or a dedicated negative-pressure chemical store. Fume alarms earn their place on the shelf, especially for those with little sense of smell or researchers who come back after weekends away. Your nose isn’t a safety monitor—use real sensors for noticing a problem before it grows.
Labeling and Regular Checks
Heavy labeling stands between confusion and accidents. Bold hazard pictograms, chemical names, and storage guidance belong right on the front. No label means no use, not just a guessing game. Regular checks stop the worst before it happens. In my own work, we checked containers monthly for cap corrosion and chemical crust. If a bottle looks different—bulged sides, crystals on threads, or a weeping cap—it goes straight into a sealed drum and off for professional disposal. No heroics, just good habits paying off over time.
Emergency Planning as Routine
It only takes one slip to turn careful storage into cleanup mode. Every facility storing Boron Trifluoride Dihydrate should back up its plans with readily available spill kits, clear escape routes, and trained staff ready for leaks. This isn’t just paperwork. Cleaning up a spill safely means knowing where neutralizing powders and fresh respirators stay, not searching under cabinets while fumes start to spread.
Final Thoughts on Safe Practice
Storing Boron Trifluoride Dihydrate safely isn’t about hoarding it behind locked doors. It rests on steady routines, clear communication, and a respect for history—both of successful storage and close calls. Good training, strong containers, segregation, and regular inspection stop mistakes from piling up. The bottom line: Treat it with attention and planning, and it stays a tool instead of a ticking hazard.
What is the chemical formula and structure of Boron Trifluoride Dihydrate?
Breaking Down Boron Trifluoride Dihydrate
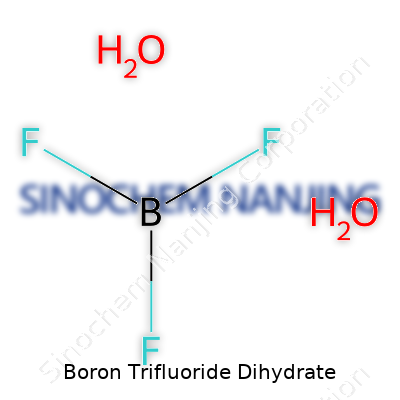
Boron Trifluoride Dihydrate shows up in chemistry labs as a compound with the formula BF3·2H2O. The boron trifluoride part brings a punch—this gas, on its own, is harsh, reactive, and not exactly something you want floating around. Adding two water molecules transforms it, giving us a more manageable solid with unique chemistry. I’ve worked with BF3 at the bench, and it demands respect; water helps control it. Hydration can turn a dangerous fume into a form you can weigh and handle.
What the Structure Looks Like
The real structure deserves a closer look. It isn’t simply a pile of boron trifluoride molecules stacked next to two water molecules. In solid BF3·2H2O, the boron atom forms coordinate bonds with fluoride ions and water molecules. Boron sits at the center, accepting electron pairs. Picture boron joined by three fluorine atoms—each tugging at boron’s electron cloud. Water molecules interact by hydrogen bonding and by donating electron density to boron. This twists the atomic geometry away from textbook trigonal planar (seen in anhydrous BF3), pulling the molecule into something a bit more complicated.
Why This Matters Outside the Lab
I learned firsthand that BF3·2H2O brings safety and practicality. Pure BF3 comes as a pressurized, corrosive gas. Hydrated forms turn it into a solid, so the risks drop and storage gets easier. Industrial chemists favor it for this reason—instead of wrangling gas cylinders, the hydrated version stores in simple containers and weighs out on a balance.
Boron trifluoride’s story goes beyond safety. The dihydrate’s coordination chemistry lets it serve as a Lewis acid in solution, helping kickstart reactions that might stall otherwise. Organic synthesis—the backbone of pharmaceuticals, plastics, agricultural products, and even flavors—depends on getting blends like BF3·2H2O just right. I’ve seen this hydrated form help smooth out alkylation and polymerization reactions that stumble under harsher conditions.
Health, Safety, and Environmental Concerns
Even hydrated, boron trifluoride demands precautions. Its reaction with water isn’t neutral; hydrofluoric acid (HF) can form, leading to serious injuries. Chemical burns from HF don’t feel like regular acids—they sneak up on you and run deep. My lab mentor insisted we keep calcium gluconate gel within arm’s reach for exactly this reason. Proper training, gloves, face shields, and fume hoods aren’t optional—they’re simply the way things work safely.
Disposal matters, too. Washing BF3·2H2O down the drain or letting it evaporate carries risk. Environmental regulators track fluoride levels for a reason. Following best practice in neutralization and waste collection protects more than just the individual in the lab.
Solutions and Best Practices
Industry and research settings can keep risk to a minimum. Storing the hydrate instead of the anhydrous gas cuts accident chances. Well-ventilated fume cupboards reduce exposure possibilities. Training remains effective—new chemists should learn hands-on protocols from someone with direct experience, not just a manual. For larger scale operations, companies use sealed systems, scrubbing any off-gases safely.
Innovation in packaging and transfer devices keeps making handling safer. Single-use ampoules, self-sealing bottles, and remote dispensing all help keep exposure low. It’s worth keeping up with chemical supplier guidance and regulatory updates—those details really do change how chemists work day-to-day.
Why BF3·2H2O Keeps Its Place
Boron trifluoride dihydrate isn’t flashy. Still, its blend of chemical muscle and practical form helps chemists bend tough reactions to their will, all while avoiding the worst risks of its anhydrous cousin. Thanks to hydration, boron trifluoride finds its way onto lab shelves across pharmaceutical, industrial, and research fields, quietly powering much of the modern world’s chemical transformations.
Is Boron Trifluoride Dihydrate toxic or hazardous to health?
Real-World Risks from the Lab to the Workplace
Boron trifluoride dihydrate shows up in laboratories and some manufacturing processes. This chemical carries risks that go beyond lab jargon. Many folks remember their first brush with serious chemicals as a rite of passage. Boron trifluoride dihydrate never makes that kind of impression for good reasons—it can harm people quickly.
Breathing in boron trifluoride fumes can irritate the eyes, nose, throat, and lungs. Most people won’t mistake the feeling: stinging eyes, burning in the chest, a raw cough. Direct skin contact could give nasty burns. Even without splashes or spills, fumes find their way around goggles or gloves if workers aren’t careful. Splashes in the eyes call for immediate rinsing; any delay can mean permanent damage. The Centers for Disease Control and Prevention warns that boron trifluoride compounds are corrosive, eating away at tissues after just brief contact. Its toxic reputation comes from both the acid it forms on contact with moisture and the fluoride ion itself, which disrupts normal cell functions.
What Makes Boron Trifluoride Dihydrate Especially Hazardous?
Boron trifluoride isn’t just another lab chemical. Its volatility and acidity set it apart. Even short exposures at the wrong concentration can bring on headaches, breathing trouble, and confusion. Sometimes accidents surprise experienced hands—vent hoods do their job, but leaks or careless storage can lead to real harm. The National Institute for Occupational Safety and Health (NIOSH) pegs the exposure limit at a strict level: 1 ppm for boron trifluoride. This tells you agencies take this chemical’s toxicity seriously despite far more toxic compounds out there.
Once it hits water, boron trifluoride hydrolyzes fast, turning into boric and hydrofluoric acids. Anyone who works in industry knows hydrofluoric acid burns sink deep and keep on hurting for hours or days. It’s a race against the clock to prevent lasting tissue loss. Medical teams keep calcium gluconate on standby because fluoride ions steal calcium from the body, sometimes leading to fatal heart trouble. Recognizing symptoms and acting fast—washing the chemical off, flushing eyes, getting medical help—makes all the difference in recovery.
Improving Safety and Preventing Accidents
Companies store boron trifluoride dihydrate in tight containers under ventilation, but accidents trace back to rushed jobs and forgotten procedures. My experience working with dangerous chemicals as a young technician taught me respect for every label, no matter how familiar a bottle seems. No shortcuts—teams run drills for spills every season. Keeping safety showers, eyewash stations, and proper PPE at arm’s reach marks a workplace that looks out for its people.
Some organizations put extra training behind every new hire, and seasoned workers reinforce the routines daily. Reporting close calls helps everyone avoid false confidence. Replacing boron trifluoride dihydrate with safer alternatives, if the process allows, gives peace of mind. Not every task can get a substitute, but even switching out glassware or improving ventilation makes a real difference.
What’s the Bottom Line?
Boron trifluoride dihydrate delivers serious hazards despite not being a household name. Its mix of corrosive action, toxicity, and the threat it poses to skin, lungs, and eyes keeps it near the top of any chemical safety training. Folks who work with chemicals ride a constant balance between getting the job done and making it home safe. Everyone benefits by putting more weight on the safety side from the start.
| Names | |
| Preferred IUPAC name | Dihydroxidodifluoroboron |
| Other names |
Boron trifluoride, dihydrate Boron trifluoride hydrate Boron trifluoride · 2H2O |
| Pronunciation | /ˈbɔːrɒn traɪˈfluːəraɪd daɪˈhaɪdreɪt/ |
| Identifiers | |
| CAS Number | 13319-75-0 |
| Beilstein Reference | 358906 |
| ChEBI | CHEBI:86460 |
| ChEMBL | CHEMBL1230873 |
| ChemSpider | 88829 |
| DrugBank | DB14506 |
| ECHA InfoCard | 06dbebfc-a6d1-49b3-9fb3-845692a7a0e8 |
| EC Number | 231-569-5 |
| Gmelin Reference | 82189 |
| KEGG | C16236 |
| MeSH | D002019 |
| PubChem CID | 166827 |
| RTECS number | ED3325000 |
| UNII | R7RZ957J8G |
| UN number | UN1744 |
| Properties | |
| Chemical formula | BF3·2H2O |
| Molar mass | 105.82 g/mol |
| Appearance | Colorless crystalline solid |
| Odor | Pungent |
| Density | 1.37 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | -0.4 |
| Vapor pressure | 14 mmHg (20 °C) |
| Acidity (pKa) | -0.4 |
| Basicity (pKb) | 8.66 |
| Magnetic susceptibility (χ) | -1.2e-6 |
| Refractive index (nD) | 1.333 |
| Viscosity | 2.35 cP (20 °C) |
| Dipole moment | 1.81 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1276.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2049.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AN02 |
| Hazards | |
| Main hazards | Reacts violently with water. Toxic if swallowed. Causes severe skin burns and eye damage. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or inhaled. H314: Causes severe skin burns and eye damage. |
| Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-2-W |
| Lethal dose or concentration | LD50 oral rat 178 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 100 mg/kg |
| NIOSH | ED3400000 |
| PEL (Permissible) | PEL: 1 ppm (3 mg/m³) as BF₃ (OSHA) |
| REL (Recommended) | 2 mg/m³ |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Boron trifluoride Boron trifluoride etherate Boron trifluoride methanol complex Boron trifluoride acetonitrile complex |

