Bismuth Citrate: In-Depth Commentary
Historical Development
Bismuth citrate represents a rather curious chapter in the larger story of inorganic compounds shaping modern science and medicine. Early chemists in the 19th and 20th centuries, while searching for cleaner alternatives to heavy metals like mercury and lead, discovered that bismuth salts offered some therapeutic value, especially for gastrointestinal conditions. Bismuth subsalicylate gained a reputation as a remedy for stomach ailments, leading researchers to explore compounds like bismuth citrate for similar purposes. With time, advancements in analytical techniques, such as X-ray crystallography, gave scientists better understanding of the structure and reactivity of bismuth citrate, pushing its applications from the apothecary to the laboratory and even further into industrial spaces.
Product Overview
Most people haven’t heard of bismuth citrate outside specialized settings, but its role has quietly expanded beyond early antacid formulations. Available as both a monobasic and tribasic salt, bismuth citrate shows up in pharmaceuticals, personal care products, and even as a pigment. Commercial producers list the compound under several names, depending on the hydrate level and bismuth to citrate ratio. In my own research, I've come across it in dental rinses and some topical treatments designed to harness its antimicrobial profile. Each lot typically comes as a fine, white or slightly off-white powder, packed tightly to prevent moisture absorption because of its tendency to clump.
Physical and Chemical Properties
Pure bismuth citrate doesn’t dissolve much in water, though it breaks apart a bit better in acidic solutions. A standard sample carries a molecular weight around 704 g/mol with the basic formula BiC6H5O7. Powder X-ray diffraction shows a crystalline pattern, and in bulk, it reflects a slight pearly sheen. In chemical terms, it runs inert under ambient conditions but forms complexes in the presence of various ligands. Heat it, and you'll notice decomposition before melting, leaving behind a bismuth-rich residue. Handling it myself, I found it neither sticky nor noticeably reactive, and the mild, earthy scent speaks to its stability.
Technical Specifications and Labeling
Suppliers and regulators require strict documentation for labeling. Each pack notes the bismuth content, usually expressed as a minimum percentage (often above 34%), the water content, and trace levels of impurities like arsenic and lead, both heavily discouraged. Certificates of analysis list tests for pH in suspension, solubility in dilute nitric acid, and presence of residual citrate. In pharmaceutical environments, every container comes tagged with storage instructions, hazard pictograms, and a batch-specific certificate to ensure traceability. From experience, these records help track any problems back to a specific manufacturing run, which comes in handy during recalls or audits.
Preparation Method
Industrial preparation takes place through a reaction of basic bismuth nitrate with citric acid under controlled temperature and pH. Chemists start by suspending bismuth nitrate in deionized water and then add citric acid slowly, paying attention to the rate of addition to avoid local overheating or premature precipitation. The resulting mix is stirred vigorously, and once the correct proportion is achieved, filtration removes any undissolved material. The filtrate gets evaporated under reduced pressure, and solid bismuth citrate is washed and dried thoroughly at 60–80°C. Scale-up requires careful management, as high local concentrations of reagents may induce a gummy product that's difficult to purify.
Chemical Reactions and Modifications
Bismuth citrate acts as a mild Lewis acid with the potential to coordinate with donor ligands, making it attractive to researchers exploring new hybrid materials. Reaction with strong acids regenerates free citric acid and forms bismuth salts such as bismuth chloride. Under alkaline conditions, partial hydrolysis yields insoluble basic bismuth salts and a more fragmented organic matrix. By adding chelating agents, the compound converts into bismuth coordination complexes with varied solubility profiles, opening pathways to adjust its release in drug formulations. In some of my lab work, this property allowed us to tune both the dissolution rate and the interaction with biological fluids for customized release.
Synonyms and Product Names
No single name covers all commercial variants. Beyond “bismuth citrate,” you see terms like “bismuth(III) citrate,” “citrate of bismuth,” and supplier-specific brands such as “BiCitro,” or “Bismocit.” Pharmacopoeia entries sometimes refer to its monobasic, dibasic, or tribasic forms, as well as their hydrates. Each version points to subtle differences in stoichiometry or particle size. Customers must pay attention to this detail, as substitution without checking these parameters shifts everything from texture in pastes to bioactivity in formulations.
Safety and Operational Standards
Bismuth citrate needs respect in the lab and factory, but it doesn’t carry acute toxicity like some heavy metals. Chronic exposure, though, can have mild renal impacts, according to the US National Institutes of Health, especially from poor-quality samples containing trace toxins. Material safety data sheets warn workers to wear particulate masks and gloves, emphasizing ventilation around larger reactors to prevent inhalation of airborne dust. If swallowed, bismuth citrate typically passes through the system, but in larger amounts and vulnerable populations, it can trigger mild but persistent gastrointestinal discomfort. Facilities that handle the compound run regular air sampling, and strict segregation from incompatible acids and alkalis limits accidents. Having worked around both organic and inorganic bismuth compounds, I learned that spills stay relatively easy to clean up compared to some solvents, provided you don't allow dust to accumulate in the air filters.
Application Area
Clinically, bismuth citrate gained attention in eradication regimes for Helicobacter pylori, working with antibiotics to treat resistant ulcers without promoting excessive bacterial resistance. Many over-the-counter topical products cite its antimicrobial action in formulations for acne and minor wounds. Dental rinses leverage its therapeutic effects on oral bacteria, and some hair dyes use it as a pigment stabilizer. Non-medical uses include its deployment in specialty pigments for ceramics and even a minor role as a catalyst in organic synthesis. Teachers use simple bismuth citrate compounds to demonstrate coordination chemistry for students, as their color changes and low acute toxicity provide a safe, instructive introduction.
Research and Development
Ongoing studies look to exploit bismuth citrate’s gentle toxicity profile and strong complexation capacity. Research teams are examining its ability to form nanoparticles that deliver drugs to targeted microbiological sites without harming human cells. In the context of resistance, a few groups in China and Europe have found synergies between bismuth citrate and antibiotics, showing upticks in antibacterial potency, even against multidrug-resistant strains. As analytical detection limits drop, interest grows in mapping the pathways bismuth compounds follow in both the human body and environmental runoff — critical knowledge in the face of tighter waste regulations. Manufacturers tweak the synthesis to minimize impurities because even low-level arsenic and lead contamination undermines both safety and trust.
Toxicity Research
Researchers routinely test bismuth citrate in rodents and cell cultures. Most acute exposures result in little observable harm, especially in doses aligned with what consumers get from typical medications or cosmetics. Chronic toxicity, while rare, pops up more in patients with kidney impairment, and there’s a push for screening at-risk groups prior to prescribing high-dose formulations. Compared to other heavy metals, bismuth’s affinity for organic acids helps chelate and remove it from the body relatively quickly, but long-term accumulation in tissues remains a topic for closer scrutiny, especially after a January 2022 case report highlighted mild encephalopathy linked with long-term over-the-counter use. More hospitals and health authorities now ask for post-market surveillance of adverse reactions tied to all bismuth salts, expanding registries and pharmacovigilance protocols to catch subtle trends early.
Future Prospects
Looking forward, the bismuth citrate market is branching out past classic stomach medicines. Polymer chemists see a role in smart drug delivery systems, leveraging its coordination chemistry to fine-tune release rates. Environmental scientists are experimenting with bismuth citrate-based materials for water purification, banking on low toxicity and high binding affinity for heavy metals like arsenic and lead. Calls for greener and safer chemicals encourage more industrial formulators to swap out more hazardous alternatives for bismuth citrate in niche pigments and catalysts. Regulatory pressures will likely increase the focus on purity and low contamination, pushing forward both production and analytical standards. Academic labs stay busy screening new derivatives for antifungal, antiviral, and even anticancer properties, inspired by how some related bismuth salts disrupt pathogen metabolism without harming host cells. The next big leap seems to rest with refining selectivity, lowering costs, and showing consistent long-term safety for both humans and the environment.
What is Bismuth Citrate used for?
What is Bismuth Citrate?
Bismuth citrate comes from combining bismuth, a metal known for its low toxicity, with citric acid, a common food and medicine additive. The result lands in a grayish-white powder that gets tucked away in many bathroom cabinets but rarely grabs attention unless someone battles a stomachache or colors their hair.
Taking on Upset Stomachs
Many folks might not know they already trust bismuth citrate. Some trusted over-the-counter remedies for heartburn, indigestion, nausea, or diarrhea feature this ingredient. My grandmother used to reach for a pink bottle when her stomach gave her trouble; it turns out that soothing coat on her stomach stemmed partly from the bismuth compound inside. Bismuth compounds, especially bismuth subsalicylate and bismuth citrate, play a role in fighting bacteria that bring on stomach lining irritation or occasional traveler's diarrhea. Lab tests show bismuth helps knock down Helicobacter pylori bacteria, one of the sneakiest culprits in peptic ulcers.
Protective Coating on the Stomach
Doctors often suggest adding a bismuth compound to a combination of antibiotics if someone gets diagnosed with an ulcer caused by bacteria. Bismuth citrate works by forming a barrier over injured tissue, keeping acids and enzymes from digging the wound deeper. This isn’t just textbook stuff; I’ve watched friends come back from gastritis, reporting meals no longer bring anxiety thanks to the numbing and shielding effect provided by bismuth found in their prescribed medicine.
A Surprising Role in Cosmetic Products
This is not just about stomachs—bismuth citrate shows up in some unexpected places. Hair dye manufacturers use this compound as a color additive, especially in products for those wishing to darken gray hair without harsh chemicals. Because bismuth citrate reacts with the sulfur in hair’s natural proteins, it helps bring a darker, softer tan without quick fading or excessive scalp irritation. Anyone who’s noticed metal ingredients on hair tint bottles has likely seen bismuth citrate quietly contributing there. The U.S. Food and Drug Administration (FDA) lists it as a color additive for these specific uses, and safety is monitored closely.
Safety Considerations: What We Know
If used as directed, bismuth citrate ranks as a safe choice for most people, but it doesn’t suit everyone. Heavy or prolonged dosage may cause black stool or tongue, something many first-timers find alarming but turns out to be harmless. Medical sources warn that those with kidney disease or pregnant individuals need to talk to their doctor before taking products containing bismuth compounds. Responsible companies label their bottles and packets with clear dosing instructions. A few years ago, I learned the importance of respecting these limits the hard way after a friend ignored them, thinking extra would mend their stomach quicker—only to end up with temporary nervous system symptoms that cleared up after stopping the medicine.
Looking Forward: Smarter Use and Public Awareness
Mixing age-old remedies with modern science, bismuth citrate earns its place in both medicine cabinets and hair product shelves. Pharmacists and doctors continue to educate patients on what works safely and what crosses a line. Improved packaging now spells out exact uses and warnings better than ever. Safety isn’t just on the user, but also on those who sell and regulate these solutions. A little knowledge and a little caution keep bismuth citrate helpful and safe for many households.
Is Bismuth Citrate safe for human consumption?
Looking at Bismuth Citrate's Place at the Table
Bismuth citrate pops up in the ingredient lists for some medications, especially those that aim to settle stomach troubles. You might have seen it in products used to ease heartburn, diarrhea, or nausea. Years back, my own doctor suggested a bismuth-based medicine during a rough patch of gastritis. People often wonder about the safety of compounds like this, especially since headlines about food and health risks tend to spark concern among families.
The Track Record So Far
Researchers and doctors have been using various bismuth compounds for more than a century. Bismuth itself doesn’t fall into the same camp as heavy metals like mercury or lead. It’s a heavier element, but it behaves much differently in the body. Most bismuth doesn’t dissolve well, so only a little of what you swallow gets absorbed. The rest heads out of the body with your waste.
Looking at studies in medical journals and FDA data, serious side effects from typical doses of bismuth citrate show up rarely. In most cases, people might experience some blackening of the tongue or stools, but these changes disappear when you stop taking the medicine. The National Institutes of Health and ConsumerLab reports reach similar conclusions: standard use in medications and food isn’t considered a health hazard for most adults.
Who Should Stay Cautious?
Like so many compounds, safety depends on who’s taking it and how much ends up in the body. Folks with kidney problems may want to avoid bismuth citrate, since their bodies have a harder time clearing out extra minerals. Medical reports show that, in rare cases and with unusually high or long-term use, bismuth compounds can build up and irritate the nervous system, causing confusion or trouble walking. This kind of risk rarely applies to people using over-the-counter stomach remedies as directed, but it makes sense to talk with a doctor if you have ongoing health problems.
Pregnant people also get reminders from doctors to avoid extra chemicals when possible, including bismuth compounds, mostly because safety data for these cases are thin.
Looking at Labels and Dosage
The FDA approves bismuth citrate for certain uses such as treating stomach issues, so products containing it meet requirements for quality and labeling. Reading the dosing instructions on medicine bottles matters. I’ve seen patients take more than the recommended dose, thinking it would work faster, only to end up with side effects they could have avoided. Stick to the dose, pay attention to any new symptoms, and check with a pharmacist or healthcare provider if you feel unsure.
What About Long-Term Use?
Most people use bismuth citrate for a week or two at a time, usually during a short burst of illness. There’s no good evidence to show that taking these compounds for short stretches leads to lasting problems in healthy people. Problems really begin with very high or long-term exposure, such as someone taking large doses daily for many months or years.
Making Smart Choices
Any food or medicine can cause trouble in excess or in the wrong hands. Bismuth citrate isn’t something to fear, but thoughtful use helps avoid problems. I’ve learned from experience and through conversations with pharmacists that having good information makes a difference. Informed decisions—not panic—go furthest in protecting health. So far, experts and regulators agree: for short-term, label-directed use, bismuth citrate remains a safe option for most adults.
What are the possible side effects of Bismuth Citrate?
Real-World Perspective on Bismuth Citrate
A bottle of bismuth medicine usually sits in a bathroom cabinet, waiting for that rough bout of indigestion or the unwelcome arrival of diarrhea. People choose products with bismuth citrate because they trust it will settle the stomach and help relieve upset. It’s found in over-the-counter relief medicines—those familiar black liquids or chalky pink syrups. As helpful as it feels when heartburn or an upset stomach hits, no medicine stays completely risk-free.
Spotting Side Effects Early
Some changes show up soon after taking bismuth citrate. The most common sign often surprises people the first time: blackened stool or a darkened tongue. The color change comes from harmless bismuth sulfide formed in the gut, but it can still raise concern if you’re not expecting it. Doctors hear stories about this all the time, and reassurance helps—regular handouts offered at clinics include this side effect at the top of the list. Folks can expect their stool or tongue to return to normal after stopping use.
Dry mouth or a mild constipation sometimes follows a dose of bismuth medicine. Staying hydrated often takes care of these symptoms, though folks who live with constipation know it doesn’t always fade so easily. On rare occasions, nausea or vomiting shows up, making a stressful situation even tougher. Each time, experience tells me it’s worth paying attention and giving the body a break if these issues don’t settle after a dose or two.
More Serious Reactions Need Attention
Certain symptoms demand a call to a healthcare provider. Trouble with breathing, sudden rashes or itching, dizziness, or confusion show that the body isn’t taking kindly to bismuth citrate. These can signal an allergic reaction—nobody should wait to see if it passes. Even outside obvious allergies, a feeling of persistent tiredness or troubles with coordination signals something is off. The nervous system can get involved if bismuth builds up in the system, though this rarely happens with normal short-term use.
People using bismuth citrate for more than a few days—especially those with kidney problems—face a higher risk of toxicity. Trouble walking, slurred speech, and mood changes have cropped up in published medical cases. These side effects underscore why some medicines shouldn’t be prolonged unless a doctor weighs in.
Risk Factors and Extra Caution
Not every stomach issue fits neatly into the same box. Anyone with pre-existing kidney problems finds themselves at greater risk. The kidneys clear bismuth out of the body, so slowdowns here can push bismuth levels too high. Children face a higher risk as well—their bodies handle elimination differently, so pediatricians prefer milder options unless absolutely needed.
Using other medicines brings up another layer. Aspirin or similar salicylates in the medicine cabinet can boost the risk of side effects. Layered medicines may seem harmless, but unexpected interactions cause real problems. My own experience with an elderly family member taking several medications showed me that even small ingredients add up quickly.
Staying Safe with Bismuth Citrate
Short-term use for familiar stomach issues works for most people, but reaching for the bottle over and over means the underlying cause might need a closer look. Doctors advise sticking to labeled instructions and stepping back after a few days if symptoms linger. For those living with chronic medical conditions, a routine talk with the pharmacist or doctor brings peace of mind. Medicines can bring relief, but keeping an eye out for changes—big or small—remains the best way to stay safe.
How should Bismuth Citrate be stored?
Why Proper Storage Matters
People often overlook how storage impacts chemical safety and quality. Bismuth citrate, found in a range of medicines and personal care items, stands out as a compound that suffers from common mishandling. I’ve seen careless storage lead to wasted product and avoidable hazards, both in labs and clinics. I want to share lessons learned through firsthand experience and reliable scientific sources.
Keep It Dry or Problems Start
Moisture invites trouble with most powders and salts, and bismuth citrate is no exception. Humidity clumping up a powder may seem minor at first glance, but the real danger lies in chemical change. I watched an unopened bottle turn lumpy over a damp summer, just because it sat on a window sill. The product lost its punch because moisture triggered degradation. Once that happens, the compound won’t work the same way—it’s like baking bread with spoiled yeast.
A dry environment truly saves money and headaches. That means sealed containers and a dedicated shelf or cabinet. Desiccant packs offer extra insurance. Each time you open the container, close it tightly right away. Even a few minutes of exposure can do harm if it’s a muggy afternoon.
Heat Isn’t a Friend
From storage rooms above a radiator to hot laboratory benches, I’ve run into heat-damaged supplies more times than I can count. High temperatures break down chemicals faster. Bismuth citrate holds up best at room temperature, away from sunlight or direct heat. Sunbeams through a window raise the temperature fast, sometimes enough to alter sensitive powders.
Many people skip over this and leave containers in trucks or mailrooms over the weekend. Once the contents get too hot, there’s no going back. The simple act of keeping things in a dark, cool cabinet extends shelf life.
Contamination Destroys Value
I've seen more than a few labs ruined by a single touch from dirty gloves or spilled solvent. Bismuth citrate should always live in clearly labeled containers—no mystery jars or old jam bottles. Throw away any ambiguous or open packages. Cross-contamination causes reactions you don’t expect, and those can throw off clinical results or even create safety hazards.
Choose glass or high-grade plastic containers with tight seals. Avoid metal scoops and utensils, which sometimes react with the compound. A cheap label maker or a big black marker solves a lot of future confusion, making sure no one guesses what’s inside.
Keep Kids and Pets Out
It’s simple: store bismuth citrate high up and out of reach if you keep it at home. I learned this lesson after my curious nephew tried to open a “cool powder” he found in the bathroom. Although not the most toxic material, unintentional exposure is never worth the risk. Locked cabinets or high shelves do the trick, especially in homes with children or animals.
Easy Solutions Make a Big Difference
Safe storage of bismuth citrate comes down to a handful of habits. Keep it dry, protect from heat, label every container, and guard against contamination. These steps, drawn from years of trial and error, protect people and save product. Simple precautions keep this useful compound working as intended.
Can Bismuth Citrate interact with other medications?
Everyday Medicines, Unexpected Overlaps
Plenty of people reach for bismuth citrate to calm a queasy stomach or handle digestive trouble. It’s in more than a few over-the-counter products. Folks don’t always pause to think about possible problems with other pills. I’ve seen neighbors stack antacids, prescription medication, and supplements, figuring nothing in the drugstore can trip them up. Experience tells me that’s risky. Bismuth compounds look tame, but chemistry inside the gut doesn’t care much for convenience.
How Bismuth Citrate Can Get In the Way
Drug interactions get messy fast, especially in the digestive tract. Bismuth citrate doesn’t stay quiet. It binds with other substances, sometimes stopping your body from absorbing what you need. Tetracycline antibiotics are a clear example. Studies have shown that bismuth can reduce the effectiveness of tetracycline, making infection harder to treat. It doesn’t take a medical degree to know that’s a bad trade-off.
That’s not the only trouble spot. Mixing bismuth citrate with medicines like digoxin or thyroid hormones can change how much of the drug actually reaches the bloodstream. Doctors have seen problems stack up for patients with heart or thyroid conditions simply because two pills crossed paths in the gut. Iron supplements and some anti-fungal drugs, such as ketoconazole, join the list as well. Each one competes with bismuth for absorption or gets chemically tangled up.
Not Just About Pills: Vitamins and Foods in the Mix
Plenty of folks don’t think twice about adding a multivitamin or calcium tablet in a daily routine. Bismuth citrate can trip up absorption for essential minerals and vitamins—not just prescription drugs. One cup of milk or a calcium-rich food slows bismuth absorption, which means those soothing effects you expect could suddenly go missing. Extra iron from a supplement gets bound up with bismuth, slipping right past your gut and onto the bathroom floor. That’s money—and nutrients—down the drain.
Symptoms Nobody Wants
Bismuth itself can build up if the body isn’t clearing it fast enough, especially if kidneys aren’t working at their best. Folks end up with black stools, confusion, and balance problems—something I saw once with an older relative who liked his antacids a little too much. Even though the FDA sets strict rules for product labeling, real-world use doesn’t always match safe use. It’s tough to spot these symptoms if you’re not on the lookout.
Sensible Steps: Talking, Timing, and Teamwork
Juggling prescriptions, supplements, and a bottle from the drugstore shelf just adds risk. Health professionals, especially pharmacists, have deep knowledge on these interactions. When I picked up a new prescription once, a sharp-eyed pharmacist caught a clash with a stomach remedy I’d planned on using. That conversation saved me a world of trouble.
Spacing out doses is simple, but powerful. A two-hour gap gives most medicines and supplements a fighting chance to get where they need to go, with less meddling from bismuth citrate. Writing down every product you use, even ones you buy without a prescription, arms your healthcare team with the information needed to spot trouble early and adjust your routine if needed.
Why It’s Worth Paying Attention
Every medicine in your cabinet works with or against everything else you swallow. Bismuth citrate finds its way into dozens of routines, but nobody gains much by adding mishap to the mix. Checking with the doctor, keeping the local pharmacist in the loop, and reading labels with fresh eyes go a lot further than gambling on a gut feeling.
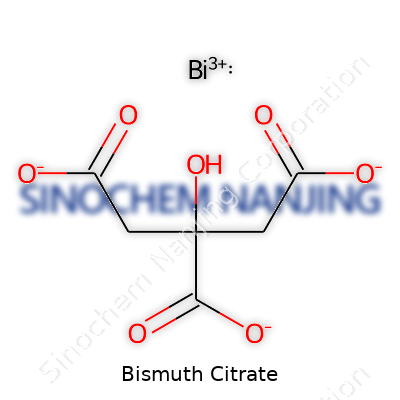
| Names | |
| Preferred IUPAC name | bismuth(3+) 2-hydroxypropane-1,2,3-tricarboxylate |
| Other names |
Bismuth(3+) citrate Bismuth(III) citrate Bismuth citrate (3:2) Citrate de bismuth Tribismuth dicitrate |
| Pronunciation | /ˈbɪz.məθ ˈsɪ.trət/ |
| Identifiers | |
| CAS Number | 813-93-4 |
| Beilstein Reference | 3581836 |
| ChEBI | CHEBI:60156 |
| ChEMBL | CHEMBL1201512 |
| ChemSpider | 12249 |
| DrugBank | DB10966 |
| ECHA InfoCard | ECHA InfoCard: 100.033.945 |
| EC Number | 262-182-4 |
| Gmelin Reference | Gmelin Reference: **3790** |
| KEGG | C18794 |
| MeSH | D001622 |
| PubChem CID | 16211219 |
| RTECS number | EB5580000 |
| UNII | 1DI56Q4489 |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID5023293 |
| Properties | |
| Chemical formula | C6H5BiO7 |
| Molar mass | 586.070 g/mol |
| Appearance | White to almost white crystalline powder |
| Odor | Odorless |
| Density | 2.1 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | -1.5 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~3.1 |
| Basicity (pKb) | 8.8 |
| Magnetic susceptibility (χ) | −1.66e-4 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.49 D |
| Pharmacology | |
| ATC code | A02BX05 |
| Hazards | |
| Main hazards | May cause respiratory tract, eye and skin irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| Lethal dose or concentration | LD50 oral rat 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Bismuth Citrate: "565 mg/kg (rat, oral) |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 3 mg/kg bw |
| Related compounds | |
| Related compounds |
Bismuth subsalicylate Bismuth subgallate Bismuth subcarbonate Bismuth subnitrate Bismuth oxychloride |

